Abstract
Serum interleukin-17 is closely related to inflammatory liver injury after hepatitis B virus (HBV) infection. The objective is to study the expression of interleukin-17 (IL-17) in different immune phases in patients with chronic hepatitis B. Whole venous blood was obtained from normal controls (n = 20) and four patient groups (n = 213): immune tolerance phase (IT, n = 57), immune clearance phase (IC, n = 59), non (low)-replicating phase (LR, n = 44), and reactivation phase (RA, n = 53). We measured IL-17, IL-6, and tumor necrosis factor alpha (TNF-α) levels in serum samples and measured IL-17, IL-6, and TNF-α messenger RNA (mRNA) levels in peripheral blood mononuclear cell (PBMC). Compared to normal controls, IL-17, IL-6, and TNF-α protein levels in serum and mRNA levels in PBMC were significantly higher in the four groups. IL-17 protein levels in serum and mRNA levels in PBMC were positively correlated with alanine aminotransferase (ALT), aspartate aminotransferase (AST), IL-6, and TNF-α. Compared to patients in the IT phase and LR phase, patients in the IC phase exhibited the highest serum IL-17 and PBMC mRNA levels. The IL-17 mRNA levels of patients in RA phase were higher than those in patients in IT phase. IL-17 plays an important part in the various immune phases of chronic hepatitis B (CHB).
Introduction
Around 2 billion individuals in the whole world have been infected with the hepatitis B virus (HBV), and at present, the chronic infection individuals are approximately 350 million. 1 Approximately 650,000 persons died of the aforementioned disease caused by HBV infection every year. 2 Although effective vaccination strategies have been implemented, the rate of hepatitis B infection in China’s general population is 10%, which is the highest prevalence in the world. 3
In recent years, cytokines have become a research hotspot in knowing the pathogenesis of chronic hepatitis and liver cirrhosis. Interleukin-17 (IL-17) can participate in host’s anti infection immunity and resist extracellular pathogens, also it can induce the expression of other inflammatory factors such as interleukin-6 and interleukin-8 (IL-6 and IL-8). According to reports, IL-17 and IL-17-producing cells have participated in the pathogenesis of various diseases, notably by inducing and mediating proinflammatory responses.4,5 Studies have suggested that IL-17 is implicated in inflammatory liver damage after HBV infection. There is positive correlation between the level of IL-17 and serum alanine transaminase, the same is also between IL-17 and HBV DNA. 6 Du et al. 7 found that the serum level of IL-17 was apparently higher in the liver cirrhosis group than in the chronic hepatitis B (CHB) group. It was thought that IL-17 acted as a leading role in occurring and growth of liver fibrosis. Studies have demonstrated that there is a good positive correlativity between the intrahepatic Th17 cytokines or the levels of serum IL-17 in hepatitis B patients and the progression of hepatocirrhosis. 8 IL-17 probably critically taken part in the pathogenesis of hepatitis B-related liver cirrhosis, so can be used to predict the degree of progression of liver cirrhosis.9,10
According to the characteristics of immune response, CHB can be generally divided into four different clinical phases, the immune tolerance phase (IT), immune clearance phase (IC), non (low)-replication phase (LC), and reactivation phase (RA). 11 However, there are few studies about IL-17 in the different immune phases of CHB. In this study, we detected the expression of IL-17 and its related cytokines IL-6 and tumor necrosis factor alpha (TNF-α) in CHB patients at different immune phases. Our objective was to inquire into the role and clinical significance of IL-17 in the progression of CHB immune phases.
Methods
Ethics statement
The research plan has abided by the Helsinki declaration and protected the rights and interests of its subjects. We obtained the permission of the Ethics Committee of Qilu Hospital of Shandong University, China. Date of approval by the ethics committee is 22 January 2017. Participation taken part in our study was totally voluntary, and participants have signed informed consent.
Design and participants
This study was carried out between January 2017 and ended in October 2017. Patients were recruited from the Qilu Hospital of Shandong University, Jinan Central Hospital of Shandong University, and Jinan Infectious Disease Hospital; the three hospitals are all public-level-three hospitals. In this study, 275 patients were initially contacted but 62 were excluded. 62 excluded patients include non-B hepatitis virus (n = 15), alcohol abuse (n = 9), hepatocellular carcinoma (n = 5), co-infection with other liver disease (n = 16), hematologic disorders (n = 4), and history of anti-HBV treatment within 1 year (n = 13). Finally, a total of 213 CHB patients were enrolled and reviewed, and they were defined as the positivity of HBsAg for more than 6 months and no history of anti-HBV treatment within 1 year, including 57 cases of the IT, 59 cases of the IC, 44 cases of LR, and 53 cases of the RA. We also recruited 20 normal control subjects. The participants have received no treatment that could affect the clinical condition before this study.
