Abstract
Background
Molnupiravir (MOV) is an orally bioavailable ribonucleoside with antiviral activity against all tested SARS-CoV-2 variants. We describe the demographic, clinical, and treatment characteristics of non-hospitalized Danish patients treated with MOV and their clinical outcomes following MOV initiation.
Method
Among all adults (>18 years) who received MOV between 16 December 2021 and 30 April 2022 in an outpatient setting in Denmark, we summarized their demographic and clinical characteristics at baseline and post-MOV outcomes using descriptive statistics. Outcomes were emergent hospitalization and all-cause mortality during the 28 days after MOV initiation. We estimated the odds ratios (OR) of outcomes by time from positive test to treatment using logistic regression.
Results
We identified 3691 MOV-treated patients, of whom 45.8% were male and mean age was 70.1 years. Most patients (76.2%) initiated MOV within 0–2 days after a positive SARS-CoV-2 test and 16.8% within 3–5 days. Over a 28-day period, rates for all-cause, respiratory- or COVID-19-related, and COVID-19-related hospitalization were 4.8%, 2.6% and 1.5%, respectively. All-cause mortality was 1.6%. Initiation of MOV 3–5 days after a positive SARS-CoV-2 test compared to 1–2 days was associated with an increased risk of all-cause (OR 1.85, 95% CI 1.29–2.67) and respiratory or COVID-19-related (OR 1.78, 95% CI 1.07–2.94) hospitalization, and all-cause mortality (OR 2.90, 95% CI 1.64–5.15).
Conclusion
MOV was primarily prescribed to vaccinated elderly persons with multiple comorbidities. The all-cause hospitalization and mortality rates in this population were low. Early initiation of MOV reduced the risk of hospitalization and death compared with late initiation.
Introduction
With more than 750 million confirmed cases and nearly seven million deaths globally, including 3.4 million confirmed cases and 8700 deaths in Denmark, the COVID-19 pandemic had overwhelmed healthcare systems around the world, and the admission of large numbers of patients to general wards and intensive care units (ICU) caused a significant drain on healthcare resources. 1 In most cases, infection with SARS- CoV-2 causes a mild respiratory disease that resolves without the need for intervention, but the elderly and those with underlying chronic medical conditions are at high risk of progression to severe disease that can result in hospitalization and/or death. 2 Vaccines have, to an extent, helped to curtail the spread of COVID-19, and reduced its associated morbidity and mortality. 3 However, possibly due to a waning efficacy of vaccines as well as emerging virus strains such as Omicron, the numbers of break-through infections in fully vaccinated individuals are high. 4 Therefore, there is still a need for effective oral antiviral drugs for treatment of early-stage COVID-19 in the outpatient setting to prevent progression to severe disease, hospitalization and/or death in high-risk patients.
The Danish Health Authorities recommendations for treatment with molnupiravir December 2021.

With the increasing uptake of vaccines, antiviral treatment, and emergence of new variants, COVID-19 epidemiology continues to evolve quickly. 10 In addition, due to decisions of the healthcare authorities and practitioners, patient characteristics in real-world healthcare settings may not always reflect the specific inclusion/exclusion criteria of randomized controlled trials. Thus, it is important to understand the real-world use and clinical and societal benefits of MOV in preventing COVID-19 disease progression resulting in additional healthcare resources utilization, hospitalization, and death.
The aims of this nationwide real-world study are to describe the baseline demographic, clinical, and treatment characteristics of non-hospitalized Danish patients treated with MOV, and the clinical outcomes over a 28-day period following MOV initiation.
