Abstract
Background
Lenacapavir (LEN) is a first-in-class inhibitor of human immunodeficiency virus type 1 (HIV-1) capsid function for the treatment of heavily treatment-experienced people with HIV (PWH) harbouring multidrug resistance in combination with an optimized background regimen (OBR). Here, we describe in vitro analysis of the interplay between entry inhibitors (EI; enfuvirtide, fostemsavir, ibalizumab, and maraviroc) susceptibility and LEN susceptibility in samples from 72 participants in the phase 2/3 CAPELLA study, as well as the emergence of resistance in CAPELLA through 52 weeks.
Methods
The phenotypic susceptibility to EIs of screening samples from participants was analysed using entry assays, and susceptibility to LEN was generated. Genotypic and phenotypic resistance to LEN was evaluated for subjects with virological failure through Week 52.
Results
Overall, viruses with resistance to EIs showed no cross-resistance to LEN, with a mean fold change from wild type close to 1.0. Of the 22 participants analysed for resistance through Week 52, 9 participants (13%) had emergence of capsid resistance mutation(s) while the remaining 13 participants (18%) had no change in the capsid sequence.
Conclusion
The gag sequence from EI-resistant isolates did not affect LEN susceptibility. The lack of cross-resistance to LEN across ARV-resistant isolates supports the use of LEN in PWH regardless of their treatment history. During the second half-year period of the CAPELLA Study, development of LEN resistance was rare and was overall associated with functional LEN monotherapy due to either nonadherence or resistance-driven non-susceptibility to OBR.
Keywords
Introduction
Lenacapavir (LEN) is a novel antiretroviral (ARV) agent that inhibits HIV-1 replication by interfering with the essential functions of HIV-1 capsid (CA) at multiple stages of the viral lifecycle. 1 LEN is formulated as a solution administered subcutaneously every 6 months and was approved in multiple geographies (including the United States, Europe, Canada, and Japan) for the treatment of HIV-1 infection in combination with other ARVs in people with HIV (PWH) with multidrug resistance (MDR). Adding LEN to an optimized background regimen (OBR) in chronically viraemic PWH with MDR led to >80% virological suppression in the CAPELLA study (NCT04150068) after 26 weeks of treatment, 2 which was maintained through 52 weeks of treatment. 3 This study demonstrated the critical impact of LEN in this difficult-to-treat population of PWH with MDR who had limited to no options to achieve viral suppression.
Per protocol, participants in CAPELLA had to show evidence of resistance to ≥3 of the 4 main ARV classes with resistance to at least 2 agents per class. As a result, 46% of the participants in the study had resistance to the 4 main ARV drug classes with 1/3 of them having exhausted all drugs from ≥3 of the 4 main classes. 4 The OBR of participants also included drugs inhibiting virus entry, including the HIV-1 fusion inhibitor synthetic peptide enfuvirtide (ENF), the HIV-1 attachment inhibitor fostemsavir (FTR, prodrug of temsavir), the CD4 monoclonal antibody ibalizumab (IBA), and the C-C chemokine receptor type 5 (CCR5) antagonist maraviroc (MVC), 5 which were used in 7%, 11%, 24%, and 14% of the OBRs, respectively. 3 Resistance or lack of susceptibility to these drugs was frequent at baseline (found in 67%, 31%, 29%, and 9%, for MVC, FTR, IBA, and ENF, respectively). 3
Resistance to LEN in CAPELLA was associated with the emergence of CA mutations M66I, Q67H/N, K70H/N/R/S, N74D/H, A105T, and T107N/A/C, which were observed in 8 participants in the Study through 26 weeks of treatment. 4
Here, we describe the emergence of LEN resistance through 52 weeks of LEN treatment in the CAPELLA Study. Additionally, we provide phenotypic analyses characterizing the lack of cross-resistance to LEN in isolates harbouring resistance or lack of susceptibility to entry inhibitors (EIs).
Materials and methods
Phenotypic susceptibility to HIV-1 EIs
The phenotypic susceptibility of screening samples from enrolled participants to EIs (ENF, FTR, IBA, and MVC) was analysed using a combination of the Trofile® and PhenoSense® entry assays (Monogram Biosciences Inc., South San Francisco, USA). These assays measure the co-receptor tropism and the susceptibility to the inhibitors, respectively, based on the HIV-1 envelope from the participant’s sample. Co-receptor tropism of X4 (CXCR4) or mix of X4 and R5 (CCR5) indicated a lack of susceptibility to MVC, while R5 tropism indicated susceptibility to MVC. 6 Susceptibility to ENF was a direct result of the assay with values either ‘sensitive’ or ‘reduced susceptibility’. Susceptibility to FTR was based on the EC50 value from the PhenoSense® entry assay, where samples with EC50 <100 nM were sensitive to FTR and samples with EC50 ≥100 nM were resistant to FTR. 7 Susceptibility to IBA was based on percent maximal inhibition (PMI) in the PhenoSense® entry assay, where samples with PMI 83% were resistant and samples with PMI >83% were sensitive. 8
Resistance analysis at Week 52
As previously described, 4 participants experiencing virological rebound (HIV-1 RNA 50 copies/mL at two consecutive visits after achieving HIV-1 RNA <50 copies/mL, or two consecutive visits with HIV-1 RNA 1 log10 above nadir) or with HIV-1 RNA ≥50 copies/mL at Week 52 were analysed for resistance development in capsid, protease/RT, and integrase using resistance assays from Monogram Biosciences (GenoSure Gag-Pro, PhenoSenseGT, and GenSeq/PhenoSense integrase, respectively). For a few samples, genotyping of CA was also conducted at Seq-IT GmbH & Co. KG (Kaiserslautern, Germany).
