Abstract
Keywords
Introduction
Antiretroviral therapy (ART) has been crucial in controlling the human immunodeficiency virus (HIV) epidemic, reducing morbidity and mortality, and leading to a life expectancy that is similar to age-matched peers. 1 As HIV is now regarded as a treatable, chronic condition, research focus has shifted to ensuring ART’s long-term efficacy, safety and tolerability. 2 An important milestone in this pursuit was discovering and approving integrase strand inhibitors (INSTIs). This includes the first-generation INSTIs raltegravir and elvitegravir, the second-generation agents dolutegravir (DTG), bictegravir (BIC), and the most recently approved cabotegravir.
Bictegravir was approved for the treatment of HIV-1 infection by the U.S. Food and Drug Administration (FDA) in March 2018 3 and is featured in the U.S. and European antiretroviral treatment guidelines.3,4 BIC is co-formulated as a fixed-dose combination (FDC) tablet with the nucleos(t)ide reverse transcriptase inhibitors (NRTIs) emtricitabine and tenofovir alafenamide (B/F/TAF) for once-daily treatment. Approval of this regimen was supported by several landmark phase III trials, in which it was demonstrated to be non-inferior to DTG-based regimens in establishing virological suppression over 96 weeks in treatment-naïve adults, 5 and was also shown to be non-inferior when switching to B/F/TAF compared to remaining on a DTG-based or boosted EVG- or protease inhibitor-based regimen in preventing virological rebound over 48 weeks in treatment-experienced patients. 6 Notably, no resistance emerged to any of the antiretroviral drug (ARV) regimen in the FDC in these studies. B/F/TAF is currently indicated for adults who have no ARV treatment history and to replace a current ARV regimen if the patient is virologically suppressed on a stable regimen for 3 months or more with no history of treatment failure and no known resistance to the FDC components. 7
INSTI-based regimens offer several advantages. For example, these regimens have activity against hepatitis B virus and HIV-1 non-B subtypes. Moreover, they do not require pre-treatment HLA-B5701 testing, making such regimens preferable for ‘rapid start’ treatment. Furthermore, INSTI-based regimens have a low potential for drug–drug interactions, as well as simple administration regarding meals. When compared to first-generation INSTI-based regimens, B/F/TAF has multiple benefits. B/F/TAF is the smallest-sized INSTI-based FDC for treatment-naïve patients, making it the favourite choice for patients with swallowing difficulties. Moreover, B/F/TAF has a relatively high barrier to resistance. The choice between DTG and BIC as part of triple therapy is often based upon the desired NRTI companion drugs present in the FDC.1,8,9
Most evidence for B/F/TAF use is based on phase III randomised clinical trials (RCTs), funded, and sponsored by Gilead Sciences.6,10–12 The internal validity of such trials is often achieved at the expense of uncertain generalisability, as populations enrolled in these studies may differ significantly from those typically using the intervention in practice. This is apparent with B/F/TAF, as although two large RCTs of the regimen confirmed efficacy, safety and absence of treatment-emergent drug resistance, at least 90% of participants were male, and 57% were White.5,6 By contrast, those affected by the worldwide HIV epidemic are 52% female and 72% Black, both factors associated with weight gain with INSTIs. 13 One exception is the BRAAVE 2020 study, a multi-centre, open-label study sponsored by Gilead Sciences, which evaluated switching from a regimen of two NRTIs plus a third agent, to B/F/TAF. Importantly, the study population consisted of virologically suppressed, HIV-1 infected African Americans, a historically under-represented group in HIV research. The study concluded that switching to B/F/TAF was non-inferior to continuing a variety of regimens, including for those with pre-existing NRTI mutations. 14
In response to such disparities, this scoping review aims to collate evidence on the efficacy, safety and tolerability of bictegravir-based ART, focusing on studies that Gilead did not sponsor but may have funded, such as investigator-led studies. Through this review, we hope to report on real-world evidence on the effectiveness, safety and tolerability of B/F/TAF. In addition to this, this review will also establish the characteristics of included studies, along with baseline participant characteristics.
Materials and methods
This scoping review used the PRISMA extension for scoping reviews checklist as a framework. 15
Information sources and search strategy
A series of pilot searches were conducted via Healthcare Database Advanced Search (HDAS) to identify relevant search terms and databases. The pilot searches were discussed with a university librarian specialising in medical science and literature searches. The following search terms and truncations were identified: efficac*, effect*, safe*, side effect*, adverse effect*, bictegravir* and biktarvy. Searching bictegravir OR biktarvy on HDAS revealed results from PubMed, EMBASE, EMCARE, Medline, CINAHL and BNI. These databases were then searched individually as part of the final search. In addition to these databases, Cochrane Library was also explored. Due to time and personnel constraints, a grey literature search was not performed. Reference searching did not yield any additional relevant sources.
The final search strategy used was (Bictegravir* OR biktarvy) AND (efficac* OR safe* OR effect* OR tolerab* OR ‘side effect*’ OR ‘adverse effect*’). The last search was performed on the 12th of August 2021.
Inclusion and exclusion criteria
Studies were eligible if they reported on the efficacy, effectiveness, safety or tolerability of bictegravir-based ART. Due to the emerging nature of evidence, relevant conference abstracts under 24 months old were included in addition to full-text articles. Studies were excluded if Gilead Sciences sponsored them, if the total population taking B/F/TAF was <20 or if B/F/TAF was used off-label for other conditions. Review articles, meta-analyses, case reports, preclinical studies and non-English publications were also excluded.
Results
After de-duplication, 333 studies were selected from peer-reviewed articles, and 27 were selected for full-text assessment after reviewing their title and abstract. Seventeen studies fulfilled the inclusion criteria after a full-text review. Data was extracted for inclusion and reporting by one author (EP), using Rayyan and Covidence and checked by the second author (CI). Figure 1 is a PRISMA flow diagram displaying the rationale behind study exclusion. PRISMA flow diagram (Adapted from Moher et al. 2009).
