Abstract
Background
In Nigeria, the effect of Hepatitis B virus (HBV) on long-term liver outcomes in persons with HIV (PLH) has not been described. We determined changes in liver stiffness measure (LSM) using transient elastography over 6 years in HIV mono-infected and HIV-HBV co-infected Nigerians initiating antiretroviral therapy (ART) and factors associated with LSM decline.
Methods
This single centre, cohort study enrolled ART-naïve HIV mono- and HIV-HBV co-infected adults (≥18 years) at the APIN Public Health Initiatives–supported HIV Care and Treatment Centre at Jos University Teaching Hospital, Nigeria, from 7/2011 to 2/2012. LSM at baseline, Years 3 and 6 were analysed using longitudinal models to estimate changes over time and their predictors.
Results
Data from 100 (31%) HIV-HBV co-infected and 225 (69%) HIV mono-infected participants were analysed. Median LSM at baseline was 6.10 (IQR: 4.60–7.90) kPa in co-infected and 5.10 (IQR: 4.40–6.10) kPa in mono-infected participants. In adjusted analyses, average LSM was not significantly different between Year 0 and 3 (β = 0.02, −0.22 to 0.26, p = 0.87 and Year 0 and 6 (β = −0.02, −0.23 to 0.27, p = 0.88) in both groups (p>0.05), but co-infected participants had significantly higher LSM than mono-infected throughout follow-up (β = 0.018, 0.019–0.28, p < 0.001). Year 3 LSM differed according to ART initiation status by Year 3 (initiators - non-initiators: −0.87, −1.70 to −0.29).
Conclusion
In this cohort, LSM remained higher among HIV-HBV co-infected versus HIV mono-infected participants throughout follow-up. Our findings emphasize the continuing need for monitoring of liver outcomes in HIV-HBV co-infected populations on ART and the importance of preventing HBV infection among PLH to optimize liver health.
Introduction
Liver diseases are one of the major non-AIDS causes of mortality among people living with HIV (PLH) [1,2]. Although HIV itself damages the liver, co-infection with Hepatitis B virus (HBV) significantly increases the risk for liver-related morbidity and mortality [3–5]. The use of antiretroviral therapy (ART) containing HBV active drugs (tenofovir (TDF) and lamivudine (3TC) or emtricitabine (FTC)) has led to significantly improved health outcomes in co-infected individuals [6–8]. However, large cohort studies have continued to report elevated risk of liver-related mortality among HIV-HBV co-infected participants compared to HIV mono-infected despite treatment [9–12]. In sub-Saharan Africa and other low and middle-income countries (LMICs), several cross-sectional studies reported elevated rates of significant fibrosis and cirrhosis in HIV-HBV co-infected [13–15]; however, there are fewer longitudinal assessments of liver fibrosis after ART initiation. Most have been limited to 1–3 years of follow-up and report early significant declines in liver fibrosis, likely due to ART-induced short-term reductions of HIV-related liver inflammation [8,16–18]. Long-term evaluations of liver fibrosis in both HIV mono-infected and HIV-HBV co-infected populations are necessary to understand how HBV interacts with HIV to cause liver damage and gain insights on how to preserve liver health among PLH. These studies are particularly important in Nigeria where ∼15% of PLH are HBV co-infected [19,20] and other competing causes of liver disease (e.g. aflatoxin exposure) exist.
In this study, we assessed changes in liver stiffness (LSM), obtained by transient elastography (TE), over 6 years among HIV mono-infected and HIV-HBV co-infected participants enrolled in an observational study at Jos University Teaching Hospital (JUTH) in Jos, Nigeria. Previously, we found significant declines in LSM among HIV-HBV co-infected participants to levels comparable to those with HIV, after 3 years of observation [17]. In this follow-up, we examined whether changes in LSM were sustained and assessed factors associated with LSM changes including the independent effects of ART.
Patients and methods
Study design and population
229 HIV mono-infected and 101 HIV-HBV–co-infected (at least one positive hepatitis B surface antigen (HBsAg) test; [Alere Determine™ HBsAg] patients were enrolled into a prospective longitudinal research cohort at APIN-supported HIV Care and Treatment Clinics at JUTH between July 2011 and February 2012. Patients were included if they were ≥18 years, HIV antibody seropositive, ART-naive and hepatitis C virus antibody-negative. Also, they had to have a known HBsAg status and baseline TE. Patients were excluded if they had evidence of ascites, decompensated liver disease and/or known hepatocellular cancer. No patients were excluded due to unreliable TE measurements. Patients receiving ≥2 weeks ART prior to enrolment were also excluded. We obtained written informed consent and the study was approved by the JUTH Ethics Committee and the Northwestern University Institutional Review Board. All analysis was done in R 4.0.3 (see details in Additional File).
