Abstract
Background
Avian infectious bronchitis virus (IBV), a coronavirus, causes a huge economic loss to the poultry industry. Andrographolide (APL) is a compound with a variety of pharmacological properties, including antiviral and anti-inflammatory effects. In this study, APL was evaluated for antiviral activity by its anti-apoptotic, anti-pyroptosis, and anti-inflammatory effects.
Methods
The cytotoxicity of APL was determined by the MTT method. We investigated the therapeutic impact of APL on IBV through a plate assay. We explored that APL inhibited IBV-induced apoptosis, pyroptosis, and inflammation in HD11 cells by RT-qPCR and immunofluorescence. Also, it was verified in the clinical chicken embryo trial.
Results
We found that APL down-regulated apoptosis-related genes Caspase-3, Caspase-8, Caspase-9, Bax, Bid, and Bak, down-regulated pyroptosis gene DFNA5, and down-regulated inflammation-related genes (NF-κB, NLRP3, iNOS, TNF-α, and IL-1β). In addition, APL reduced the reactive oxygen species (ROS) production in cells. Finally, clinical trials showed that APL inhibited IBV-induced apoptosis, pyroptosis, and inflammation, as well as reduced the mortality and malformation of chicken embryos.
Conclusions
In this study, we delved into the antiviral properties of APL in the context of chicken macrophage (HD11) infection with IBV. Our findings confirm that andrographolide effectively inhibits apoptosis, pyroptosis, and inflammation by IBV infection. Furthermore, this inhibition was verified on chicken embryos in vivo. This inhibition suggests a substantial potential for APL as a therapeutic agent to mitigate the harmful effects of IBV on host cells.
Introduction
Avian Infectious bronchitis (IB) is an acute high-contact respiratory infectious disease caused by the infectious bronchitis virus (IBV). IBV is a gammacoronavirus belonging to the Coronaviridae family, and its virion contains a single copy of a positive-sense, single-stranded 27.6-kb RNA. 1 The virus can affect the upper respiratory tract and the reproductive tract, some strains can cause nephritis and affect chickens of all ages, leading to decreased egg production and even death of sick chickens, causing huge economic losses to the poultry industry. 2 Currently, both classic and variant IBV serotypes have been identified in most countries, with little or no cross-protection between the serotypes thus making IB control and prevention a global challenge.3,4
Andrographis paniculata (AP) is a therapeutic herb that has been discovered and practiced as an effective herbal immuno-drug in traditional medicine systems. 5 The main constituent of A. paniculata is andrographolide, a diterpene that contains a γ-lactone ring that is present in leaves. 6 A decade of proliferating scientific research with APL based on the active components established its multifarious pharmacological effect as an anti-bacteria, anti-virus, and anti-inflammation.7,8 A large number of experimental studies have shown that andrographolide has significant antiviral activity against the dengue virus,9,10 Zika virus, 10 chikungunya virus, 11 and SARS-CoV-2.7,12 Shi found that Andrographolide and its derivatives mainly inhibit the activity of Sars-Cov-2 protease, thus inhibiting viral replication. 13 Andrographolide reduces H1N1 virus-induced cell death by inhibiting virus-induced activation of RLRs signaling pathway. 14 Whether Andrographolide has an inhibitory effect on IBV-induced apoptosis, pyroptosis, and reactive oxygen species production have not been reported.
Apoptosis is programmed cell death which is essential for the development and survival of living organisms. 15 Apoptosis is thought to be a natural defence mechanism for the host to disrupt viral replication by eliminating virus-infected cells. 16 The balance between pro-apoptotic and anti-apoptotic protein regulators is the key to determining whether cells undergo apoptosis. 17 In contrast to apoptosis, pyroptosis is a form of necrotic and inflammatory programmed cell death induced by inflammatory caspases. 18 Gasdermin E also known as deafness, autosomal dominant 5 (DFNA5) is a second member of the conserved gasdermin family that displays a similar membrane-perforating function resulting in pyroptotic-like cell death. 19 Studies have shown that caspase-8 can also cleave GSDMD, leading to caspase-8-mediated GSDMD-dependent cell death in response to the extrinsic trigger of apoptosis.20,21 Pyroptosis has been considered inflammatory, as cells undergoing pyroptosis release pro-inflammatory molecules and organelles, such as nucleotides, IL-1 family cytokines, HMGB1, nucleic acids, and mitochondria. 22
In this study, we delved into the antiviral properties of APL in the context of chicken macrophage (HD11) infection with IBV. We studied the effects of APL on IBV-relative mRNA expression levels, including apoptosis-related genes, and ROS production in IBV-infected HD11 cells. Our results demonstrated the antiviral activity of APL against IBV infection, including inhibition of apoptosis-induced apoptosis and ROS production through the cellular mitochondrial pathway in HD11 cells. This provides an approach for the treatment of IBV infection. This interesting finding will contribute to further research and development of relevant therapeutic treatments.