The inclusion criteria for the object of study were (1) voluntary participation, (2) age of 18 years and older, and (3) meets the definitions established by the guideline of prevention and treatment for chronic hepatitis B (2015 Update). 12 The case group was divided into four groups in the light of the natural history of CHB. The IT was marked by serum HBsAg+ and HBeAg+, serum anti-HBe−, high levels of serum HBV DNA (>106 IU/mL), normal serum ALT, with liver histological evidence of mild or no liver necroinflammation, and no progression or only slow progression of hepatic fibrosis. The IC was characterized by serum HBsAg+ and HBeAg+, serum anti-HBe−, serum HBV DNA level > 2000 IU/mL, persistent or intermittent elevation in serum ALT, and moderate or severe inflammation and necrosis observed in hepatic histology. The LR was characterized by serum HBsAg+, serum HBeAg− and anti-HBe+, low or undetectable serum HBV DNA level, ALT within the normal range, and no inflammation or evidence of only mild inflammation in the hepatic histology. Finally, the RA was characterized by serum HBsAg+, negativity for HBeAg, positivity for anti-HBe, moderate and high HBV DNA replication (>2000 IU/mL), and sustained or repeatedly abnormal ALT.
The exclusion criteria for the object of study were (1) severe cognitive impairment; (2) combination of HIV, other types of hepatitis virus infection, metabolic liver disease, hepatocellular carcinoma, liver transplantation, or end-stage non-hepatitis B-related illnesses; (3) excessive alcohol consumption (>30 units/week), illegal drugs, or pregnant; (4) other severe chronic diseases of the organ systems; and (5) refused consent.
Specimen collection
In the early morning, 5-mL samples of fasting venous blood were drawn from all groups, both patients and normal controls; the blood were all gathered in ethylenediaminetetraacetic acid (EDTA) tubes. Next, we centrifuged all samples (2000 r/min for 5 min at room temperature) and got the upper serum, stored them at −80°C until they were used for analysis.
Detection of IL-17, IL-6, and TNF-α level in serum
IL-17, IL-6, and TNF-α levels in serum were detected by the enzyme-linked immunosorbent assay (ELISA) method; the ELISA kits were bought from R&D Systems (Minneapolis, MN, USA). The measurements were carried out in accordance with the manufacturer’s instructions. The specimen was balanced to room temperature. Standard holes were set for both samples and controls, and 100 µL of standard or control samples to be tested were added to each hole. Samples were incubated for 2 h at room temperature, the liquid was discarded, and the plates were washed four times and dried. Then, 100 µL of the anti-biotin antibody was added into each pore. The mixture was incubated for 1 h at room temperature, the liquid was discarded, and the plate was washed four times and then dried. Next, 100 µL horseradish peroxidase was used to label the avidin working fluid. This was incubated for 45 min at room temperature, the liquid was discarded, and the plates were washed four times and dried. Then, 100 µL of chromogenic solution was added to each hole and was incubated at room temperature for 20 min. The termination reaction of the 50-µL terminating solution was then added. Immediately, the automatic enzyme labeling instrument was used to detect the absorbance of each hole at 450 nm and to automatically convert absorbance into concentration values.
Isolation of peripheral blood mononuclear cells
Peripheral blood mononuclear cells (PBMC) were separated by density gradient centrifugation from the peripheral blood anti-coagulated with EDTA via Ficoll-Paque Plus (GE Healthcare, Uppsala, Sweden).