Data collection
Clinical care of all PLH at JUTH follows Nigerian and World Health Organization (WHO) guidelines [21,22] and was updated as guidelines changed. As part of standard care, participants were evaluated monthly to every 3 months in the clinic (on ART) or every 6 months (when not on ART), and at each visit, patients are provided with ART refills and treatment adherence counselling where applicable. Laboratory testing, including haemoglobin (g/dL), CD4+ T cell count (cells/mm3), alanine aminotransferase (IU/L) and HIV RNA [Roche Diagnostics, GmbH, Germany; lower limit of detection (LLOD) <200 copies/mL], were performed every 6–12 months. Recommended ART at the time of study enrolment (2011) included the combination of TDF or zidovudine (AZT) plus 3TC or FTC and a non-nucleoside reverse transcriptase inhibitor and was initiated in participants according to criteria in the Nigerian and WHO guidelines [21,23]. Criteria for ART initiation at the beginning of the study included CD4+ T cell count<350 cells/mm3, WHO stage 3 or 4 irrespective of CD4+ T cell count, tuberculosis co-infection, and those requiring treatment for HBV. In 2015, criteria for ART in HIV and HIV/HBV co-infected were modified to initiate ART in all individuals regardless of CD4+ T cell count [23].
Study participants received an annual study visit in addition to standard care visits. At each study visit, patients underwent a clinical assessment, and additional laboratory studies including HBV DNA (Roche Diagnostics, GmbH; LLOD <20 IU/mL), hepatitis B e antigen (HBeAg) and anti-HBe (Abbott Architect i2000 kits, Abbott Laboratories). TE was performed at baseline and every 3 years (non-fasting). One trained technician performed TE using the M probe on all patients (FibroScan™, Echosens, France) to obtain LSM. TE was considered reliable if 10 successful measurements are obtained with a success rate >60% and IQR/median LSM ≤30%. Patients who did not meet the criteria for successful measurement were asked to come back for a repeat scan or were excluded from the study.
Definitions, outcomes and data analysis
Liver stiffness measure as a continuous variable
The primary outcome was LSM. We analysed changes in LSM as a continuous variable and as a binary variable (significant LSM decline (≥30% change) relative to Year 0 LSM). To model LSM as a continuous variable, we applied a log transformation (to reduce skew) and then used random intercept (for person) linear models. Since this is an observational study, baseline LSM is treated as an outcome rather than a predictor following recommend statistical practice [24]. We started with a base model that included time, co-infection status and a time x co-infection status interaction term to assess if rate of change in LSM differed between co-infected and mono-infected. We then conducted forward stepwise fitting guided by Type III ANOVA and AIC to build the final model. Assessed variables were age at baseline, weight, BMI, serum alanine aminotransferase, creatinine, haemoglobin, platelet count, CD4+ T cell count (baseline or at time of TE), HIV viral load (baseline or at time of TE, continuous or categorical (≤200 vs >200 copies/mL)) and ART use before time of follow-up TE (as binary – on ART vs not on ART before TE).
We also conducted a sensitivity analysis where ART was expressed as a continuous variable in terms of duration of ART use in months by the time of TE was also conducted. Finally, we performed a subgroup analysis that included only HIV-HBV co-infected individuals to assess if HBV-related variables (TDF and 3TC or FTC containing regimen, HBV viral load, hepatitis B e antigen and antibody) could predict LSM during follow-up.
Decline (30%) in LSM during follow-up
For modelling significant LSM decline relative to Year 0 LSM (binary variable (≥30% change)), we used generalized estimating equations and followed a similar model fitting procedure described previously. LSM declined if the measured LSM for a follow-up year (Year 3 or Year 6) was 30% lower LSM relative to Year 0. We initially planned to perform a multivariable analysis using METAVIR stage change but limited eligible samples (n with F1/F2 = 143 at Year 0) and stage increase events (n = 4 in Year 3 and n = 10 in Year 6) made this analysis infeasible. The sample size was also too small (n = 50 with F3/F4 at Year 0) to examine predictors of stage decrease.