Materials and methods
Reagents
Andrographolide (Solarbio, Beijing, China) with a molecular weight of 350.45 was diluted to 100 mM with DMSO and used as the stock solution. Serum-free Dulbecco’s Modified Eagle’s Medium (DMEM) (Gibco, USA) was used to dilute the APL master mix. Thiazolyl blue tetrazolium bromide (MTT) was sourced from Solarbio in Beijing, China, and Dimethyl sulfoxide (DMSO) was obtained from Sigma-Aldrich in Shanghai, China. Additionally, DMEM 2X High Glucose with
Virus and cell cultures
Chicken macrophages were purchased from the cell bank of the Chinese Academy of Science (Shanghai, China). HD11 cells were grown in 10% Fetal Bowen’s Serum (FBS) (Gibco, Australia) and RPMI Medium 1640 basic (Gibco, USA) containing penicillin and streptomycin and incubated at 37°C in a 5% CO2 incubator.
IBV Beaudette strain was kindly provided by Sichuan Agricultural University and propagated in 10-day-old specific-pathogen-free (SPF) chicken embryos (Beijing Boehringer Ingelheim Vital Biotechnology Co., Ltd). The allantoic fluid of embryos infected at 37°C was collected after 48 h. The collected virus was adapted to Vero cells after three passages.
Cytotoxicity assay
The cytotoxicity of APL was tested according to the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) methods used by Ren X et al. 23 HD11 cells were inoculated into 96-well plates. HD11 cells were treated with different concentrations of APL and placed in an incubator at 37°C for 48 h. The supernatant was discarded, and 20 μL of 5 mg/mL MTT solution was added to each well. The wells were incubated at 37°C for 4 h 150 μL of DMSO was added to each well and incubated for 10 min at 37°C in the dark. The OD value at 490 nm was measured with a microplate reader, and cell viability was calculated according to the formula: (OD490 drug)/(OD490 control) × 100%.
Virus titration and infection
Virus titrations were calculated by median tissue culture infective dose (TCID50) with the Reed and Muench method. 24 Cells were seeded into 96-well plates. The virus solution was diluted in a 10-fold gradient, a total of 7 gradients were diluted, each gradient had 8 wells, and 100 μL was added to each well. And set up a negative control. After incubation at 37°C for 1 h, 200 μL of maintenance medium was added to each well and incubated for 48 h to observe the results and calculate the virus titre.
Virus growth kinetics
HD11 cells were initially seeded in 12-well plates and cultured in a cell culture incubator at 37°C with 5% CO2. The cells were then exposed to IBV virus at a viral load of 100 TCID50. Inoculation took place once the cells had reached approximately 90% confluence. After a 1-h incubation at 37°C, the viral supernatant was removed, and fresh maintenance medium was added. Samples, we collected at specified time intervals to assess the levels of IBV-N mRNA.
Plaque assay
When the culture cell density reached 90%, the virus was added to the cells and incubated at 37°C for 1 h. After washing with PBS, APL overlay medium containing 2% MC was added, and after 48 h at 37°C, the crystalline violet staining solution was added, and the number of plaques was counted.
Cells treatment
To determine the effect of APL on IBV Beaudette-infected HD11 cells, the HD11 cells cultured on 6-well plates were treated with different concentrations of APL for 36 h at 37°C after infecting with 100 TCID50 IBV for 1 h. The antiviral effect of APL was evaluated by cytopathic observation and the relative mRNA expression levels of the IBV-N gene.