Methods
Study design and population
The study is designed as a retrospective observational cohort study using de-identified patient level data from national public health registries in Denmark. The study population includes all individuals ≥18 years of age who received MOV in Denmark in the period 16 December 2021 to 30 April 2022 and who were not hospitalized at the time of MOV initiation. One of the criteria for prescription of MOV in Denmark was a current positive SARS-CoV-2 test. The date of MOV prescription redemption serves as the index event. Guidelines for when to initiate treatment is based on onset of symptoms. We do not have information on onset of symptoms and therefore use date of prescription redemptions as proxy for date of symptom onset. Because the registries do not capture whether or when a patient took their MOV medication, we assume that patients initiated MOV treatment the same day the MOV prescription was redeemed. The baseline period is the span 1-day to 24 months before the index event. Participants were retrospectively followed-up for 28 days after the index event. The study population consists of unique individuals. If a patient had more than one redemption, only the first redemption was included. Patients who were younger than 18 years old, died on the index date, had evidence of pregnancy in the 3 months prior to the index event (7 patients), or were treated with any antiviral medication for COVID-19 other than MOV in the 30 days before MOV initiation (27 patients) were excluded. To ensure that the population only consisted of non-hospitalized patients, patients who had a hospital contact (of any cause) on the day of the index event (245 patients) were also excluded.
Study data sources
The data for this study were extracted from multiple national registries, which contain individual-level health data linked to the unique identification numbers for all individuals living in Denmark. All inhabitants in Denmark are assigned a unique social security number (Central Person Register [CPR] number) at birth or immigration. All contacts with the health system, including drug prescription, are linked with the unique CPR number, and are captured in national public health registries. Patients’ medication history was extracted from the Danish National Prescription Registry (DNPR), which records the Anatomical Therapeutic Chemical (ATC) codes and dates for all prescriptions redeemed from Danish pharmacies. Patients’ medical, clinical, and hospital data were extracted from the Danish National Patient Registry (NPR), which records data using ICD-10 codes. COVID-19 vaccination history was extracted from the Danish Vaccination Registry (DVR). Data on a positive a SARS-CoV-2 tests were extracted from the COVID-19 surveillance registry. Any occurrence of death among the study population was identified from the Danish Civil Registration System (CRS). Demographic data, such as age, sex, civil status, and address, are also linked to CRS. Crosslinking the registries provided demographic characteristics, vaccination status, comorbidities, and concomitant medications, as well as hospitalization and mortality data.
Exposure
Adult patients (aged ≥18 years) treated with MOV in an outpatient setting. MOV treatment was defined by the first redeemed prescription for MOV identified through the Danish National Prescription Registry.
Outcomes
Primary outcomes were all-cause hospitalization, respiratory illness- or COVID-19-related hospitalization, COVID-19-related hospitalization, and all-cause mortality over a 28-day follow-up period after MOV initiation. Hospitalization was defined as inpatient admission that was emergent and lasting more than 24 hours to exclude planned contacts such as previously scheduled check-up visits. To control for potential misclassification of diagnosis, we specified two different COVID-19-related outcomes – one that includes hospitalizations due to COVID-19 and another that includes hospitalization due to COVID or other respiratory illnesses.
Covariates
List of ICD-10 and ATC codes.

*Identified through cancer diagnosis instead of ATC, since most cancer drug treatment takes place at a hospital.
Statistical analysis
Patients’ demographic and clinical characteristics at baseline and post-MOV initiation were summarized using descriptive statistics of means and distribution across characteristics. We estimated the odds ratio and 95% confidence intervals (CI) of outcomes by time from positive test to treatment and vaccination status using logistic regression, controlling for age, gender, region of residence, comorbidities, and comedication. We did not control for previous infection due to too few cases. A p-value of <0.05 is considered statistically significant. Analyses were carried out in STATA 17.
Results
Baseline characteristics
Patient characteristics at the time of MOV initiation.
