Abstract
Background
Darunavir/cobicistat/emtricitabine/tenofovir alafenamide (D/C/F/TAF) fixed-dose combination (FDC) was developed as a once-daily, complete antiretroviral (ARV) regimen therapy to address the need for simplified protease inhibitor-based ARV regimens. This study assessed the swallowability and acceptability for long-term use of scored placebo tablets matching the D/C/F/TAF FDC tablets in children living with HIV-1.
Methods
This study (NCT04006704) was a Phase 1, open-label, randomized, single-dose, 2-period, 2-sequence crossover study in children living with HIV-1, aged ≥6 to <12 years and weighing ≥25 to <40 kg, on a stable ARV regimen for ≥3 months. Participants were asked to swallow whole (size, 21 × 11 × 7 mm) and split matching placebo D/C/F/TAF tablets. Swallowability of the matching placebo D/C/F/TAF tablets (primary endpoint) was assessed by observers. Acceptability of taking matching placebo D/C/F/TAF tablets and current ARVs was evaluated by participants using a 3-point questionnaire. Participants rated the acceptability for long-term daily use of the placebo D/C/F/TAF tablets, and observers assessed how easily caregivers could split a scored tablet by hand, using 3-point questionnaires.
Results
Among the 24 participants who enrolled and completed the study, 95.8% (23/24) were able to swallow the whole and split matching placebo D/C/F/TAF tablets after 1 or 2 attempts. Most participants (>70%) rated the acceptability of tablets for long-term daily use as acceptable or good to take. Breaking the tablets was considered easy or OK by 79.2% (19/24) of caregivers.
Conclusion
Scored D/C/F/TAF FDC tablets are swallowable – with whole favoured over split – and considered at least acceptable for long-term daily intake in children living with HIV-1 aged ≥6 to <12 years.
Trial registration
ClinicalTrials.gov Identifier: NCT04006704.
Introduction
For individuals living with HIV, single-tablet regimens can increase adherence, increase virologic suppression, decrease health care utilization and costs, and improve quality of life compared with multitablet regimens.1–4 The development of simplified antiretroviral (ARV) regimens, such as single-tablet regimens, that reduce pill burden is a high priority for pediatric populations living with HIV. 5 Pediatric guidelines recommend the use of simplified regimens, such as darunavir/cobicistat/emtricitabine/tenofovir alafenamide (D/C/F/TAF), to optimize adherence, which is key to achieving and maintaining viral suppression and preventing medication resistance.6,7 The D/C/F/TAF fixed-dose combination (FDC) was developed as a once-daily, complete ARV therapy addressing the need for simplified protease inhibitor-based ARV regimens.8–11 Bioequivalence of D/C/F/TAF FDC relative to combined administration of the separate commercially available agents has been demonstrated in both adult and pediatric populations.12–14 As such, D/C/F/TAF is approved at doses of 800/150/200/10 mg in several countries for the treatment of HIV-1 infection in adults and adolescents weighing ≥40 kg15,16 and is under development as a scored D/C/F/TAF 675/150/200/10 mg tablet containing 675 mg of darunavir (as ethanolate), 150 mg of cobicistat, 200 mg of emtricitabine, and 10 mg of tenofovir alafenamide (as fumarate) for pediatric patients ≥6 to <18 years who weigh ≥25 to <40 kg.
Patient acceptability, which may include palatability and swallowability, should be evaluated as part of pediatric development programs, as it is likely to have a significant impact on treatment adherence and therapeutic outcomes.17,18 The objective of this study was to assess the swallowability, ease of swallowing, and acceptability of long-term daily intake of scored D/C/F/TAF 675/150/200/10 mg FDC tablets in children living with HIV-1.
