Abstract
Background
Drug-resistant Mycobacterium tuberculosis strains have challenged efforts to combat tuberculosis (TB), a major global killer. C-reactive protein (CRP) shows promise as a biomarker for TB screening, particularly in HIV-positive cases, with demonstrated sensitivity and specificity in meta-analyses.
Methods
We performed a meta-analysis to assess the accuracy of CRP for screening HIV-associated PTB in outpatients, combining the sensitivities and specificities of diagnostic tests. PubMed, Web of Science, and SCOPUS were searched for articles that were published until April 2024. Quality assessment was done using the QUADAS-2 scale, and analysis was conducted using the random-effect model in STATA 17.
Results
Eighteen studies, primarily from Africa (2013–2023), were analysed from an initial pool of 1186. These studies included 5625 HIV patients, 1248 of whom had PTB coinfection. Using a CRP threshold of 10 mg/L, 17 studies (5109 patients) showed 84% sensitivity (95% CI: 72%–91%) and 67% specificity (95% CI: 52%–79%) with I2 = 84.91%. At 8 mg/L, nine studies (3631 patients) reported 77% sensitivity (95% CI: 65%–86%) and 81% specificity (95% CI: 69%–89%) with I2 = 86.75%.
Conclusions
Our study showed that CRP may aid in screening for PTB in PLHIV but requires clinical assessment and additional tests. Its high sensitivity can rule out PTB, but low specificity necessitates further investigation.
Introduction
Tuberculosis (TB) remains a leading cause of mortality among individuals living with HIV/AIDS, particularly in resource-limited settings. 1 Despite the World Health Organization (WHO) goal of reducing TB incidence by 90% by 2035, the global TB burden remains significant. According to the latest update, in 2023, there were 18.8 million people worldwide diagnosed with TB (134 cases per 100,000 population) with 56% of cases concentrated in five high-burden countries, including India (26%), Indonesia (10%), the Philippines (6.8%), China (6.8%), and Pakistan (6.3%). In 2023, 10.8 million new TB cases were reported; among them, 6.1% (662,000 patients) were documented as TB/HIV-coinfected. However, regional epidemiological studies in some developing countries have reported a higher prevalence of HIV/TB coinfection between 10.92 and 15.54%. 2 Despite the existence of new cases annually, TB-caused mortalities were slightly decreased by 2023 in comparison to the previous year (1.25 million vs 1.32 million), among them, 161,000 deaths were HIV-positive individuals. The increased TB-caused mortality in 2022, with 1.3 million deaths among HIV-negative individuals and an additional 214,000 among HIV-positive individuals, was likely due to the COVID-19 pandemic restricting access to healthcare and treatment.3–5 This means that TB remains a serious health problem among populations. To combat this ongoing burden, the WHO recommends systematic TB screening for all people living with HIV (PLHIV) irrespective of symptoms. The objectives of TB screening in PLHIV are to (1) detect active TB early to reduce the risk of severe outcomes and transmission and (2) identify those eligible for preventive therapy to significantly reduce the incidence of TB. However, a significant challenge in implementing systematic TB screening is the absence of a screening test that meets the WHO’s target product profile, which requires a sensitivity of ≥90% and a specificity of ≥70% criteria that no current test or algorithm has yet met for PLHIV.6,7
Current screening methods, such as symptom-based screening and chest radiography, have significant limitations in terms of sensitivity and specificity. There is an urgent need for more accurate, accessible, and affordable screening tools to improve the early detection of TB, including both pulmonary and extrapulmonary forms, among PLHIV. 8
C-reactive protein (CRP) is an emerging biomarker with significant potential as a screening tool for TB in PLHIV. 9 Research indicates that CRP offers a specificity that is two to six times greater than that of symptom-based screening methods, while still maintaining high sensitivity. Notably, CRP testing is already accessible as a quick, straightforward, and affordable point-of-care test, suggesting it could be a scalable option for resource-limited environments.10,11
This research aimed to assess the diagnostic accuracy of CRP in detecting pulmonary tuberculosis (PTB) among people living with HIV (PLHIV) who have been diagnosed as positive based on the WHO symptom screen. The outcomes of this study could significantly influence the development of more efficient TB screening methods in resource-limited areas with high rates of HIV/TB coinfection.
