Abstract
Contemporary antiretroviral therapy (ART) regimens have high barriers to the development of drug resistance. However, resistance to earlier antiretrovirals and uncommon cases of resistance to contemporary ART illustrate the continued need for good clinical management of HIV drug resistance. Here, we describe HIV drug-resistance mechanisms, the interaction of HIV drug-resistant mutations and the patterns of drug resistance to contemporary ART. We then provide guidance on the management of HIV drug resistance, including how to limit the development of resistance and manage virologic failure that is complicated by resistance. To complement this, links to resources and treatment guidelines are provided that can assist with the interpretation of HIV drug resistance test results and optimal ART selection in the clinic.
Introduction
Appropriate clinical assessment and management of HIV drug resistance remain important with contemporary antiretroviral therapy (ART) to ensure people living with HIV (PLWH) can continue to achieve lifelong viral suppression (<50 copies/mL plasma).1,2 Resistance will almost inevitably develop to any effective HIV drug used in isolation but is markedly reduced with the use of multidrug regimens. Ideally, to minimize drug resistance, an ART regimen will contain fully active antiretrovirals, each with a high genetic barrier to resistance. Additionally, the regimen should maintain virologic suppression and enable good treatment adherence. The prevalence of HIV drug resistance has declined in recent years owing to the use of ART regimens that have a higher barrier to resistance than historical regimens.3,4 Despite this, drug resistance to contemporary antiretrovirals has been demonstrated in vitro and is evident, although in small numbers, in clinical trials and real-world settings.5–15 Drug resistance is likely to reduce ART options and so reduce the likelihood of achieving virologic suppression. If virologic failure is not managed appropriately, HIV can become more resistant and lead to deterioration of the immune system and the development of clinical sequelae (AIDS and non-AIDS morbidities), with a continued risk of onward transmission. Therefore, it remains critical for healthcare professionals to understand how HIV drug resistance develops and how it can be prevented and managed.2,16 Here, we provide an overview of HIV-1 drug resistance and its clinical management in middle- and high-income countries.
