Abstract
Background
Cytomegalovirus (CMV) infection is a frequent complication in haematopoietic cell/solid organ transplant (HCT/SOT) recipients. Previous studies report all-cause mortality rates of 31% and 50% in HCT/SOT recipients post-treatment initiation with conventional anti-CMV therapies for refractory or resistant CMV.
Methods
This was a multi-country, retrospective medical chart review study of HCT/SOT recipients with refractory CMV infection with or without resistance (R/R) who were randomized to the maribavir arm in the open-label Phase 3 SOLSTICE trial. Patients came from 21 SOLSTICE sites across 6 countries; each site randomized ≥3 patients to the maribavir arm. Patients were followed for 52 weeks (SOLSTICE trial period: 20 weeks; follow-up chart review period: 32 weeks). The primary outcomes were mortality and graft status.
Results
Of 234 patients who were randomized and received maribavir in SOLSTICE, chart abstraction was completed for all 109 patients enrolled across 21 trial sites (SOT, 68/142; HCT, 41/92). At 52 weeks, overall mortality was 15.6% (17/109) and survival probability was 0.84. Among SOT recipients, survival probability was 0.96, and 3 (4.4%) deaths occurred during the chart review period. For the HCT recipients, survival probability was 0.65 with 14 (34.1%) deaths; 8 occurred during SOLSTICE and 6 during the chart review period. No new graft loss or re-transplantation occurred during the chart review period.
Conclusions
Overall mortality at 52 weeks post-maribavir treatment initiation in this sub-cohort of patients from the SOLSTICE trial was lower than that previously reported for similar populations treated with conventional therapies for R/R cytomegalovirus infection.
Introduction
Cytomegalovirus (CMV) infection is a frequent complication in haematopoietic cell/solid organ transplant (HCT/SOT) recipients1,2 and is associated with poor clinical outcomes such as graft failure and mortality.3–7 Recipients’ risk of CMV infection post-transplantation is affected by several factors including donor/recipient serostatus, level of immune suppression, and transplant type. 5 Conventional antivirals such as ganciclovir, valganciclovir, foscarnet, and cidofovir are limited by potential toxicities and development of treatment resistance.8–11 Previous literature reports mortality estimates of 31% (HCT: 10/12 [83%]; SOT: 2/12 [17%]) and 50% (HCT: 4/6 [67%]; SOT 4/10 [40%]) post-treatment initiation in HCT and SOT recipients with refractory CMV infection with or without resistance (R/R).8,10
Maribavir is an oral antiviral with multimodal anti-CMV activity12–15 recently approved for the treatment of post-transplant CMV infection/disease that is refractory (with or without resistance) to treatment with ganciclovir, valganciclovir, cidofovir, or foscarnet. Approval was based on the open-label Phase 3 SOLSTICE trial (NCT02931539), with a duration of 20 weeks. Transplant recipients with R/R CMV were randomized 2:1 to maribavir 400 mg twice daily or investigator-assigned therapy (IAT) for 8 weeks with 12 weeks follow-up. After ≥3 weeks of treatment, patients in the IAT group meeting pre-specified criteria could enter a maribavir rescue arm. The primary and key secondary endpoints were met, with 55.7% of patients in the maribavir group achieving primary endpoint of CMV viraemia clearance at the end of Week 8 versus 23.9% of patients in the IAT group, and 18.7% versus 10.3% of patients maintaining CMV viraemia clearance and symptom control to Week 16 (key secondary endpoint), respectively. 16 Fewer patients discontinued treatment due to treatment-emergent adverse events with maribavir than IAT. 16 Data on longer-term outcomes such as mortality and graft outcomes were limited from the clinical trial setting due to trial design and follow-up duration. Therefore, the objectives of this observational, retrospective chart review study were to evaluate mortality and graft status at 52 weeks post-maribavir treatment initiation in a sub-cohort of HCT and SOT recipients from the 20-week SOLSTICE trial.