Methods
Study design
This study (TMC114FD2HTX1006; ClinicalTrials.gov Identifier: NCT04006704) was a Phase 1, open-label, randomized, single-dose, 2-period, 2-sequence crossover study conducted in the United States and Spain. The study consisted of a screening phase and an administration phase of 1 day during which the intake of 2 matching placebo D/C/F/TAF 675/150/200/10 mg FDC tablets (1 taken whole and 1 taken as a split tablet) was evaluated. Participants were randomized 1:1 by computer-generated randomization to 1 of 2 treatment sequences (whole then split or split then whole), stratified by age category (≥6 to <9 years and ≥9 to <12 years).
Placebo tablets matching the size and shape of the active FDC tablets were used so as not to interfere with participants’ active ARV regimen and as taste of the core tablet is not expected to be perceived while swallowing a film-coated whole or split tablet. The matching placebo D/C/F/TAF tablets were sized 21 × 11 × 7 mm and were composed of lactose monohydrate, microcrystalline cellulose, croscarmellose sodium, magnesium stearate, polyvinyl alcohol, polyethylene glycol, talc, and titanium dioxide. Participants could indicate their preferred method for tablet swallowing (i.e. taken with approximately 15 ml of semi-solid food followed by ≤150 ml of liquid or taken with ≤150 ml of liquid only). Participants’ caregivers reviewed a standardized instruction for swallowing the placebo tablets (Table S1) and could develop an individualized instruction based on their pre-study experience in coaching their child at home to swallow tablets; the standardized or developed individualized instruction was used consistently for both tablet intakes. Caregivers split the tablets by hand (i.e. tablet cutters were not permitted).
This study was conducted in compliance with the current International Conference on Harmonization guidelines on Good Clinical Practice, consistent with the Declaration of Helsinki principles. All participants provided written informed consent.
Participants
Eligible participants were children ≥6 to <12 years of age who weighed ≥25 to ≤40 kg, had documented HIV-1 infection with <400 HIV-1 RNA copies/ml within 6 months prior to screening, and were on a stable ARV regimen for ≥3 months prior to screening. Participants were excluded if they were not willing to attempt swallowing tablets or had any active condition (e.g. oral infection, significant physical or psychological disease or other findings, such as concomitant medications) that could prevent the participant from swallowing or limit or confound the protocol-specified assessments and outcomes. Participants’ parent(s) or their legally acceptable representative provided signed informed consent. Children capable of understanding the nature of the study (typically ≥7 years of age) also provided assent.
Assessments
The primary endpoint was the ability to swallow the matching placebo D/C/F/TAF tablets, irrespective of the mode of intake, based on a questionnaire completed by study site observers. The secondary study endpoints were acceptability of swallowing, acceptability of long-term intake, and ease of splitting the tablet as assessed by questionnaires completed by 3 sets of individuals: participants, caregivers, and study site observers. Participants completed a 3-point questionnaire to document the ease of taking their current ARV regimen during screening and the whole and split matching placebo D/C/F/TAF tablets after drug intake. In addition, participants rated the acceptability of taking the whole and split matching placebo D/C/F/TAF tablets for long-term daily use. Caregivers completed a 3-point questionnaire on the ease of taking the whole and split matching placebo D/C/F/TAF tablets. Observers completed a questionnaire based on observation of the participant to record swallowing difficulties after drug intake. Additionally, observers recorded the ease of breaking the tablet based on observation of the caregiver. Observers were permitted to assist in completing the questionnaire if the participant and their caregiver had difficulties. Safety and tolerability were evaluated throughout the study from signing of informed consent until the last study-related procedure.
Data analyses
As there was no formal hypothesis tested in this study, sample size was calculated based on precision. With a study population of 24 participants, assuming that ≥20/24 could swallow the tablet, it could be determined with 80% confidence that the ability to swallow would be ≥76%.
The ability to swallow the whole and/or split matching placebo D/C/F/TAF tablets (primary endpoint) was reported as proportions with 95% confidence intervals (CIs; based on the Wilson method); for other questionnaire outcomes, frequencies were tabulated.