Chicken embryos treatment
Nine-day-old SPF chicken embryos were randomly divided into four groups (n = 13): mock-infected group, IBV-infected group, high-concentration APL group (50 mM), and low-concentration APL group (15 mM). 100 μL of 100 TCID50 virus solution was injected into the allantoic fluid of each chick embryo. After incubation for 1 h, each group was injected with different concentrations of APL. On the third day, chick embryos were dissected and allantoic fluid and chorioallantoic membrane (CAM) were collected. The viral load in allantoic fluid and mRNA level of inflammatory factors in CAM were detected by RT-qPCR. The growth of chicken embryos was observed until the 9th day, and the daily embryo death was recorded. And the growth of embryos in each group was compared. The survival curves were plotted with GraphPad 8.0.
Reverse transcription-quantitative polymerase chain reaction
Primer sequences used for real-time PCR assays to detect gene expression.

Detection of intracellular ROS production
The reactive oxygen species kit (Beyotime, China) was used to detect the changes of reactive oxygen species in cells caused by IBV infection. The apoptotic changes after IBV infection were observed with the Hoechst 33,342 staining solution (Solarbio, China). HD11 cells were cultured in six-well plates. After 1 h of IBV infection in HD11 cells, the supernatant was discarded and washed with PBS buffer. The samples were incubated with 10 μM, 25 μM, 50 μM APL, and 2 mM NAC for 2 h, washed with PBS buffer, and added with 2% maintenance medium for 36 h. After the supernatant was discarded, 1 mL DEFH-DA was added to each well for 20 min at 37°C. The supernatant was discarded, washed three times with DMEM, and then 1 mL Hoechst 33342 was added into each well. After incubation at 37°C for 20 min, the results were observed under the fluorescence microscope.
Statistical analysis
All experiments were independently replicated three times. Statistical analysis was performed using GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA). A p-value was considered significant as the following: *p < .05; **p < .01; ***p < .001; ****p < .0001. Figures were created with Image J software.
Results
Cytotoxicity of APL on HD11 cells
Inverted microscopy was used to observe the changes in the morphology of HD11 cells at different concentrations of APL, and the cytotoxicity of APL was further verified by the MTT method. Under the inverted microscopy, it was observed that none of the HD11 cells produced cytopathic effects (CPE) at concentrations of APL within 50 μM. The morphology of HD11 cells was altered at 75 μM of APL, and a significant CPE was observed at 200 μM (Figure 1(a)). The results of MTT showed that the cell viability was greater than 90% at APL below 50 μM (Figure 1(b)). Consequently, the maximum non-toxic concentration of APL was 50 μM. Cytotoxic effects of APL on HD11 cells. (a) Observed the morphological changes of HD11 cells under an inverted microscope. (b) The MTT colorimetric assay was used to determine cell viability. The survival rate of HD11 cells under APL treatment at different concentrations was above 90% and was considered as the maximum non-toxic concentration of APL. The results are expressed as the mean ± SEM (n = 3, **p < .01, ****p < .0001 by t test).
Therapeutic effect of APL on IBV
To investigate the therapeutic effect of APL on IBV-infected HD11 cells, inverted microscopic observation, plaque assay, and RT-qPCR methods were used. As the time of IBV infection increased, the virus titre increased, reaching a maximum at 36 h (Figure 2(a)). Microscopic observations in the inverted view revealed that as the drug concentration increased, the cell condition improved. In contrast, the group without the added drug exhibited a higher number of dead cells compared to the group with the drug, as illustrated in Figure 2(b). Plaque experiments showed a dose-dependent decrease with increasing drug concentration (Figure 2(c)). With increasing drug concentration, the IBV inhibition rate of the drug increased significantly (Figure 2(d)). The virus inhibition rate is (number of empty spots in the attack group – number of empty spots in the treatment group)/number of empty spots in the attack group × 100%. The RT-qPCR method was employed to assess the mRNA expression level of the IBV-N gene. As illustrated in Figure 2(e), it is evident that the IBV-N gene expression level declined with the rise in drug concentration. Overall, APL has a treatment effect on IBV, and the best treatment results are seen at 50 µM. APL has a treatment effect on IBV-infected HD11 cells. HD11 cells were infected with 100 TCID50 IBV, incubated at 37°C for 1 h, and then treated with different concentrations of APL for 24 h and 36 h. (a) The growth kinetics of IBV Beaudette in HD11 cells following infection at 100 TCID50. (b) The effects of different concentrations of APL on IBV-infected HD11 cells observed by inverted microscopy (c) Inhibitory effect of APL on plaque formation of IBV infection. (d) Inhibition rate of IBV by post-treatment APL. (e) The expression level of IBV-N mRNA was detected with different drug concentrations by RT-qPCR. The results are expressed as the mean ± SEM (n = 3, *p < .1, **p < .01, ***p < .001, ****p < .0001 by t test).