16
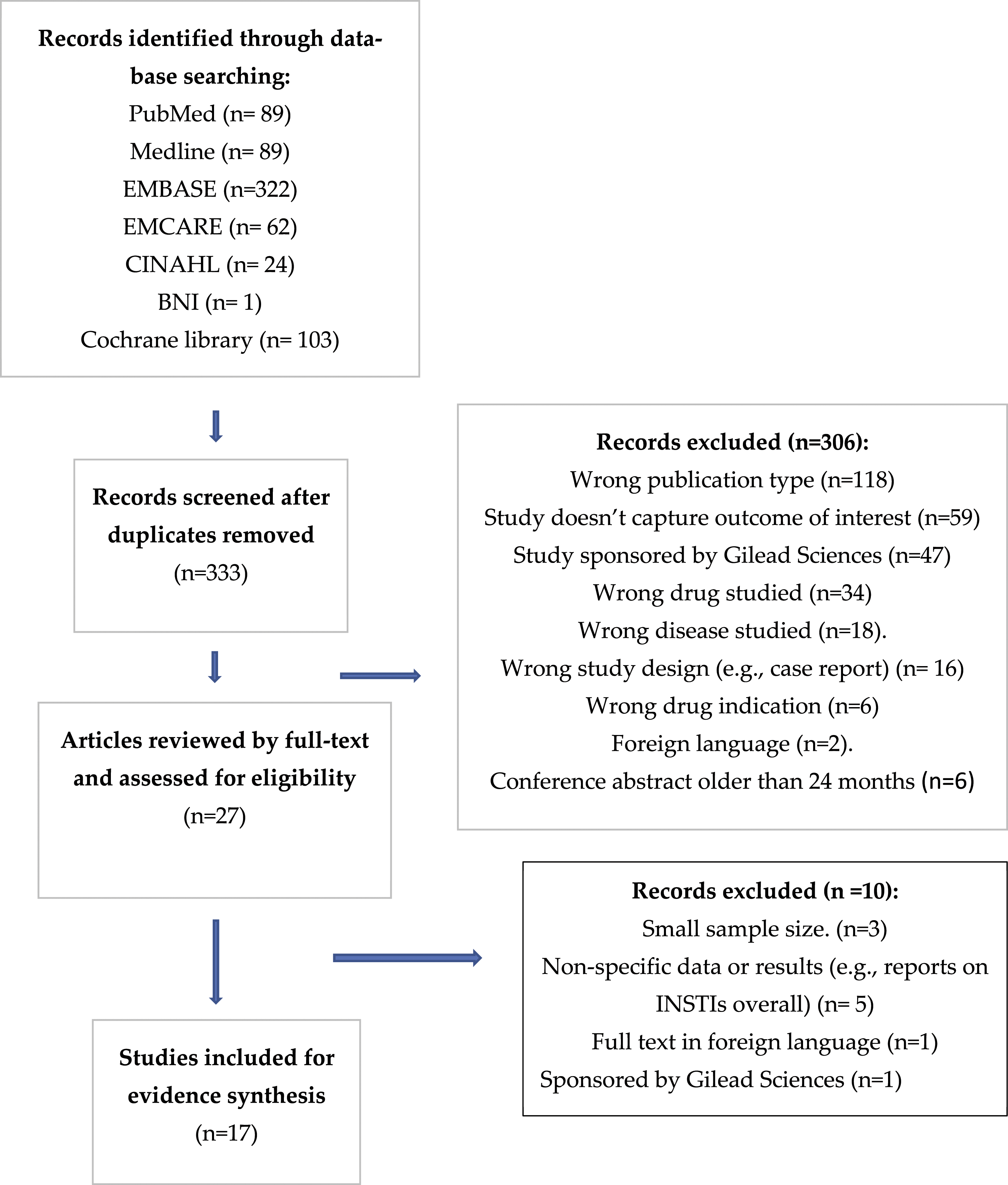
Study characteristics
Study characteristics and baseline participant information.
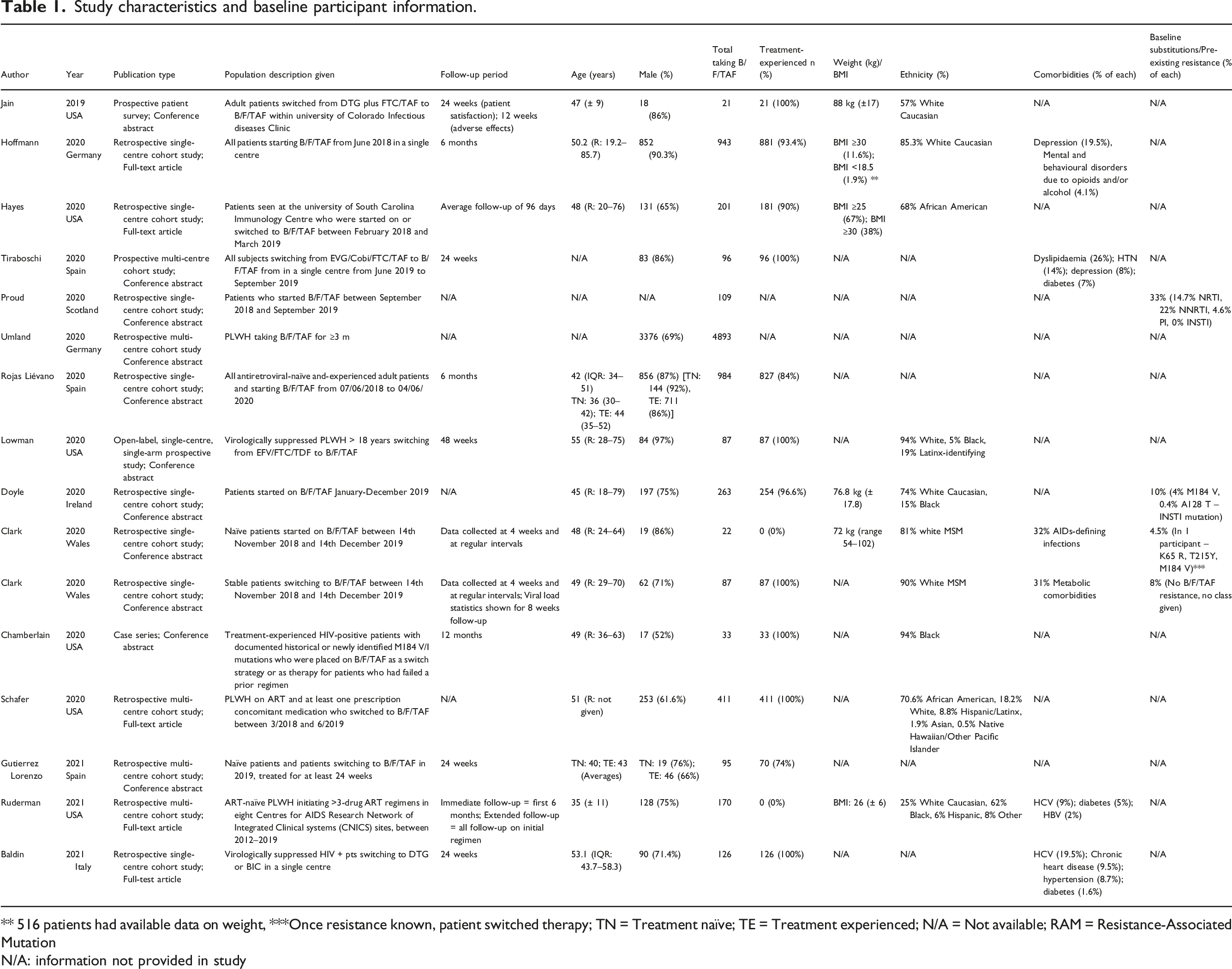
** 516 patients had available data on weight, ***Once resistance known, patient switched therapy; TN = Treatment naïve; TE = Treatment experienced; N/A = Not available; RAM = Resistance-Associated Mutation
N/A: information not provided in study
Baseline participant characteristics
This analysis included 8847 people living with HIV (PLWH). Of those with data on HIV treatment history, the majority were treatment-experienced (3380),17,18,21–24,26–32 whilst 465 participants were treatment-naïve.21,23,25,27–29,33 Two studies did not specify the percentage of treatment-naïve versus treatment-experienced PLWH, meaning 5002 PLWH in this analysis could not be categorised in this way.19,20
Switch information from studies with treatment-experienced participants.