The ease of swallowing their current ARV regimen and the matching placebo D/C/F/TAF tablets, rated by participants, were jointly analyzed by a repeated proportional odds logistic regression model based on cumulative logits, adjusted for sequence, age group (≥6 to <9 years and ≥9 to <12 years), and sex.
Results
Participants
Out of 27 participants screened, 24 were enrolled and completed the study. Three patients were excluded as they did not meet one of the eligibility criteria, which was unrelated to the willingness to attempt swallowing tablets or the presence of an active condition that may affect swallowing. Two participants had weights >40 kg; however, both participants were aged ≥6 years to <12 years and conclusions of the study were not considered to be impacted by this deviation.
Baseline demographics and characteristics.
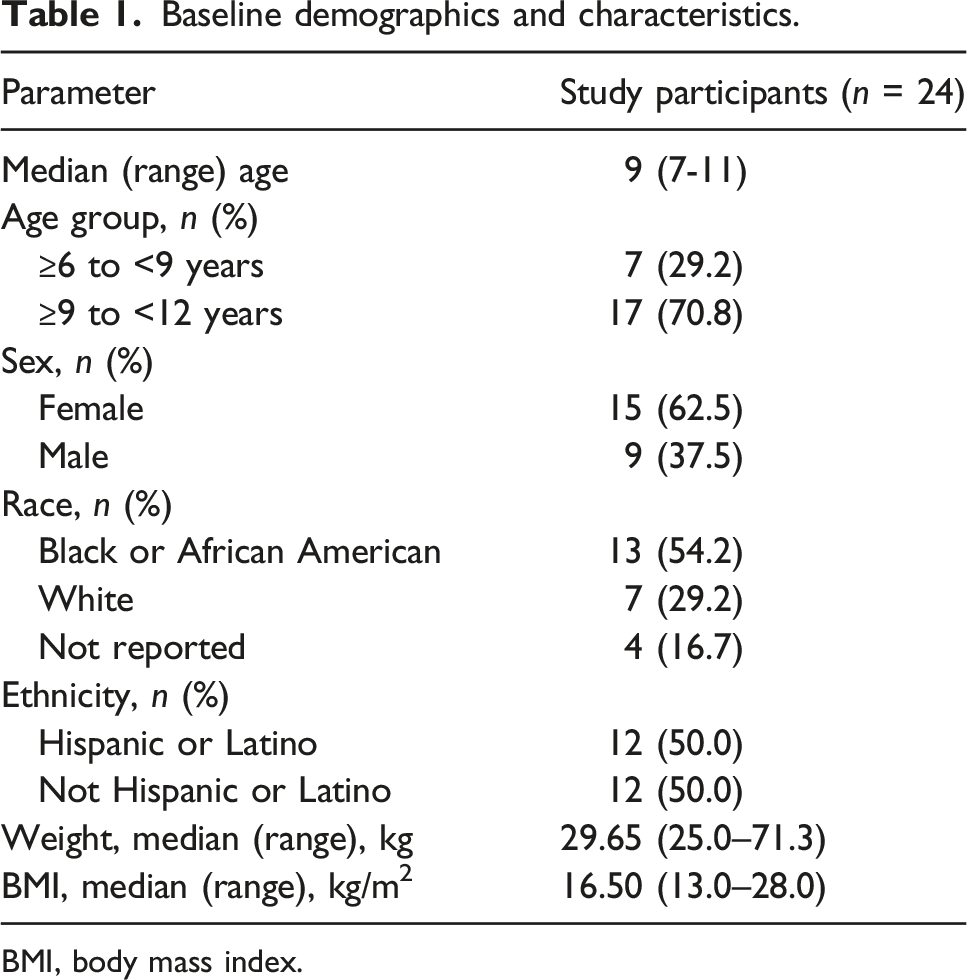
BMI, body mass index.
Current ARV regimens.