APL inhibits IBV-induced apoptosis and ROS production in vitro
As can be seen in Figure 3(a) and (b), compared to the IBV group, APL was able to significantly reduce the levels of mRNA of apoptosis-mediated Caspase-8, Caspase-9, and Caspase-3, the mitochondrial apoptosis-related pro-apoptotic genes Bid, Bax, and Bak. With an increased dose of APL, the mRNA expression of apoptosis-related genes decreased in a dose-dependent way. APL inhibits IBV infection-induced apoptosis and ROS production in HD11 cells. HD11 cells were infected with 100 TCID50 IBV and incubated at 37°C for 1 h and then treated with different concentrations of APL for 36 h. And the cells were lysed to extract total RNA. (a) The mRNA levels of Bid, Bax, Bak, and Cyt C were assessed by RT-qPCR. (b) The mRNA levels of Caspase-3, Caspase-8, and Caspase-9 were assessed by RT-qPCR. (c) The level of ROS and the degree of apoptosis were observed by fluorescence microscopy. Mock control was used as the negative control, 100 TCID50 IBV as the viral control, and 2 mM NAC as the ROS inhibitor. (d) The Relative Fluorescence Intensity of ROS. The results are expressed as the mean ± SEM (n = 3, ***p < .001, ****p < .0001 by t test).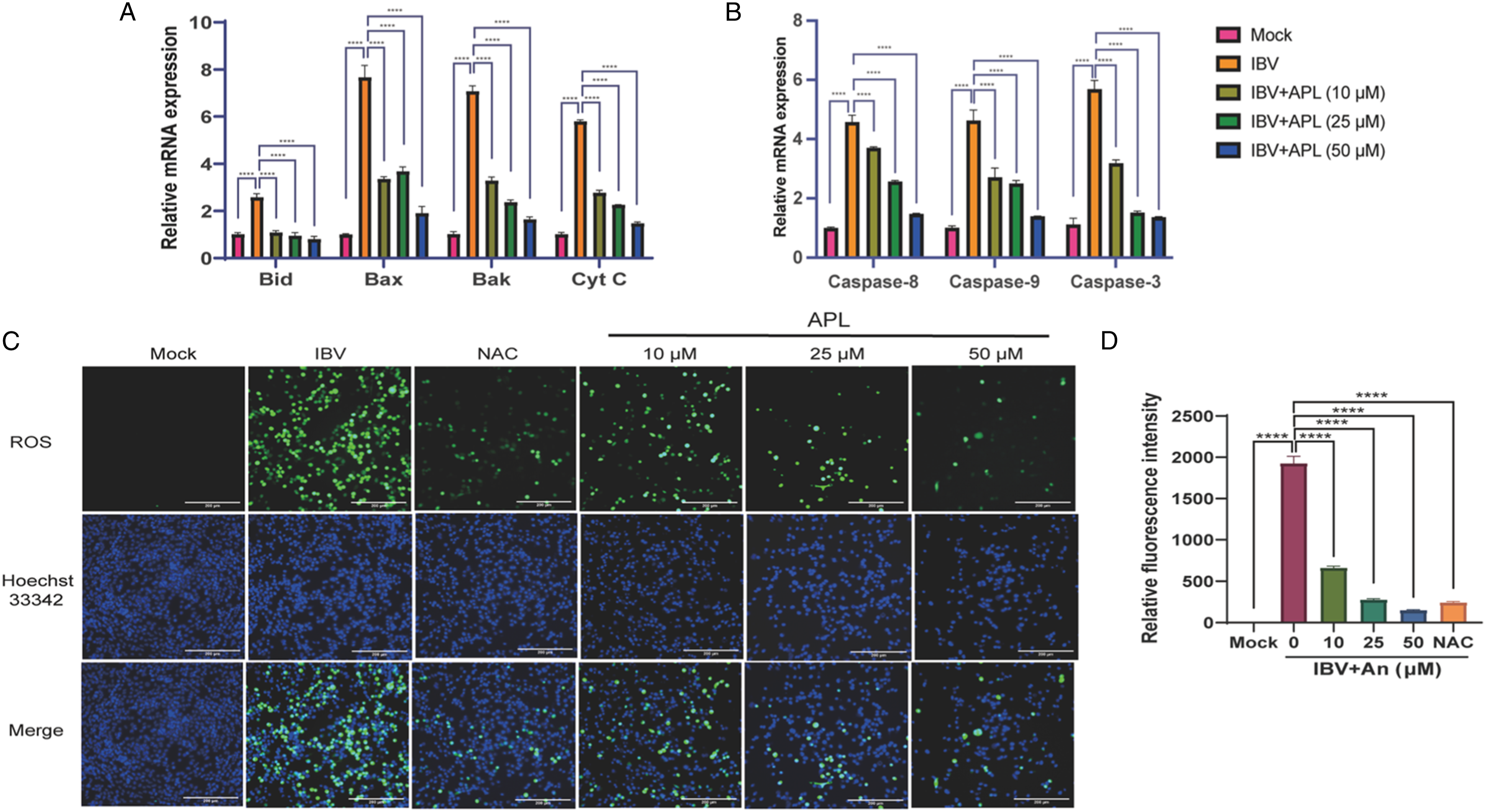
To explore whether APL affects ROS and apoptosis, the ROS changed under different concentrations of APL treatment and were observed under fluorescence microscopy using the DCFH-DA method. As shown in Figure 3(c), APL significantly inhibited ROS production by HD11 cells induced by IBV infection. The fluorescence signal of ROS decreased in a dose-dependent manner as the concentration of APL increased. NAC is a ROS inhibitor. The inhibitory effect of APL treatment on ROS was similar to the level after NAC treatment. Following staining with Hoechst 33342, apoptosis occurred when apoptotic cells were