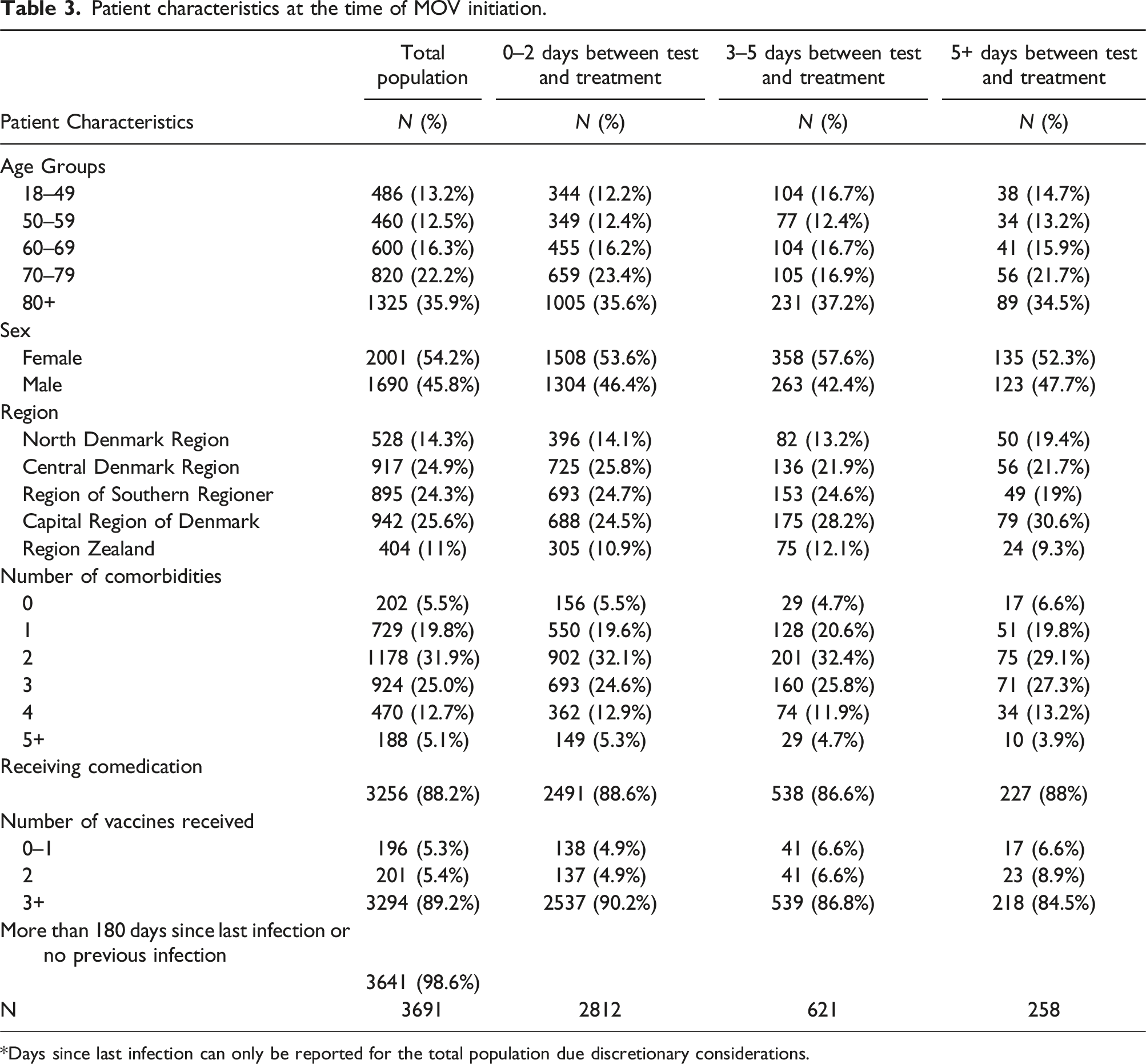
*Days since last infection can only be reported for the total population due discretionary considerations.
The correlation between covariates and treatment initiation.

*RC: Reference category.
*- : Omitted due to too little variation.
*The number of observations vary due to a few individuals not having information about what region they live in and categories being omitted due to too little variation.
Most patients (93%) initiated MOV within 5 days after a positive SARS-CoV-2 test: 76.2% within 2 days and 16.8% within 3–5 days (Table 3). Time from a positive SARS-CoV-2 test to MOV initiation was not different by age, sex, or burden of comorbidity (Table 4). Fully vaccinated patients were more likely to start treatment within both 2 (OR 1.41, 95% CI 1.109–1.802, p = 0.005) and 5 days (OR 1.61, 95% CI 1.103–2.357, p = 0.014) (Table 4).
Outcomes
During the 28-day follow-up period, rates for all-cause, respiratory- or COVID-19-related, and COVID-19- related emergent hospitalization were 4.8%, 2.6%, and 1.5%, respectively. All-cause mortality rate was 1.6%.
The effect of time of treatment initiation on health outcomes.