Methods
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 2020. 12
Search strategy
Three electronic databases (PubMed, Scopus, and Web of Science) were systematically searched for English-language published articles from their inception to April 15, 2024. The searches were conducted utilizing keyword combinations such as ‘HIV’ AND ‘CRP’ and ‘TB’. No synonyms or related terms were excluded from the search strategy. Additionally, the references of the included studies were screened to identify potentially eligible articles.
Eligibility criteria
We included observational studies on the diagnostic value of CRP levels in HIV-positive patients with TB. Only studies that provided true positive (TP), true negative (TN), false positive (FP), and false negative (FN) data against a reliable reference standard were included.
Furthermore, we excluded studies that met the following criteria: (1) Inadequate data for quantifying the desired data for the diagnostic value of CRP. (2) Replication of studies or sharing of participant data. (3) Studies categorized as reviews, editorials, conference papers, case series/reports, secondary analyses, or animal experiments. (4) Studies employing qualitative research methodologies.
Study selection
Two authors (YG and QC) independently screened the titles and abstracts of the potentially eligible studies using EndNote. For studies considered possibly eligible, different authors independently reviewed the full texts. Disagreements regarding the study design, methods, or inclusion decisions were resolved through a consensus meeting.
Data extraction
General characteristics of all included studies.

Abbreviations: TB, tuberculosis; CRP, C-reactive protein; WHO-4SS: World health Organization 4-symptoms screening; SSM: sputum-smear microscopy; MTB/RIF: Mycobacterium tuberculosis/resistance to rifampicin; CS: cross-sectional; ELISA: enzyme-linked immunosorbent assay; CR: chest radiography; POC: point of care; NR: not reported; CC: case–control.
Risk of bias assessment
Using the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool, the quality of studies was appraised. QUADAS-2 tool helps comparison of four domains: patient selection, index test, reference standard, and flow and timing.
Quantitative analysis
A meta-analysis to estimate pooled sensitivity and specificity using a bivariate random-effects model was conducted, selecting to accommodate variability in test accuracy across studies. The TP, FP, TN, and FN values were either extracted or obtained from the studies. Forest plots were plotted to display the individual’s study results, exhibiting sensitivity and specificity estimates alongside 95% confidence intervals. Studies were categorized by CRP measurements at two distinct thresholds of 8 mg/L and 10 mg/L, utilizing mycobacterial culture, composite reference standards, or bacterial microscopic examination as reference standards. Key parameters of the summary ROC curves and summary sensitivity–specificity points were examined. Heterogeneity was evaluated by visually inspecting the forest and ROC plots. Descriptive statistics comprised pooled sensitivity, pooled specificity, diagnostic odds ratio (DOR), Higgins I2 (to assess consistency, with 0% indicating no observed heterogeneity), and Cochran’s Q statistic. Heterogeneity in sensitivities and specificities was assessed using a chi-squared test, with a significance level of 0.05.
Results
Selection of studies
After removing 348 duplicates by endnote from the initial 1186 articles, only eighteen6,8,10,11,13–26 articles remained after title/abstract screening and full-text screening. Further details are provided in Figure 1. Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flow diagram of search results.
Study characteristics
A total of eighteen studies conducted between 2013 and 2023 were included in the analysis. With the exception of two studies, all were conducted in Africa. Eight studies were conducted in South Africa, four in Uganda, and one each in Kenya, Zambia, India, and Ethiopia. Two studies were multicenter, one conducted in Zambia and South Africa and the other in South Africa, Cambodia, Peru, Georgia, and Vietnam. Twelve studies were cohort studies, six were cross-sectional, and one was a case–control study. The sample size varied from 93 to 1177 HIV-positive patients. All studies, except one, were conducted in adults, with the mean age ranging from 32 to 46.2 years old.
Risk of bias within studies
The results of the risk of bias assessment within the included studies through the QUADAS-2 tool are presented in Figure 2. This tool evaluates the four key domains for risk of bias and concerns regarding applicability, including (1) patient selection (low risk = 61.12%, some concerns = 27.77%, and high risk = 11.11%); (2) index test (all were low risk); (3) reference standard (low risk = 83.34%, some concerns = 11.11%, and high risk = 5.55%); (4) flow and timing (low risk = 83.34%, some concerns = 11.11%, and high risk = 5.55%). Overall risk of bias assessment results among eighteen included studies were ten studies with low risk (55.57%), five studies with some concern scores (27.77%), and three studies (16.66%) with a high risk of bias (Figure 2). Risk of bias and applicability concerns summary: review authors’ judgments about each study.
