Abstract
Background
This in vitro study aimed to investigate the effect of several phenolic compounds, including doxorubicin, quercetin, and resveratrol, on HSV-1 infection.
Methods
The cytotoxicity of the drugs was assessed on Vero cells using the MTT assay. HSV-1 was treated with the drugs, and the supernatants were collected at various time points. TCID50% and qPCR tests were conducted on the supernatants to determine viral titration post-inoculation.
Results
The TCID50% assay showed significant changes in viral titration for acyclovir, doxorubicin, and quercetin at most concentrations (p-value < .05), while no significant changes were observed for resveratrol. The qPCR results demonstrated that drug-treated HSV-1 exhibited a significant reduction in DNA titers at various time points compared to non-treated HSV-1 infected Vero cells, except doxorubicin (0.2 µM) and acyclovir (5 µm). However, over time, DNA virus levels gradually increased in the drug-treated groups. Notably, at certain concentrations of doxorubicin and quercetin-treated groups, virus titer significantly declined, similar to acyclovir.
Conclusions
Our findings suggest that quercetin at concentrations of 62 and 125 µM significantly reduced HSV-1 infectivity, as well as these two concentrations of quercetin showed a significant difference in virus reduction compared with acyclovir (10 µM) at certain time points. The anti-inflammatory properties of quercetin, in contrast to acyclovir, make it a potential candidate for anti HSV-1 treatment in life-threatening conditions such as Herpes encephalitis. Additionally, doxorubicin, an anticancer drug, showed meaningful inhibition of HSV-1 at non-toxic concentrations of 2 and 8 µM, suggesting its potential interference with HSV-1 in viral-oncolytic therapy in cancer treatment.
Introduction
Globally, Herpes Simplex Virus type 1 (HSV-1) is one of the most common encountered human pathogens.
1
It is estimated that approximately 3.7 billion (67% of the world's population) are living with HSV-1. Annually, it is responsible for infecting around 23 million new hosts.2,3 While HSV-1 infection is often asymptomatic and self-limiting in healthy individuals, it can cause a range of symptomatic and potentially life-threatening manifestations in immunocompromised patients. In these patients, a range of diseases including mild disorders such as cold sores, herpetic whitlow, and gingivostomatitis, to severe complications like encephalitis, meningitis, keratitis
Transmission of HSV-1 occurs through close contact with individuals shedding the virus from peripheral sites, mucosal surfaces, or in genital or oral secretions. Infection requires the inoculation of the virus on to sensitive surfaces such as the pharynx, cervix, conjunctiva, or a minor skin abrasion. The most significant mode of transmission is through exposure to the virus in carrier saliva.6,7 During primary infection, the virus establishes latency in sensory neurons, often within the ganglia of trigeminal nerves, and can reactivate to cause recurrent infections, particularly in the presence of immune impairment. The neurotropic and neuroinvasive properties of HSV-1 contribute to the development of neurological diseases and the establishment of long-life and incurable infections.8,9
The current management approach for HSV infection focuses on preventing viral transmission, recurrence, clinical manifestations, shedding, and complications. 10 The utilization of nucleoside analogues has greatly influenced the advancement of healthcare and the creation of anticancer and antiviral medications. 11
The present management of HSV infection depends on the administration of various potent and targeted medications, including acyclovir, and another nucleoside analogue like penciclovir, valacyclovir, trifluridine, and vidarabine. 9 Cidofovir has also been shown efficacy in the treatment of acyclovir-resistant HSV infections. 5 Acyclovir, a leading antiviral medication, is the current standard for treating viral infections due to its exceptional selectivity, minimal toxicity, and ability to inhibit the proliferation and transmission of the herpes virus. The administration route of acyclovir, whether oral, topical, or intravenous, depends on the severity and location of the outbreak and aims to target the virus at its source.10,12