seen with dense staining of the nuclei or fragmented dense staining. Compared with the IBV group, with increasing concentration of APL nuclear staining of apoptotic cells decreased. The relative fluorescence intensity showed that the fluorescence intensity of ROS decreased with the increase in APL concentration (Figure 3(d)). The fluorescence intensity was proportional to the viral infection and inversely proportional to the APL concentration.
APL inhibits IBV-induced pyroptosis and inflammatory factor release
DFNA5 is an important gene in pyroptosis. APL significantly inhibited pyroptosis at 50 µM, and DFNA5 decreased in a dose-dependent manner with increasing drug concentration (Figure 4(a)). Pyroptosis can induce inflammation factor production.
25
To further clarify whether APL can inhibit IBV-induced inflammation in cells, the levels of mRNA expression of inflammatory factors in cells, including NF-κB, NLRP3, iNOS, TNF-α, IL-1β, and IL-6, were detected by RT-qPCR. The results showed that APL dose-dependently significantly inhibited the expression of inflammatory factors (Figure 4(b) and (c)). These results showed that APL could inhibit IBV-induced pyroptosis as well as pyroptosis-induced inflammatory response. APL inhibits IBV-induced pyroptosis and cellular inflammatory responses in vitro. HD11 cells were infected with 100 TCID50 IBV for 1 h after. APL at different concentrations was used to treat HD11 cells for 36 h. Cells were lysed and total RNA was extracted from the cell lysates. (a) The mRNA levels of DFNA5 were assessed by RT-qPCR. (b) The mRNA expression levels of NF-κB, NLRP3, iNOS, and TNF-α were detected by RT-qPCR. (c) The mRNA expression levels of IL-1β and IL-6 were detected by RT-qPCR. The results are expressed as the mean ± SEM (n = 3, *p < .1, **p < .01, ***p < .001, ****p < .0001 by t test).