Effect of ART on LSM
At the time of enrolment into this study, not all participants were eligible for ART. Therefore, we were able to use our data to estimate the effect of ART on LSM after accounting for non-random ART initiation. We used marginal structural modelling using weighted generalized estimating equations to test the hypothesis that those who initiated ART before Year 3 TE (actual or estimated from Year 0 TE) would have lower LSM than those who did not initiate ART. Due to nearly everyone being on ART by Year 6, we limited the analysis to change in LSM from Year 0 to Year 3.
Assessing impact of missing data
For all models, except the analysis on the Effect of ART on LSM and the HBV co-infected subgroup analyses, we used multiple imputation to assess robustness of findings assuming missing at random (MAR). We also conducted sensitivity analysis that relaxed the MAR assumption and assess some scenarios where missingness is not random. Loss to follow-up might be more likely among those with worse health and thus they would have worse LSM scores. To assess the impact of this scenario, we increased or decreased imputed LSM by up to 20% of what was imputed and used these adjusted values in the modelling. Related to this, we also explored the impact of differential loss to follow-up by repeating the analyses with inclusion of censoring weights using general estimating equations.
Results
Description of the cohort
Of the 330 with baseline TE, 325 were included in this analysis (100 HIV-HBV co-infected, 225 HIV mono-infected). Five (4/5 HIV mono-infected) were excluded because they had been on ART for ≥2 weeks by the time of baseline TE. By Year 3, 66.8% [66% HIV-HBV; 67.1% HIV] remained active in the study and 59% by Year 6 [58% HIV-HBV; 60% HIV]. Having an LSM during follow-up varied in the cohort with 54% (176/325) [74% (74/100) HIV-HBV; 45% (102/225) HIV] of the sample having LSM in Year 3 and 46% (148/325) [43% (43/100) HIV-HBV; 47% (105/225) HIV] for Year 6. Two people died during follow-up (both were HIV-HBV co-infected). (Supplementary Additional Figure 1)