Understanding HIV drug resistance
Wild-type HIV has evolved to be the most genetically fit version of the virus. However, the very nature of HIV makes it primed to mutate and circumvent drug inhibition.1,17,18 The high replication rate, error-prone reverse transcription and high recombination rate of HIV give rise to a genetically diverse, heterogeneous virus population that is distributed as a quasispecies.1,17,18 In vivo, the HIV mutation rate is estimated to be 4.1 × 10−3 per base per cell.
19
During viral replication, mutations are generated that have the potential to either increase or reduce viral replication capacity.
18
Mutations that counteract ART-mediated inhibition of viral replication are known as drug-resistant mutations (DRMs). DRMs can develop during selective drug pressure (treatment-emergent) or can be acquired during initial HIV exposure (transmitted).1,20 The likelihood of HIV drug resistance developing is increased by certain HIV-1 characteristics, patient-specific factors and the choice of ART (Figure 1).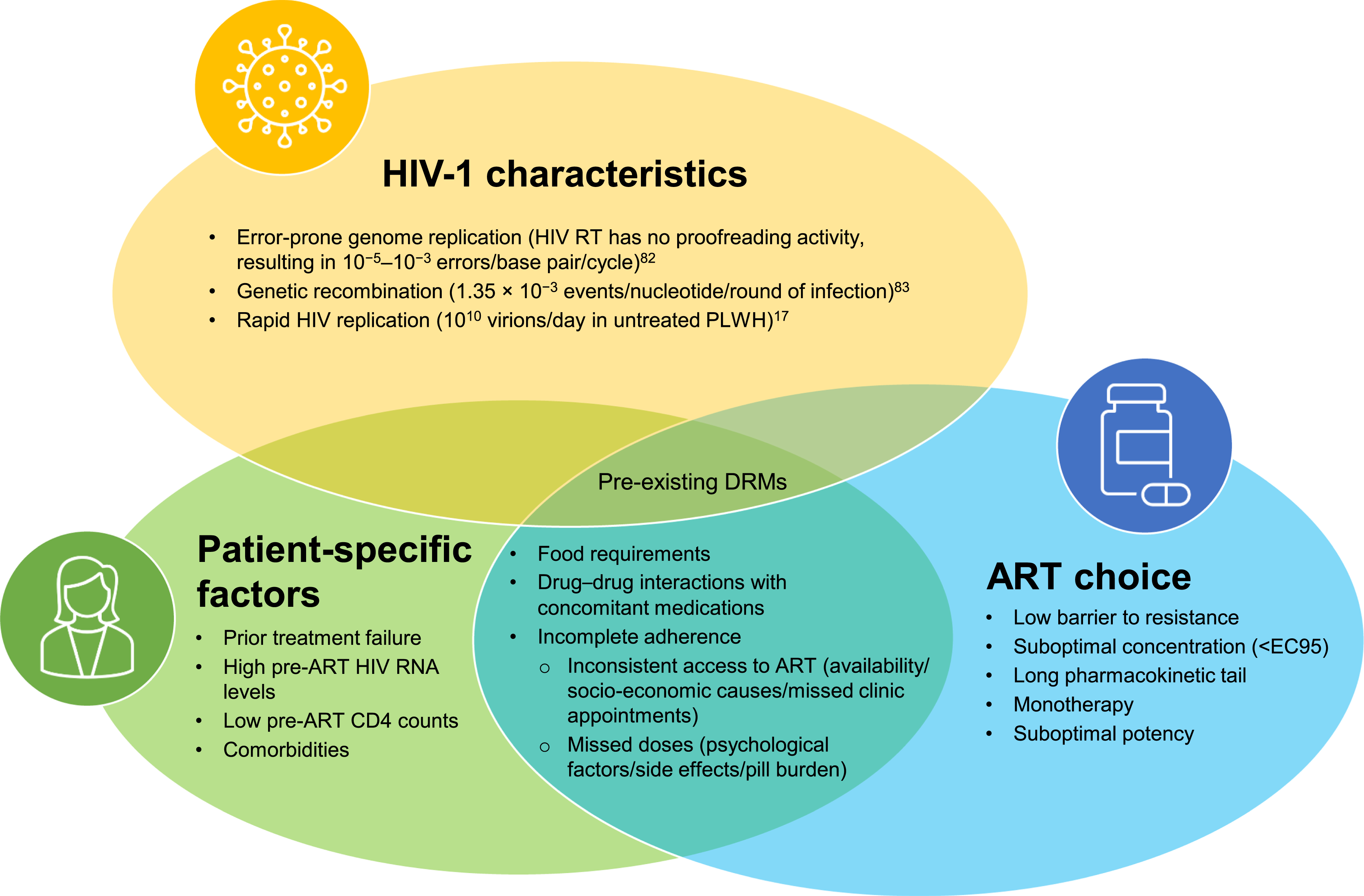
HIV drug classes
Common, clinically significant DRMs within each drug class and their worldwide prevalence. a
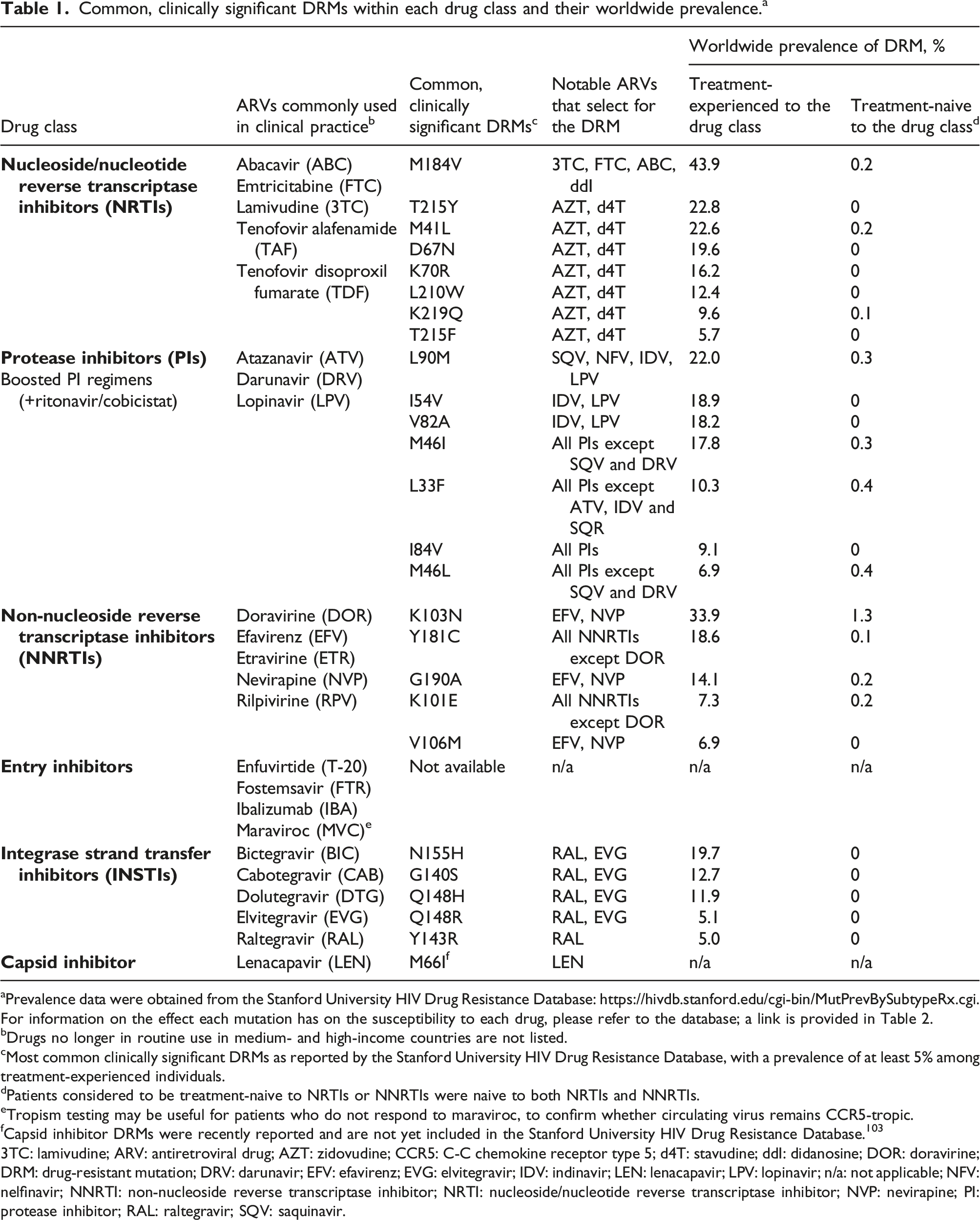
aPrevalence data were obtained from the Stanford University HIV Drug Resistance Database: https://hivdb.stanford.edu/cgi-bin/MutPrevBySubtypeRx.cgi. For information on the effect each mutation has on the susceptibility to each drug, please refer to the database; a link is provided in Table 2.
bDrugs no longer in routine use in medium- and high-income countries are not listed.
cMost common clinically significant DRMs as reported by the Stanford University HIV Drug Resistance Database, with a prevalence of at least 5% among treatment-experienced individuals.
dPatients considered to be treatment-naive to NRTIs or NNRTIs were naive to both NRTIs and NNRTIs.
eTropism testing may be useful for patients who do not respond to maraviroc, to confirm whether circulating virus remains CCR5-tropic.
fCapsid inhibitor DRMs were recently reported and are not yet included in the Stanford University HIV Drug Resistance Database. 103
