Abstract
Background
Monoclonal antibody therapy (MAT) received Food and Drug Administration (FDA) Emergency Use Authorization (EUA) for mild to moderate COVID-19 treatment in adults at a high-risk for progression to severe disease in November 2020. This study assessed the impact of MAT on clinical outcomes.
Methods
We conducted a single-center, retrospective study comparing 30-day COVID-19-related emergency department (ED) visits, admissions, and mortality in patients receiving MAT (bamlanivimab, bamlanivimab-etesevimab, or casirivimab-imdevimab) between 16 November 2020 and 19 June 2021, compared to a control group of high-risk adults diagnosed with mild to moderate COVID-19 prior to MAT availability between 16 May 2020 and 15 November 2020. Statistical analysis used logistic regression analysis with backward selection to determine the odds ratios and 95% confidence interval evaluating the relationship between clinical characteristics and outcomes.
Results
1187 patients who received MAT were compared to 1103 patients not treated with MAT. Multivariable regression model adjusted for possible confounders showed patients who received MAT had lower rates of ED visits (3.2% vs 7.4%, OR = 0.46, 95% CI = 0.31–0.70, p < .001) and hospital admissions (4.3% vs 7.8%, OR = 0.42, 95% CI = 0.29–0.62, p < .001) compared to the control group. After adjusting for confounders, MAT was associated with decreased mortality (OR = 0.36, p = .035). In the MAT group, those treated within 2 days of COVID-19 diagnosis had lower mortality than those treated more than 2 days post-diagnosis (unadjusted OR = 0.152, 95% CI = 0.031–0.734, p = .019).
Conclusions
Individuals treated with MAT had lower rates of 30-day COVID-19-related ED visits and hospital admissions compared to those not receiving MAT. Early MAT resulted in lower 30-day mortality compared to receipt >2 days post COVID-19 diagnosis.
Summary of key points
Individuals who received bamlanivimab, bamlanivimab-etesevimab, or casirivimab-imdevimab for COVID-19 had lower rates of ED visits and hospitalizations compared to an untreated cohort. Monoclonal antibody therapy (MAT) within 2 days of COVID-19 diagnosis resulted in lower 30-day mortality compared to receipt >2 days post-diagnosis.
Introduction
Since the emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in December 2019, SARS-CoV-2 has spread rapidly worldwide leading to the onset of a global pandemic resulting in over 774 million cases leading to over 7 million deaths worldwide as of March 2024, with over 1.1 million deaths in the United States alone. 1 SARS-CoV-2 is the viral etiology of the coronavirus disease 2019 (COVID-19) which presents as a spectrum of disease from asymptomatic infection to more severe illness including acute respiratory distress syndrome, multiorgan failure, or death. Despite the astonishing spread and severity of infections secondary to SARS-CoV-2 worldwide over the previous 4 years, few therapeutic options exist for the treatment of patients diagnosed with SARS-CoV-2, with remdesivir, an intravenous antiviral medication, is the only therapy approved by the US Food and Drug Administration (FDA) for treatment of SARS-CoV-2, and its use limited to hospitalized patients during the time of this study.