Demographic and clinical variables according to co-infection status.
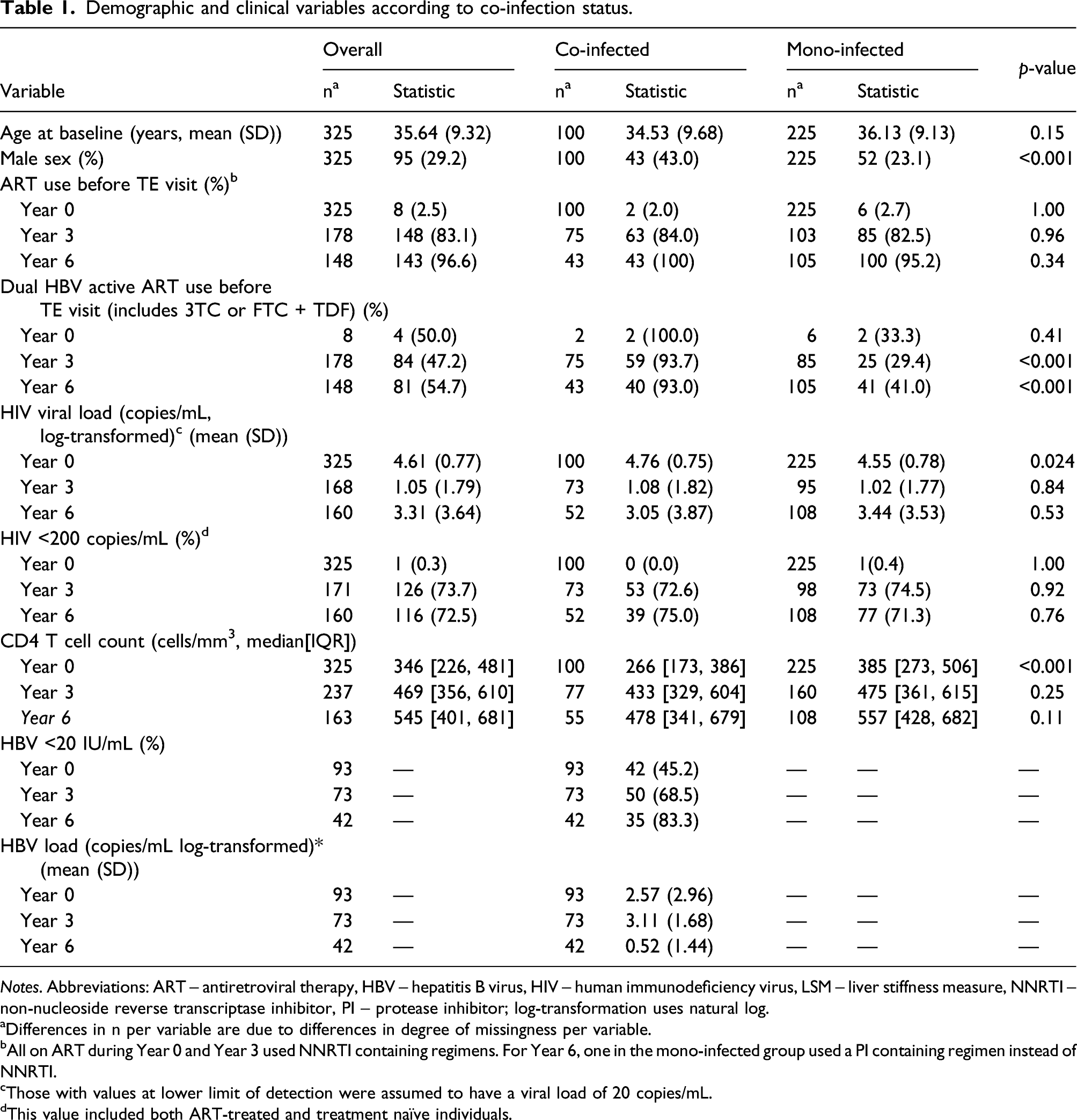
Notes. Abbreviations: ART – antiretroviral therapy, HBV – hepatitis B virus, HIV – human immunodeficiency virus, LSM – liver stiffness measure, NNRTI – non-nucleoside reverse transcriptase inhibitor, PI – protease inhibitor; log-transformation uses natural log.
aDifferences in n per variable are due to differences in degree of missingness per variable.
bAll on ART during Year 0 and Year 3 used NNRTI containing regimens. For Year 6, one in the mono-infected group used a PI containing regimen instead of NNRTI.
cThose with values at lower limit of detection were assumed to have a viral load of 20 copies/mL.
dThis value included both ART-treated and treatment naïve individuals.
Changes in liver stiffness measurement
Liver stiffness measure at each time point.

Notes: Abbreviations: HBV – hepatitis B virus, HIV – human immunodeficiency virus, kPa – kiloPascals, LSM – liver stiffness measure.
ap-values are test of differences between mono-infected and co-infected.
bDifferences in n per variable are due to differences in degree of missingness per variable.
crefers to METAVIR staging.
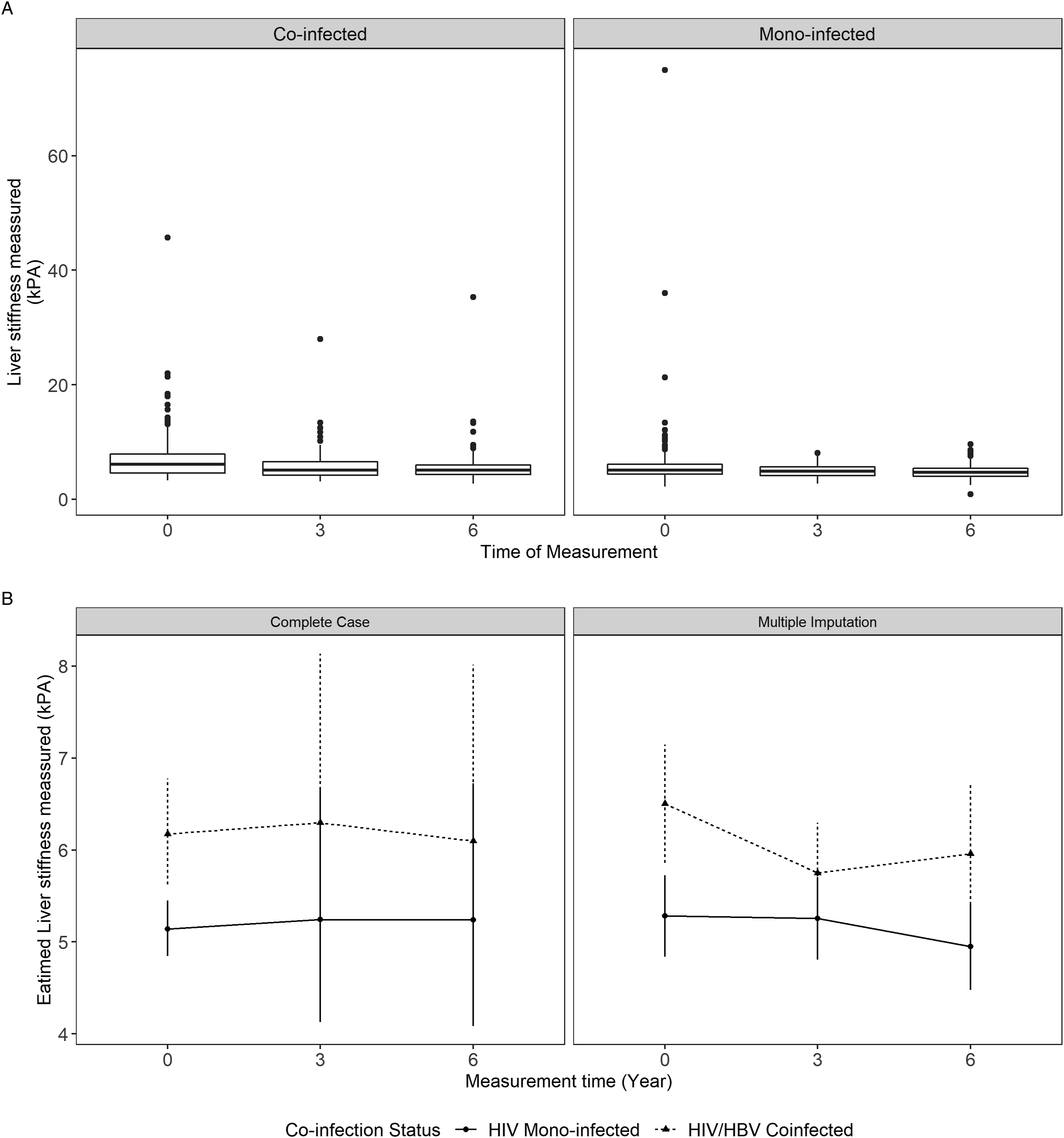
Liver Stiffness Measure over time according to co-infection status: (A) Observed mean (B) marginal means from multivariable model. Note: Marginal means were obtained from the final random-intercept model that adjusted for age, sex, ART use, CD4 count, HIV viral load, time, and co-infection status and includes a time x co-infection status interaction term.
Predictors of Liver Stiffness Measure (Natural Log Transformation) Over Time.

Notes: Abbreviations: HIV – human immunodeficiency virus, HBV – hepatitis B virus, TE – transient elastography.