3TC: lamivudine; ARV: antiretroviral drug; AZT: zidovudine; CCR5: C-C chemokine receptor type 5; d4T: stavudine; ddI: didanosine; DOR: doravirine; DRM: drug-resistant mutation; DRV: darunavir; EFV: efavirenz; EVG: elvitegravir; IDV: indinavir; LEN: lenacapavir; LPV: lopinavir; n/a: not applicable; NFV: nelfinavir; NNRTI: non-nucleoside reverse transcriptase inhibitor; NRTI: nucleoside/nucleotide reverse transcriptase inhibitor; NVP: nevirapine; PI: protease inhibitor; RAL: raltegravir; SQV: saquinavir.
HIV drug resistance mechanisms
Mechanisms of HIV drug resistance are class- or drug-specific, but DRMs primarily prevent the interaction of an antiretroviral drug with its binding site. NRTI and NNRTI DRMs both arise in the HIV reverse transcriptase (RT) gene. NRTI DRMs either reduce the efficiency of NRTI binding to the RT active site or facilitate unblocking of NRTI-terminated primers.17,18,22 For the NNRTIs, DRMs inhibit the interaction or reduce access of an NNRTI to the NNRTI binding pocket in RT.17,18,22 Resistance to PIs occurs via mutations in the HIV protease active site but can be partly modulated by HIV gag polyprotein cleavage site mutations.1,23 Most INSTI DRMs cause conformational changes at the catalytic pocket of HIV integrase, while others occur in the C-terminal domain of integrase. 24 Resistance to INSTIs may also occur from mutations in genes other than integrase, such as the viral 3′ polypurine tract, or through the accumulation of multiple mutations in the envelope glycoprotein gene (Env).25,26 This may explain rare cases of virologic failure in patients who are receiving second-generation INSTIs with no known DRMs in the integrase gene.
Entry inhibitor DRMs occur in the genes of HIV envelope proteins, but mutation sites vary depending on the subclass of drug. DRMs conferring resistance to fusion inhibitors occur in HIV gp41. 27 DRMs in the gp120 envelope protein have been reported following treatment with pre-attachment inhibitors, post-attachment inhibitors and CCR5 antagonists, but these occur at different sites such that DRMs for one subclass are not reported to confer resistance to other entry inhibitor subclasses.28–30 Capsid inhibitor DRMs cause conformational changes in the HIV capsid hexamers, resulting in steric hindrance that impedes the binding of the capsid inhibitor. This prevents capsid inhibitor-mediated stabilization of the viral core and subsequent blocking of viral integration.31,32
DRM prevalence
Key tools and guidelines to support the clinical management of virologic failure and HIV drug resistance.