Neutralizing monoclonal antibodies targeting the SARS-CoV-2 spike protein have been evaluated for efficacy in treating high-risk patients with mild to moderate COVID-19 symptoms due to their ability to potentially inhibit viral attachment by blocking the SARS-CoV-2 spike protein from binding to the angiotensin-converting enzyme 2 receptor in the respiratory cells, thereby decreasing the infectivity of the virus. 2 Previous studies have identified male gender, older age, cardiovascular disease, lung disease, hypertension, diabetes, and obesity as risk factors for progression to severe COVID-19. 3 When evaluating patient outcomes, several studies have illustrated decreased hospitalizations and death in patients with mild to moderate infections who received treatment with monoclonal antibody therapy (MAT), including in the immunocompromised population.4–10 The BLAZE-1 randomized, placebo-controlled trial showed accelerated viral load clearance following treatment with bamlanivimab, and also found decreased hospitalizations at day 29 for patients treated with bamlanivimab compared to those who received placebo with an absolute risk reduction of 4.8%, although this study had too small a sample size to draw meaningful conclusions.4–6 Additionally, Ambrose et al. also noted decreased all-cause emergency department (ED) visits, hospitalizations, and deaths in patients treated with MAT but noted variations in effectiveness based on patient risk factors and variants. 7
Bamlanivimab became the first MAT to be granted EUA by the FDA for treatment of those with mild to moderate COVID-19 considered at a high-risk for progression to severe disease on 9 November 2020, with casirivimab-imdevimab becoming the second MAT approved shortly thereafter on 21 November 2020.11,12 Randomized control trial data demonstrated decreased viral load at day 7 in participants randomized to receive casirivimab-imdevimab compared to those who received placebo, and additionally showed that MAT appeared more effective when given earlier in the disease course. 13 Following these initial EUAs, a third MAT, bamlanivimab-etesevimab, was granted EUA approval on 9 February 2021. 14
Several observational studies have assessed the safety and real-world efficacy of MAT for treatment of mild to moderate COVID-19 demonstrating that treatment with MAT resulted in decreased ED visits and hospitalizations in patients treated with MATs compared to untreated high-risk patients.15–17 Although clinical efficacy in reducing all-cause ED visits and hospitalizations in patients diagnosed with mild to moderate COVID-19 has been illustrated, further studies are needed to determine the efficacy of MAT in COVID-19-specific outcomes in those who received MAT versus those who did not. Data is also lacking to determine if the timing of receipt of MAT from the time of COVID-19 diagnosis influences outcomes. The purpose of this study was to assess the impact of MAT on clinical outcomes in adult outpatients with mild to moderate COVID-19.
Methods
Study design
This was a single-center, retrospective study at a large, tertiary care institution comparing outcomes of adult outpatients diagnosed with mild to moderate COVID-19 between 11 May 2020, and 10 November 2020, with a high-risk for progression to severe disease who would have qualified for MAT based on EUA criteria if it had been available at that time to those diagnosed with mild to moderate COVID-19 between 16 November 2020 and 19 June 2021, who were treated with bamlanivimab, bamlanivimab-etesevimab, or casirivimab-imdevimab. This study was approved by the Institutional Review Board at The Ohio State University.
Institutional practices
Patients who tested positive for SARS-CoV-2 after initial EUA authorization of MAT on 9 November 2020 and met EUA criteria were referred for MAT through an electronic consult process previously described in a separate publication.
18
Patients were considered eligible for MAT if they tested positive for SARS-CoV-2 via either polymerase chain reaction (PCR) or rapid nasopharyngeal nucleic acid amplification testing (NAAT), had onset of symptoms less than 7 days prior to referral for MAT, and were considered high-risk for disease progression based on having at least 1 of the following: age
Patient population
Medication administration records were used to identify patients who received MAT with either bamlanivimab, bamlanivimab-etesevimab, or casirivimab-imdevimab between 16 November 2020 and 19 June 2021 in the outpatient setting. All patients who received MAT in the outpatient setting during this time period were included in the treatment group. Patients who received MAT during inpatient admission or in the ED were excluded from this study. A query of The Ohio State University Information Warehouse was performed to identify patients with a positive SARS-CoV-2 PCR or rapid NAAT in the outpatient setting from 11 May 2020 through 10 November 2020. The electronic medical record (EMR) for each patient was reviewed to collect demographic information, medical co-morbidities, SARS-CoV-2 test results, MAT details, duration of symptoms, and outcomes for each patient. Demographic data collected included age, race, ethnicity, sex, and co-morbidities, BMI
Outcomes
Medical records were reviewed to assess for all-cause ED visits, inpatient admissions, or mortality within 30 days from a positive SARS-CoV-2 test in the control group and from date of MAT administration in the treatment group. Outcomes were reviewed and designated as COVID-19-related or not based on documentation in the EMR.