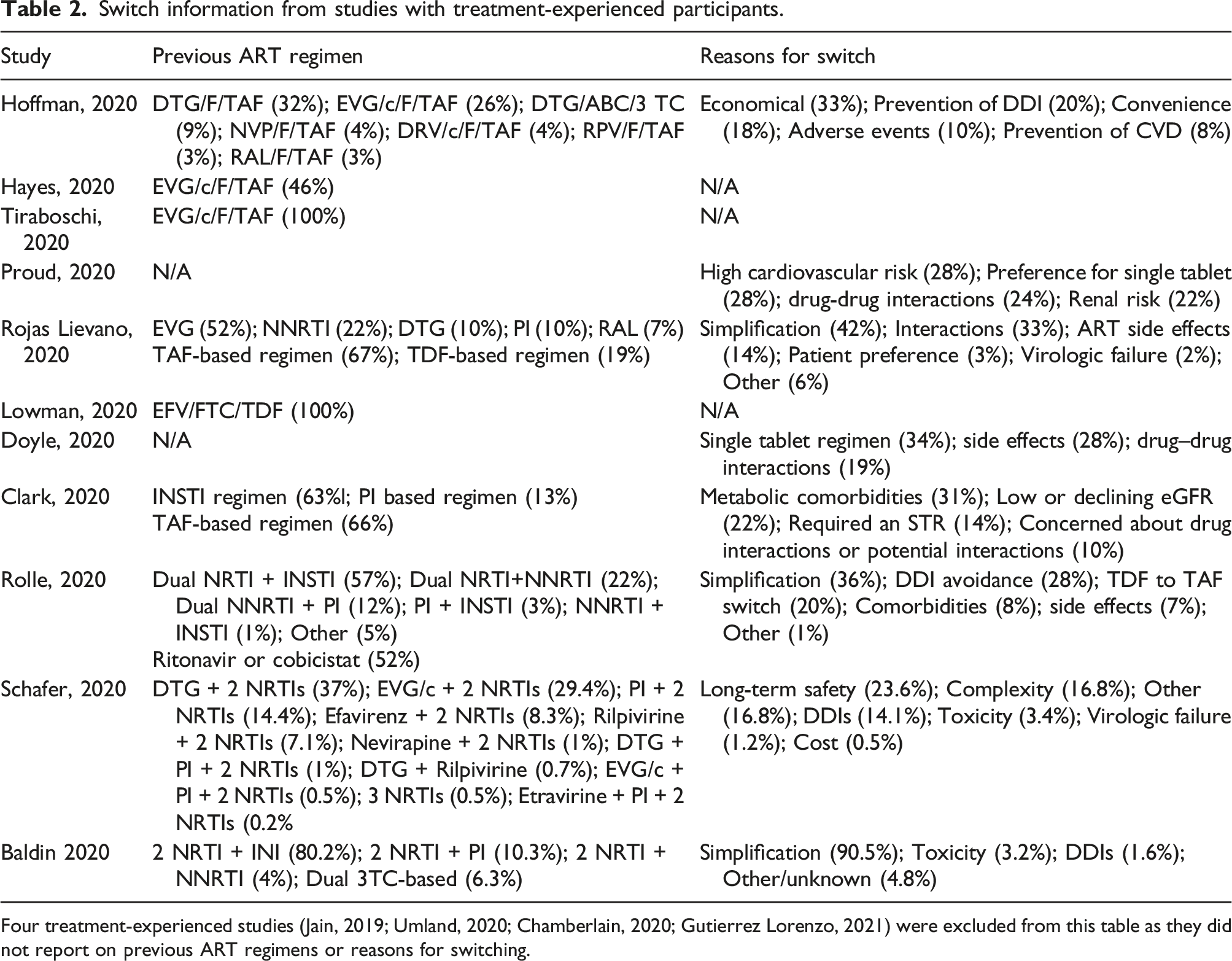
Four treatment-experienced studies (Jain, 2019; Umland, 2020; Chamberlain, 2020; Gutierrez Lorenzo, 2021) were excluded from this table as they did not report on previous ART regimens or reasons for switching.