Synthesis of results
Eighteen studies measured CRP levels in PTB and HIV coinfection. The study included 5625 HIV patients, and 1248 of them had PTB coinfection. Among the included studies in our meta-analysis, there were seventeen studies that estimated the sensitivity and specificity of CRP at a threshold of 10 mg/L. In addition, its sensitivity and specificity at a cut-off threshold of 8 mg/L were also evaluated in nine studies.
The sensitivity estimation for the CRP at a cut-off threshold of 10 mg/L was variable, ranging between 20%
16
and 97%,
25
while its specificity estimation ranged from 8%
21
to 96%.
14
In an illustrative diagnostic meta-analysis using a forest plot, there were 5109 involved patients across seventeen studies (CRP threshold of 10 mg/L), the pooled sensitivity was calculated at 84% (95% CI: 72%–91%), and the pooled specificity was obtained at 67% (95% CI: 52%–79%). Heterogeneity testing resulted in an I2 value of 84.91% (Figure 3). Forest plot and SROC curve (HIV patients, using the CRP threshold of 10 mg/L).
In the diagnostic meta-analysis comprising nine studies with 3631 patients, for the CRP at a cut-off threshold of 8 mg/L, the sensitivity estimation varied between 24%
16
and 94%,
23
while the specificity estimation ranged from 49%
23
to 96%.
6
The pooled sensitivity of 3631 patients, who participated across nine studies (considering CRP at a threshold of 8 mg/L), was estimated at 77% (95% CI: 65–86%), and the pooled specificity was 81% (95% CI: 69–89%). The heterogeneity test yielded an I2 value of 86.75% (Figure 4). Forest plot and SROC curve (HIV patients, using the CRP threshold of 8 mg/L).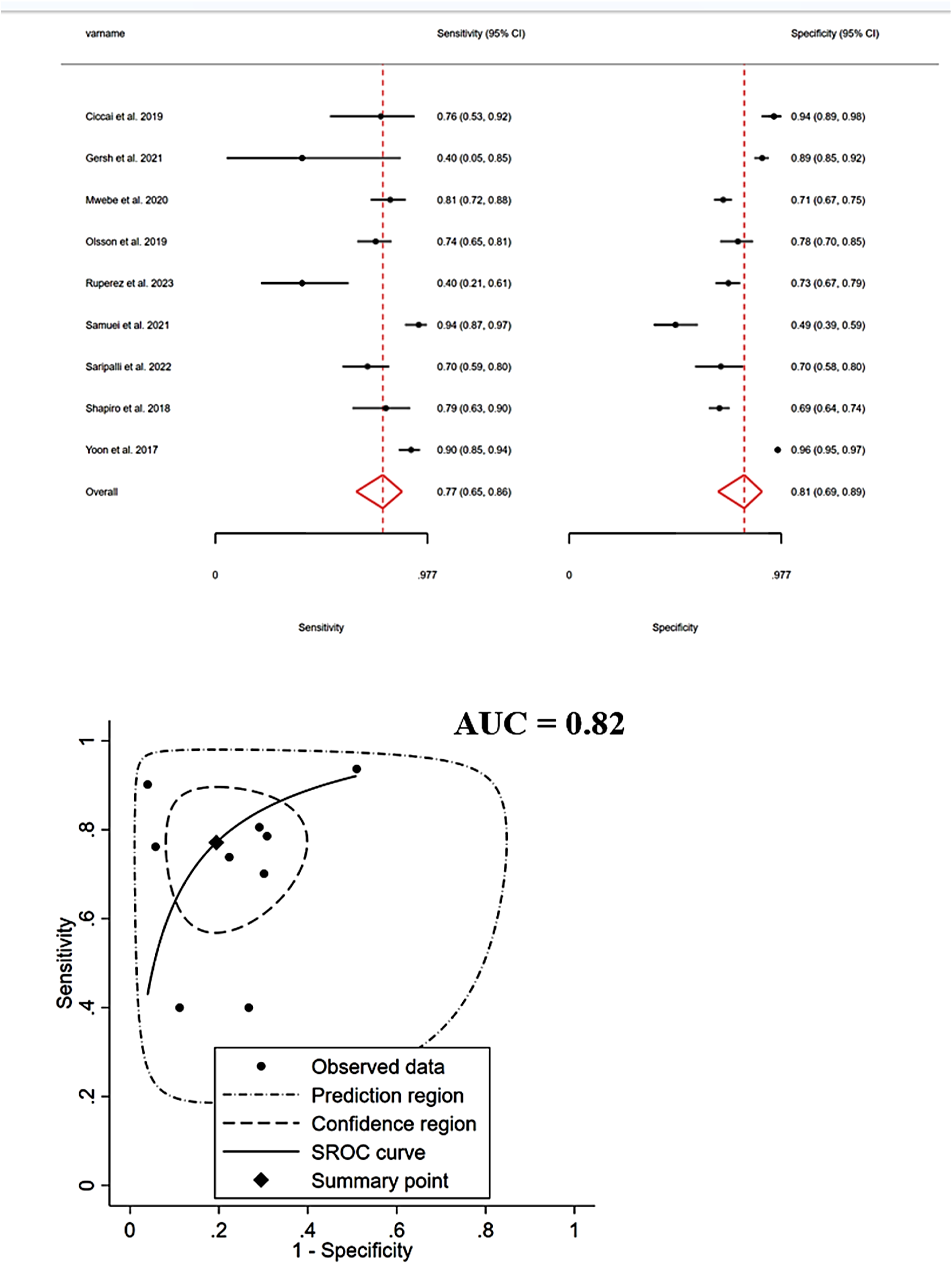
Discussion