Statistical analysis
Descriptive characteristics, symptoms, and outcomes were compared using T-tests and Chi-square tests (for continuous and dichotomous variables respectively). Odds ratios (OR) were calculated to compare descriptive characteristics by patient outcomes and recipient of MAT. Logistic regression was used to model ED visits, admission, and mortality. A backwards selection procedure was used for these models. All analyses were conducted in the STATA version 17. p-values <0.05 were considered statistically significant.
Results
Patient population
Descriptive characteristics for patients who received monoclonal antibody therapy (MAT) versus patients who did not receive MAT.

Unless otherwise noted, data presented as N (%).
1Data Presented as mean ± standard deviation.
2Duration of symptoms is duration of symptoms prior to receipt of MAT in the MAT group and duration of symptoms prior to positive SARS-CoV-2 test in the no MAT group.
Outcomes
30-Day outcomes in patients who received monoclonal antibody therapy (MAT) versus patients who did not receive MAT.
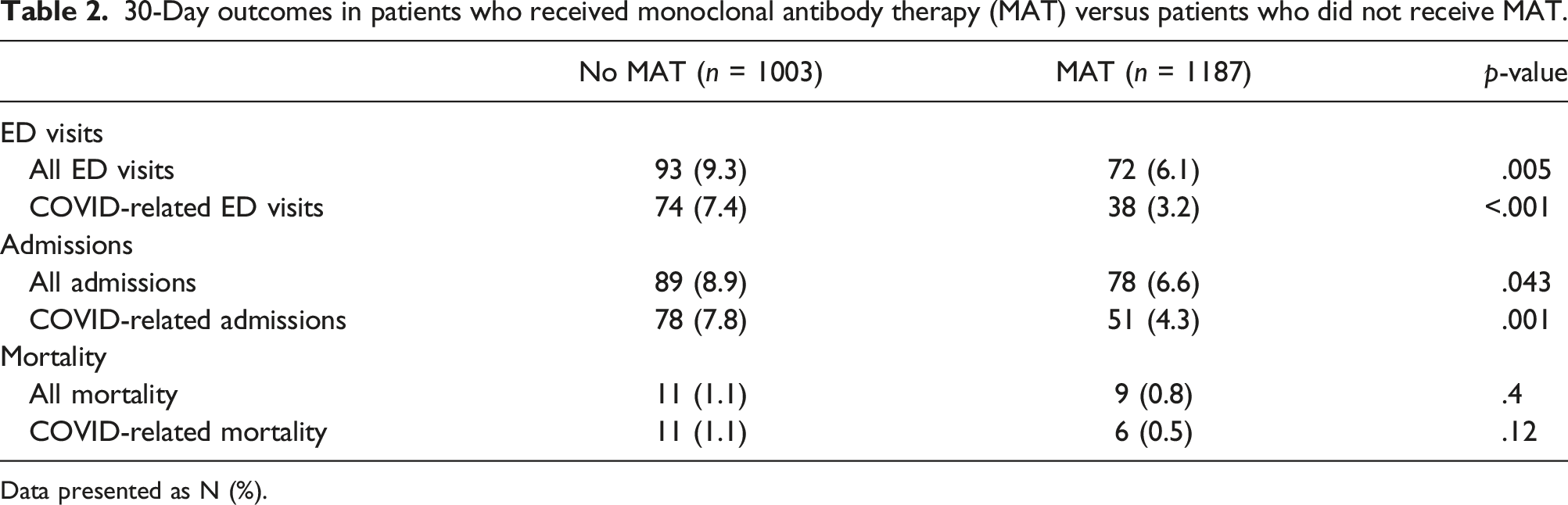
Data presented as N (%).
Descriptive characteristics and outcomes by emergency department (ED) visit or by admission.