Methods
Patients and study design
This was a multi-country, observational, retrospective medical chart review study of a cohort of HCT and SOT recipients with R/R CMV infection who were randomized to the maribavir treatment arm in SOLSTICE. Patients in this cohort came from 21 sites across 6 countries (Belgium, Canada, France, Spain, UK, and USA); each site randomized ≥3 patients to the maribavir arm. Data for all SOLSTICE participants who were randomized to maribavir at each participating site were extracted in this chart review study (Supplementary Material). Due to the retrospective nature of this chart review, exemption of informed consent was requested to the independent ethics committees, except if required in accordance with the site or local country regulations. This study was approved by the institutional review boards or independent ethics committees at each site (Supplementary Table 1).
This study included the SOLSTICE trial period (20 weeks [8 weeks treatment; 12 weeks follow-up]) and the follow-up chart review period (32 weeks; starting 1 day after the SOLSTICE trial period) (Figure 1). The index date was defined as the date of randomization to the maribavir arm in SOLSTICE, and the end of follow-up was defined as 52 weeks post-index date or patient death/loss to follow-up, whichever occurred first. Study design. Notes. aTrial analysis period was truncated at 20 weeks if the last patient visit recorded occurred 20 weeks after the index event, bDuration of chart review study period depended on the duration of follow-up in the SOLSTICE trial (e.g. patients who withdrew from the trial at <20 weeks may have had longer post-trial follow-up in chart review study period), and cChart abstractions occurred once and patients had accrued at least 32 weeks of post-trial follow-up.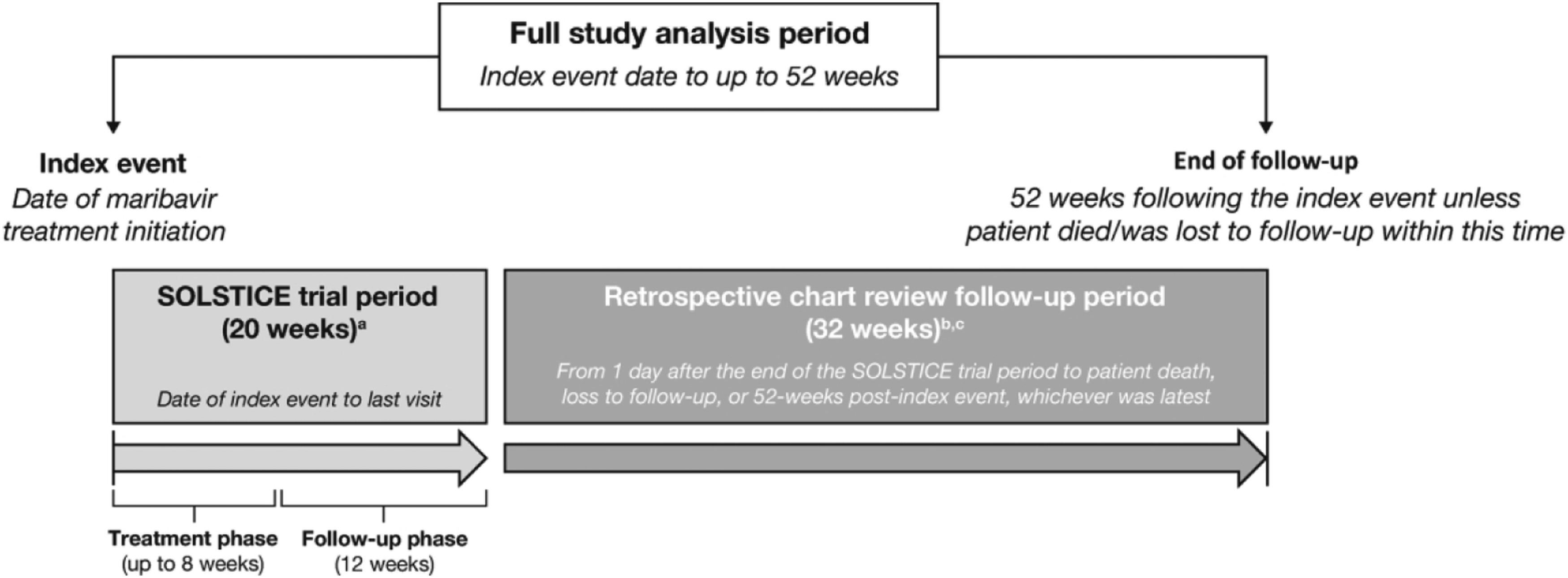
Endpoints and statistical analyses
This retrospective study examined overall survival (OS) at 52 weeks post-maribavir initiation for the chart review sample overall and for the HCT and SOT subgroups. Additionally, graft outcomes in the HCT and SOT cohorts were examined. Overall survival at 52 weeks was analyzed from the time of randomization to maribavir in the SOLSTICE trial using the Kaplan–Meier method, and the survival probability at 52 weeks was provided with the 95% confidence interval (CI); patients who were lost to follow-up within the 52-week observation period were censored at the last contact date, as were those who were alive at week 52. Descriptive statistics were used to summarize graft outcomes in SOT and HCT recipients. A weighted sensitivity analysis was performed to assess the impact of minor imbalances between the trial and chart review patients on important characteristics such as transplant type (HCT vs SOT), age (≥65 years old vs <65 years old), sex, and CMV tissue-invasive disease on OS.
Results