APL inhibits IBV-induced apoptosis and pyroptosis in chicken embryo
Compared with the IBV group, the mRNA expression levels of apoptosis and pyroptosis-related signaling pathways Caspase-3, Caspase-8, and Caspase-9 were significantly reduced in the APL-treated group (Figure 5(a)). Similarly, the expression of the pyroptosis gene DFNA5 gradually decreased (Figure 5(a)). And the expression level of the pro-apoptotic factor Bax mRNA was dose-dependently reduced, while the expression of the anti-apoptotic gene Bcl-2 was dose-dependently increased (Figure 5(b)). APL inhibits IBV infection-induced apoptosis and pyroptosis in chicken embryo cells. Chicken embryos infected with 100 TCID50 IBV were treated with different concentrations of APL, and the embryonic allantoic fluid and CAM were collected after 72 h. (a) The mRNA expression levels of Caspase-3, Caspase-8, Caspase-9, and DFNA5 in the urinary bladder fluid were detected by RT-PCR. (b) The expression levels of mRNA of pro-apoptotic gene Bax and anti-apoptotic gene Bcl-2 in the allantoic fluid were detected by RT-PCR. The results are expressed as the mean ± SEM (n = 3, *p < .1, **p < .01, ****p < .0001 by t test).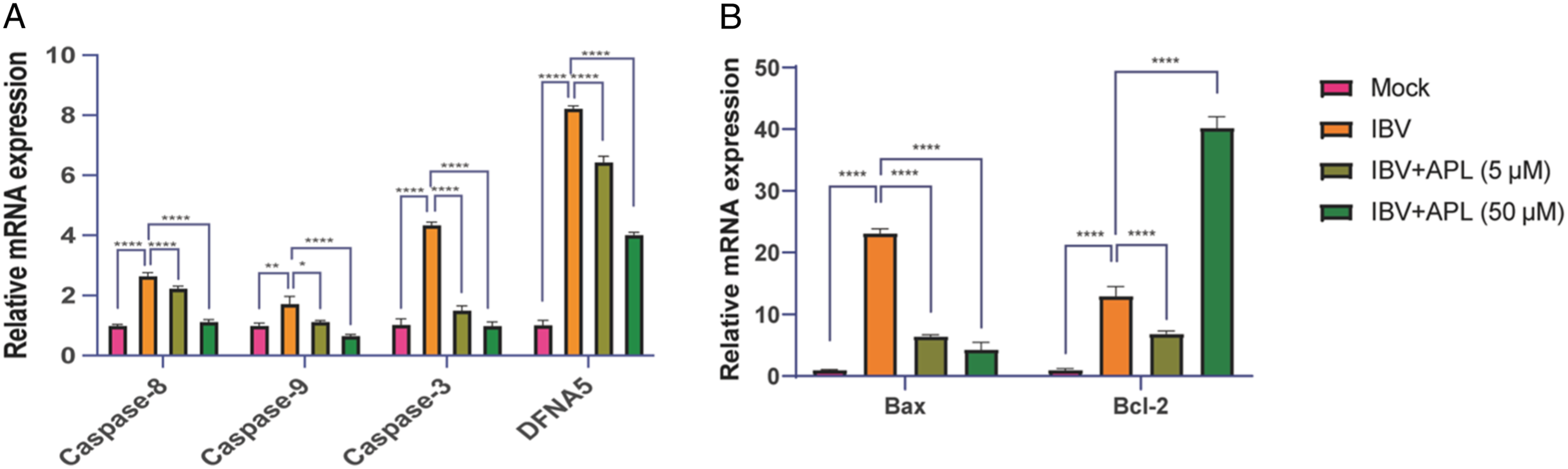
APL has an anti-IBV induced inflammatory response in chicken embryos
The mRNA expression of inflammation-related genes (NF-κB, NLRP3, iNOS, TNF-α, IL-1β, and IL-6) were dose-dependently reduced in chick embryos infected with IBV (Figure 6(a) and (b)). Meanwhile, APL inhibited the mortality of chicken embryos (Figure 6(c)). At 3 d. p.i, the chick embryo survival rate was 0% in the IBV group, at 4 d. p.i, the APL survival rate was 0% in the high dose group, and at the 6th d. p.i, the chick embryo survival rate was 0% in the low dose group. As shown in Figure 6(d), the chicken embryos in the IBV group had obvious inflammatory phenomena, showing reddening and deformation. With the increase in APL dose, the inflammation decreased. It showed that APL could inhibit the inflammatory response of IBV-infected chicken embryos. APL inhibits inflammatory factor release in chick embryos. Chicken embryos were infected with 100 TCID50 IBV and were treated with different concentrations of andrographolide. After 72 h. p.i of infection, chorionic allantoic membrane (CAM) and allantoic fluid were collected. (a-b) The mRNA expression levels of inflammation-related genes in CAM inflammation were detected by RT-qPCR. (c) The survival rate of chicken embryos was recorded each day until the 6th day. (d) The embryo growth and development of each group of embryos on the 6th day. The results are expressed as the mean ± SEM (n = 3, *p < .1, ****p < .0001 by t test).
Discussion
Several studies have shown that the IBV Beaudette strain is susceptible to chicken macrophages and can replicate in HD11 cells in large numbers and produces CPE. 26 Chicken macrophages play a role in virus infection. Therefore, HD11 cells were selected as cells for IBV infection in vitro in this study.