Abbreviations: BMI, Body Mass Index.
Unless otherwise noted, data presented as N (%).
aData presented as mean ± standard deviation.
Logistic regression models for ED visits, inpatient admissions, and mortality.

Table 3 details the association of descriptive characteristics and inpatient admissions. Significant descriptive characteristics identified were placed in a multivariable logistic regression model for inpatient admissions (shown in Table 4). In this model, age (OR = 1.30 per 10 years increase in age, 95% CI = 1.15-1.47, p < .001), CKD (OR = 2.29, 95% CI = 1.42–3.67, p = .001), COPD (OR = 2.16, 95% CI = 1.21–3.86, p = .009), CAD (OR = 1.58, 95% CI = 1.08–2.34, p = .02), history of transplant (OR = 2.34, 95% CI = 1.12–4.88, p = .02), diabetes (OR = 1.79, 95% CI = 1.27–2.51, p = .001), and malignancy (OR = 1.77, 95% CI = 1.17–2.70, p = .007) were associated with an increased risk of inpatient admission, while receipt of MAT was significantly associated with a decreased risk of inpatient admission (adjusted OR = 0.56, 95% CI = 0.40–0.79, p = .001). The association of MAT and COVID-19-specific admissions was also considered and in a multivariable regression model adjusting for other effects (including age, race, gender, and relevant medical comorbidities), a similar OR was calculated (adjusted OR = 0.42, 95% CI = 0.29–0.62, p < .001).
In Table 4, an analysis of descriptive characteristics and mortality was undertaken. There was no statistically significant association between crude 30-day mortality and MAT (unadjusted OR = 0.69, 95% CI = 0.28–1.67, p = .4). Age, however, was a negative confounder of the association of MAT and mortality. As noted earlier, individuals who received MAT were 5.4 years older on average than those who did not. Age was associated with increased mortality as well (unadjusted OR = 2.73, 95% CI = 1.93–3.86, p < .001 per 10-year age increase). Adjustment for age was conducted to determine the true association of MAT and mortality. After adjusting for age, obesity, and CKD, MAT was associated with reduced mortality (adjusted OR = 0.36, 95% CI = 0.14–0.93, p = .035). 85% (17/20) of the deaths were deemed COVID-19-related, so COVID-19-specific mortality was not modelled given limited statistical power.