Detection of IL-17, IL-6, and TNF-α mRNA in peripheral blood mononuclear cells
For IL-17, IL-6, and TNF-α gene primers, refer to previous references, with ß-actin as the internal reference; this was synthesized by the Shanghai Boya Biotechnology Corporation (Shanghai, China). The experiment used real-time polymerase chain reaction (RT-PCR) 96-hole plates. Two micrograms of total RNA were reversely transcribed into cDNAs using RevertAid First Strand cDNA Synthesis Kit (Fermentas, Vilnius, Lithuania). The following primers were used:
Forward and reverse primers of human IL-17, 5′-AGAGATATCCCTCTGTGATC, and 5′-TACCCCAAAGTTATCTCAGG-3′;
Forward and reverse primers of human IL-6, 5′-ACCCCTGACCCAACCACAAAT-3′, and 5′-AGCTGCGCAGAATGAGATGAGTT-3′;
Forward and reverse primers of human TNF-α, 5′-AAGCCTGTAGCCCATGTTGT-3′, and 5′-CAGATAGATGGGCTA-3′;
Forward and reverse primers for human β-actin, 5′-CATGTACGTTGCTATCCAGGC-3′, and 5′-CTCCTTAATGTCACGCACGAT-3.′
The polymerase chain reaction (PCR) cycle parameters were 95°C—10 min, 95°C—30 s, 60°C—45 s, 72°C—1 min, 45 cycles, and 5 min at the last 72°C. After mixing the reaction system, centrifugation was set at 3000 r/min for 3 min. LightCycler 480 software was used to analyze the RT-PCR instrument. The specificity of the amplified products was determined by dissolution curves and agarose gel electrophoresis. ß-actin was used as a reference to calculate the relative expression level of mRNA gene.
Determination of serum liver tests
The activities of alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), and albumin (ALB) in the serum were all determined using standard autoanalyzer methods with an Hitachi Automatic Analyzer (Hitachi Inc., Tokyo, Japan), and HBV DNA concentrations were tested using a fluorescence quantitative PCR.
Statistical analysis of results
SPSS software program version 13.0 (SPSS Inc., Chicago, IL, USA) was applied to all the statistical analyses. Kruskal–Wallis H test was used for comparison among multiple groups. The two groups were compared with the Mann–Whitney U test. Correlation analysis was carried out by Spearman rank sum correlation analysis. Bilateral tests with
Results
Demographic characteristics
A total of 213 patients with CHB were comprised in this study. There were 170 males and 43 females, with a mean age ± standard deviation of 36.41 ± 14.58 years. Among the 20 healthy controls, there were 14 males and 6 females, with a mean age ± standard deviation of 34.00 ± 11.59 years. It shows that two groups with age and sex have no statistical difference. The levels of ALT, AST, TBIL, and ALB were significantly different in different clinical phase groups (
Demographic characteristics in chronic hepatitis B and healthy controls.

ALT: alanine aminotransferase; AST: aspartate aminotransferase; TBIL: total bilirubin; ALB: albumin.
Demographic characteristics in immune phases of chronic hepatitis B.
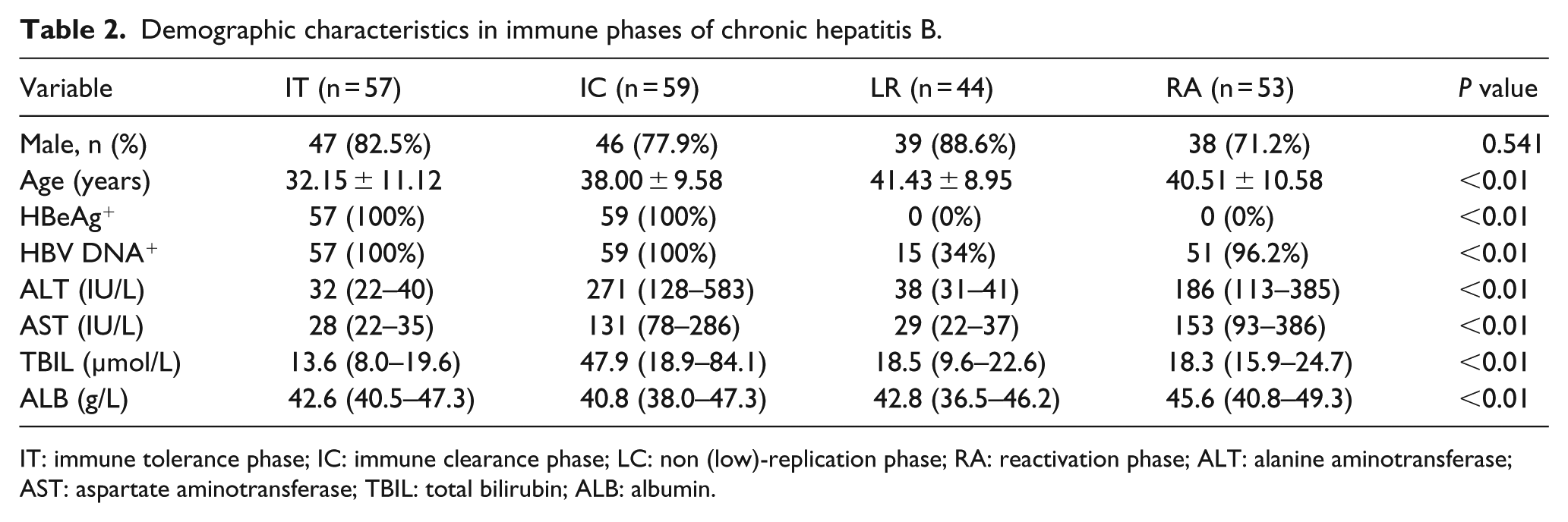
IT: immune tolerance phase; IC: immune clearance phase; LC: non (low)-replication phase; RA: reactivation phase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; TBIL: total bilirubin; ALB: albumin.