This systematic review and meta-analysis aimed to evaluate the diagnostic accuracy of CRP for the detection of PTB in PLHIV. The review included 18 studies that measured CRP levels in 5625 HIV patients, of whom 1248 had PTB coinfection. For a CRP threshold of 10 mg/L, the pooled sensitivity was 84%, and specificity was 67%; while at a lower CRP threshold of 8 mg/L, the pooled sensitivity was 77%, and specificity was 81%. This suggests that CRP is a useful biomarker for PTB, but its lower specificity at higher doses means positive results must be interpreted cautiously.
The wide variability in the sensitivity and specificity of CRP across the included studies suggests that its diagnostic accuracy is heavily influenced by various factors. These factors may include the underlying patient population, stage of the disease, presence of comorbidities or opportunistic infections, and laboratory methods used to measure the CRP levels. For instance, the sensitivity of CRP may be lower in patients with HIV or other immunocompromised conditions, where the inflammatory response and CRP levels may be attenuated. Similarly, concurrent infections, such as viral or fungal infections, can also affect the CRP levels and influence its diagnostic performance. The heterogeneity observed in the meta-analyses highlights the importance of considering the clinical context and patient-specific factors when interpreting the CRP results. A single CRP threshold may not be universally applicable, and the interpretation of CRP levels should be tailored to the individual patient’s clinical presentation and risk factors.
Furthermore, the variability in the laboratory methods used to measure CRP, such as the type of assay or analytical sensitivity of the test, can also contribute to the differences in the reported diagnostic performance. Standardization of CRP measurement techniques may help reduce the heterogeneity observed across studies.
