Abstract
Enteroviruses can cause severe, chronic infections in patients with primary and secondary humoral immunodeficiencies. These patients may benefit from anti-enteroviral therapy. Here, we report a patient with mantle cell lymphoma treated with chemotherapy followed by autologous stem cell transplantation and rituximab maintenance therapy, who presented with echovirus 7-associated deafness and myositis leading to severe disability. She showed marked clinical improvement and enterovirus clearance from faeces and blood after treatment with intravenous immunoglobulins (IVIgs) followed by remdesivir. We demonstrated efficacy of IVIg and remdesivir against echovirus 7 using virus neutralization and cell culture assays, which supports a potential contribution to the treatment success for both therapies.
Introduction
Enteroviruses of the
Effective treatment is urgently needed for the small group of patients with protracted, severe enterovirus infections. Sporadic cases have been treated with antiviral agents with known
Case report
The patient is a 70-year-old woman with a medical history of stage IV mantle cell lymphoma (MCL). For this, she was treated with rituximab/cyclophosphamide/doxorubicin/vincristine/prednisolone (R-CHOP) and rituximab/dexamethasone/cytarabine/cisplatin (R-DHAP) and subsequently with carmustine/etoposide/cytarabine/melphalan (BEAM) and autologous stem cell transplantation followed by 2-monthly rituximab maintenance treatment for a total duration of three years. Six months before hospital admission, she experienced acute bilateral perceptive hearing loss that was steroid-refractory and progressed to deafness. No tumour was found on magnetic resonance imaging (MRI) of the cerebellopontine angle. Auto-immune serology was negative. Liquor puncture revealed polyclonal pleocytosis with a positive real-time 5′UTR PCR for enterovirus (Ct value 27) and otherwise negative polymerase chain reactions (PCR) for neurotropic viruses. At that time, it was considered unlikely that the patient’s deafness was caused by enterovirus infection, because enteroviruses are highly prevalent in the population, do not commonly cause deafness and the patient tested negative by PCR on several earlier nasopharyngeal swabs obtained while she was already symptomatic.
Two months later, she was re-admitted to the hospital for progressive clinical deterioration with severe limb girdle weakness, episodic fever, weight loss and pancytopenia. An 18F-fluorodeoxyglucose positron emission tomography-computed tomography (PET-CT), bone marrow biopsy and repeated MRI of brain and spine did not demonstrate MCL recurrence nor hemophagocytosis. We refrained from a new lumbar puncture because asymptomatic hygromas were detected and a suboccipital liquor puncture was considered too risky. Serum creatine phosphokinase (CPK) was normal, but MRI of the large muscle groups of all extremities showed extensive oedema. Antinuclear antibodies (ANA) and myositis immunoblots were negative. A MRI-guided skin-muscle-fascia biopsy demonstrated a myositis with lymphocytic cellular infiltrate and striking mitochondrial abnormalities. Myxoma resistance protein 1 (Mxa) staining on muscle tissue was positive in addition to a strong interferon type 1 signature in the circulation and upregulation of Interleukin-18, suggestive of a chronic viral infection. Infectious work-up showed a positive PCR for enterovirus (Ct value 34) on feces, in absence of diarrhoea or abdominal complaints. Enterovirus PCRs on muscle tissue as well as on EDTA plasma were also found positive (Ct value 28 and 35, respectively), but again negative on a nasopharyngeal swab. The enterovirus initially found in the cerebrospinal fluid was typed as echovirus 7 (E7) by genotyping of a 350–400 bp sequence fragment of the viral capsid protein (VP)1 gene. 10 We diagnosed this immunocompromised patient with enterovirus-associated deafness and myositis. We started treatment with IVIg of 2 mg/kg body weight divided in 5 daily dosages of 30 g. After completion of IVIg, no significant clinical improvement was observed and enterovirus PCR on EDTA plasma remained positive (Ct value 38). One week later, we also started remdesivir (a loading dose of 200 mg, followed by 100 mg once daily, for 5 days in total). On week 2 of treatment, we noted a significant clinical and neurological improvement and enterovirus PCR was negative on blood and feces. After rehabilitation, she regained the ability to walk unsupported, although some balance issues remained. Deafness also persisted, for which she received a cochlear implant. She continued on maintenance IVIg every 3 weeks and enterovirus remained undetectable by PCR in blood and feces. We discontinued rituximab maintenance treatment for MCL.