aART users are those that start ART before the TE of interest so people who started ART between Year 3 and Year 6 are non-users when analysing Year 3 LSM but are users when it comes to Year 6 data.
bCD4 and HIV viral load are time-updated variables such that CD4 and HIV viral load at Year 3 was used for the Year 3 LSM and the corresponding year 6 values for Year 6 LSM.
Other predictors of liver stiffness measurement in all patients during follow-up
In addition to HBV co-infection, other predictors of LSM during follow-up were observed. In both complete case and multiple imputation models, higher CD4+ T cell counts (at time of TE) were associated with lower LSM at each time point. Multiple imputation analysis also showed male gender was associated with higher LSM (Table 3). HIV viral load or virologic suppression was not significant predictors of LSM. ART use status prior to TE was also not associated with LSM. Sensitivity analyses where ART use was specified as duration in months by time of TE (instead of binary) did not change these findings (Supplementary Additional Table 2).
Predictors of LSM during follow-up in HIV-HBV co-infected
In HIV-HBV co-infected participants, we explored predictors of LSM during follow-up focusing on hepatitis B biomarkers. Our initial models (adjusting for time and a predictor) showed that age, sex, creatinine, HBeAg positivity, HBV DNA load, CD4+ T cell count and alcohol consumption at baseline may be predictors of LSM. In multivariable models, adjusting for all these potential predictors, only HBV DNA was significantly associated with LSM at each time point during follow-up. HBV suppression (HBV DNA <20 IU/mL) was associated with a significantly lower LSM across time periods [0.87 (SE: 0.0283, p < 0.001)] compared to individuals with the population average log HBV DNA values (2.02 IU/mL). Use of dual HBV-active ART (vs being not on ART) was not associated with significantly lower LSM across time periods.
