Abstract
Background
In the DAWNING study, dolutegravir + 2 nucleoside reverse transcriptase inhibitors (NRTIs) demonstrated superior efficacy at Week 48 and a favourable safety profile compared with lopinavir/ritonavir + 2 NRTIs in adults with HIV-1 failing first-line therapy of a non-nucleoside reverse transcriptase inhibitor + 2 NRTIs.
Methods
Participants at 58 centres in 13 countries were randomised (1:1) to 52 weeks of open-label treatment with dolutegravir or lopinavir/ritonavir combined with 2 investigator-selected NRTIs, including at least one fully active NRTI based on screening resistance testing. The primary endpoint was the proportion of participants achieving HIV-1 RNA <50 copies/ml at Week 48 (Snapshot algorithm). Post-hoc efficacy analyses were performed based on baseline NRTI resistance profile and second-line NRTI use.
Results
Of 624 participants randomised and treated, 499 (80%) received <2 active NRTIs at Baseline. NRTI resistance was present in 561 participants (90%). Among participants receiving lamivudine or emtricitabine in the presence of M184V/I, 85% (187/220) of participants on dolutegravir versus 72% (152/210) on lopinavir/ritonavir had HIV-1 RNA <50 copies/ml at Week 48 (difference, 12.6%; 95% CI: 4.9–20.3%). High responses were also observed in the dolutegravir group, when zidovudine or tenofovir disoproxil fumarate were included in the background regimen in the presence of thymidine analogue mutations or K65R, respectively; however, participant numbers in these subgroups were small.
Conclusions
Response rates were high in participants receiving dolutegravir + 2 NRTIs as second-line treatment regardless of pre-existing resistance to one of the NRTIs, including in participants using lamivudine or emtricitabine in the presence of M184V/I.
Keywords
Introduction
The DAWNING study evaluated the efficacy and safety of dolutegravir + 2 nucleoside reverse transcriptase inhibitors (NRTIs) versus the (at the time) World Health Organization (WHO)-recommended regimen of lopinavir/ritonavir + 2 NRTIs in participants with HIV-1 failing first-line therapy of a non-NRTI (NNRTI) + 2 NRTIs (ClinicalTrials.gov: NCT02227238). Dolutegravir + 2 NRTIs was superior in the proportion of participants achieving viral suppression (plasma HIV-1 RNA <50 copies/ml) at Week 48 compared with lopinavir/ritonavir + 2 NRTIs [1].
The WHO now recommends dolutegravir + 2 NRTIs as a preferred first-line regimen and dolutegravir with an optimised NRTI backbone among people for whom a non–dolutegravir-based first-line regimen has failed [2–4]. NRTI resistance (especially M184V/I and K65R) is common following virologic failure on first-line regimens in resource-limited settings, and an optimised NRTI backbone is recommended in second-line treatment, such as zidovudine following tenofovir disoproxil fumarate (TDF) or abacavir failure and TDF following zidovudine failure plus recycled lamivudine or emtricitabine [4].
This post-hoc analysis examines efficacy outcomes in DAWNING for different NRTI combinations in the presence of existing NRTI resistance, including lamivudine or emtricitabine in the presence of M184V/I.
Methods
DAWNING is a phase 3b, open-label, parallel-group, non-inferiority, active-controlled trial conducted at 58 sites in 13 countries. Eligible adults were aged ≥18 years and had experienced virologic failure (confirmed HIV-1 RNA ≥400 copies/ml) during ≥6 months of treatment with a first-line regimen containing an NNRTI and 2 NRTIs (methodology described previously) [1]. All participants were protease inhibitor (PI) and integrase strand transfer inhibitor (INSTI) naive and received an investigator-selected dual NRTI background regimen for second-line treatment, including ≥1 fully active NRTIs based on genotypic resistance testing at screening using Q2 Solutions (Morrisville, NC, USA) genotypic testing and their proprietary algorithm. NRTIs showing no evidence of genotypic resistance at screening were considered fully active.
Monogram Biosciences (South San Francisco, CA, USA) did genotypic and phenotypic resistance testing using the PhenoSense GT assay for protease and reverse transcriptase for all baseline samples. For participants meeting confirmed virologic withdrawal (CVW) criteria [1], baseline samples and the first of 2 samples required to meet CVW criteria were tested for protease and reverse transcriptase and for integrase genotype and phenotype using PhenoSense Integrase and GenoSure. If the PhenoSense GT or Integrase assays were unsuccessful, the PhenoSense GT Plus Integrase assay was used. Participants who met CVW criteria discontinued the study; information on the post-study regimen and treatment response of these participants was not collected.
Written informed consent was obtained from each participant before study entry. The study protocol was approved by appropriate ethics committees and institutional review boards in accordance with the Declaration of Helsinki and the International Conference on Harmonisation Guideline for Good Clinical Practice.
Participants were randomised 1:1 to receive either oral dolutegravir (50 mg once daily) or lopinavir/ritonavir (800 mg lopinavir plus 200 mg ritonavir once daily or 400 mg plus 100 mg twice daily), plus 2 investigator-selected NRTIs of which ≥1 was fully active.