ARV, antiretroviral; FDC, fixed-dose combination.
aIncludes both current ARV medication taken as tablet formulation and other concomitant medication taken as liquid formulation.
Ability to swallow as assessed by observers (primary endpoint)
Ability to swallow the matching placebo D/C/F/TAF tablet and details for each attempt according to study site observers.
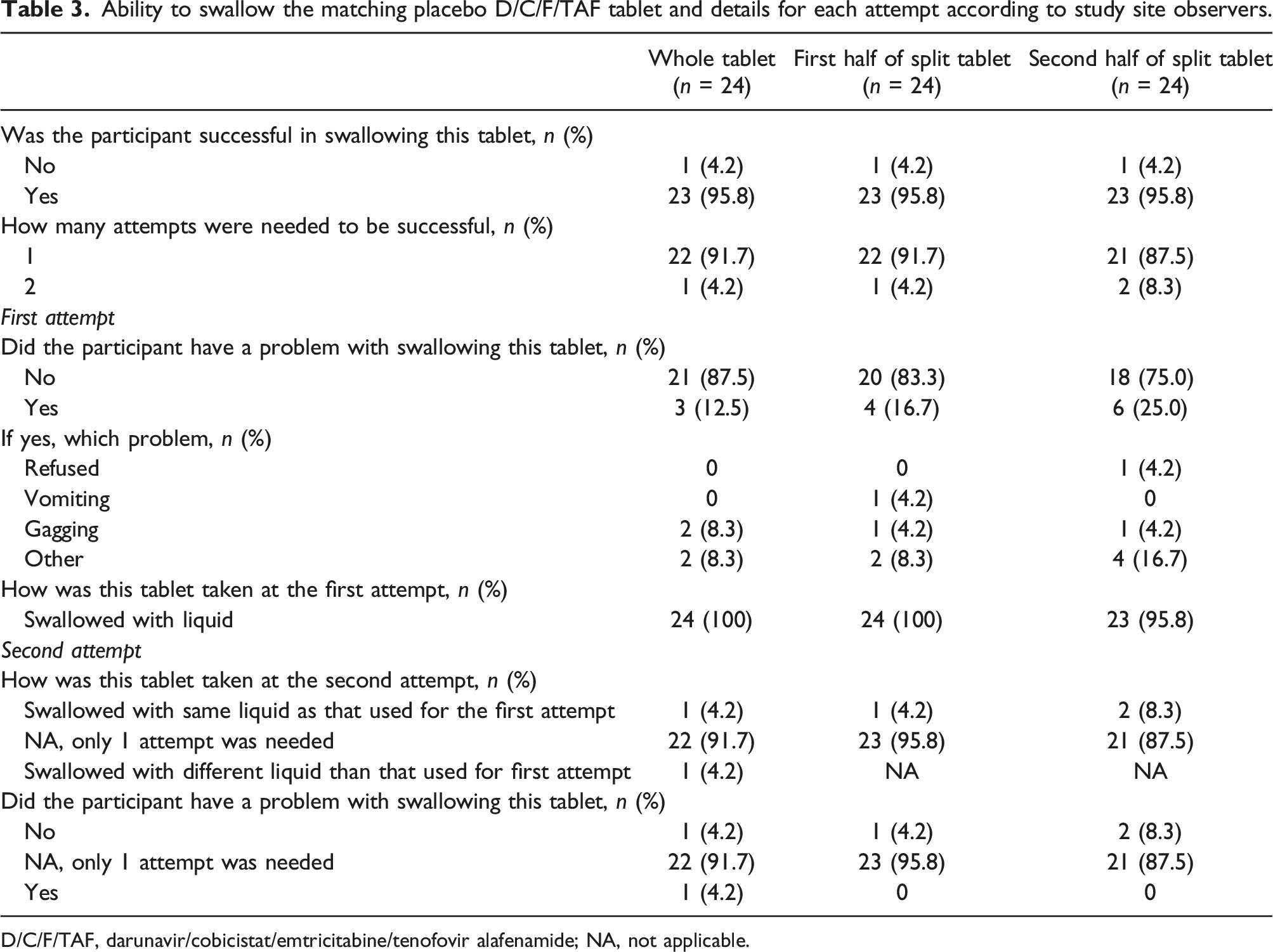
D/C/F/TAF, darunavir/cobicistat/emtricitabine/tenofovir alafenamide; NA, not applicable.