Within the MAT group, 30-day outcomes were also compared for those who received MAT within 2 days of SAR-CoV-2 diagnosis versus those receiving MAT more than 2 days after diagnosis. No association was seen between the outcomes of ED visits or admissions between the two groups. However, 30-day mortality was lower if patients received MAT within 2 days of COVID-19 diagnosis (unadjusted OR = 0.152, 95% CI = 0.031–0.734, p = .019; age-adjusted OR = 0.205, 95% CI = 0.041–1.01, p = .052). As shown in Figure 1, the risk of mortality increased continuously up to day 5 (p-value for trend = 0.02). Death rate at 30 days based on time to monoclonal antibody therapy. Figure 1 illustrates the death rate 30 days after COVID-19 diagnosis based on the timing in days of receipt of monoclonal antibody therapy after COVID-19 diagnosis. Abbreviations: MAT, monoclonal antibody therapy.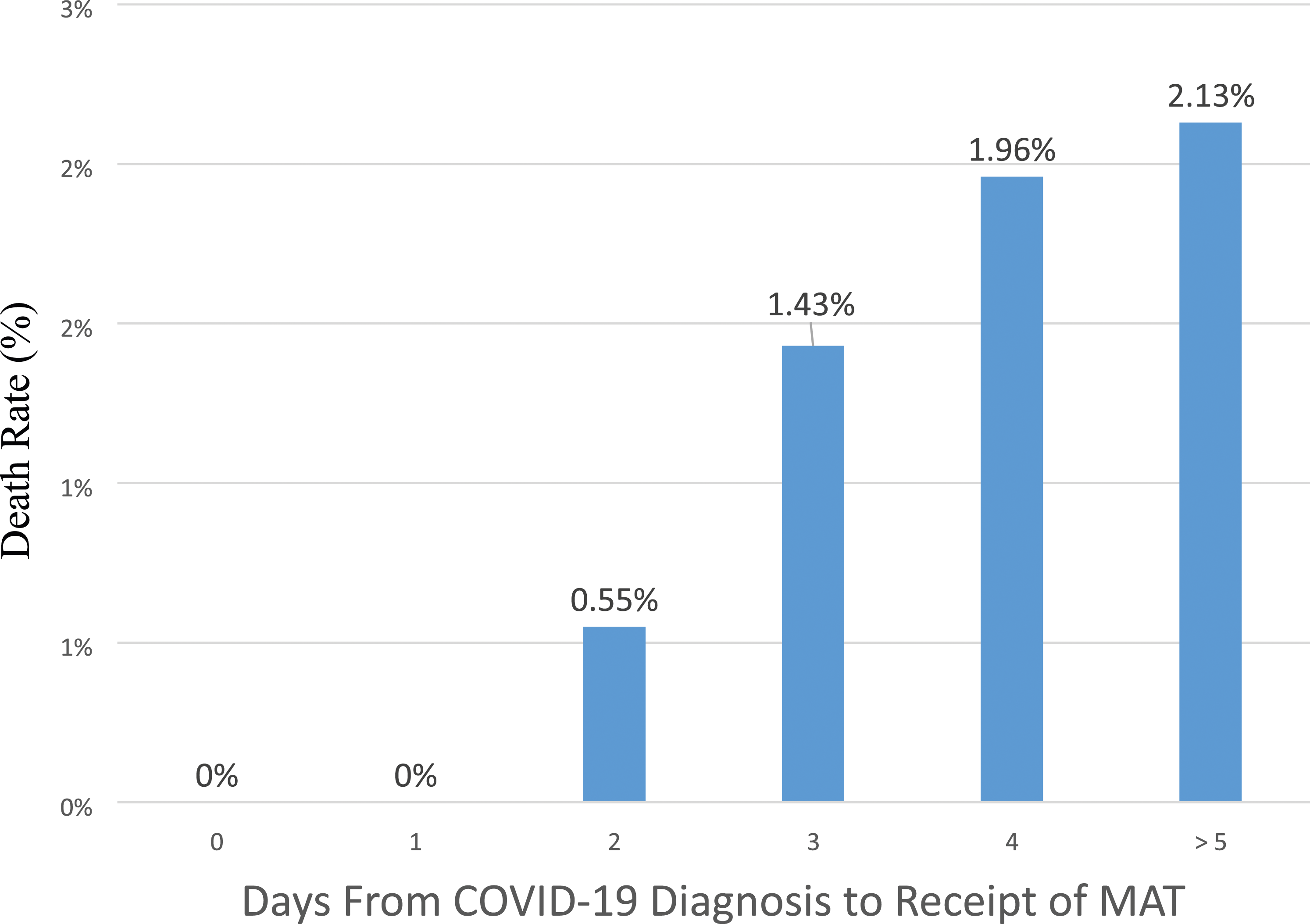
Comparison of outcomes by MAT
Within the MAT group, 803 patients (67.6%) received bamlanivimab monotherapy or bamlanivimab-etesevimab. Neither age, race, ethnicity, nor obesity were significantly associated with these MAT therapies. Those who received any bamlanivimab therapy were more likely to be male compared to those who received casirivimab-imdevimab (OR = 1.46, 95% CI = 1.14–1.88, p = .003). Receipt of any bamlanivimab therapy was not significantly associated with ED visits (OR = 0.74, 95% CI = 0.45–1.20, p = .2), admission (OR = 1.15, 95% CI = 0.70–1.91, p = .6), or death (OR = 0.60, 95% CI = 0.16–2.22, p = .4) compared to those who received casirivimab-imdevimab. No significant difference in COVID-19-specific outcomes was noted based on MAT received (data not shown).