The primary endpoint was the proportion of participants achieving viral suppression (plasma HIV-1 RNA <50 copies/ml) at Week 48 according to the Snapshot algorithm in the intention-to-treat–exposed population.
Proportion of participants with HIV-1 RNA <50 copies/ml at Week 48 (Snapshot algorithm) was compared between dolutegravir and lopinavir/ritonavir groups by second-line background NRTIs and baseline NRTI resistance.
Results
Of 968 people screened, 624 were randomised and treated, 312 each in the dolutegravir and lopinavir/ritonavir groups. As previously reported, baseline demographics were similar between groups; approximately one-third of participants had a history of AIDS (Centers for Disease Control and Prevention category C at Baseline) and approximately half had CD4+ cell counts <200 cells/μl [1,5]. All participants had ≥1 active NRTI in their second-line background regimen based on screening resistance testing.
Most participants (561/624 [90%]) had NRTI resistance-associated mutations (RAMs) at Baseline, with M184V/I (alone or plus additional NRTI RAMs) present in 84% (261/312) in the dolutegravir group and 81% (252/312) in the lopinavir/ritonavir group (Additional file 1). K65R was present in 30% (95/312) and 29% (92/312) and ≥1 thymidine analogue mutations (TAMs; M41L, D67N, K70R, L210W, T215F, T215Y, and K219E/Q) in 23% (71/312) and 26% (81/312) of participants in the dolutegravir and lopinavir/ritonavir groups, respectively. In the dolutegravir group, 71% (220/312) of participants used lamivudine or emtricitabine in the presence of M184V/I versus 67% (210/312) in the lopinavir/ritonavir group. Seven participants in the dolutegravir group and eight in the lopinavir/ritonavir group received TDF in the presence of K65R; of these, 4 and 6, respectively, also received zidovudine in their background regimen, while, respectively, 11% (35/312) and 16% (51/312) of participants took zidovudine with ≥1 TAM present.
Overall, 84% (261/312) of participants on dolutegravir versus 70% (219/312) on lopinavir/ritonavir achieved HIV-1 RNA <50 copies/ml at Week 48 (adjusted difference, 13.8%; 95% CI: 7.3–20.3%; P < 0.001 for superiority) [1]. Overall efficacy was consistent across subgroups defined by number of fully active NRTIs (<2 or 2) in the second-line background regimen [1].
Response rates were consistently higher in the dolutegravir group regardless of the presence of M184V/I and use of lamivudine or emtricitabine (Figure 1). Among participants receiving lamivudine or emtricitabine in the presence of M184V/I, 85% (187/220) of participants on dolutegravir versus 72% (152/210) on lopinavir/ritonavir had HIV-1 RNA <50 copies/ml at Week 48 (difference, 12.6%; 95% CI: 4.9–20.3%). This higher response rate was observed regardless of whether lamivudine or emtricitabine use included zidovudine (86% [96/112] versus 73% [76/104]; difference, 12.6%; 95% CI: 1.9–23.3%) or TDF (84% [91/108] versus 72% [79/109]; difference, 11.8%; 95% CI: 0.9–22.6%) as the second NRTI. High responses were also observed in the dolutegravir group when zidovudine or TDF were included in the background regimen in the presence of TAMs or K65R, respectively; in the ‘TDF use with K65R’ subgroup, response rates were similarly high between groups (Figure 1). However, participant numbers in these subgroups were small. Snapshot response rates at Week 48 in the ITT-E population 
In the presence of M184V/I and additional NRTI mutations (including K65R and/or other International Antiviral Society–USA major NRTI substitutions), more participants using lamivudine or emtricitabine in the dolutegravir group achieved viral suppression at Week 48 versus the lopinavir/ritonavir group; response rates were similar to overall rates (Figure 2). Few participants with both M184V/I and K65R received lamivudine or emtricitabine; zidovudine was included in the background regimen in 94% (46/49) of these cases. Snapshot response rates at Week 48 in the ITT-E population by presence of M184V/I with or without additional NRTI mutations at Baseline. CI, confidence interval; ITT-E, intention-to-treat–exposed; NRTI, nucleoside reverse transcriptase inhibitor; RAM, resistance-associated mutation; TAM, thymidine analogue mutation. aM184V/I alone or plus additional NRTI mutations.
Emergent resistance in participants meeting confirmed virologic withdrawal criteria.