ANRS-MIE: National Agency for AIDS Research – Emerging Infectious Diseases; BHIVA: British HIV Association; DHHS: Department of Health and Human Services; DRM: drug-resistant mutation; EACS: European AIDS Clinical Society; GeSIDA: Spanish AIDS Study Group.
Effect of DRMs on drug susceptibility and viral fitness
The interaction of all DRMs present in a specific virus population determines the level of drug resistance or susceptibility to a specific drug and/or regimen. Individual mutations confer varying levels of resistance to different drugs within the same class. For instance, the protease mutation I84V strongly reduces susceptibility to atazanavir but only slightly reduces susceptibility to lopinavir and darunavir (DRV). The development of certain DRMs and their effect on viral fitness can also vary between subtypes of HIV-1. DRMs selected for by a particular drug may also make the viral population less susceptible to other drugs within the same class (cross-resistance). 1 For example, the integrase mutations G140S and Q148H/K/R, which may be selected for by raltegravir (RAL) or elvitegravir (EVG), in combination can contribute to resistance against dolutegravir (DTG) and bictegravir (BIC). 34
In contrast to cross-resistance, some DRMs conferring resistance to one drug may cause the virus population to be more susceptible to a different drug within the same class. For instance, the RT mutation M184V elicits high-level resistance to lamivudine (3TC) and emtricitabine (FTC), while increasing susceptibility to zidovudine (AZT) and tenofovir in vitro. 35 This was thought to be a potential mechanism for the sustained efficacy of AZT/3TC combination therapy, but AZT sensitization was shown to be a transient effect of M184V in the clinic.36,37 In some cases, this increased susceptibility can also occur between drug classes. For example, the RT mutation F227C, which is selected for by the NNRTI doravirine (DOR), confers hypersusceptibility to the investigational nucleoside RT translocation inhibitor islatravir in vitro. 38
Mutations that confer relatively high-level drug resistance (primary DRMs) often also cause a reduction in viral fitness. 23 Accessory or secondary DRMs can act synergistically to generate greater and broader resistance and/or compensate for the loss of viral fitness incurred from a primary DRM. 1 Primary DRMs usually precede secondary DRMs.
Many DRMs impair viral fitness, so reversion of a viral population with DRMs to wild type can occur upon removal of the selective drug pressure. 39 Reversion can arise owing to the activation of archived wild-type virus from latently infected cells, which rapidly outcompetes the mutated virus. In cases of transmitted drug resistance, where there is an absence of archived wild-type virus, reversion occurs more slowly through back mutation of the mutated strain. 40 The rate of reversion may vary depending on: the number of back mutations required; the effect on viral replication capacity from the individual DRM; and the relative fitness of the mutant and back-mutated viruses (which can be affected by secondary DRMs). 39 In addition, the evolutionary route to reversion to wild type can be blocked in mutants that have multiple mutations and a reduced replication capacity via a mechanism known as compensatory fixation. For example, prolonged persistence (up to 4 years) of multiple PI DRMs has been observed in patients after PI discontinuation and is likely attributed to subsequent secondary mutations that have already partly compensated for the initial loss in replication capacity. 41
Despite the viral population potentially reverting to wild type upon removal of selective drug pressure, DRMs are lifelong once they are present in a person living with HIV because HIV integrates into the host cell genome. 42 DRMs can be archived as proviral DNA in a person’s cellular DNA and may re-emerge in the circulating viral population upon reintroduction of the selective drug pressure. 42 When constructing a new regimen for a person who has experienced virologic failure, a complete treatment history, the reasons for virologic failure and the results of previous resistance tests should be taken into account, whenever possible. 42 However, if a complete treatment history is unavailable, then a cautious choice of antiretroviral regimen may be the optimal approach, taking into consideration the barrier to resistance of the new regimen and possible resistance mutations that were selected for in previous treatment failures. In this context, a proviral DNA genotypic assay could be considered, particularly if resistance is suspected, because it may be able to provide information on archived viral resistance. While the use of such assays has increased in recent years, they have not been validated for clinical management in a randomized trial.42,43
Genetic barrier to resistance
The distinct genetic barrier to resistance of an antiretroviral drug or regimen refers to the number and type of viral mutations required to confer resistance. The genetic barrier to resistance can be increased by using multiple potent drugs in a treatment regimen, which will likely contain drugs from different drug classes. 2 A multidrug regimen is preferable to monotherapy for suppressing the development of drug resistance because any effective regimen will likely require multiple primary DRMs in a single virion to result in the emergence of resistance.