In all sixteen studies reporting data on sex, the majority of PLWH were male (range: 52–97%).17,18,20–33 In the eleven studies reporting ethnicity data, seven consisted primarily of White/Caucasian PLWH.17,22–25,28,30 However, four studies were exceptions to this:26,29,31,33 the first included 68% African American participants; 29 the second included 94% Black participants; 26 the third included participants that were 70.6% African American, 18.2% White, 8.8% Hispanic, 1.9% Asian and 0.5% Native Hawaiian/Pacific Islander; 31 and the fourth included participants that were 72% Black, 25% White, 6% Hispanic and 8% other. 33
The mean or median age of study participants ranged between 35 and 58 years, with three studies not giving data on age.19,20 Seven studies included data on comorbidities; the most common comorbidities were metabolic abnormalities (two studies reporting, 26–31%),18,24 depression (two studies reporting, 8–19.5%),18,28 hypertension (two studies reporting, 8.7–14%),18,32 diabetes (three studies reporting, 1.6–7%),18,32,33 and chronic heart disease (one study reporting, 9.5%). 32 Moreover, one study focusing on treatment-naïve patients reported that 32% of participants had AIDs-defining infections, including Pneumocystis jirovecii pneumonia, cerebral toxoplasmosis, Kaposi’s sarcoma and cryptococcal meningitis. 25 Seven studies gave details on weight or body mass index (BMI).17,23,25,28–30,33 Of the three studies giving detail on weight, the average weight ranged between 72–88 kg.17,23,25 Of the four studies giving detail on BMI, the first had 11.6% of participants with BMI ≥30 and 1.9% < 18.5; the second had 67% of participants with BMI ≥25 and 38% ≥30; the third had an average BMI of 27.9 and the fourth had an average BMI of 26.28–30,33
Among the fifteen studies including treatment-experienced PLWH, nine gave information on previous ART regimens.18,21,22,24,28–32 The most common prior regimens were DTG-based and EVG-based. Eight studies detailed participants’ reasons for switching to B/F/TAF, the most common of which included simplification/convenience (eight studies reporting, range 18–90.5%), avoidance of drug–drug interactions (eight studies reporting, range 10–33%) and adverse events (eight studies reporting, range 3.2–28%)19,21,23,24,28,30–32 (Table 2).
Five studies reported the prevalence of baseline resistance-associated mutations (RAMs) or prior ART resistance, ranging between 4.5–35%.19,23–25,30 The first reported that 33% of participants had some form of prior antiretroviral resistance – 14.7% to NRTIs, 22% due to NNRTIs, 4.6% to PIs and 0% to INSTIs. 19 The second reported that 10% of participants had prior resistance mutations – 4% had the M184 V mutation, conferring resistance to lamivudine and emtricitabine, whilst 0.4% had the A128 T mutation, conferring resistance to INSTIs such as BIC. 23 The third study, focusing on treatment-experienced participants, had an 8% prevalence of RAMs, although none conferred resistance to B/F/TAF. 24 In the fourth study focusing on older adults, 27% of participants had historical genotype resistance data available of whom 31% had % ≥1 NRTI RAM, 31% had ≥1 NNRTI RAM, 35% had ≥1 PI RAM and 2% had % ≥1 INSTI RAM. 30 The fifth study focusing on treatment-naïve participants found three RAMs in one patient (K65 R, T215Y, M184 V) who had started ART before resistance testing results. After resistance was found, they were switched from B/F/TAF 25 (Table 1).
Efficacy of B/F/TAF
Efficacy of Biktarvy.
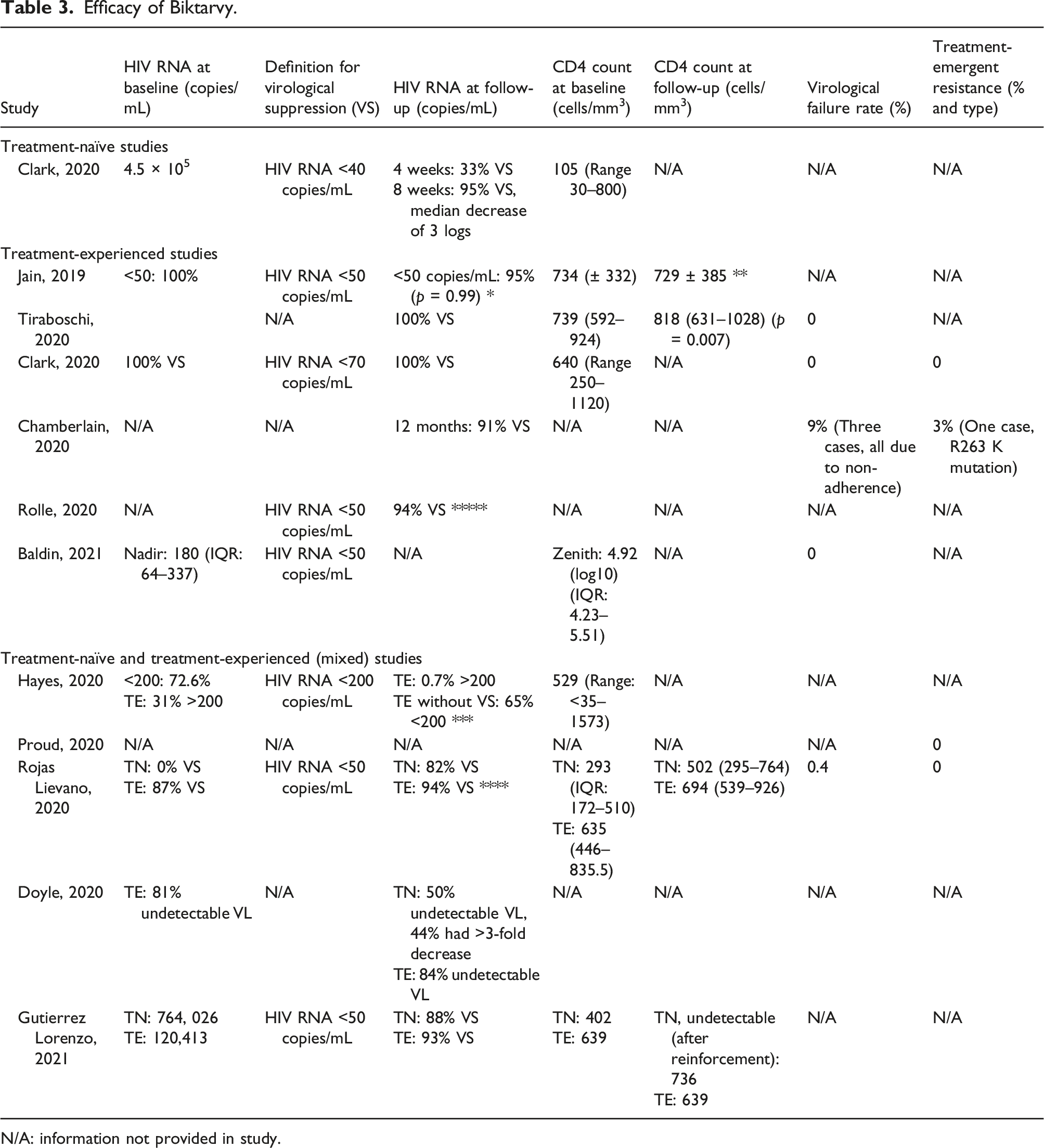
N/A: information not provided in study.