Andrographolide has a variety of biological activities. Some studies have shown that andrographolide has antiviral effects against RNA viruses.27,28 APL has potential anti-coronavirus effects such as SARS-CoV-2. 29 However, there has been no study showing that APL has an inhibitory effect on IBV. In the present study, we investigated the effects of andrographolide on IBV-induced inflammatory response, antiviral response, ROS pathway-mediated apoptosis, and pyroptosis. In addition, the effect of APL was further validated by chicken embryos. These results suggest that andrographolide is a potential anti-IBV drug with a spectrum of antiviral activity.
Apoptosis has an impact on virus infection and replication, as it can eliminate virus-infected cells and terminate virus replication. 30 Also, some virus can use the induction of apoptosis as a way to release and spread the progeny virus. 31 Pyroptosis, a highly inflammatory form of programmed cell death, can be induced by inflammasomes and acts as a defence against pathogenic infection. 32 Pyroptosis is a form of macrophage death that also promotes the release of pro-inflammatory cytokines. 33 IBV infection and replication can induce apoptosis26,34 and inflammatory responses. 35 Some studies show that caspase/granzyme-induced apoptosis can be switched to pyroptosis by the expression of GSDMs.22,36 Reactive oxygen species (ROS) play an essential role in the regulation of various physiological diseases. Infection with viruses causes an increase in intracellular ROS, thereby blocking the body's natural immune signaling pathways. 37
We aimed to investigate the potential of APL to inhibit apoptosis and pyroptosis through the mitochondrial pathway. Our findings revealed that APL had a notable down-regulatory effect on pro-apoptotic cytokines while up-regulating the expression of anti-apoptotic cytokines. A related study has shown that APL attenuates apoptosis-induced by Chikungunya virus.38,39 Furthermore, APL demonstrated a down-regulatory impact on the pyroptosis-associated gene DFNA5. This suggests that APL effectively inhibits apoptosis in vitro, consequently suppressing pyroptosis and the ensuing inflammatory response by preventing apoptosis (Figure 7). Avian infectious bronchitis virus is a coronavirus. Andrographolide inhibits IBV-induced apoptosis, pyroptosis, inflammation, and ROS. APL down-regulated apoptosis-related genes Caspase-3, Caspase-8, Caspase-9, Bax, Bid, and Bak, down-regulated pyroptosis gene DFNA5, and down-regulated inflammation-related genes. Meanwhile, APL reduced ROS release from mitochondria in HD11 cells.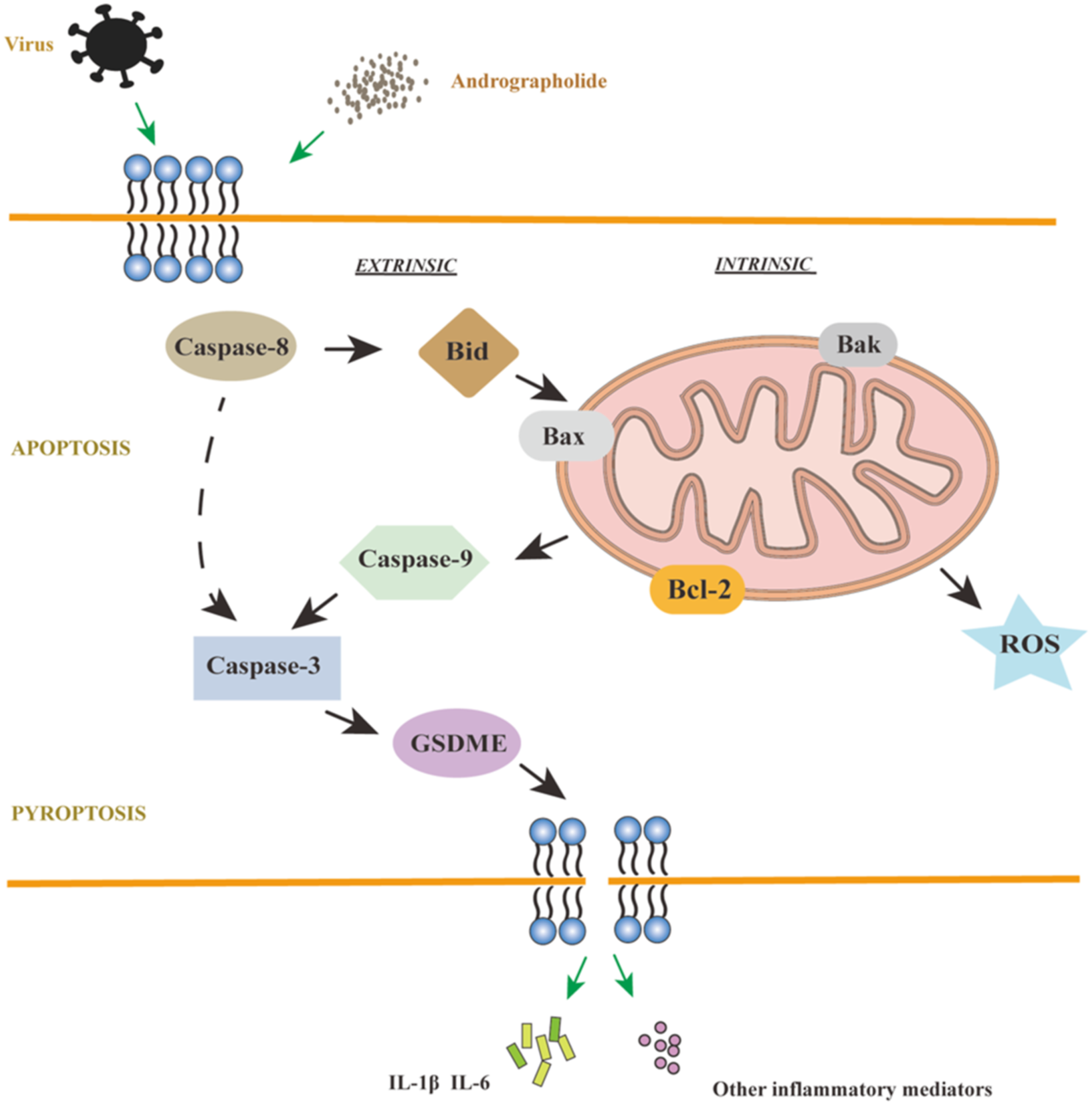