We tested the Efficacy of IVIg and remdesivir against clinical enterovirus isolates. (a) Neutralizing antibody (nAb) titers against echovirus 1 (E1), echovirus 30 (E30) and four strains of echovirus 7 (E7) in IVIg from four different batches (shades of purple), including the one that the patient received (IVIg patient), and in six sera (S, shades of green) from the patient before (S1, obtained in 2022) and during the infection (S2-6, obtained between April and July 2024). nAb titers are averages of duplicate measurements, calculated using the Reed Muench method. nAb titers ≥16 and ≥32 were considered positive and protective, respectively. (b) Enterovirus A71 (EV-A71), E7 (3) and E30 cytopathogenic effect (CPE) in single measurements on RD cells after 24, 48 and 72 h of incubation without (shades of red) and with (shades of blue) 4 μM remdesivir (REM). Lower CPE indicates stronger inhibition of the virus.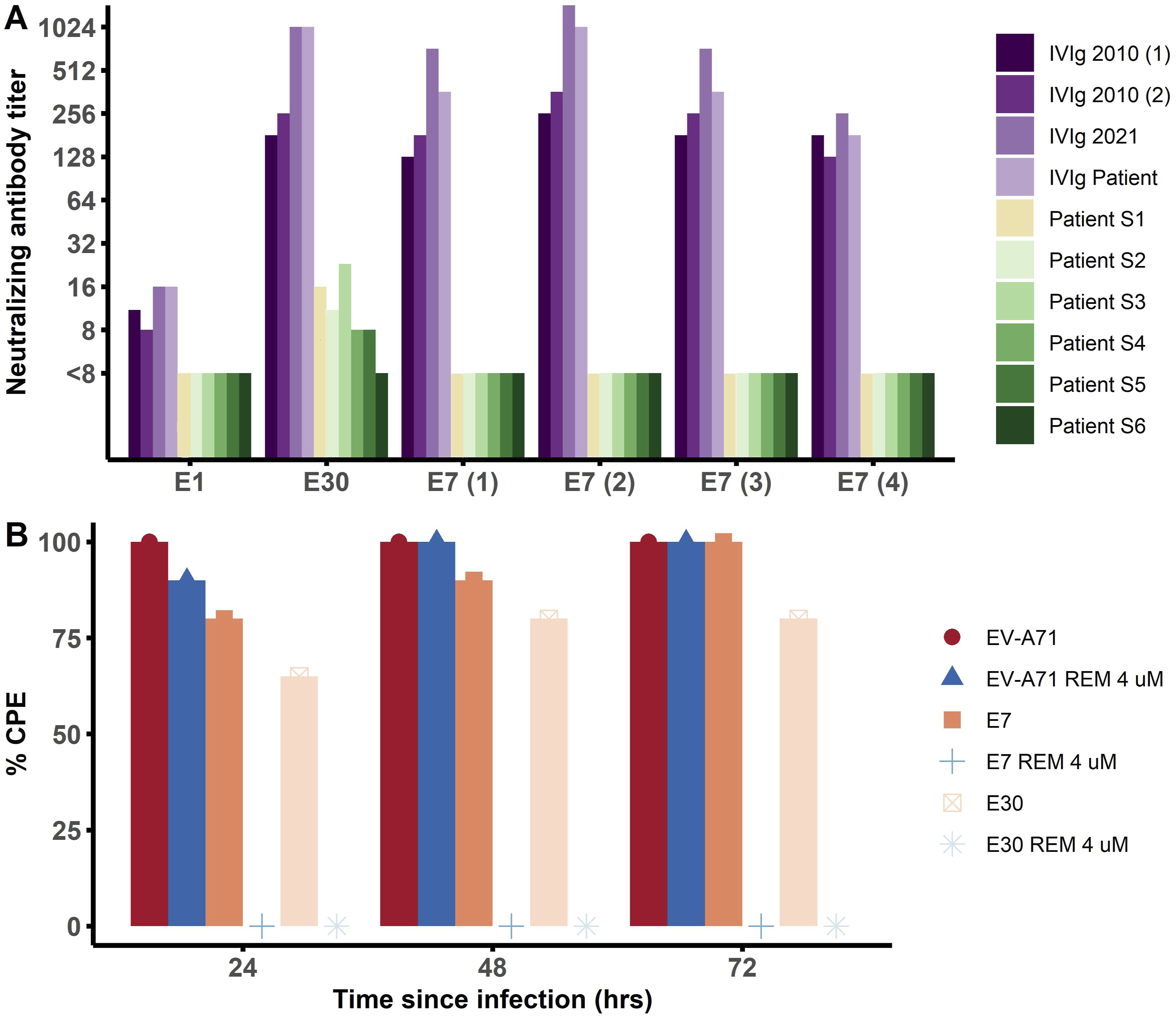
Discussion
This case report demonstrates clinical and virological success of an experimental therapy combining high dose IVIg with the broad-spectrum antiviral remdesivir in an immunocompromised patient with enterovirus-associated deafness and myositis following rituximab. Although these symptoms are not classically attributed to enterovirus infection, similar cases with deafness and/or myositis as presenting symptom have been described previously.2,14,15 In particular, we found one case report of a 67-year-old female on maintenance rituximab therapy for MCL, who presented with hepatitis, bilateral hearing loss and myalgias and had enterovirus (serotype unknown) detected in cerebrospinal fluid by PCR.
4
She was treated with IVIg and showed clinical improvement after the first infusion, though deafness persisted.
4
In our patient, timing of clinical improvement and virus clearance from blood and feces only after starting remdesivir suggests that the antiviral was an important component of the treatment. Nonetheless, we demonstrated
Supplemental Material
Supplemental Material - Enterovirus-associated deafness and myositis in an immunocompromised patient with in vivo and in vitro efficacy of intravenous immunoglobulins and remdesivir: Case report
Supplemental Material for Enterovirus-associated deafness and myositis in an immunocompromised patient with in vivo and in vitro efficacy of intravenous immunoglobulins and remdesivir: Case report by Emma M. de Koff, Harmen van Andel, Hanna K. de Jong, Anneke J. van der Kooi, Carlemi Calitz, Gerrit Koen, Arjan J. Kwakernaak, and Katja C. Wolthers in Antiviral Therapy.
Footnotes
Ethical considerations
We comply to practice guidelines on research integrity and publishing ethics and the committee on Publication Ethics.
Consent for publication
Patient provided written informed consent for participation in this study and publication of this case.
Author contributions
EdK wrote the manuscript together with AJK and KCW. AJK also provided clinical immunology care of the patient at the in- and outpatient clinic. HvA, HKdJ and AJvdK provided haematological, infectious disease and neurology consultation, respectively. EdK and KCW provided virological consultation. CC and GK performed laboratory investigations under the supervision of KCW. The manuscript was revised and approved by all authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the European Union’s Horizon 2020 Research and Innovation 362 Programme under the grant agreement GUTVIBRATIONS [grant number 953201 to KCW and CC].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data available on request from the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