The initial phase involves the conversion of acyclovir to acyclovir-monophosphate through the action of viral thymidine kinase within the infected cell. Subsequently, acyclovir-monophosphate is transformed into its active form, acyclovir triphosphate, by cellular kinase. This active compound effectively disrupts viral DNA polymerase activity during the synthesis of new viral DNA strands, leading to the cessation of genome transcription and inhibition of viral replication. 13
Inevitably, long-term treatment with ACV and its derivatives particularly in immunosuppressed individuals such as those with organ transplantation recipients, malignancy, and those infected with HIV can potentially result in the development of drug resistance. The incidence of ACV resistance in immunocompetent patients has been reported to range from 0.1% to 0.7%, while in immunocompromised patients, the range is higher, varying from 3.5% to 10%. In specific cases, such as recipients of hematopoietic stem cell transplantation, resistance rates as high as 36% have been documented. 14
Acyclovir resistance primarily occurs due to genetic mutations or alterations in viral thymidine kinase (TK) or viral DNA polymerase. These mutations can result in a decreased ability of TK to phosphorylate acyclovir or a reduced affinity of viral DNA polymerase for acyclovir triphosphate. Among the three recognized mechanisms of ACV resistance associated with thymidine kinase, TK deficiency is the most prevalent. TK deficiency is observed in around 95% of clinically resistant HSV isolates to ACV. The remaining two mechanisms involve a decline in viral TK activity and TK mutants, which phosphorylate thymidine but not acyclovir. Additionally, there have been rare instances of impaired DNA polymerase activity associated with ACV resistance, although these cases are infrequently reported.14,15
Given that HSV can lead to severe illness in individuals with compromised immune systems, it is necessary to consider alternative treatment options for patients who do not respond to ACV.
They have a wide variety of biological activities, and their derivatives can be good sources of antiviral compounds against human herpesviruses. Further research and discoveries in this area can help develop new derivatives and therapeutic leads, 16 for instance, terpene extracts from the Persian lilac tree leaves have shown good antiviral activity against HSV-1 in cell culture experiments. Additionally, this agent can inactivate the HSV-1 DNA polymerase during DNA replication. 17 Furthermore, the peptide A-3302-B extracted from the marine bacterium Micromonospora sp. has been found to prevent viral egress from host cells and inhibit HSV-2 infection. 18 Additionally, the therapeutic development through repurposing drugs targeting HSV-1 has shown promising results. These drugs, although originally designed for different primary functions, have demonstrated favorable antiviral effects in both clinical and laboratory evaluations when used specifically for HSV-1 treatment. 19 Consequently, there is a demand for new antiviral medications that possess diverse antiviral effects, such as targeting novel antiviral mechanisms, utilizing novel antiviral targets and introducing novel antiviral molecules. 20
Materials and methods
Cell and virus culture
The Vero (African green monkey kidney) cell line was obtained from the Pasteur Institute of Iran. The cells were cultured and sustained with Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, Grand Island, NY, USA) and 10% fetal bovine serum (FBS; Gibco™, Thermo Fisher Scientific, BR, USA), IU/ml penicillin, and 1 μg/ml streptomycin (Bioidea, Iran). The cells were incubated at temperature of 37°C in a 5% CO2 and 90% humidified incubator. HSV-1 KOS strain was provided by the Department of Virology, Tarbiat Modares University, Tehran, Iran. The virus seed was prepared by culturing the virus with a multiplicity of infection (MOI) of about 0.01 and was quantified using plaque assay twice. The virus seeds were stored in a −70°C freezer. Resveratrol and quercetin drugs were purchased from MOLEKULA (Molekula group, Darlington, England), while acyclovir was obtained from EXIR (Exir Pharmaceutical Company, Borujerd, Iran) in the form of sodium salts. Doxorubicin was obtained from ACTOVER (Actoverco Pharmaceutical Company, Karaj, Iran) as hydrochloride.
Cell cytotoxicity
The MTT assay colorimetric dye reduction method was used to evaluate the cytotoxic effects and determine the optimal concentrations for cell viability of all drugs.