Virological effectiveness
Four studies reported on percentage of virological suppression in treatment-naïve participants, ranging between 50 to 95%21,23,25,27 (Table 3). When using on-treatment analysis, the first study reported that 82% of treatment-naïve participants had HIV RNA <50 copies/mL at 6 months follow-up, and 12% had HIV RNA between 50 to 200 copies/mL. 21 The second study found that on follow-up, nine of the 18 treatment-naïve participants had an undetectable viral load and 8 had a >3-fold decrease in HIV RNA. However, no follow-up time or definition for undetectable viral load was given. 23 The third study found that at 4 weeks, 33% had viral suppression, increasing to 95% at 8 weeks of follow-up (HIV RNA <40 copies/mL). In this study, there was a median HIV RNA decrease of 3 logs. 25 Moreover, the last study reported that after 24 weeks of follow-up, 88% had an undetectable viral load (HIV <50 copies/mL), whilst the remaining 12% failed due to poor adherence. When adherence was reinforced, these participants achieved undetectable viral load at subsequent follow-up. 27
Eight studies reported on percentage of treatment-experienced participants with virological suppression, ranging between 65 to 100%.17,18,21,23,24,26,29,30 The first study found that among the 95% of participants that had HIV RNA data available at 6 weeks follow-up after switching from DTG + 3 TC/TAF, there was no significant decrease in the percentage of participants with HIV RNA <50 copies/mL (HIV RNA < 50 copies/mL: 100% vs. 95% [
One study reported percentage of virological failure in treatment-naïve participants as 0%, after 24 weeks follow-up. 27 Four studies reported on percentage of treatment-experienced patients with virological failure, ranging between 0 to 8%.18,24,26,32 The first study found that in a cohort of patients switching to B/F/TAF, no virological failures occurred at 24 weeks follow-up, defined as two consecutive HIV RNA measurements of >50 copies/mL or a single determination of >1000 copies/mL. 32 Similarly, the second study found that at 24 weeks follow-up after switching from EVG/c/F/TAF, there was no instances of virological failure, although no definition was given. 18 The third study also found no virological failure, defined as HIV RNA >70 copies/mL, at follow-up, which ranged between 4 to 24 weeks. 24 However, the last study, which focused on treatment-experienced PLWH with the M184 V/I mutation, had a virological failure rate of 8%, although no definition was given. However, all cases of virological failure were attributed to treatment non-adherence. 26 Another study which did not differentiate between treatment-naïve and treatment-experienced patients had a virological failure rate of 0.4%. 21
Two studies reported on change in CD4 cell count in treatment-naïve patients, ranging between +209 to +334 cells/mm3.21,27 The first study found that after 6 months, median CD4 cell count increased from 293 to 502/mm3 (IQR: 295–764). 21 The second study found that in a treatment-naïve population with a median CD4 count of 402 cells/mm3 at baseline, the CD4 count increased to 736 cells/mm3 at 24 weeks of follow-up after adherence was reinforced. 27
Four studies reported on changes in CD4 cell count in treatment-experienced patients, ranging between −5 to +78 cells/mm3.17,18,21 At 12 weeks follow-up after switching from DTG + 3 TC/TAF, the first study found no significant difference between baseline and post-switch CD4 count ([mean ± SD (Standard Deviation)]: 734 ± 332 cells/mm3 vs. 729 ± 385 cells/mm3 [
Treatment-emergent resistance
No studies reported the percentage of treatment-emergent resistance in treatment-naïve patients. One study reported the percentage of treatment-emergent resistance in a cohort of treatment-experienced patients with documented M184 V/I mutations – one patient (3%) developed an R263 K mutation, conferring low-level resistance to bictegravir. 26 Two studies reporting the percentage of treatment-emergent resistance did not differentiate between treatment-naïve and treatment-experienced participants, with both studies reporting 0% – the first with unknown follow-up, 19 and the second with 6 months follow-up 21 (Table 3).
Safety of B/F/TAF
Safety of Biktarvy.
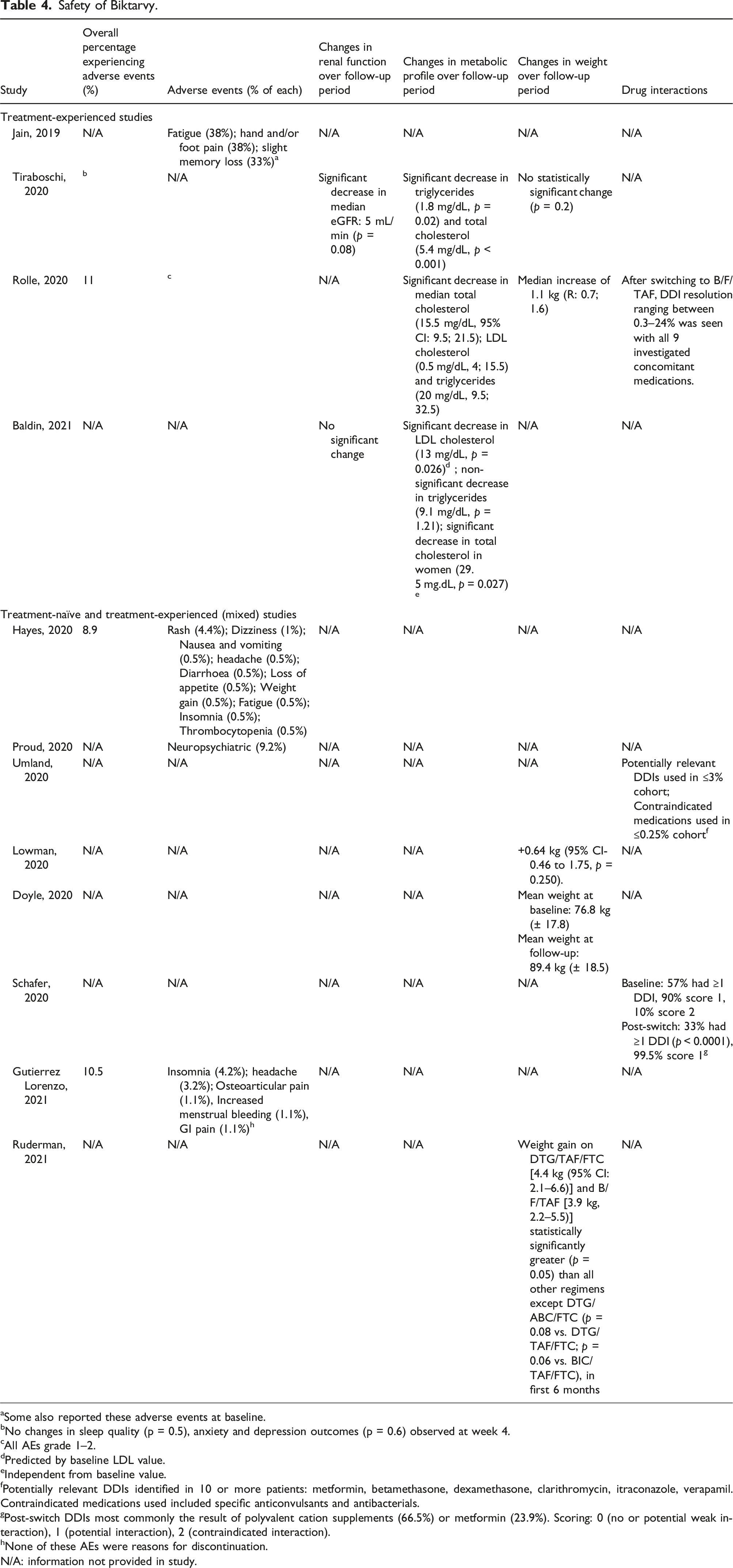
aSome also reported these adverse events at baseline.