Patients
Key baseline characteristics among patients enrolled in the chart review study.

CMV, cytomegalovirus; D+/R+, donor positive/recipient positive; D−/R+, donor negative/recipient positive; D+/R−, donor positive/recipient negative; D−/R−, donor negative/recipient negative; HCT, haematopoietic cell transplant; SD, standard deviation; Q, quartile; SOT, solid organ transplant.
aTime from transplant was computed in months by subtracting the date of transplantation from the patient’s index date.
bDuration of maribavir treatment was computed in days by subtracting the patient’s index date from the date of the last dose.
Overall survival at 12 months post-maribavir treatment initiation
Overall survival, all-cause mortality, and graft loss at 12 months.

CI, confidence interval; CMV, cytomegalovirus; HCT, haematopoietic cell transplant; SOT, solid organ transplant.
aIncludes anaemia from lung transplant with underlying cause of idiopathic pulmonary fibrosis (n = 1), worsening renal failure and debilitation while on home intravenous foscarnet, refused readmission to the hospital and requested transition to hospice (n = 1).
bIncludes progressive disease (acute lymphoblastic leukaemia; n = 1), septic shock (n = 1), and underlying disease relapse (n = 4).
Graft outcomes during the full 12-month study period
No patients had graft failure/loss or graft re-transplantation during the chart review period (Table 2). Overall, 24 (22.0%) patients experienced graft complications during the full study period.
Of the 68 SOT recipients, 8 (11.8%) recipients had graft complications during the full study period (SOLSTICE trial period: 3; chart review period: 5). Of these patients, 4 (50.0%) had an acute rejection, 2 (25.0%) had a chronic rejection, and 2 (25.0%) had other types of complications. Five complications occurred in kidney transplant patients, while the remaining occurred in lung (n = 2) and heart (n = 1) transplant patients. At 52 weeks, 61 (89.7%) patients had a functioning SOT and 6 (8.8%) patients had a functioning SOT with ongoing complications. One (1.5%) patient had an unknown complication and there were no patients with non-functioning SOT.
Of the 41 HCT recipients, 16 (39.0%) patients experienced 17 graft complications, with 1 patient experiencing acute graft-versus-host disease (GVHD) and relapse of leukaemia during the full study period. Of the 17 complications, 11 (64.7%) were acute GVHD, 5 (29.4%) were chronic GVHD, and 1 (5.9%) complication was reported as relapse of leukaemia.
For SOT and HSCT graft events, patient’s death may be considered as a competing risk for graft outcomes since death impedes the occurrence of a graft issue. Therefore, as a sensitivity analysis, a competing risk analysis was also performed on the time-to-event graft outcomes. The results of this analysis showed minimal impact on the graft outcome estimates.