Predictors of ≥30% LSM decline
By Year 3, 32 (18.2%) HIV mono-infected and HIV-HBV co-infected experienced ≥30% decline in LSM from baseline which increased slightly to 39 (26.4%) by Year 6. Declines were more common among co-infected than mono-infected for both Year 3 (25.7% vs. 12.7%) and Year 6 (39.5% vs. 21%) (Table 2). In multivariable analysis, after adjusting for confounders, only LSM at baseline and CD4+ T cell count at TE were associated with odds of ≥30% LSM decline, whereas co-infection was not. Use of ART was also not associated with 30% decline on follow-up even if ART use was specified in terms of duration (months) of ART use by time of TE. Participants with higher LSM at baseline had higher odds of experiencing significant LSM decline during follow-up [OR 1.81 (95% CI 1.44–2.26); p < 0.01], as well as those with higher CD4+ T cell counts at time of TE (rescaled to be between 0 to 1) [OR 6.31 (95% CI 1.12–35.58); p = 0.04]. In all patients, the odds of decline by Year 6 were not significantly different compared to Year 3. Similar results were seen in complete case and in multiple imputation analysis (Supplementary Additional Table 3A and B) as well as in the model that used censoring weights (Supplementary Additional Table 5B).
Effect of Initiating ART before Year 3 on LSM
Variance in ART initiation allowed analysis as a natural experiment to test if starting ART before actual or scheduled Year 3 LSM led to lower LSM by Year 3 compared to those who had no record of not initiating ART. Adjusting for non-randomized ART initiation and non-random censoring at Year 3, we found there was a significant difference in LSM at Year 3 between those started ART prior to Year 3 compared to those who did not start ART by that time (Year 3 LSM initiators – Year 3 LSM non-initiators = −0.87 kPa, 95% CI: −1.70 to −0.29). (Supplementary Additional Table 4)
Discussion
In this observational study of liver disease in HIV mono-infected and HIV-HBV co-infected Nigerians, we observed non-significant declines in LSM after 6 years of follow-up. However, LSM scores, which were significantly higher in HIV-HBV co-infected versus HIV mono-infected at baseline, remained significantly higher in this group throughout follow-up, even after adjusting for other potential confounders.
Co-infection with HBV is a well-known risk factor for liver fibrosis in persons living with HIV (PLH) based on several large cross-sectional studies from the United States, Europe and LMICs [13,15,18,25–27]. Since the widespread use of HBV-active ART, several studies have reported significant declines in liver fibrosis scores (APRI, FIB-4, TE) among HIV-HBV co-infected individuals following ART initiation [16,18,28]. However, most of these declines have been reported in early follow-up among participants newly initiated on ART and likely reflect the beneficial effects of treatment on HIV-associated inflammation, rather than fibrosis. In this study, we observed an independent effect of ART on Year 3 LSM which would support this, in addition to other studies showing positive associations between fibrosis scores and HIV viral load before and after ART initiation [16,29,30].
Although median LSM scores in both patient groups were relatively low, the finding of persistently elevated LSM among HIV/HBV co-infected throughout follow-up is concerning and could reflect ongoing liver inflammation in this population, despite early initial declines in LSM with ART. We also observed a higher proportion of HIV-HBV co-infected versus HIV mono-infected with an increase in METAVIR equivalent stage or ≥30% from baseline, although those differences were not significant and overall numbers were small. Further studies are needed to evaluate whether these persistently elevated scores are responsible for the increased risk of overall mortality, liver-related mortality, hospital utilization rates and risk of hepatocellular carcinoma that continue to be observed in HIV-HBV co-infected individuals [1,30–33].
Factors associated with long-term liver-related outcomes have not been explored in great depth in either HIV or HIV-HBV co-infected individuals. In studies of HBV mono-infected, HBV viral load has been shown to be strongly associated with end-stage liver disease, cirrhosis and hepatocellular carcinoma [34] and may be contributory in co-infected populations who often have higher HBV viral loads prior to treatment compared to those with HBV alone [16]. Even on HBV-active ART, a significant proportion of HIV-HBV–co-infected patients fail to achieve HBV DNA suppression and it is not unusual to see detectable levels of HBV in patients with HIV virologic suppression and excellent adherence [35–37]. Pre-therapy HBV-DNA levels have also been shown to be associated with sub-optimal HBV response [38]. In our study, only 75% HIV-HBV co-infected achieved HBV DNA suppression and notably HBV suppression (HBV DNA <20 IU/mL) was the only independent predictor of lower LSM in our HIV-HBV co-infected cohort.