Sex, number of comorbidities, and comedication were not significantly associated with the risk of all-cause, respiratory-, or COVID-19-related hospitalization or all-cause mortality. There were no differences in outcomes among the five Danish Regions (Table 5).
As a sub analysis, we examined the outcomes of fully vaccinated patients (≥3 vaccinations per Danish standards) separately. Since 89.2 % of the population is fully vaccinated the results are very similar to those of the full population. Initiation of MOV 3–5 days after a positive SARS-CoV-2 test compared to 1–2 days had higher odds of all-cause (OR 2.05 95% CI 1.4–3.01, p < 0.001), respiratory or COVID-19-related (OR 2.04, 95% CI 1.12–3.47, p = 0.009) and COVID-19 related (OR 2.12 95% CI 1.07–4.23, p = 0.033) emergent hospitalization, and all-cause mortality (OR 2.93, 95% CI 1.61–5.35, p < 0.001). Of note, time between MOV initiation and COVID-19-related emergent hospitalization was significant in the fully vaccinated population but not in the full population. The ORs for initiation of MOV later than 6 days compared to 1–2 days remains not statistically significant.
As a second sub analysis, we examined the population of patients with concomitant cancer. 15.1 % of the MOV-treated population had concomitant cancer. Patients with cancer had a higher risk of all cause hospitalization compared to patients without cancer (6.8% vs 4.7%, p-value: 0.035). There were no significant differences between cancer and non-cancer patients with regards to COVID-19-related, COVID-19-related or respiratory-related hospitalizations, and death. Additionally, we found no significant difference in outcome comparing initiation of MOV at 1–2 days to initiation at 3–5 days for patients with cancer (Table 5).
Discussion
In December 2021, the Danish Medicines Agency approved MOV for pre-license use. To reduce the number of COVID-19-related hospitalizations and deaths, the Danish Health Authorities recommended that MOV be offered to patients with mild-to-moderate COVID-19 with symptoms for ≤5 days and an increased risk of progression to severe disease 9 (Table 1). During the study period, MOV was the only available oral antiviral treatment for COVID-19 in Denmark and could be prescribed by both general physicians and hospital doctors. The treatment was tax-funded and free of charge for the patients.
The MOVe-OUT trial was carried out in an unvaccinated population during the period of Delta variant predominance.6,10 Given the dynamic nature of the COVID-19 pandemic, with emergence of new variants of concern, and, implementation of COVID-19 vaccine programs, there is considerable uncertainty about whether the clinical benefit of treatments observed in clinical trials are realized in the real world in Denmark and the rest of the world. Therefore, in this nationwide real-world study, we aimed to describe the baseline demographic, clinical, and treatment characteristics of non-hospitalized Danish patients treated with MOV and their clinical outcomes over a 28-day period following a redeemed prescription of MOV.
Our study population consist of MOV treated, predominantly vaccinated, elderly patients with multiple comorbidities likely infected with the Omicron variant. The population characteristics show that not all MOV prescriptions were in accordance with the recommendations from the Danish Health Authority (Table 1). 13% of the population were younger than 50 years of age and not all patients had a positive SARS-CoV-2 test registered. However, home-tests are not in the registries, and the few patients without a positive SARS-CoV-2 test visible in the registries are assumed to have a positive home-test.11,12
We found that the rates of all-cause (4.8%), respiratory- or COVID-19-related (2.6%), and COVID-19 related (1.5%) emergent hospitalization and all-cause mortality (1.6%) were low. However, hospitalization rates increased with age among people with COVID-19, and were 2.9% for people aged 65–79 and 10.7% for people aged 80+ according to national surveillance data, which both are significantly higher than the estimates from our study. 13 MOV treatment, high vaccine coverage, predominant infection with the Omicron variant or a combination of these factors may explain low overall hospitalization rates. However, data on the background population during the study period cannot be directly compared with the results from our MOV-treated study population. Among other things we restricted the study population to people not hospitalized immediately before or at the time of a positive test, whereas the reported statistics include them. Therefore, it cannot be concluded whether MOV treatment contributed to the low hospitalization and death rates seen in the study population. In a sub analysis, we found that patients with cancer had a higher risk of all-cause hospitalization, but not respiratory- or COVID-19-related, and COVID-19 related emergent hospitalization and all-cause mortality compared to patients with no cancer. A study examining hospitalization rates and mortality among MOV-treated patients with haematological malignancies found substantially higher rates of hospitalization (20 %) and mortality (4 %) in the 28 days after breakthrough COVID-19 diagnosis during the same time period as our study. 