21
Vero cells were seeded in 96-well plates until reaching a confluency of 70%–80%. Different concentration of the drugs was prepared with 2-fold serial dilution from 500 µM resveratrol, 500 µM quercetin, 10 µM acyclovir, and 290 µM doxorubicin. The diluted drugs were added to 96-well plates and incubated at 37°C for 72 hours in a 5% CO2 and 90% humidified incubator. MTT powder (5 mg/ml) was solved in DMEM and thoroughly vortexed. Then, 100 μl of the solution was added to each well, and the plate was incubated for around 3–4 hours at 37°C. After incubation, the MTT and culture media were discarded, and 50 μl of dimethyl sulfoxide (DMSO) was added and incubate for 20 minutes. All procedures were performed in a dark room. The optical density (OD) was measured at 540 nm (with differential filter 620 nm) by a microtiter plate reader. The cell cytotoxicity was determined as the 50% inhibitory concentration (IC50%)
HSV-1-drug treatment assay
Vero cell line was cultured in 24-well plates for 48 hours to reach about 100% confluence. The cells were infected with HSV-1 KOS at MOI 0.01 and were incubated for 1 h at 37°C. Two or three different dilutions of drugs were added to each well. The concentrations used include acyclovir 5, 10 µM, resveratrol 10, 20, and 30 µM, quercetin 7.8, 62, and 125 µM, and doxorubicin 0.2, 2, 8 µM. The supernatant of infected Vero cells was collected at different time points including 24, 48, 72, and 96 hours post-inoculation and stored at −20°C. HSV-1/Drug treatment was measured by TCID50% and qPCR.
Tissue culture cell cytotoxicity 50% assay (TCID50%)
The virus titration was performed using the Median Tissue Culture Infectious Dose (TCID50%) assay, which estimates the viral titer by determining the dilution at which 50% of Vero cells become infected. To conduct the assay, serial dilutions were prepared in ten-fold steps from the supernatant of HSV-1 infected Vero cells in various drug concentrations at specific time points of 24, 48, 72, and 96 hours using serum-free DMEM. Initially, Vero cells were cultured in 96-well plates until they reached approximately 100% confluence. Subsequently, the medium was discarded, and 100 µl of each dilution was added to the wells in eight replicates. The cells were then incubated for 72 hours at 37°C with 5% CO2. A control with non-infected cells was included in each plate for comparison. Following the incubation period, the plates were examined under an inverted microscope to assess the cytopathic effects (CPE) of HSV-1 up to 96 hours post-infection, and the wells were scored for the presence or absence of CPE. Finally, the TCID50% titer for each treatment was calculated using the Reed and Muench method. 22
Quantitative polymerase chain reaction (qPCR)
The sequences, amplicon size, and gene location of primers and probe used in the present study.
Initially, the viral stock was tittered using a plaque assay on two separate occasions, and the average titer was calculated. Subsequently, a ten-fold serial dilution of the viral stock was prepared, and each dilution was extracted by DNA extraction kit (BehGene Biotechnology Company Manufacturer, Shiraz Especial Economic Zone, Fars, Iran) according to the manufacturer's protocol. For qPCR analysis, a reaction mixture containing 10 μl of PCR master mix, 0.3 μl probe, 0.7 μl of each primer, and 2 μl of DNA template from different dilutions has been done by Light Cycler Roche 96 machine. A standards curve was generated by plotting the Ct values against the ten-fold dilutions. The qPCR efficiency, correlation coefficient, and slope were determined. The concentration of the virus in the HSV-1/Drug treatment was interpolated by the standard curve.
Statistical analysis
For the statistical analysis, we employed the Reed–Muench method to calculate the virus titers as TCID50% per ml. To compare the TCID50% titer between the HSV-1 treated and drug-treated groups at different time points, non-linear regression was utilized. The standard curve of different virus concentrations was calculated by linear regression and the unknown concentration of different drug-treated and untreated groups in HSV-1 infection was interpolated by linear regression. One-way ANOVA was employed for multiple group comparisons. A significance level of p < .05 (two-sided) was measured statistically significant. Non-linear regression was used to calculate the IC50%, and the OD values were normalized to survival percentages. All graphs and statistical analysis have been done by GraphPad Prism version 8.