Moreover, our investigation revealed that IBV-induced the production of reactive oxygen species (ROS) in HD11 cells, as evidenced by DCFH-DA staining. Importantly, APL was found to attenuate the levels of ROS. These findings collectively indicate that APL holds promise as an effective therapeutic agent for the treatment of IBV infections, primarily by mitigating apoptosis and, consequently, suppressing pyroptosis and the associated inflammatory response.
In our study, we aimed to investigate the potential clinical application of APL in inhibiting IBV-induced disease, an area that has not been extensively explored. To address this, we conducted experiments using IBV-infected chicken embryos as a model system. We found that APL down-regulates pro-apoptotic and pyroptotic genes and up-regulates anti-apoptotic gene (Bcl-2) mRNA expression levels in chick embryos (Figure 5). IBV-infected chicken embryos have bleeding spots and redness on the surface, and some chicken embryos showed malformation and even died. These pathological symptoms decreased in a dose-dependent manner with increasing APL concentrations, meanwhile, the survival time was increased in the chicken embryos. The results showed that APL could reduce chicken embryo mortality by inhibiting the inflammation caused by IBV infection.
In summary, our study presents promising insights into the potential therapeutic application of APL in mitigating IBV-induced disease in a chicken embryo model. However, viruses can develop resistance to antiviral drugs over time. Continuous monitoring and combination therapies may be necessary to counter this challenge. Meanwhile, further research is needed to elucidate the detailed mechanisms of APL’s action in this context, which could pave the way for the development of effective treatments for IBV infections in poultry.40,41
Footnotes
Authors’ contributions
Software, data curation, experimentation, and writing-original draft preparation, J.S. and Q.X.; J.S.and Q.X. contribute equally; methodology, L.C.; data curation, X.C.; visualization, R.S. and Z.Z.; validation, L.Z. and Y.W.; conceptualization, supervision, writing-reviewing and editing, X.H. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the This study was supported by the National Natural Science Foundation of China (No. 31372485).
Institutional review board statement
Chicken embryo: The animal study protocol was approved by the Institutional Ethics Committee on Beijing Agricultural College Experimental Animal Welfare and Ethics Committee (Approval number: 2022-020B) (Approval Date: March 10th, 2021).