For the whole tablet, 22 (91.7%) participants were able to swallow it on the first attempt and 1 (4.2%) on the second attempt (Table 3). Problems were reported for 3 participants: 1 participant gagged, 1 participant needed multiple sips to swallow the tablet, and 1 participant gagged and was not able to swallow the tablet.
For the split tablet, 22 (91.7%) participants were able to swallow the first half on the first attempt and 1 (4.2%) on the second attempt. For the second half of the tablet, 21 (87.5%) participants were able to swallow on the first attempt and 2 (8.3%) participants required 2 attempts. Problems were reported for 6 participants: 1 participant found the tablet too big (first half) and preferred smaller pills (second half), 1 participant needed multiple swallows to move the tablet down their throat (second half), 1 participant said the tablet was bothering them while swallowing (second half), 1 participant reported scraping while swallowing (first and second half), 1 participant reported gagging (first and second half), and 1 participant reported vomiting (first half) and refused to take the tablet (second half; Table 3).
Ease of swallowing as assessed by participants
Ease of swallowing and acceptability according to study participants and caregivers.

ARV, antiretroviral; D/C/F/TAF, darunavir/cobicistat/emtricitabine/tenofovir alafenamide; NA, not applicable.
Acceptability of long-term use
Most participants reported the whole or split tablets as acceptable or good to take for a longer period of time (Table 4). Four (16.7%) participants said the whole tablet was not acceptable to take for a long period of time, the same proportion as for the split tablet.
Logistic regression model
No differences of acceptability were observed between the whole (p = 0.44) or split (p = 0.19) matching placebo D/C/F/TAF tablets compared with current ARV therapy as the reference treatment. There were no effects of age group (p = 0.23), sex (p = 0.75), or treatment sequence (p = 0.057).
For the analysis with the whole tablet as the reference treatment, no differences of acceptability between current ARV therapy were observed (p = 0.44). However, for the split tablet, a less favourable acceptance was reported compared with the whole tablet (p = 0.0031).
Ease of swallowing and acceptability as assessed by caregivers
Reports from caregivers regarding swallowability agreed with those from participants and observers, with 3 (12.5%) reporting the whole tablet as hard for the child to take, and 1 (4.2%) and 2 (8.3%) reporting split 1 and split 2, respectively, as hard for the child to take (Table 4).
Ease of splitting by caregivers as assessed by observers
Ease of splitting matching placebo D/C/F/TAF tablets by caregivers as assessed by observers.

Safety
Safety data were analyzed for the intent-to-treat population (i.e. all participants who were randomized and received ≥1 dose of treatment). No treatment-emergent adverse events (AEs) were reported during the study.
Discussion
This study examined the swallowability and acceptability of matching placebo, scored, film-coated D/C/F/TAF tablets in a pediatric population of participants living with HIV. Most (95.8%) participants successfully swallowed the tablets after 1 or 2 attempts. The majority of participants found it easy to take the matching placebo D/C/F/TAF tablets and rated the acceptability of these tablets for long-term daily use as acceptable or good to take. Study site observers’ and caregivers’ assessments matched those from participants. The results from this study add to those from previous studies reporting that simplified ARV regimens have greater acceptability in the pediatric population compared with those that are more complex.19–22 Previous studies have also reported a preference for tablets over pellets or syrup formulations in children living with HIV.20,22
Adherence to HIV regimens is critical to keep virus levels undetectable, prevent the development of medication resistance, and reduce the risk of transmission to others.6,7 Simplified ARV regimens, such as single-tablet regimens, have been shown to improve adherence in the pediatric population.5,20 However, such regimens need to be acceptable to children for long-term daily dosing with respect to swallowability and palatability.17,22,23 Therefore, assessing the acceptability of single-tablet regimens in the pediatric population is important to long-term adherence and outcomes in children living with HIV.