14 This could potentially be explained by the higher vaccination rates in our population, as only half of the participants in the other study had received a booster. Of note, early treatment with MOV was associated with reduced risk of hospitalization and death, indicating that initiation of MOV within 1–2 days of a positive SARS-CoV-2 test influences the clinical outcome of COVID-19. Viral replication is highest in the initial stage of the infection, so treatment with MOV has the best effect during this period. 7 An observational study from Australia suggests that treatment with MOV within 1 day of diagnosis provided the greatest benefit. 15 No benefit in reducing the risk of hospitalization was seen if treatment was initiated two or more days after diagnosis and for death four or more days after diagnosis. 15 However, since we cannot control for confounding factors, such as disease severity at the time of MOV initiation, we cannot conclude whether the difference in outcomes are caused by earlier initiation of treatment or that patients initiating treatment earlier were less likely to have severe symptoms. In our study being vaccinated significantly reduced the risk of being hospitalized, but not all-cause mortality. This is, however, most likely due to 95% of our population being fully vaccinated and a very low event rate of all-cause mortality. Previous studies have shown that vaccines are effective at preventing COVID-19 related death. 16 In our sub analysis, consisting of individuals with ≥3 vaccines, initiation of MOV within 3-5 days of a positive SARS-CoV-2 test compared to 1–2 days had higher risks of hospitalization and all-cause mortality; odds ratios for all outcomes were higher compared to the full population. However, based on this analysis we cannot determine whether this is because early initiation of MOV treatment is more effective if patients have received a third booster vaccine dose or if there are other differences between the groups such as disease progression or symptoms.
Observational studies from Hong Kong and Israel have suggested that treatment with MOV is associated with reduced risk of hospitalization and death in patients over 65 years of age.17,18 PANORAMIC is the largest study including over 25,000 participants who were randomized to MOV plus usual care or only usual care. The study population was young (mean age 56.6 years) and 94% had received at least three doses of COVID-19 vaccines. Early treatment with MOV did not reduce the low risk (1%) of the composite outcome of hospitalization or death, but MOV reduced time to recovery and reduced viral load. 19 A meta-analysis including six studies during the Omicron wave found that MOV only had clinical effectiveness on disease progression and all-cause mortality in patients with low vaccine coverage. However, MOV was associated with faster reduction in viral load than placebo. 20 A recent systematic review and meta-analysis including nine randomized clinical trials concluded that MOV has no effect on mortality and likely no effect on hospitalization and death in a predominantly immunized population of COVID-19 patients and its effect on symptom resolution is uncertain. 21 In accordance, an observational study from Israel found that only older and inadequately vaccinated patients had clinical benefit of treatment with MOV. 22
In clinical trials with unvaccinated adults at high risk of progression to severe COVID-19, early treatment with nirmatrelvir/ritonavir (NMV/r) or remdesivir reduced the relative risk of hospitalization or death by 89% and 87%, respectively.23,24 In addition, the neutralizing monoclonal antibodies (nMAb) casirivimab and imdevimab and sotrovimab have been used for treatment of early COVID-19. 24 Remdesivir and nMAb can only be administered intravenously and NMV/r has a high potential for drug-drug-interactions. 8 Further, the emergence of new SARS-CoV-2 variants, including the Omicron variant, have been found to evade neutralization by nMAb. 25 In a recent retrospective cohort study from Wales, it was shown that prompt outpatient treatment of high-risk patients with COVID-19 with MOV, NMV/r, or sotrovimab was associated with a significant reduction in hospitalization and death within 28 days of infection. No difference in effectiveness between treatments were observed. The rates of hospitalization or death were 4.1% among treated and 10.9% among untreated. The study was conducted in the same period as our study when Omicron was the dominant variant and 95.6% of participants were vaccinated. 26 The hospitalization rate in the treated group is comparable with our findings in MOV-treated patients. Mutoh et al. compared the effectiveness of MOV and NMV/r in a real-world community during the surge of the Omicron BA.5 subvariant in Japan. COVID-19 related hospitalization (2.8% and 3.5%, respectively) and death (0.4% and 3.5%, respectively) did not differ significantly between the MOV and NMV/r groups. 27 The mean age of the high-risk participants was 71.7 years and 71.7% had received at least three COVID-19 vaccine doses. The low hospitalization and death rates are comparable with our findings. Most patients receiving MOV are older and have comorbidities or comedications that made them otherwise ineligible for treatment with NMV/r. 8 This treatment selection bias could result in a pool of eligible participants with very high risk for hospitalization and death, thus masking the effectiveness of MOV treatment.