Discussion
In our study, we found that treatment with MAT for COVID-19 resulted in a significant reduction in ED visits and admissions at 30 days when compared to a control group who did not receive MAT. When considering specifically COVID-19-related ED visits and hospital admissions, those treated with MAT maintained a significant decrease in these outcomes when compared to the control group. These findings are in line with what other studies have shown illustrating that treatment with MAT for COVID-19 resulted in reduced risk of ED visits and admission, both all-cause and COVID-19-related outcomes.15,16,19–23
Additionally, we found that after adjusting for age, obesity, and CKD, MAT was also associated with decreased mortality (OR = 0.36, 95% CI = 0.14–0.93, p = .035). While a previous randomized clinical trial of casirivimab-imdevimab showed a mortality benefit for MAT versus placebo, 24 a mortality benefit of MAT had not been demonstrated in previous real-world observational studies.
This study also found that asthma, African American race, male gender, and leukaemia were risk factors associated with increased ED visits, while older age, CKD, COPD, CAD, history of transplant, diabetes mellitus, and malignancy were noted to be risk factors for admission in patients with COVID-19. While previous studies have found similar risk factors, our study evaluated risk factors not just for admission, but for ED visits as well, showing that risk factors for these two outcomes may differ while both are decreased by receipt of MAT.25–27 Identifying risk factors that increase an individual’s risk for ED visits or admissions following diagnosis with COVID-19 may result in earlier identification of those patients most likely to progress to severe disease to prioritize them for MAT.
When we compared outcomes of ED visits, admissions, and mortality between patients who received either bamlanivimab or bamlanivimab-etesevimab versus those who received casirivimab-imdevimab, we noted no difference in outcomes based on the specific MAT patients received. This is in contrast to a previous study showing improved outcomes when patients were treated with casirivimab-imdevimab compared to those who received either bamlanivimab or bamlanivimab-etesevimab. 28 While our study did not observe a difference in 30-day outcomes for patients based on the MAT they received, these results may have been limited by the sample size of the patients included in the two MAT treatment groups and changes in the predominant variant during the clinical study period.
While monoclonal antibodies targeting the COVID-19 spike protein have previously proven useful in reducing ED visits and hospitalizations in high-risk patients, as the COVID-19 virus has evolved and new variants have emerged, the neutralization activity may become compromised or make specific monoclonal antibodies less useful.29–31 During the time period for this study, the EUA for bamlanivimab was revoked on 15 April 2021, and although outside the window of inclusion for this study, the EUAs for bamlanivimab-etesevimab and casirivimab-imdevimab were limited on 25 June 2021 and 26 January 2022, respectively.32,33 These EUAs were revoked based on decreasing the efficacy of each specific MAT with the emergence of new COVID-19 variants. 31 As this pattern continued, there was a continued need to evaluate the impact of different MATs to target the current circulating SARS-CoV-2 virus variant(s). Although currently there is no approved MAT available for the treatment of COVID-19, it is still imperative to understand both the utility and the benefits of MAT so that these lessons can be applied in the future, whether in the management of another potential viral pandemic or through extrapolation of these therapies to apply to the treatment of other viral infections. Additionally, though alternative oral agents are available for treatment of COVID-19, MAT may still be an important therapeutic option for high-risk or immunocompromised patients unable to receive other oral agents due to potential drug–drug interactions. While logistically it is complex for institutions to make these therapies readily available to the most at-risk patient population, there is a clear benefit to institutions in providing these therapies, since these treatments can result in decreased ED visits and hospitalizations, thereby decreasing the strain on the health care system. Our data highlights the importance of administering MATs rapidly, as those who received MAT earlier following COVID-19 diagnosis had lower mortality. Even with the availability of oral therapeutic antiviral options for COVID-19, monoclonal antibodies will still be an important therapeutic option for high-risk patient populations given possible limitations on oral antiviral therapy due to significant drug–drug interactions and renal clearance limitations. Monoclonal antibodies also have considerably longer half-lives than currently available oral antiviral treatments and may be particularly beneficial for severely immunocompromised individuals who may be at risk for prolonged viral replication and shedding.