Results
Cell cytotoxicity
The cytotoxic effect of each drug on Vero cells was investigated. Vero cells were treated with various concentrations of the drugs to assess their cytotoxicity. Cell viability was evaluated using the MTT assay to determine the IC50%, which represents the average 50% inhibition of drug viability by non-linear regression. The IC50 values for resveratrol, quercetin, and doxorubicin on Vero cells were 60 μΜ, 150 μM, and 10 μΜ, respectively. Additionally, acyclovir was tested at different concentrations, but no toxicity was observed. Therefore, we conclude that acyclovir is not dose-dependent and does not exhibit toxicity on the Vero cell line (Figure 1). Dose-response curves with MTT assay for resveratrol, quercetin, doxorubicin, and acyclovir on Vero cells. Data in some cases were normalized to create of cell survival percent.
Cytopathic assay of HSV-1-drug treatment
Vero cells were infected with HSV-1 KOS at MOI 0.01 after that treated with each drug at various concentrations, lower than IC50% were selected. The infected cells were examined for cytopathic effect (CPE) with a microscope at time points of 24, 48, 72, and 96-hour post-infection. This experiment was repeated twice to ensure the reliability of the results. Acyclovir was used at two concentrations: 5 μΜ and 10 μΜ (Figure 2C & 2(D)). The CPE was significantly reduced in the acyclovir-treated group compared to the infected control (Vero cells infected with KOS at MOI 0.01) (Figure 2(B)). A mock treatment was used as a negative control (Figure 2(A)). CPE of infected cells. A) Vero cell non-treated, B) Vero cell infected KOS MOI 0.01, C, D) Vero cell infected HSV-1, treated by acyclovir 5 and 10 μM, respectively.
Quercetin was used in three concentrations: 7.8 μΜ, 62.5 μΜ, and 125 μΜ. CPE was clearly observed at the concentration of 7.8 μΜ (Figure 3(C)), similar to the infected control. However, at the concentrations of 62.5 μΜ and 125 μΜ, CPE was dramatically reduced compared to the infected control (Figures 3D & 3(E)). CPE of infected cells. A) Vero cell non-treated, B) Vero cell infected KOS MOI 0.01, C, D, and E) Vero cell infected HSV-1, treated by quercetin 7.8, 62, and 125 μM, respectively.
Resveratrol was used in three concentrations: 10 μM, 20 μM, and 30 μM. There were no significant differences in CPE between the resveratrol-treated group and the positive control (Figures 4(C), 4(D) & 4(E)). Resveratrol failed to inhibit HSV-1 in infected cells in vitro. CPE of infected cells. A) Vero cell non-treated, B) Vero cell infected KOS MOI 0.01 C, D, and E) Vero cell infected HSV-1, treated by resveratrol 10, 20, and 30 μM, respectively.
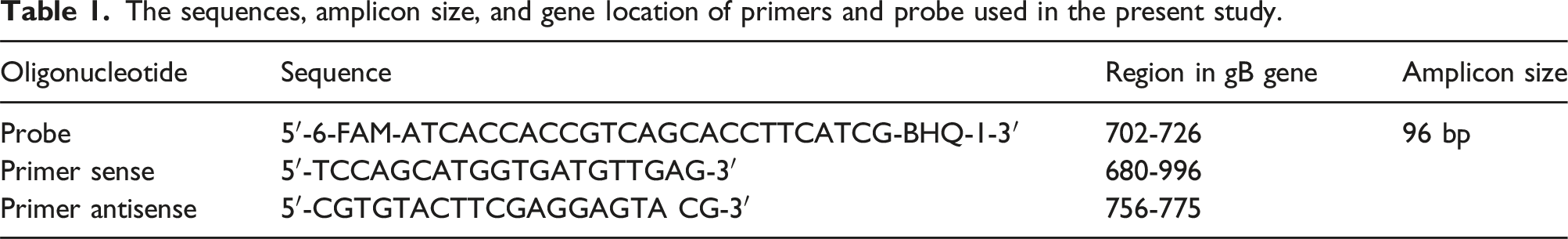
Doxorubicin was used in three concentrations: 0.2 μM, 2 μM, and 8 μM, these concentrations are lower than the toxic dose. It seems that CPE is dose-dependent, with increasing drug dose, and a decrease in CPE was observed (Figure 5). At 0.2 μΜ, there was no difference between the treated and untreated groups, but at 2 μΜ and 8 μΜ, CPE was significantly reduced compared to the positive control. CPE of infected cells. A) Vero cell non-treated, B) Vero cell infected KOS MOI 0.01 C, D, and E) Vero cell infected HSV-1, treated by doxorubicin 0.2, 2, and 8 μM, respectively.