Results
Resistance to EIs and LEN cross-resistance with EIs
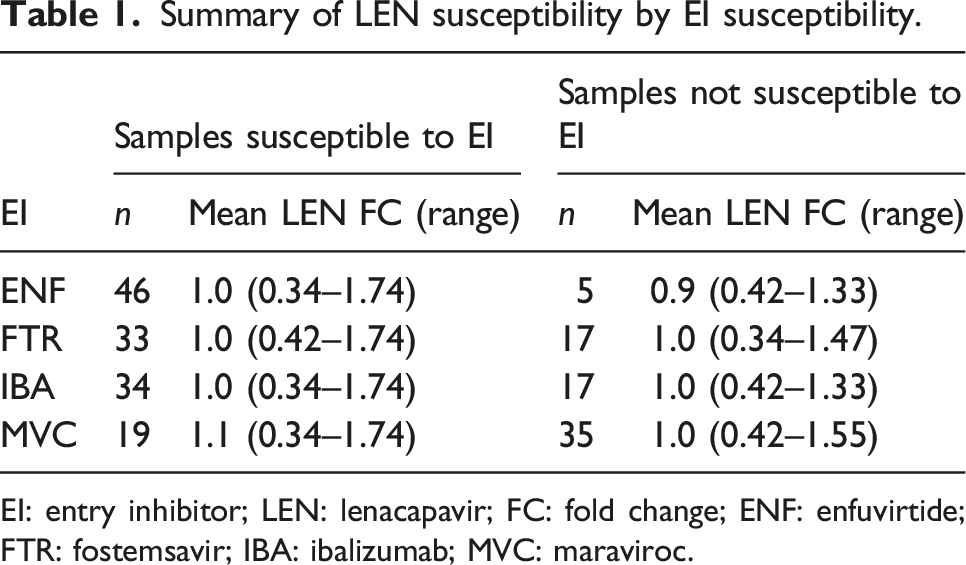
As part of the baseline analysis, viral samples from the 72 participants in CAPELLA were phenotypically evaluated against LEN, resulting in data for 62 participants. Out of these 62 samples, 17 were resistant to FTR, 17 were resistant to IBA, 41 were not susceptible to MVC, and 5 were resistant to ENF (Figure 1, Table 1, Table S1). Overall, viruses with resistance to ENF, FTR, IBA, and MVC showed no cross-resistance to LEN, with a mean fold change (FC) from wild type (WT) of 0.92, 0.97, 1.0, and 0.97, respectively (Table 1), indicating that the gag sequence from EI non-susceptible isolates did not affect LEN activity. Baseline susceptibility to LEN by EI susceptibility. The Trofile® and PhenoSense® entry assays (Monogram Biosciences, Inc.) were used to determine the co-receptor tropism and the susceptibility to the respective entry inhibitor based on the participant’s HIV envelope. (a) Susceptibility to enfuvirtide (ENF) was a direct output of the assay, with ‘susceptible’ corresponding to an overall susceptibility score (OSS) of 1 and ‘reduced susceptibility’ corresponding to an OSS of 0. (b) Susceptibility to fostemsavir (FTR) was based on the EC50 value from the PhenoSense® entry assay, with EC50 < 100 nM corresponding to an OSS of 1 and EC50 ≥ 100 nM corresponding to an OSS of 0. (c) Susceptibility to ibalizumab (IBA) was based on percent maximal inhibition (PMI) in the PhenoSense® entry assay, with PMI >83% corresponding to an OSS of 1 and PMI ≤83% corresponding to an OSS of 0. (d) Susceptibility to maraviroc (MVC) was determined from the Trofile® co-receptor tropism assay, with ‘R5’ corresponding to an OSS of 1 and ‘X4’ or ‘dual mix’ corresponding to an OSS of 0. Summary of LEN susceptibility by EI susceptibility. EI: entry inhibitor; LEN: lenacapavir; FC: fold change; ENF: enfuvirtide; FTR: fostemsavir; IBA: ibalizumab; MVC: maraviroc.