Comparison of IL-17 and its related cytokines in CHB group and healthy controls
The levels of IL-17 protein and mRNA in CHB patients were higher than those in the healthy control group (
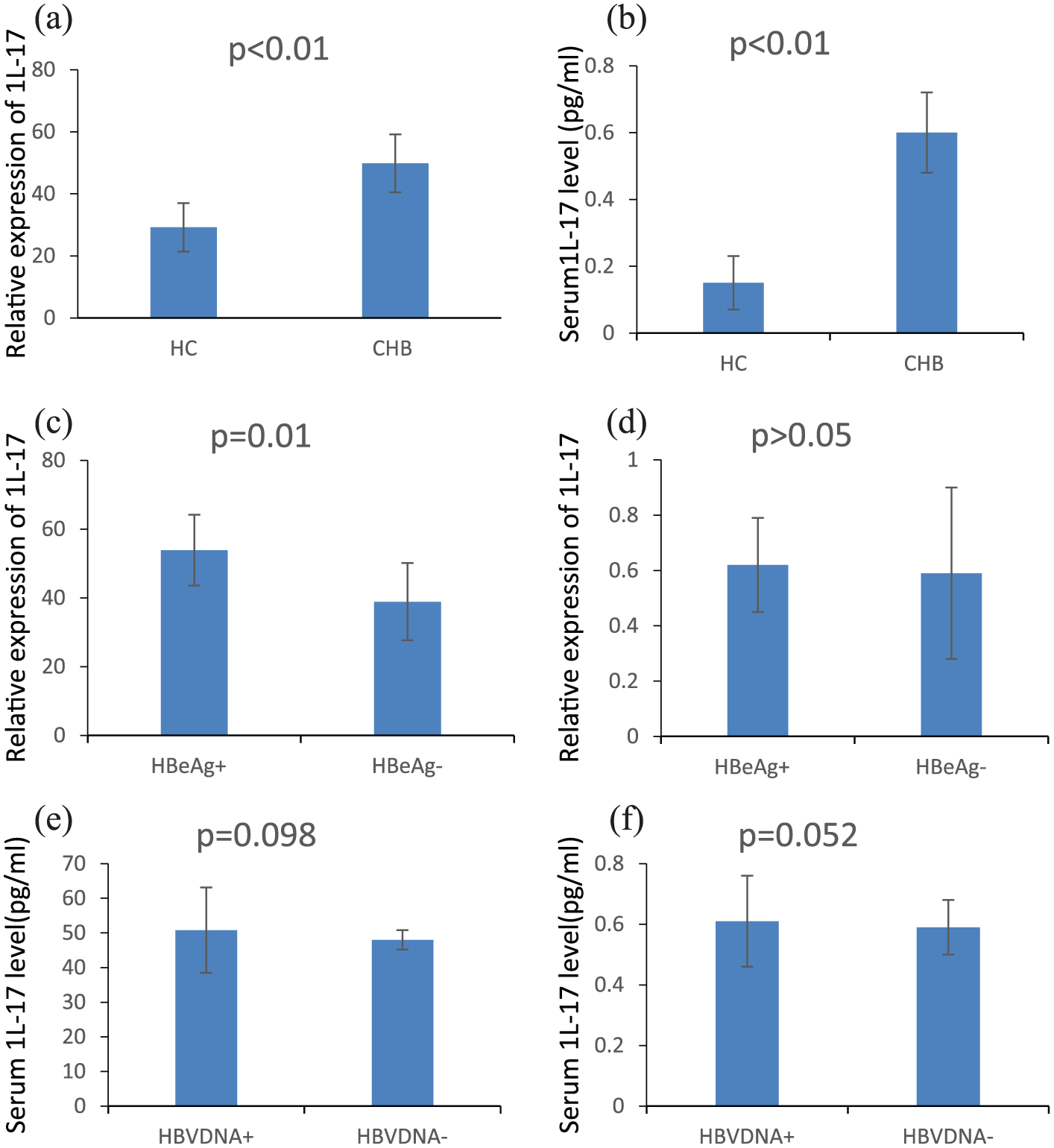
IL-17 protein concentration in serum and IL-17mRNA content in PBMC. Comparison of IL-17 in CHB group and healthy controls.