Immunosuppression related to HIV may also play an important role in determining long-term liver outcomes in both HIV and HIV-HBV co-infected populations. We found a strong inverse correlation between TE score and CD4+ cell count taken near the time of TE but not with HIV viral load in all patients. Higher CD4+ T cell counts were also associated with higher odds of ≥30% TE decline. Several other studies have reported associations between low CD4+ T cell counts liver fibrosis and liver fibrosis progression both prior to and after ART initiation [17,26,28,39]. Low CD4+ T cell counts have been shown to predict liver cirrhosis and other liver outcomes in HIV mono-infected populations with and without viral co-infection, [40–42] and a slower recovery of CD4+ T cell counts after ART has been reported in some, although not all, studies of HIV-HBV [43,44]. Low CD4+ T cell counts have also been shown to be associated with higher HBV viral loads and greater HBV disease activity [45,46]. A number of mechanisms explaining how HIV infection accelerates the progression of HBV-related liver disease have been proposed, all of which may be enhanced in patients with more severe immunosuppression [3]. Finally, other risk factors such as male gender, alcohol use and low BMI have also been found to be associated with liver fibrosis progression in viral and non-virally infected populations. Male gender was the only other independent predictor of LSM that was supported by our present analysis. Our prior observation of an inverse association between BMI and LSM at Year 3 [17] was not observed with longer follow-up.
Our study has some limitations including the small number of patients, high attrition, a large number of incomplete serial TE data and limited clinical outcomes data. Although only two deaths were reported, some participants lost to follow-up could have also died as deaths were only passively reported. The attrition could have resulted in a retention of only the healthiest individuals which could have biased our results. The small HIV-HBV co-infected sample limited robust analysis of differences in risk factors and clinical outcomes associated with LSM in this group and a number of other risk factors (drug use, herb use, HDV co-infection, HBsAg sero-conversion [47]) were not examined. The finding of a lack of ART effect may have also been limited by the small number of patients. We were also unable to do multivariable analysis to model LSM stage decline or increase in all participants due to too little sample sizes and variation. Finally, although TE data are still lacking in HIV-HBV co-infected populations [48], TE has been well validated in HBV populations, demonstrating high accuracy for the detection of liver fibrosis and cirrhosis and good diagnostic accuracy for predicting liver-related events [49,50].
Conclusion
In one of the longest observational studies of liver outcomes in HIV and HIV-HBV co-infected, HBV co-infection was associated with higher LSM scores that persisted for up to 6 years. Both HIV (immunosuppression) and HBV (viral load) related factors were strongly associated with elevated LSM emphasizing the importance of both early treatment and use of potent HBV active agents that suppress HBV viral load. The association between LSM and long-term liver complications in HIV-HBV co-infection needs to be examined in larger cohort studies.
Supplemental Material
sj-pdf-1-mpx-10.1177_13596535211058262 – Supplemental Material for HBV co-infection is associated with persistently elevated liver stiffness measurement in HIV-positive adults: A 6-year single-centre cohort study in Nigeria
Supplemental Material, sj-pdf-1-mpx-10.1177_13596535211058262 for HBV co-infection is associated with persistently elevated liver stiffness measurement in HIV-positive adults: A 6-year single-centre cohort study in Nigeria by Adovich S Rivera, Stephen Machenry, Jonathan Okpokwu, Bola Olatunde, Placid Ugoagwu, Muazu Auwal, Halima Sule, Patricia Agaba, Oche O Agbaji, Chloe L Thio, Robert Leo Murphy and Claudia Hawkins in Antiviral Therapy
Footnotes
Acknowledgements
We thank APIN, JUTH for permission to use the patients’ data and the reviewers for their thoughtful and constructive feedback.
Author’s Note
Parts of the data have been previously presented at the 2019 Conference on Retroviruses and Opportunistic Infections, March 4-7 2019, Seattle, WA, USA.
Author Contributions
ASR and CH led the writing of the current manuscript. ASR conceptualized and performed the statistical analysis. SM, JO, BO, PU, MA, HS and PA contributed to data collection and curation. CH, CT, OA and RM designed the original cohort of this study. All authors provided feedback regarding the analysis and critically revised and approved the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was facilitated, in part, by the US Department of Health and Human Services, Health Resources and Services Administration (U51HA02522- 01-01) which funded HIV/AIDS treatment and care services at JUTH, Jos. The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding organizations.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