HSV-1 drug-trearment assay by TCID50%
The Vero cells were infected with HSV-1 KOS at MOI 0.01 and incubated with the mentioned drugs at the time points of 24, 48, 72, and 96 hours. The observations were reported as follows:
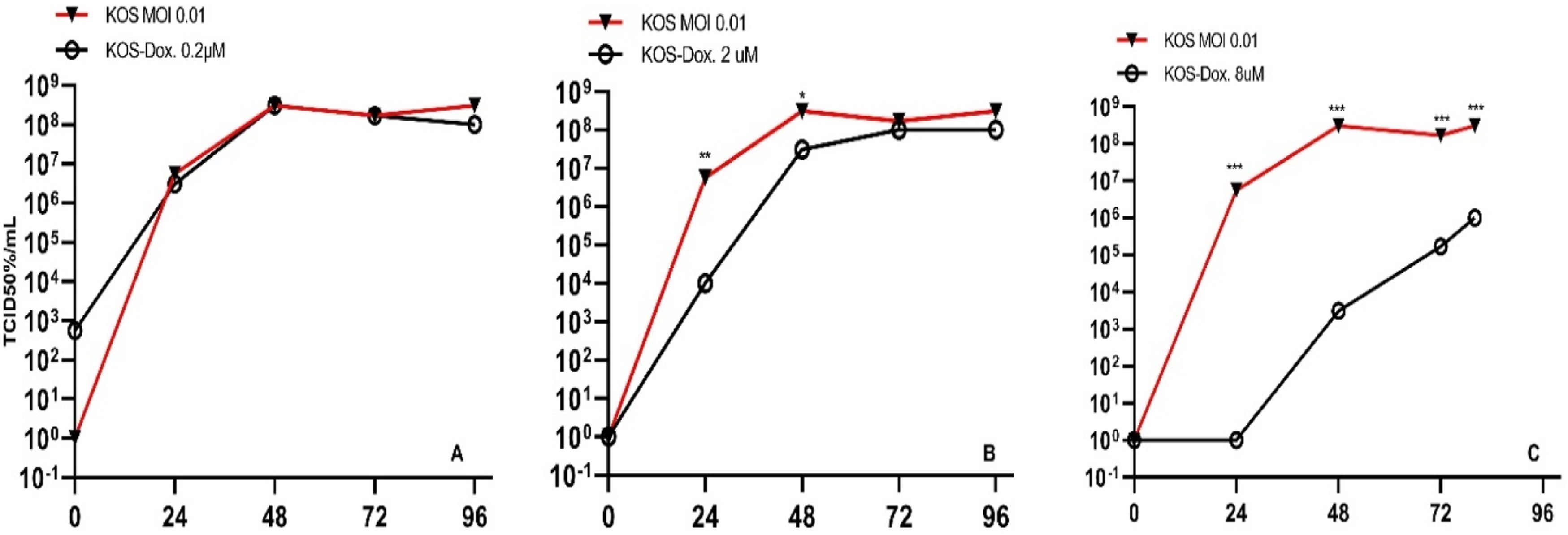
Acyclovir 5 μΜ showed significant changes (p-value < .05) at 24, 48, and 96 hours, but no significant changes were observed at 72 hours. Acyclovir 10 μΜ showed significant changes (p-value < .05) at all time points compared to Vero cell infected KOS at MOI 0.01(Figure 6). Quercetin 125 μM and 62 μΜ showed significant changes (p-value < 0.05) in virus titration at all time points (24, 48, 72, and 96 hours) compared to Vero cells infected with KOS at MOI 0.01. However, Quercetin 7.8 μΜ showed no significant changes at none of the four time points (Figure 7). Resveratrol at concentrations of 10 μM, 20 μM, and 30 μΜ did not have significant cytopathic effects at each of the time points (24, 48, 72, and 96 hours) (Figure 8). Doxorubicin at a concentration of 0.2 μΜ did not show significant changes in the reduction of virus titration. However, significant changes (p-value < .05) were observed at a concentration of 2 μΜ at 24, 48 hours, as well as at a concentration of 8 μΜ at all time points (24, 48, 72, and 96 hours) in viral titration (Figure 9). Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 infected cells compared to HSV-1 treated by acyclovir 5 (A) and 10 (B) μM, respectively. Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 infected cells compared to HSV-1 treated by quercetin Que. 7.8 (A), 62 (B), and 125 (C) μM treated cells, respectively. Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 infected cells compared to HSV-1 treated by resveratrol 10 (A), 20 (B), and 30 (C) μM, respectively. Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 infected cells compared to HSV-1 treated by doxorubicin 0.2 (A), 2 (B), and 8 μM (C), respectively.