The barrier to resistance is also determined by drug pharmacokinetics, such as the rate of dissociation from the target viral protein and the rate of elimination, because these can affect the maintenance of therapeutic drug concentration levels.44,45 For instance, DTG and BIC have a longer dissociation rate from HIV-1 integrase DNA complexes than either EVG or RAL, which may contribute to their higher barrier to resistance. 45 Cabotegravir (CAB) has a long elimination half-life but a potentially lower barrier to resistance, as demonstrated by the rates of resistance seen in the small numbers of individuals who experience virological failure in clinical trials. 46 In the case of boosted regimens, the booster can increase gut absorption of the PI and inhibit its metabolic breakdown in the liver, thereby maintaining therapeutic concentrations. 47 For this reason, boosted PIs have higher potency and a higher genetic barrier to resistance than unboosted PIs.10,44 Drugs with a long pharmacokinetic tail can lead to suboptimal levels in the body after discontinuation; therefore, if HIV is not subsequently suppressed, there is a risk of DRMs developing. 48 In most regimens, the antiretrovirals will have different length pharmacokinetic tails. Therefore, the pharmacokinetic properties of each drug in a regimen must be considered if ART has been stopped or needs to be stopped for any reason.
Regimens containing the second-generation INSTIs DTG or BIC, and boosted PI regimens, such as DRV with ritonavir or cobicistat, have a high genetic barrier to resistance. In general, NRTIs have a low-to-medium barrier to resistance; however, they can form part of the backbone of three-drug regimens with much higher barriers to resistance.1,44 The first-generation NNRTIs nevirapine and efavirenz have a low barrier to resistance, whereby just one DRM can be sufficient to significantly reduce HIV susceptibility. 44 The second-generation NNRTIs etravirine and rilpivirine have slightly higher barriers to resistance due in part to higher conformational flexibility.17,44 The more recently approved NNRTI DOR has a distinct resistance profile among its class.49,50 For example, the RT mutation K101P shows a high level of cross-resistance among NNRTIs, but does not reduce susceptibility to DOR. 51 Nonetheless, the long pharmacokinetic tail of the NNRTI class, in particular that of efavirenz, means that DRMs may develop in situations of poor adherence or unplanned treatment interruption. 52
Patterns of drug resistance to contemporary ART
Resistance to second-generation INSTI BIC- and DTG-based regimens is rare in clinical trials and real-world studies enrolling patients who are either treatment-naive or treatment-experienced and virologically suppressed.3,8,13,15,53–60 From such studies and in vitro passage studies using clinical isolates, multiple synergetic DRMs were generally shown to be required before high-level resistance can be generated against BIC or DTG. For instance, Q148H/K/R confers resistance to RAL, EVG and CAB, but alone has a minimal effect on BIC or DTG activity.5,8,14,15 However, Q148H/K/R together with E138K/A or G140S/A/C reduces RAL and EVG susceptibility by more than 100-fold, CAB susceptibility by 10-fold, BIC susceptibility by 2- to 5-fold and DTG susceptibility by 2- to 10-fold, with resistance increasing further still with additional mutations such as N155H.15,34 In the small number of PLWH who develop virologic failure while receiving DTG, R263K is commonly seen, and this mutation appears to reduce susceptibility to DTG, BIC and CAB.15,34 Additionally, the presence of NRTI DRMs and the use of DTG monotherapy were each associated with an increased risk of resistance to DTG compared with no NRTI DRMs and the use of at least a three-drug regimen, respectively, in a pooled cohort study. 61 CAB has a higher barrier to resistance than EVG or RAL in vitro; however, its barrier to resistance is lower than that of BIC or DTG. 14 In rare cases of virologic failure in clinical trials using CAB and rilpivirine, the failure was usually associated with emergent integrase and NNRTI resistance mutations.14,62,63
PI resistance is uncommon and slow to develop in previously PI-naive people with virologic failure receiving a regimen containing boosted PIs (atazanavir, DRV or lopinavir). 64 There are only very rare cases of treatment-emergent resistance in clinical trials of boosted DRV-based regimens in treatment-naive and virologically suppressed treatment-experienced PLWH, with slightly more cases in individuals who were treatment-experienced and had active viral replication at study entry. 12 Virologic failure while receiving DRV-based regimens usually requires multiple PI DRMs and DRV-specific DRMs, such as V32I, I84V, I54L/M, I50V and L76V. 64
Compared with INSTIs and PIs, treatment-emergent resistance is more common for NRTIs, which are frequently used as the backbone of contemporary treatment regimens. M184V alone and M184V in combination with K65R are the two most frequent NRTI mutation patterns that emerge in patients experiencing virologic failure while receiving a tenofovir-containing first-line regimen.65,66 Interestingly, K65R is detected approximately twice as frequently in patients with subtype C HIV-1 as in those with other viral subtypes. 67 M184V is selected for by 3TC and FTC, reducing susceptibility to both drugs by more than 100-fold in vitro, while also reducing viral replication rates and RT enzyme processivity in vitro.35,68 K65R reduces abacavir (ABC) and tenofovir susceptibility by 2-fold, while 3TC and FTC susceptibility decreases by 5- to 10-fold. 35 Thymidine analogue mutations (TAMs), selected for by AZT and stavudine, may need to be considered in treatment-experienced patients. Type 1 TAMs (M41L, L210W and T215Y) induce a greater reduction in susceptibility to ABC- or tenofovir disoproxil fumarate-containing regimens than type 2 TAMs (D67N, K70R, T215F and K219Q). 35