Correlation analysis between serum IL-17 protein, mRNA level and IL-17-related cytokines in CHB patients
Spearman rank sum correlation analysis showed that, at the gene level, the PBMC IL-17 mRNA in CHB patients was positively correlated with the level of IL-6 protein and mRNA (r = 0.185,
At the protein level, the level of serum IL-17 in CHB patients was positively correlated with the level of IL-6 protein and mRNA (r = 0.525,
The relationship between IL-17 and IL-17-associated cytokines and the liver function index in CHB patients at the gene- and protein-expression level.

CHB: chronic hepatitis B; Interleukin-17; ALT: alanine aminotransferase; AST: aspartate aminotransferase; TBIL: total bilirubin; ALB: albumin; TNF-α: tumor necrosis factor alpha.
Relationship between serum IL-17 protein, mRNA level, and liver function in CHB patients
Serum IL-17 protein and mRNA levels in CHB patients were positively correlated with serum ALT (r = 0.256,
Comparison of protein and mRNA levels of IL-17 and its related cytokines in different immune phases of CHB patients
The Kruskal–Wallis analysis showed that serum IL-17 and mRNA had statistical significance in different immune phase groups (
As shown in Figure 2, we demonstrated that PBMC IL-10 mRNA in the IC phase was significantly higher than in the IT phase (

Expression of IL-17, IL-6, and TNF-α in different immune phases of CHB.
Discussion
It is well known that HBV infection can directly stimulate the secretion of TNF-α and IL-6 in the immunocompetent cells. These cytokines mediate liver cell damage and induce differentiation and proliferation of all kinds of cells, thus causing liver inflammation to persist and lead to liver fibrosis. Our results showed that, compared to normal controls, IL-17, IL-6, and TNF-α protein levels in serum and mRNA levels in peripheral blood mononuclear cells were significantly higher in the four groups. It suggests that IL-17 is involved in the occurrence and development of CHB. We also found that the levels of serum IL-17 and PBMC mRNA in CHB patients were positively correlated with the level of serum ALT and AST. Meanwhile, the expression level of IL-17 was positively correlated with the IL-6 level, and that is the same when comparing with TNF-α. Furthermore, we also studied the relationship between IL-17 and HBV replication. It was found that the serum IL-17 protein level in the HBeAg+ group was significantly higher than that in HBeAg− group. However, there was no significant correlation between the HBV DNA and IL-17 expression level. These results indicated that IL-17 has no definite relationship with virus replication.
We found that the levels of serum IL-17 and PBMC IL-17 mRNA in the immune scavenging period were higher than those in the IT and the LC. The level of PBMC IL-17 mRNA in the RA was significantly higher than that in the IT. This is associated with immune activation in the IC and low immunity during the IT.
To sum up, IL-17 played an important role in the development of CHB. IL-17 mediates viral response after HBV infection. Our study of IL-17 may be help to give new insights in treating CHB. It may provide a more comprehensive theoretical basis for the prevention and treatment of CHB, and may provide the basis for immune intervention therapy. However, before IL-17 is used as a new intervention target to treat CHB and cirrhosis, there are still many other functions of IL-17 in the liver that required further research, and the role of IL-17 in complex immune systems needs further exploration. Also, there are also shortcomings in our research, especially there had only a small number of cases in this study; therefore, further studies with enlarged samples and extended follow-up are warranted.
Footnotes
Acknowledgements
The authors greatly appreciate the time and effort devoted by the study participants in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Science and Technology Planning Project of Shandong Province, China. The authors declare that funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