In this study, nearly all participants reported their current ARV as neither hard nor easy or easy to take. As their current ARV, 20 out of 24 participants were taking FDC regimens; none of these participants reported their current regimen as hard to take. One participant reported her current multiple dose ARV of abacavir + atazanavir + ritonavir as hard to take and the whole matching placebo D/C/F/TAF tablet as easy to take. This observation is consistent with results from previous studies demonstrating children’s preference for simpler ARV regimens.19,20
Pill swallowability can be influenced by tablet size: in a study of children 4 to 12 years of age, tablet size was noted as the most important factor for taking a tablet every day. 24 The matching placebo D/C/F/TAF tablet used in this study is 21 × 11 × 7 mm in size, which is similar to the larger FDC pills taken by participants as current ARV. The participant who was unable to swallow the D/C/F/TAF FDC tablet either whole or split, was used to taking bictegravir/F/TAF as their current ARV, which is a smaller FDC tablet (15 × 8 mm) than the D/C/F/TAF FDC tablet. 25
In the present study, no differences in acceptability were observed between the whole or split matching placebo D/C/F/TAF tablets versus participants’ current ARV regimens; however, participants reported that taking the split matching placebo D/C/F/TAF tablet was less favourable than taking the whole matching placebo D/C/F/TAF tablet. Participants who reported a preference for the whole versus the split tablet indicated that the split tablet was bothering or scraping the throat. Taking a split tablet requires swallowing twice, which may be less favourable to many children.
However, many individuals, especially children, have difficulty swallowing larger pills; therefore, split tablets might improve adherence for some patients. In the current study, breaking the matching placebo D/C/F/TAF tablets was considered easy or OK for the majority of caregivers in the absence of tablet splitters. No relevant differences in bioavailability have been observed between the adult split and whole D/C/F/TAF 800/150/200/10 mg tablets. 14
The limitations of this study included small study size, which may limit generalizability, in particular for patients aged 6 to 9 years.
In conclusion, scored D/C/F/TAF FDC tablets were generally considered acceptable to swallow and acceptable for long-term daily intake in children living with HIV. Simplified medication regimens are expected to be associated with improved adherence and outcomes.
Supplemental Material
Supplemental Material - Assessment of swallowability and acceptability of scored darunavir/cobicistat/emtricitabine/tenofovir alafenamide (D/C/F/TAF) fixed-dose combination tablets (FDC) in HIV-1–infected children aged ≥6 to <12 years, using matching placebo tablets: A randomized study
Supplemental Material for Assessment of swallowability and acceptability of scored darunavir/cobicistat/emtricitabine/tenofovir alafenamide (D/C/F/TAF) fixed-dose combination tablets (FDC) in HIV-1–infected children aged ≥6 to <12 years, using matching placebo tablets: A randomized study by Sandy Van Hemelryck, Erika Van Landuyt, Veerle Hufkens and Simon Vanveggel in Antiviral Therapy.
Footnotes
Acknowledgements
This study was sponsored by Janssen Research & Development, LLC, and Janssen-Cilag S.A. Medical writing support was provided by Miranda Tradewell, PhD, of Lumanity Communications Inc., and was funded by Janssen Scientific Affairs.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SVH, EVL, VH, and SV are or were employees of Janssen Pharmaceuticals and may hold stock / stock options in Johnson & Johnson.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored and funded by Janssen Research & Development.
Data availability statement
Although these data are not currently publicly available for sharing, requests for sharing can be sent to the corresponding author and will be evaluated on an individual basis.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