Acyclovir, as a nucleoside analog, has long been considered the conventional therapy in HSV-1 treatment. Therefore, the reduction of virus replication in acyclovir was compared with other drugs at different concentrations. Quercetin at concentrations of 62 μM and 125 μΜ showed comparable performance to acyclovir 10 μM at all time points (24, 48, 72, and 96 hours), while Quercetin 7.8 μΜ showed a higher virus titer than acyclovir 10 μΜ at different time points (Figure 10). Resveratrol at all three concentrations (10 μM, 20 μM, and 30 μM) showed significant changes compared to acyclovir 10 μΜ and resulted in a higher virus titer (Figure 11). Doxorubicin at concentrations of 0.2 μM and 2 μΜ showed a higher virus titer at all time points compared to acyclovir 10 μΜ, and doxorubicin 8 μΜ had a similar effect to acyclovir 10 μΜ only at 72-hour time point (Figure 12). Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 Acy. treated, compared with Que. 7.8 (A), 62 (B), and 125 (C) μM treated cells, respectively. Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 Acy. treated, compared with Res. 10 (A), 20 (B), 30 (C) μM treated cells, respectively. Growth curve of HSV-1 in treated Vero cells. A) TCID50% HSV-1 Acy. treated, compared with Dox. 0.2, 2 (B), and 8 (C) μM treated cells, respectively.

qPCR assay for HSV-1 DNA measurement
The virus titer was measured three times using the plaque forming unit (PFU) method. The average amount of the virus in the qPCR reaction was measured at the maximum and minimum concentrations of 2.9×104 PFU/reaction and 2.9 PFU/reaction, respectively.
The concentration below 2.9 PFU/reaction is undetectable. A standard curve was plotted for this reaction, and the slope = −3.3460, R2 = 0.99, Efficiency = 1.99 were calculated (Figure 13). The standard curve used in qPCR assay.
qPCR was carried out on the supernatant of infected Vero cells to analyze the amount of viral DNA at 24, 48, 72, and 96 hours. These results were compared with the HSV-1 KOS control group.
At 24 hours post-infection, the highest amount of viral DNA was observed in relation to quercetin (7.8 µm), while the lowest viral titration was observed with quercetin (125 µm), doxorubicin (0.2,2, 8 µm), acyclovir (10 µm), and resveratrol (30 µm).