Resistance analysis population and emergence of capsid resistance mutations by Week 52
Resistance analysis population and emergence of capsid resistance mutation by Week 52 in CAPELLA.

CA: capsid; RAP: resistance analysis population; R: resistance.
aOne participant developed a T107A polymorphism in CA with no loss in LEN susceptibility (FC = 0.6 compared to WT). Another participant developed a T107S polymorphism in CA with no impact on LEN susceptibility (FC = 1.3 compared to WT). These participants are counted in the ‘No CA-R Emergence’ category, but the mutations are tallied in the T107 row.
For the first full year of study, 9 participants (13%) had emergence of CA resistance-associated mutations (RAMs) while the remaining 13 participants (18%) in the RAP had no CA RAMs, including 4 participants analysed both at Wk26 and Wk52. Of the 9 participants with CA RAMs, 6 had emergence of M66I along with other mutations that were associated with a median of 234-fold resistance to LEN compared to WT control. These include one participant who developed the additional N74D and A105T mutations at Wk52 with an increase in LEN phenotypic resistance and one participant with CA RAMs changing from M66M/I + A105A/T at Wk26 to M66I + A105T at Wk52 with a minor increase in phenotypic resistance. One participant with emergence of K70H in the Wk26 analysis (265-fold resistance to LEN) had genotypic changes at Wk52 (K70H + A105A/S/T + T107T/N at Wk26 and Q67K + K70H at Wk52) with similar phenotypic susceptibility; one participant had emergence of Q67H and K70R at Wk26 with 15-fold resistance to LEN; and the new participant in the RAP at Wk52 had emergence of Q67Q/H alone with 6-fold resistance to LEN (Table 2, Table S2). Emergence of resistance to OBR drugs from the four main drug classes through Wk52 occurred in 3 participants who re-suppressed HIV-1 RNA <50 copies/mL.
Discussion
Phenotypic studies to assess the potential for cross-resistance to LEN in isolates harbouring resistance to the four main classes of ARV drugs as well as MIs have shown the absence of cross-resistance to LEN, indicating that the resistance profile of LEN is orthogonal to that of the main classes of ARV drugs. 4 Here, we have shown that the phenotypic susceptibility to LEN was unchanged from WT regardless of the presence or absence of susceptibility to EIs in baseline isolates from participants in the CAPELLA Study. This indicates that phenotypic resistance associated with the HIV-1 envelope is not associated with resistance in the capsid gene. Therefore, this dataset complements the existing data indicating the complete absence of cross-resistance to LEN in isolates harbouring resistance to any ARV drugs, thereby confirming that LEN can be used regardless of the resistance makeup of PWH, which also correlates with the absence of pre-existing resistance to LEN in the population at large.9,10 The lack of cross-resistance across ARV classes supports the use of LEN with OBR in PWH regardless of treatment history.
Emergent on-treatment resistance to LEN was observed in our study by Wk52, with 8 participants developing resistance to LEN by Wk26, 4 and one additional participant with emergent resistance to LEN between Wk26 and Wk52. Similar to previous observations, 4 this participant was in a situation of functional LEN monotherapy at the time of the emergence of resistance, in this case due to inadequate adherence to OBR, and re-suppressed durably to undetectable levels upon resumption of OBR.
Among the 7 participants analysed at both Wk26 and Wk52, 4 participants had no CA RAMs throughout and 3 participants showed some evolution in their resistance pattern. As for the new RAP participant discussed above, these 3 participants were either not adequately adherent to their OBR or had no active agent in their OBR and as a result were receiving functional LEN monotherapy.
In conclusion, the data presented here have established that none of the baseline resistance to existing ARV drugs shows cross-resistance to LEN, indicating that the resistance profile of LEN is orthogonal to the resistance profile of other ARVs used in the clinic. In addition, we have observed only one new participant with CA RAM from Wk26 to Wk52, which was associated with LEN functional monotherapy (inadequate adherence to OBR), similarly as with the other eight participants with CA RAMs.
Supplemental Material
Supplemental Material - Cross-resistance to entry inhibitors and lenacapavir resistance through Week 52 in study CAPELLA
Supplemental Material for Cross-resistance to entry inhibitors and lenacapavir resistance through Week 52 in study CAPELLA by Nicolas Margot, Nina Pennetzdorfer, Vidula Naik, Martin Rhee, and Christian Callebaut in Journal of Antiviral Therapy.
Footnotes
Authors’ note
All authors are employees and stockholders of Gilead Sciences Inc., which funded the studies.
Acknowledgements
The authors would like to thank the patients and their families along with the investigators and their staff who participated in the study, as well as the Gilead LEN project team for their contributions to these analyses. This study was presented in part at the virtual Conference on Retroviruses and Opportunistic Infection (CROI) 2022, February 12–16, 2022 (poster presentation 102) and at IDWeek 2022 in Washington DC, October 19–23, 2022 (oral presentation 1585).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Gilead Sciences.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