DOR has a distinct resistance profile among the NNRTIs and is included as an alternative first-line regimen in the European AIDS Clinical Society and the Department of Health and Human Services guidelines in certain clinical situations.2,69 In clinical trials, treatment-emergent resistance to DOR was uncommon among virologically suppressed and treatment-naive PLWH. 49 However, DRMs associated with high resistance to DOR in vitro include Y188L (50–100-fold resistance) and V106A (10-fold resistance).49,51
Multi-class drug resistance
While not formally defined, multi-class drug resistance (MDR) typically equates to reduced susceptibility to multiple antiretroviral drug classes. The prevalence of MDR has been falling since the availability of contemporary ART, which is more potent and tolerable, often with a lower pill burden, than historical regimens. In a large cohort study in Switzerland, the estimated prevalence of three-class drug resistance halved between 1999 and 2013, while a study of 2643 patients across Europe reported that four-class drug resistance declined from 5.6% in 2008 to 2.4% in 2018.70,71
Treatment options can be severely limited when an individual develops NRTI, NNRTI, PI and/or INSTI MDR. 72 Alternative agents that may enable the selection of fully suppressive regimens in those with MDR HIV include the HIV entry inhibitors fostemsavir and ibalizumab. In the BRIGHTE trial, 53% and 60% of 272 PLWH who enrolled with MDR and received fostemsavir plus optimized background regimen (OBR), achieved virologic suppression (measured in this study as <40 copies/mL) after 24 and 96 weeks, respectively.73,74 Susceptibility to fostemsavir in some patients in clinical trials has been reduced by treatment emergence of DRMs associated with HIV-1 gp120, such as M426L or M434I.75,76 In the TMB-301 trial, 43% of 40 PLWH who enrolled with MDR and received ibalizumab plus OBR achieved virologic suppression. 77 Treatment-emergent resistance to ibalizumab can be associated with the loss of predicted N-linked glycosylation sites in the V5 loop of HIV-1 gp120. 77 Although ibalizumab is approved by the US Food and Drug Administration, European Medicines Agency marketing authorization was recently withdrawn after the European commercialization rights holder ceased operations in Europe.78,79
Also for consideration for PLWH with MDR is lenacapavir, a first-in-class capsid inhibitor, which was recently approved for use with an OBR for PLWH with MDR. The approval followed results from the CAPELLA trial; 81% and 83% of 36 PLWH who enrolled with MDR and received lenacapavir plus OBR achieved virologic suppression after 26 and 52 weeks, respectively. 80 DRMs associated with virologic failure, such as M66I and Q67H, have developed in people receiving lenacapavir in clinical trials. 81
Clinical management of HIV drug resistance
Here, we provide a brief overview of the clinical management of virologic failure and the development of HIV drug resistance, from testing through to regimen selection. Further details can be found in the relevant guidelines (Table 2). These guidelines provide recommendations for the treatment of HIV in a range of different clinical scenarios, including treatment selection for babies/children, pregnant individuals, the elderly and those with certain comorbidities, including tuberculosis, hepatitis C infection, chronic kidney disease and liver disease.2,69,82
When and how to assess HIV drug resistance
HIV drug resistance test results inform treatment decisions and ensure the selection of an optimum ART regimen. Although there are several different tests for HIV drug resistance (Supplementary Table 1), the most commonly used test in clinical practice, which is recommended in treatment guidelines, is genotypic resistance testing of viral RNA.2,69,82 Genotypic assays usually require a minimum plasma viral load of at least 500–1000 copies/mL. Sequences from genotypic testing can sometimes be obtained from PLWH with viral loads of 50–500 copies/mL; however, the chances of viral amplification decrease as the viral load decreases. 83 Genotypic assays do not reliably detect resistant viruses that constitute less than 5%–20% of the circulating virus population.69,84,85 Therefore, circulating mutant virus populations not under current selective drug pressure may be present at sufficiently low concentrations that they are not detected by standard genotypic assays of viral RNA. To overcome this, next-generation sequencing (or ultra-deep sequencing) with sensitivity thresholds as low as 1% may be beneficial, although this has not been confirmed in a randomized trial.86,87 Additionally, care must be taken when interpreting genotypic assays, as the APOBEC3-mediated cellular defence mechanism has been shown in vitro to induce M184I and E138K mutations in the HIV RT in the absence of selective drug pressure. 88