bNo changes in sleep quality (p = 0.5), anxiety and depression outcomes (p = 0.6) observed at week 4.
cAll AEs grade 1–2.
dPredicted by baseline LDL value.
eIndependent from baseline value.
fPotentially relevant DDIs identified in 10 or more patients: metformin, betamethasone, dexamethasone, clarithromycin, itraconazole, verapamil. Contraindicated medications used included specific anticonvulsants and antibacterials.
gPost-switch DDIs most commonly the result of polyvalent cation supplements (66.5%) or metformin (23.9%). Scoring: 0 (no or potential weak interaction), 1 (potential interaction), 2 (contraindicated interaction).
hNone of these AEs were reasons for discontinuation.
N/A: information not provided in study.
In the three studies that gave detail on specific neuropsychiatric adverse events, the most common were insomnia (2 reporting, 0.5–4.2%),27,29 headache (2 reporting, 0.5–3.2%)27,29 and fatigue (2 reporting, 0.5–38%).17,29 The study reporting the highest individual NPAE rates was a prospective patient survey in which 38% of participants reported fatigue, 38% reported hand and/or foot pain and 33% had slight memory loss. However, some participants reported these symptoms at baseline, and there was no information given on the overall percentage of patients reporting AEs or whether some reported multiple symptoms. 17 Importantly, no studies gave a definition for NPAEs.
Lipid and renal outcomes
Two studies reported eGFR changes – the first found a significant median decrease in eGFR of 5 mL/min, 18 whilst the second found no significant eGFR change, 32 both after 24 weeks follow-up.
Three studies of treatment-experienced participants reported on changes in metabolic profile, all of which found significant improvements from baseline in various parameters.18,30,32 The first study involved participants solely switching from EVG/cobicstat/F/TAF and found a significant decrease in triglycerides and total cholesterol. 18 In the second study, 52% of patients were switched from regimens containing ritonavir or cobicistat with a significant decrease in triglycerides, total cholesterol and LDL cholesterol. 30 The third study, with 80.2% switching from another NRTI + INSTI regimen, found a substantial reduction in total cholesterol in women and LDL cholesterol overall, and a non-significant decrease in triglycerides.14,32
Weight outcomes
Four studies reported on changes in participant body weight.18,23,30,33 The first study found a median weight increase of 1.1 kg.
30
The second study compared antiretroviral regimes and their associated weight changes and concluded that weight gain on DTG/FTC/TAF (4.4 kg) and B/F/TAF (3.9 kg) was significantly higher than all other tested regimens except DTG/ABC/FTC, in the first 6 months of starting therapy.
33
The third study found no statistically significant change in weight (
Drug–drug interactions
Two studies solely focused on drug–drug interactions in PLWH taking B/F/TAF.20,31 The first study concluded that switching to B/F/TAF reduced the incidence of drug–drug interactions amongst PLWH receiving multiple concomitant medications. 31 The second study found that contraindicated medications were used in <0.25% of the cohort, and those with potentially relevant DDIs could replace medications with other compounds in the same drug class without interaction. 20 Moreover, another study, which focused on treatment-experienced PLWH aged 50 years or over, found that after switching to B/F/TAF, DDI resolution ranging between 0.3–24% was seen with all 9 concomitant medications investigated 30 (Table 4).