Generally, at different time points, viral DNA significantly decreased in almost all treatment groups compared to HSV-1 infected without treatment, except doxorubicin (0.2 µM) and acyclovir (5 µm) treatments at 72 and 96-hours post-infection did not show a significant difference in viral DNA titrations (Figure 14 (C) and (D)). However, in drug-treated groups, as time progressed, the DNA virus increased slowly. The effects of doxorubicin, quercetin, and resveratrol in different concentrations on HSV-1 production in Vero cells during different time points.
Discussion
Considering that cancer patients who receive chemotherapy or radiotherapy have weakened immunity, the reactivation of the HSV occurs more significantly in these individuals. 23 HSV recurrence can lead to the development of visceral or central nervous system lesions, and in severe cases, it can even result in death. 24 Although acyclovir is a highly selective drug for the treatment of certain herpesviridae, such as HSV-1, alternative drugs such as quercetin, which have fewer side effects and can effectively inhibit virus replication 25 and modulate inflammation during HSV-1 infection, especially in cases of encephalitis and keratitis, can be considered. Several studies have shown that quercetin and resveratrol can modulate immune cells via NF-κB and TNF-α inhibition.26–29 Moreover, acyclovir resistance has been reported in over 30% of patients with immunocompromised conditions, which suggests that alternative approaches should be considered.20,30
Plants produce a diverse range of secondary metabolites known as phenolic compounds, which are characterized by their aromatic rings with hydroxyl substituents. To date, over 8000 different phenolic compounds have been identified across various plant families. These compounds possess potent antioxidant properties due to their phenolic hydroxyls, which help neutralize free radicals. In addition, they exhibit a wide range of therapeutic properties, including anticancer, anti-inflammatory, antibacterial, antifungal, and antiviral activities. 31 Several phenolic compounds have been found to have antiviral activity. For instance, procyanidin condensed tannins of epicatechin have shown efficacy against SARS-CoV-2. 32 Sambucus Formosana Nakai stem extract, which contains caffeic acid as a vital component, has demonstrated strong anti-CoV-NL63 potential. 33 Gallic acid and epigallocatechin gallate have been found to be highly effective against HSV-1, while pinoresinol and N-trans-ferulolyl tyramine exhibited moderate effectiveness. 34 Umesu phenolics show antiviral activity against influenza viruses in cell culture. 35 Furthermore, Umesu phenolics have shown antiviral activity against influenza viruses in cell culture. Numerous other studies have also highlighted the importance of phenolic compounds in various aspects of human health and disease prevention.
Tej N. Kaul et al. 25 have demonstrated that quercetin can decrease HSV-1 infectivity by 0% to 80% in a dose-dependent method, with doses ranging from 6 to 200 μΜ. However, a concentration of 200 μΜ of the drug seems to be toxic for cells, as it has shown a reduction in virus infectivity due to cell cytotoxicity. 36 The present experiments were designed to investigate whether quercetin, like acyclovir, can inhibit viral DNA and virus infection at doses that are not toxic to cells, using qPCR and TCID50% assays, respectively. Quercetin significantly decreased virus infectivity at concentrations of 62 and 125 μM at different time points during the kinetic assay, from 24 to 96 hours post-infection (P-value < .05). The quantity of viral DNA can be altered by drugs that affect viral DNA replication. Quercetin significantly diminished viral DNA at different time points in low (7.8 μΜ), medium (62 μΜ), and high (125 μΜ) concentrations. The virus kinetic assay of 7.8 μΜ quercetin, as measured by TCID50%, did not show a decline in virus titer compared to the DNA quantification assay, likely due to the higher sensitivity and resolution of qPCR. Interestingly, when comparing the effectiveness of quercetin and acyclovir in reduction of the virus, it was shown that quercetin at 62 μΜ can inhibit the virus as effectively as acyclovir (10 μΜ), and at 125 μΜ it significantly inhibited the virus more effectively than acyclovir.