HIV drug resistance tests should ideally be performed at the time of HIV diagnosis or, if this is not possible, before initiation of ART.2,69 Treatment need not be delayed while waiting for resistance test results but can be optimized if necessary once results are available. 69 In addition, resistance testing should be performed in patients who are receiving antiretrovirals and have confirmed viral rebound.2,69 Resistance testing in those with virologic failure is ideally performed while the individual is still on treatment or in the 4 weeks after stopping therapy because reversion of the circulating HIV population to wild type can occur in the absence of drug-selective pressure, and previously selected resistance mutations can be missed. 69 Experience in the clinic from the authors has highlighted occasional cases of PI or INSTI resistance without the presence of NRTI mutations, suggesting a possible reversion of NRTI DRMs. Where possible, resistance testing of cerebrospinal fluid should also be considered when patients present with neurological symptoms because, in rare cases, central nervous system-specific ART resistance can develop, while plasma HIV RNA remains undetectable. In such cases, resistance test results from cerebrospinal fluid HIV RNA can guide the modification of ART regimens to reduce central nervous system viremia and improve neurological symptoms. 89
A new drug resistance test should always be interpreted alongside the complete treatment history and results of prior resistance tests to assess cumulative resistance, particularly if the most recent genotype has been performed in a patient who is no longer on ART.
42
Resources such as the Stanford University HIV Drug Resistance Database: Mutations Analysis tool can be used to help with the interpretation of complex results. The user inputs multiple mutations or sequences, and the programme provides detailed information about the inferred susceptibility to commonly used antiretroviral drugs (Figure 2). Other mutation analysis tools are available (Table 2). Worked example of using the Stanford University HIV Drug Resistance Database: Mutations Analysis tool to support ART selection for a patient who has experienced virologic failure on their current regimen. Based on an anonymized patient case. 3TC: lamivudine; ABC: abacavir; ART: antiretroviral therapy; AZT: zidovudine; BIC: bictegravir; D4T: stavudine; ddl: didanosine; DOR: doravirine; DRM: drug-resistant mutation; DRV: darunavir; EFV: efavirenz; ETR: etravirine; FTC: emtricitabine; HIVdb: Stanford University HIV Drug Resistance Database; INSTI: integrase strand transfer inhibitor; NNRTI: non-nucleoside reverse transcriptase inhibitor; NRTI: nucleoside/nucleotide reverse transcriptase inhibitor; NVP: nevirapine; PI: protease inhibitor; RPV: rilpivirine; RT: reverse transcriptase; TAF: tenofovir alafenamide; TDF: tenofovir disoproxil fumarate.
Strategies to minimize the development of HIV drug resistance in PLWH
To reduce the chances of drug resistance developing in PLWH, selection of the optimal available ART regimen, ideally individualized for each patient, is fundamental. In addition to considering patient factors, this selection should take into consideration any current and possible archived drug resistance. Treatment guidelines are available to help clinicians to identify the most appropriate ART for their patients (Table 2). Treatment should be well tolerated and minimally burdensome, such as single-pill regimens that can be taken with or without food, while also being potent enough to achieve virologic suppression.2,90 Additionally, potential drug–drug interactions should be considered and can be checked using the freely available online tool, the Liverpool Drug–Drug Interaction Checker. 91
Early detection of HIV and rapid start of ART in the clinic is important for lowering the risks of virological failure and development of drug resistance. 92 In the RESPOND cohort study of patients with HIV who were treatment-naive and subsequently initiated ART, low baseline CD4 cell counts were associated with a higher risk of virological failure than high baseline CD4 cell counts. 93 Similar results are reported for those starting a first-line INSTI-based regimen, highlighting that early intervention is still important in the era of contemporary ART regimens. 94 Strategies to increase access to testing for HIV should be implemented to improve early detection of HIV and therefore decrease the risk of drug resistance development. 95
It is important to educate PLWH on the importance of good adherence in reducing the risk of virologic failure and development of drug resistance at all stages of the HIV care continuum, and to support them in achieving this goal. 2 The selection of fixed-dose combinations and once-daily regimens can reduce pill burden and improve adherence. 96 Considerations for clinics would be adherence support, family- and peer-support groups, dedicated adolescent HIV clinics, use of reminder systems (text, phone and apps) to encourage appointment keeping, and directly observed therapy.2,69,97 The Undetectable = Untransmittable (U=U) campaign and health literacy support can highlight to PLWH the importance and benefits of good adherence to treatment. 98 Other strategies that have shown promise in improving adherence include motivational interviewing and financial incentives. 69 Finally, it is important to maximize initial engagement and long-term retention in care.