The strength of this study is the nationwide design and the very high quality of the Danish national registries, which captures health information on all residents in Denmark. This makes it possible to obtain demographic and health information on all individuals with a redeemed MOV prescription. By identifying the entire population of patients with redeemed MOV prescriptions we have eliminated some of the possible selection bias. However, when looking at the outcomes for different rates of initiating treatment, there is a possibility of unobserved confounding factors. The results in Table 3 does show that, with the exception of vaccination status and living in the Central Denmark Region, there are no differences in the observable characteristics between patients whose treatment was initiated within 2 days, 5 days or later than 5 days. But there could be difference in unobservable confounding factors such as difference in disease progression and symptoms. Since medical conditions will be identified based on existing records, coding inaccuracies may lead to misclassification bias. However, this misclassification will be non-differential since this would be the case for the entire population. In addition, we cannot ascertain based on the registry data that patients perfectly adhere to the MOV regimen or took MOV at all after being prescribed the medication. However, because only redeemed prescriptions are recorded in the Danish National Prescription Registry, it is likely that most of the study participants were exposed to MOV.
The limitations are that there is no comparison group. Further, the high vaccine coverage and predominance of infection with the Omicron variant during our study period could result in lower hospitalization or death rates compared to infection with other previous variants of concern. Moreover, because there is currently no reliable distinction between COVID-19-related and non-COVID-19-related deaths, we used all-cause mortality as the outcome. This definition could also result in the underestimation of MOV effectiveness due to its lack of specificity. In addition, the lack of availability of some risk factor information (obesity, smoking, physical activity, etc.) could limit the full characterization of MOV-treated patients in Denmark.
Since 27 February 2023, the Danish Health Authorities no longer recommend MOV for treatment of early COVID-19 in high-risk patients based on EMA recommending that a marketing authorization should not be granted. 28 In consequence, the MOV European Union application for marketing authorization for treatment of COVID-19 in adult was withdrawn on 23 June 2023. According to the WHO living guideline for COVID-19 drugs, NMV/r is the preferred drug, but MOV is conditionally recommended for patients with non-severe COVID-19 at highest risk of hospitalization. 29
Conclusion
In Denmark, in the period December 2021 to April 2022 when Omicron was the predominant variant, MOV was primarily prescribed to predominantly vaccinated older patients with multiple comorbidities in accordance with the guidelines from the Danish health authorities. The all-cause hospitalization and mortality rate during 28 days of follow-up were low in this population. In most cases, treatment was initiated within 5 days after a positive test. Initiation of MOV within 2 days after a positive test reduced the risk of all-cause hospitalization and death. This indicates that even in a population of immunized elderly high-risk patients infected with Omicron prompt initiation of treatment with MOV may have a beneficial effect on the clinical course of COVID-19.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Carsten Schade Larsen (CSL) has received fees from MSD and Pfizer for lectures and participation in advisory boards. CSL is the main author of this article and has not received any funding. Nina Breinholt Stærke (NBS) has no conflicts of interest. NBS has assisted with the writing of the article and has not received any funding. The statistical analysis was funded by MSD and carried out by Caroline Louise Westergaard (VIVE) and Jakob Kjellberg (VIVE). Gui Liu is an employee of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA, Urs Arnet is an employee of MSD, Switzerland and Line Roesgaard Kantsø is an employee of MSD, Denmark and were all responsible for the study protocol.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the MSD Denmark Aps.