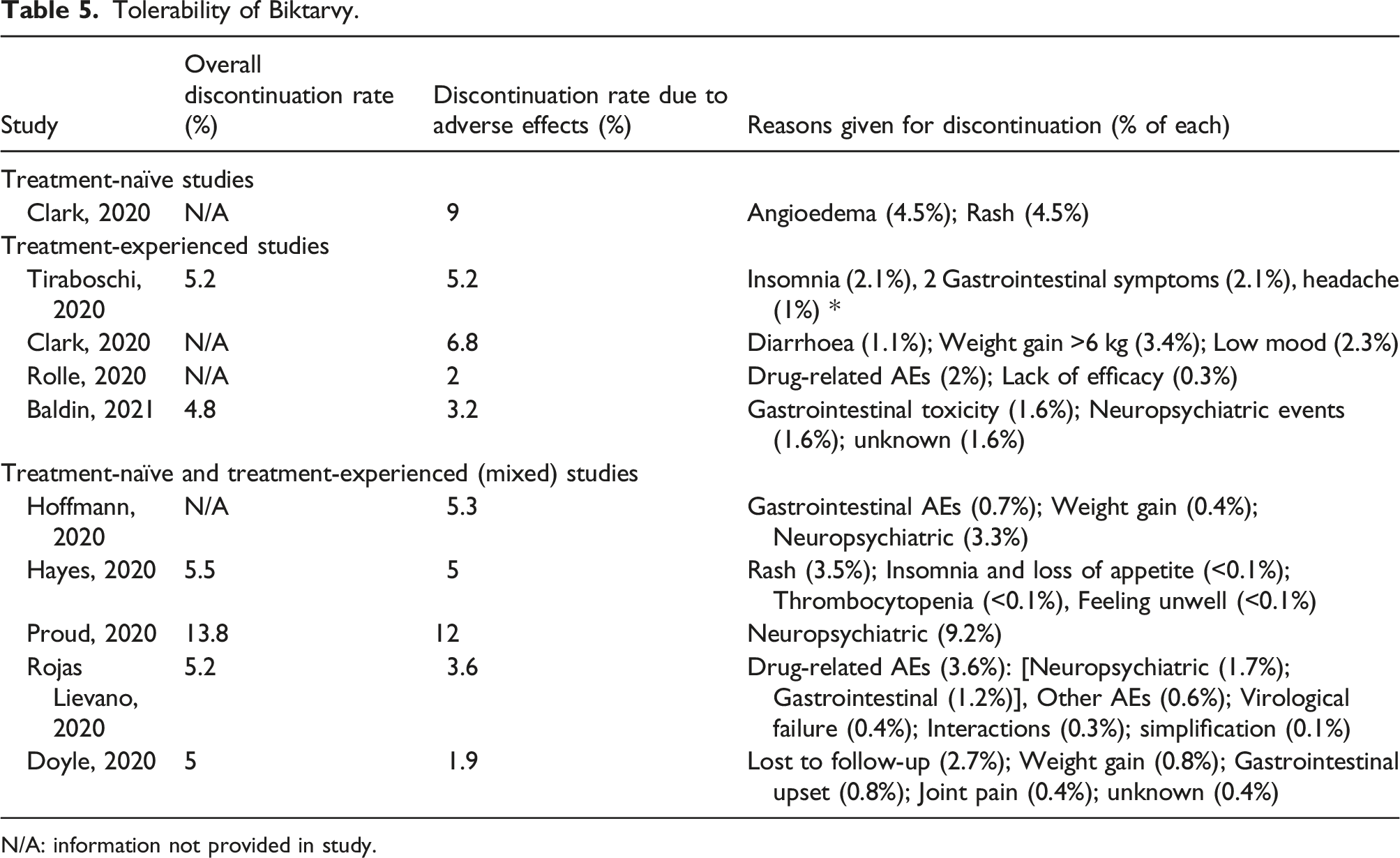
Tolerability of B/F/TAF
Tolerability of Biktarvy.
N/A: information not provided in study.
Comparison of regimens
Five studies focused on a comparison between other ART regimens and B/F/TAF.17,18,22,32,33 The first studied patients switching from dolutegravir plus FTC/TAF (2 tablets) to B/F/TAF. The prospective patient survey found that 91% of patients were ‘happy and satisfied’ with the switch. Moreover, no differences were observed when comparing baseline and post-switch laboratory parameters. 17 The second study compared virologically suppressed patients switching to B/F/TAF versus those switching to DTG + 3TC. The study found no significant difference in treatment discontinuation, or probability of treatment discontinuation due to neuropsychiatric AEs or central nervous system toxicity. Furthermore, both regimens showed favourable effects on cholesterol and triglycerides. 32 The third study found that switching from elvitegravir/cobicistat/FTC/tenofovir alafenamide to B/F/TAF did not result in significant weight gain. Moreover, no significant changes in sleep quality, anxiety or depression outcomes were observed at week 4 follow-up. 18 Similarly, the fourth study found that switching from efavirenz/emtricitabine/tenofovir disoproxil fumarate to B/F/TAF did not result in weight gain. 22 When comparing weight gain between different regimens, the last study found that in comparison to efavirenz/TDF/FTC, initiating B/F/TAF and DTG/TAF/FTC were associated with the greatest weight gain in the immediate period, followed by darunavir/TDF/FTC and DTG/TDF/FTC. 33
Discussion
Overall, this scoping review supports B/F/TAF as an efficacious, safe and tolerable regimen for both treatment-naïve and treatment-experienced PLWH. Virologic suppression in real-world studies was comparable to clinical trial data. For example, the virological suppression rates (defined as HIV-1 RNA of <50 copies/mL) for the two approval trials on treatment-naïve PLWH, studies 1489 and 1490, were 92.4% and 89.4% at week 48, respectively.10,12 In this scoping review, four studies reported on the percentage of virological suppression in treatment-naïve participants, ranging between 50 to 95%.21,23,25,27 The exception was in the study that included a small number of TN individuals, in which 50% had an undetectable viral load, the other 44% had a >3-fold decrease in HIV RNA, but no follow-up time was given. 23 Moreover, in the study where 82% of participants were virologically suppressed at follow-up, 12% had HIV RNA between 50–200 copies/mL. 21 The study where 88% had an undetectable load, the other 12% failed due to poor adherence, and after adherence was reinforced, these participants also achieved an undetectable viral load. 27
On the other hand, the virological suppression rates (defined as HIV-1 RNA <50 copies/mL) in the two approval trials of virologically suppressed PLWH switching to Biktarvy, studies 1844 and 1878, were 93.6 and 89.4% respectively.6,11 In this scoping review, eight studies reported on the percentage of treatment-experienced participants with virological suppression, ranging between 65 to 100%.17,18,21,23,24,26,29,30 The 65% virological suppression was seen in treatment-experienced patients who were not virologically suppressed at baseline prior to switching to B/F/TAF whilst another 25% did not have any follow-up viral load data to analyse. However, amongst those with virologic suppression at baseline, only one individual developed detectable viral load following a switch to B/F/TAF. 29 Moreover, in the study reporting 84% virologic suppression in treatment-experienced participants at follow-up, 81% of participants were virologically suppressed at baseline. 23
However, compared to clinical trial data, the incidence of treatment discontinuation due to adverse effects was significantly higher in real-world studies, prompting further investigation. For example, the percentage of patients discontinuing Biktarvy due to AEs in the two approval trials for treatment-naïve PLWH was 0% and 2%, with overall treatment discontinuation rates at 6.1% and 8.8%, respectively.10,12 This contrasts with the studies included in the scoping review – discontinuation rates due to adverse effects ranged between 1.9 to 12% in studies that did not specify TN versus TE participants.19,21,23,28,29 Furthermore, in one study focusing on TN patients, discontinuation due to AEs was 9%. 25 On the other hand, the percentage of patients discontinuing Biktarvy due to AEs in the two treatment-experienced approval trials were 2% and 1%, with overall treatment discontinuation at 5.3% and 5.5%, respectively.6,11 This also contrasts with the studies included in the scoping review, as discontinuation rates due to AEs in studies focusing on TE participants ranged between 2 to 6.8%.18,24,30,32
One specific AE that was found to be higher in some real-world studies compared to the clinical trials leading to Biktarvy approval was rash. For example, a study with both treatment-naïve and treatment-experienced participants reported a 4.5% incidence of rash, with 3.5% of participants discontinuing due to this. The population in this study was 68% African American and 67% overweight, which were both higher percentages than seen in most other studies. 29 Moreover, another study focusing on treatment-naïve participants reported that 4.5% of participants discontinued Biktarvy due to rash. 25 However, the Biktarvy package insert details that in the two treatment-naïve approval trials, rash was reported to occur in less than 2% of subjects, and that this was similar in the two treatment-experienced trials. 34 Interestingly, no other literature reports increased rash with B/F/TAF. Of note, low CD4 count (<200 copies/mL) has been associated with rash, such as pruritic papular eruption. 35 However, for the two studies reporting on rash incidence, the average baseline CD4 counts were within normal limits (640 [range 250–1120] 29 and 529 [range <35–1573] copies/ml. 25 It is unclear whether racial demographics or physiological differences such as weight and decreased metabolism may also predispose to rash development. To further investigate this, a larger-scale prospective study is required.