Management of virologic failure with suspected resistance
When PLWH who are receiving ART present with virologic failure, we recommend several key actions that can be conducted in parallel (Figure 3). It is important to repeat viral load testing using a fresh sample within 2–4 weeks to confirm virologic failure. To investigate the presence of DRMs, a new RNA genotypic resistance test should be conducted while the patient is still receiving the failing regimen or in the 4 weeks after discontinuation if possible (see the section: When and how to assess HIV drug resistance).
69
It is also important to rule out drug–drug interactions, including other medications and recreational drugs that may be affecting the absorption, clearance or potency of an ART regimen.2,90 Treatment history and all available resistance reports should be reviewed together when selecting a new ART regimen to identify any possible resistance not indicated by the current genotype resistance test.2,42 An HIV tropism test can be informative because it allows for the potential inclusion of tropism-specific treatments in a new regimen. For example, if the circulating virus was CCR5-tropic, the CCR5 antagonist maraviroc could be used.99,100 Actions to be taken upon virologic failure with suspected resistance.
Understanding the factors that contributed to treatment failure for a particular individual is also critical to ensuring future treatment success (Figure 1). These factors may include pill attributes, socio-economic factors, partner and peer support, education and healthcare costs. Specifically, low adherence may be related to treatment-emergent adverse events, pill burden, fear of disclosure, medication costs or lack of suitable health insurance, substance abuse disorders, concurrent mental illness, food insecurity, transportation costs, housing insecurity and economic hardship.2,90 PLWH may also require additional psychological support and other services beyond identifying a suitable regimen, such as adherence counselling or addiction services.2,90
Once virologic failure is confirmed and ideally after a resistance test is available, the regimen should be switched as soon as possible to avoid accumulation of resistance mutations. Guidance for the selection of specific ART regimens following virologic failure with resistance are provided in treatment guidelines (Table 2), including recommendations for specific pathways of resistance. For individuals with restricted options available, discussion with a multidisciplinary team or ‘virtual clinic’ is recommended. At least two and preferably three active drugs should be selected, which may include agents with novel mechanisms, as described previously. If appropriate, individuals could be enrolled in a clinical trial or referred to compassionate access programmes.
Conclusions
In the era of potent and tolerable first-line ART with a high genetic barrier to resistance, emergent resistance is now relatively rare. However, a comprehensive understanding of HIV drug resistance and how it can be managed in the clinical setting is still relevant to ensure ongoing treatment success. Selection of an ART regimen with a high barrier to resistance is a key step in preventing the development of drug resistance, but other factors, such as choosing simple, well-tolerated therapy, supporting adherence and monitoring drug–drug interactions, are equally important. Once DRMs have developed, they are lifelong and may limit treatment options within classes. Nonetheless, there are valuable resources available to support the clinical management of HIV drug resistance. Understanding the reasons for virologic failure is essential to minimize further development of resistance and to ensure the success of new therapies. Ultimately, the prevention and management of HIV drug resistance are crucial for maintaining the efficacy of contemporary antiretrovirals.
Supplemental Material
Supplemental Material - HIV drug resistance in the era of contemporary antiretroviral therapy: A clinical perspective
Supplemental Material for HIV drug resistance in the era of contemporary antiretroviral therapy: A clinical perspective by Andrew Carr, Nicola E Mackie, Roger Paredes and Kiat Ruxrungtham.
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Andrew Carr has received research funding from MSD and ViiV Healthcare; lecture and travel sponsorships from Gilead Sciences and ViiV Healthcare; and has served on advisory boards for Gilead Sciences, MSD and ViiV Healthcare. Nicola E Mackie has received fees for talks and advisory boards from Gilead Sciences, MSD and ViiV Healthcare. Roger Paredes has served on advisory boards related to HIV and/or COVID-19 therapeutics for Atea Pharmaceuticals, Gilead, GSK, Lilly, MSD, Pfizer, Roche, Theratechnologies and ViiV Healthcare, and has received research support from Gilead, MSD and ViiH Healthcare, awarded to his institution. Kiat Ruxurngtham has served as a consultant for Merck, Mylan and ViiV Healthcare, and has had paid speaking engagements with Bristol Myers Squibb, GSK, Janssen-Cilag, Merck, Mylan Lab Limited, Roche, Thai Government Pharmaceutical Organization and ViiV Healthcare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Medical writing support was funded by Gilead Sciences, Inc., Foster City, CA, USA. Gilead Sciences had no influence on the content of the manuscript or the decision to submit to Antiviral Therapy.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