TB continues to be a significant public health issue, particularly for those infected with HIV. The strong link between HIV and TB is well-known, with adults infected with HIV more frequently affected by Mycobacterium tuberculosis (M.tb). This is mainly due to the higher false-negative rates, reduced sensitivity, and limited accessibility of screening tests in TB-prevalent areas.14,15,27,28 The underlying mechanisms are complex. HIV infection impairs immune function by reducing CD4 + T cells, increasing the risk of both primary M.tb infection and latent TB reactivation. HIV-infected individuals are 26 times more likely to develop TB than those without HIV.29,30 The weakened immune response in HIV patients creates an ideal setting for the development of TB. M.tb can adapt and survive in the host despite immune responses and cytokine activation. 31 The immune deficiency caused by HIV allows TB to flourish. Notably, studies have discovered an inverse relationship between inflammatory biomarkers, such as CRP and neopterin, and the count of CD4 positive T-lymphocytes. This indicates that higher inflammatory responses and immune activation are linked to CD4 lymphocytopenia in M.tb-infected adults. 32
Therefore, accurate and timely diagnosis is crucial for effective treatment and disease management. The WHO recommends systematic screening for active TB in people infected with or living with HIV, using either the WHO-4SS assessment or chest radiography as an initial screening test, followed by more specific diagnostic methods such as mycobacterial culture, sputum smear microscopy (SSM), and the Xpert MTB/RIF test.5,33 The current WHO recommendations for TB screening in PLHIV have faced significant challenges in their implementation. Despite the recommendations, only a small fraction of the target population is actually screened for TB, and even fewer patients receive preventive therapy. 3 A major drawback of the current method is the high false-positive rate linked to the WHO symptom screen, the advised screening test. Previous studies6,34–38 have revealed that 86% of PLHIV without active TB falsely screened positive. Despite the symptom screen achieving the minimum sensitivity threshold of 90%, its low specificity of 14% makes it unsuitable for this group.
Moreover, although mycobacterial culture is the definitive method for TB diagnosis, it is frequently not employed as the initial test due to its long result turnaround time (2 to 6 weeks). In resource-limited settings with high HIV prevalence, diagnostic protocols typically involve SSM and Xpert MTB/RIF, which deliver results in less than 24 h.5,33 However, these tests may be less effective in HIV-positive individuals, causing delayed diagnosis, increased transmission, and higher mortality.14,39
According to the WHO’s target product profile, TB screening tests should prioritize sensitivity over specificity to be practical and widely adopted. 40 This is because the risks associated with false-negative results, such as the generation of drug-resistant TB, outweigh the burden of unnecessary confirmatory testing for patients with false-positive results. Recent studies have suggested that CRP could be a useful biomarker for the screening and identification of active PTB, especially in HIV-positive populations.11,33,41 CRP has been shown to have high sensitivity (up to 95%) and moderate specificity (up to 63%) for detecting active PTB when compared to culture-based methods.14,42
The use of CRP as a screening tool could be particularly beneficial in resource-limited settings, as it is a relatively inexpensive and easily accessible point-of-care test that can provide results quickly.11,14,33,42 This could help to reduce the reliance on more expensive and time-consuming diagnostic methods, such as the Xpert MTB/RIF assay, which is currently recommended by WHO as the initial diagnostic test for HIV-associated TB.4,43 Point-of-care CRP is highly effective in patients with low CD4 + T-cell counts, vital for accurate TB screening in this high-risk group. 44 Additionally, researchers have emphasized the importance of establishing appropriate cut-off values for CRP to optimize its performance as a screening tool. Increasing the CRP cut-off value can improve specificity but at the cost of reduced sensitivity (47, 49, 54). The closest cut-off point to WHO recommendations was CRP >3.3 mg/L, with a sensitivity of 80% and a specificity of 72%. 16
This study has faced several limitations. First, there was high heterogeneity among the included studies, possibly owing to variations in diagnostic tests and population characteristics. Additionally, the lack of ethnic diversity, with the majority of studies conducted in Africa, limits the generalizability of the findings beyond this region. However, the study also has several strengths. By systematically reviewing and meta-analyzing available data, this study consolidates evidence on CRP’s diagnostic significance in HIV/TB coinfection. This contributes to a comprehensive understanding of CRP’s utility in clinical practice. Future prospects for research in this field include conducting studies in regions beyond Africa to enhance diversity and broaden the applicability of the findings. Moreover, increasing the sample size in future studies can improve their statistical power and precision of estimation, leading to more robust conclusions.
Conclusion
Overall, the findings of this review suggest that CRP may play an important role as a screening tool for PTB in PLHIV, but its use should be accompanied by clinical assessment and additional diagnostic tests to confirm the diagnosis. The high sensitivity observed at certain CRP thresholds indicates that CRP could be useful in ruling out PTB, but its lower specificity means that a positive CRP result should be interpreted with caution and may require further investigation. Future research should focus on identifying the factors that influence the diagnostic performance of CRP in this setting to optimize its use in the clinical management of PLHIV with suspected PTB.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Science and Technology Project of Jilin Provincial Health Commission (No: 2016ZC020).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All information required is given in the text; other supplementary information can be obtained via email from the corresponding author.