Another AE important to investigate in a real-world context is weight gain, which has been demonstrated previously with other INSTIs. 33 Out of the five studies in this scoping review reporting weight change, four report a median increase in weight. Three of the studies reporting weight gain were performed in mixed treatment-naïve and treatment-experienced populations,22,23,33 whilst one was in a solely treatment-experienced population. 30 Moreover, three studies reported treatment discontinuation due to weight gain. Two were studies in mixed treatment-naïve and treatment-experienced populations, reporting discontinuation rates of 0.4% and 0.8%, respectively.23,28 In a solely treatment-experienced population, 3.4% discontinued due to weight gain over 6 kg. 24 These results contrast with the clinical trials leading to approval of Biktarvy, which all reported no discontinuation due to weight-related AEs.6,10–12 Despite these results, the possible mechanisms behind a proposed link between B/F/TAF and weight gain are not completely clear. Previously, it was thought that weight gain on ARVs was solely due to a ‘return to health’ effect in untreated PLWH, which may be more evident with highly efficacious ART regimens such as Biktarvy, where an increase in CD4 count is faster. However, as the prevalence of obesity in PLWH continues to increase, further investigation into other weight gain mechanisms is necessary.33,36 Importantly, no included studies reported on the development of metabolic syndrome, diabetes or cardiovascular complications. In addition to reporting on weight changes, future research should investigate these clinically important outcomes
Of note, one study of patients switching from EVG/F/TAF to B/F/TAF found a significant decrease in eGFR of 5 mL/min. Bictegravir is a known inhibitor of organic cation transporter 2 and multidrug and toxin extrusion protein 1, which decreases tubular secretion of creatinine. However, this effect does not alter glomerular function, so this study finding is unlikely to be clinically significant. 37
NPAE incidence in real-world studies was generally comparable to the clinical trials leading to the approval of Biktarvy. For example, study 1489 in treatment-naïve participants reported 11% headache, 6% fatigue and 4% insomnia, with no discontinuation due to AEs. 10 Moreover, study 1490 in treatment-naïve participants reported 13% headache, 6% fatigue and 5% insomnia, with one patient (0.3%) discontinuing due to sleep disorder, tension headache and depressed mood. Importantly, it was not determined if individual adverse events were treatment-related in these two studies. 12 On the other hand, study 1844 in treatment-experienced participants reported 2% headache, <1% abnormal dreams and <1% fatigue, all determined to be treatment-related. Treatment-related NPAEs leading to drug discontinuation were headache (<1%) and abnormal dreams (<1%). 6 Study 1878 in treatment-experienced participants reported a 5% incidence of treatment-related headache. One patient (3%) discontinued due to schizophrenia, which was determined to be treatment related. 11 Interestingly, although NPAE incidence was generally comparable with clinical trials, discontinuation due to NPAEs tended to be higher in real-world studies included in this scoping review, ranging between <0.1–9.2%,18,19,21,24,28,29,32 again prompting further investigation.
A few weaknesses must be accounted for in this scoping review. Firstly, reporting of study demographics was varied. For example, there were significant disparities in sex and ethnicity data reporting. Moreover, not all studies reported the percentage of treatment-experienced participants, leaving a substantial portion of the overall pan-study cohort unanalysable in this way. This may be because most included studies were conference abstracts. Study population sizes also varied significantly, increasing the risk of inaccuracy if comparing results directly. Significantly, when looking at efficacy outcomes, different studies used different definitions for virological suppression and virological failure, with some not giving any definition, meaning studies reporting efficacy could not be compared directly. Moreover, there was inconsistent reporting on whether AEs were treatment-related, meaning direct comparisons between studies were limited. Furthermore, no studies gave a precise definition for NPAEs.
Future real-world studies are required to better characterise the efficacy, safety and tolerability of B/F/TAF, especially within under-represented subgroups such as women, pregnant people, older adults and children. These studies should have larger sample sizes and include in-depth demographic data. Although the included real-world studies were more ethnically diverse than the phase III trials leading to the approval of B/F/TAF, most studies still had cohorts that were predominantly White Caucasian, though four studies were an exception to this pattern. Moreover, no studies were conducted in Asian or African countries – all took place in the US and Europe. Considering the known disparities in healthcare resources, it is important to characterise real-world efficacy, safety and tolerability in countries outside of the Global North.
Footnotes
Acknowledgements
Advice on literature search given by Stephen Bowman, Medical School Librarian.
Author contributions
EP did the literature search and wrote the first draft of the manuscript with guidance from CI. Both authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CI has received honoraria for consulting services, conference attendance support and research grant funding from Gilead Sciences. EP declares no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
All data are contained within the manuscript.