Limitations of this study include its retrospective nature, which limited data collection by information documented in the institutional EMR. Patients presenting to other EDs or admitted to other hospitals were not captured. Another limitation of this study included differences in the demographics of the MAT group and the control group. Most notably, patients in the group who received MAT were less likely to be African American or Hispanic, which may be secondary to inequities in access to care among these populations despite their increased risk of exposure to the SARS-CoV-2 virus and complications from COVID-19. This study is also limited by a potential immortal time bias as patients who were eligible for MAT in the treatment group were not included if they did not receive this MAT, which may have resulted in patients who qualified for MAT but were excluded due to either ED visit or admission after COVID-19 diagnosis but prior to receipt of this antiviral therapy. Additionally, as a tertiary referral hospital, some patients live at a considerable distance from our campus and may have been limited in their ability to receive MAT due to lack of transportation to be able to present to our institution for administration of MAT within the eligibility window. One final limitation of this study is that the adjusted logistic regression results are adjusted for variables other than receipt of MAT, and these variables may also be potentially confounding themselves.
In our study, African American patients were significantly less likely to receive MAT in a timely way (Δ mean = 0.30 ± 0.11 days, p = .008). To receive MAT at our institution, patients needed to have a documented positive SARS-CoV-2 NAAT and be referred via an electronic consult for consideration of this therapy by a primary care provider or subspecialist within 7 days of onset of symptoms which may have been an important barrier for those patients without established providers or easy access to SARS-CoV-2 testing.
Conclusions
Individuals who received MAT for mild to moderate COVID-19 in the outpatient setting had a statistically significant lower rate of COVID-19-related 30-day ED visits and admissions compared to those not treated with MAT, after adjusting for potential confounders. Patients receiving MAT for COVID-19 also had a lower adjusted 30-day mortality. Patients who received MAT within 2 days of COVID-19 diagnosis had statistically significant lower 30-day mortality compared to those who received it later after the onset of symptoms, illustrating the importance of early therapy with antivirals for improved efficacy of treatment. Lessons from the COVID-19 pandemic, including the importance of rapid development and implementation of effective MAT are crucial and applicable to viral illnesses in future viral infection epidemics/pandemics. MAT has been a critical component of our COVID-19 armamentarium and health systems should strive to ensure equitable access to these life-saving therapies when they are available.
Supplemental Material
Supplemental Material - Clinical outcomes in patients with mild to moderate coronavirus disease 2019 treated with monoclonal antibody therapy versus an untreated control cohort
Supplemental Material for Clinical outcomes in patients with mild to moderate coronavirus disease 2019 treated with monoclonal antibody therapy versus an untreated control cohort by Courtney N Nichols, Mark Lustberg, Mohammed Mahdee E Sobhanie, Lucia J Niermann, Melissa Gordon, Nicholas Kman, Jonathan Parsons, Mark Conroy, Michael Dick, James Allen, Erica Reed, Joy Lehman and Carlos Malvestutto in Antiviral Therapy
Footnotes
Author contributions
All listed authors conceived and designed the analysis. CN collected the data. ML performed the statistical analysis. CN, CM, and ML wrote the initial draft of the paper with revisions by all other listed authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Carlos Malvestutto’s institution received funds from Lilly Inc. to conduct a trial involving bamlanivimab and bamlanivimab-etesevimab at his institution. All other authors have no relevant conflicts of interest to report.
Funding
The work was supported by Award Number UL1TR002733 from the National Center for Advancing Translational Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