Talimogene laherparepvec is a genetically engineered strain of HSV-1 that uses an oncolytic virus. It was approved by the FDA for melanoma therapy. 37 Other forms of engineered HSV-1 oncolytic viruses are being used in clinical trials in combination with chemotherapy. 38 Doxorubicin is a chemotherapy medication used to treat different types of cancer. 39 It can block the catalytic cycle of human DNA topoisomerase II (Top2) at various stages of the enzyme. 40 HSV-1 has evolved to utilize many cellular proteins for efficient replication within host cells. Top2 has been associated with the replication process of HSV-1 genome and also blocking the enzyme significantly reduces the quantity of infectious HSV-1.
Thus, the utilization of doxorubicin can be considered as drug repurposing. Doxorubicin is a chemotherapy agent that has been widely applied in the treatment of various cancers, including solid tumors, soft tissue and bone sarcomas, and breast, ovary, bladder, and thyroid cancers. Intriguingly, recent studies have also demonstrated the antiviral effects of doxorubicin against the SARS-CoV-2 spike protein and the MERS-CoV papain-like protease (PLpro), among others.41,42
At concentrations of 2 and 8 µM of doxorubicin, virus infection was significantly diminished at different time points (P-value <.05). However, the decrease in virus infection was less pronounced at a concentration of 2 μM compared with 8 μΜ. Despite the fact that in-vitro dose-response studies use lower concentrations of IC50% compared to cancer chemotherapy, the virus infection is still affected. 43 The DNA concentration of the virus in 2 and 8 μΜ of doxorubicin declined at different time points. In comparison with acyclovir, doxorubicin 8 μΜ remarkably showed good and similar performance to acyclovir 10 μΜ at the 72-hour post-infection. Similar studies have not been seen in previous studies that can be compared with it.
In this study, absolute quantification to quantify DNA concentration by plaque forming unit or PFU assay to evaluate of the standard curve was conducted. Plaque assay is a standard method for virus quantification and determines viable viruses and defective viruses or non-capsidated genomes are not measured. The standard curve analysis of the experiment was established in different time points with interval 2 months and significant fluctuation at Ct values was not observed (data was not shown). The correlation coefficient or R, slope, and efficiency were 0.99, −0.33, and 99%, respectively, (Figure 13) that are ideal. 44 We determined at least 9 pfu per reaction at qPCR which is quite low and there is a rational correlation between pfu and viral genome copies in qPCR.
We exposed the virus to different concentrations of resveratrol (10, 20, and 30 μM) but found no significant effects on HSV-1 during the examined time period. John J. Docherty et al. 45 reported that resveratrol, at concentrations of 10, 25, and 50 μg/ml, inhibited HSV-1 replication. Concentrations of 25 and 50 μg/ml showed a significant effect on viral inhibition. We converted these concentrations to molar units, which were equal to 40, 109, and 219 μΜ, respectively. These concentrations were much higher than the ones we used. Therefore, we used lower concentrations due to the difficulty of dissolving a large amount of resveratrol powder in ethanol, which caused precipitation. Additionally, we used lower concentrations to avoid any cytotoxic effects on the cells, staying below the lethal dose of the drug. Two other studies have examined signaling pathways such as NF-κB and AMPK/Sirt1, which resveratrol affects to lessen the expression of HSV-1 genes involved in virus replication.46,47 In a study by John J. Docherty et al. 48 , who investigated the local effect of resveratrol in the form of cream on skin lesions in mice, they concluded that one of the concentrations of resveratrol reduced lesion formations. This suggests that the local effect of resveratrol may be greater than what is observed in in vitro experiments.
Footnotes
Author contributions
Shahrzad Zangooie carried out the experiments. Shahrzad Zangooie and Ali Teimoori wrote the manuscript with support from Reza Ghanbari. Farid Azizi Jalilian and Shahab Mahmoudvand helped supervise the project. Ali Teimoori designed the model and the computational framework and analyzed the data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the proposal number 140205314429 of Hamadan University of Medical Sciences (UMSHA).