Discussion
Overall mortality of 15.6% at 12 months in SOT and HCT recipients with R/R CMV infection treated with maribavir was lower than estimates reported in similar populations treated with conventional anti-CMV therapy. For instance, one study reported an all-cause mortality rate of 50% post-cidofovir initiation (HCT: 4/6 [67%]; SOT 4/10 [40%]) and another study reported a 1-year mortality rate of 31% post-foscarnet initiation (HCT: 10/12 [83%]; SOT: 2/12 [17%]) in a mixed population of SOT and HCT recipients with R/R CMV infection.8,10 Additionally, our findings indicated that, as expected, 1-year mortality was lower in SOT than HCT recipients, and that these mortality rates were generally lower than published estimates also reporting mortality by transplant type: HCT (4/6 [67%]) and SOT (4/10 [40%]); 8 HCT (10/12 [83%]) and SOT (2/12 [17%]); 10 SOT (14/37 [38%]); 11 HCT (33.3%). 17 No new graft loss or re-transplantations occurred in the chart review study.
Despite lower mortality rates in the present study, comparisons to previously published data should not be inferred due to small patient numbers and differences in patient populations across studies. Further limitations of this study include the possibility that the present chart review sample was not representative of the overall trial population due to the sampling strategy employed of targeting high enrolling sites. However, no major imbalances were found between patients enrolled in the chart review study and those not enrolled with respect to baseline demographic and clinical characteristics. Additionally, analyses of mortality between subgroups of patients (e.g. those with or without viraemia clearance, or those with refractory vs resistant CMV infection) were not performed, as the design and protocol of the study precluded acquisition of such data.
Mortality and graft loss/re-transplantation were well-documented post-transplant due to reporting requirements for transplant registries and due to the close follow-up of this patient population. As such, there was a low probability of missing data on the primary outcomes of this chart review study. Finally, given the rescue arm of the SOLSTICE trial with 18.8% of patients in the conventional therapy arm switching to maribavir, 16 only patients initially assigned to receive maribavir treatment were included in this chart review.
In conclusion, overall mortality at 52 weeks post-maribavir treatment initiation in these sub-cohorts of patients from the SOLSTICE trial was lower than mortality estimates reported for similar populations treated with conventional therapies for R/R CMV infection. These results, taken together with the observed superior efficacy of maribavir versus conventional therapies in the SOLSTICE trial, 16 suggest that the benefits of maribavir treatment for post-transplant CMV infection may carry beyond the 20 weeks of trial duration into at least 52 weeks post-treatment initiation.
Supplemental Material
Supplemental Material - Retrospective chart review of transplant recipients with cytomegalovirus infection who received maribavir in the Phase 3 SOLSTICE trial: Data at 52 weeks post–maribavir treatment initiation
Supplemental Material for Retrospective chart review of transplant recipients with cytomegalovirus infection who received maribavir in the phase 3 SOLSTICE trial: Data at 52 weeks post-maribavir treatment initiation by Marielle Bassel, Dorothy Romanus, Tien Bo, Aimee K Sundberg, Sandra Okala, and Ishan Hirji in antiviral therapy.
Footnotes
Author’s note
Under the direction of the authors, Ashvanti Valji, PhD of Caudex, London, UK provided writing assistance, and Mary Casey, PhD of Caudex, Oxford, UK, provided editorial assistance. Caudex was funded by Takeda Development Center Americas, Inc. for these medical writing and editorial services.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB, SO: employee: PPD, a Thermo Fisher Scientific company, which was contracted to provide services to Takeda for this study. DR, TB, AKS, and IH: employee: Takeda; stock/stock options: Takeda.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Takeda Development Center Americas, Inc.
Data availability statement
The datasets, including the redacted study protocol, redacted statistical analysis plan, and individual participants data supporting the results reported in this article, will be available 3 months from initial request, to researchers who provide a methodologically sound proposal. The data will be provided after its de-identification, in compliance with applicable privacy laws, data protection, and requirements for consent and anonymization. The data sharing policy of Takeda is available at https://clinicaltrials.takeda.com/. Data sharing requests can be submitted through ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