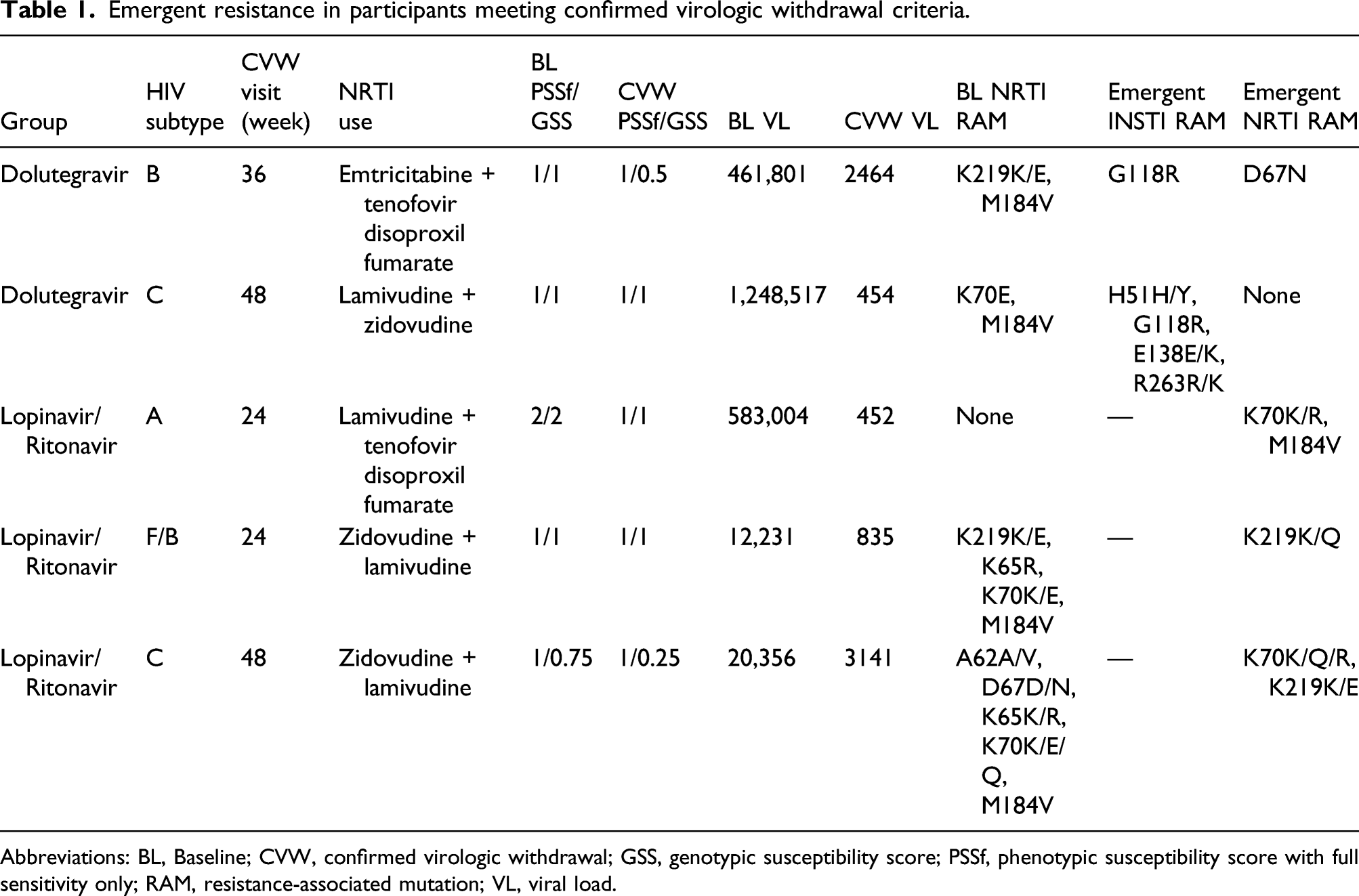
Abbreviations: BL, Baseline; CVW, confirmed virologic withdrawal; GSS, genotypic susceptibility score; PSSf, phenotypic susceptibility score with full sensitivity only; RAM, resistance-associated mutation; VL, viral load.
Discussion
The DAWNING study demonstrated superior efficacy of dolutegravir + 2 NRTIs compared with lopinavir/ritonavir + 2 NRTIs at Week 48 [1]. Response rates were high in participants receiving dolutegravir + 2 NRTIs regardless of pre-existing resistance to one of the background NRTIs. In this post-hoc analysis examining the impact of NRTI RAMs and specific NRTI use, response rates for dolutegravir were consistently high and for the majority of subgroups (except for the smallest subgroup, TDF use with K65R), were higher than for lopinavir/ritonavir, consistent with the overall study findings. Recycling of lamivudine or emtricitabine is common in second-line regimens across both resource-limited and high-resource countries, so the high response rate observed for participants receiving dolutegravir and lamivudine or emtricitabine in the presence of M184V/I is of particular relevance. Interpretation of findings in the smaller subgroups is limited by relatively small participant numbers.
DAWNING required resistance testing at screening and the inclusion of ≥1 fully active NRTI in the second-line background regimen; however, testing is not routinely done to guide second-line therapy in low-resource settings. As previously reported, results from subgroup analyses comparing participants who received or did not receive their second-line NRTI background regimen in accordance with WHO recommendations were also consistent with overall efficacy results [1].
Maintenance of lamivudine or emtricitabine in the second-line regimen was common in DAWNING; however, the majority of participants switched their second NRTI at randomisation. The study therefore does not address the important data gap regarding the efficacy of switching from a first-line regimen containing TDF plus lamivudine or emtricitabine directly to second-line TDF/lamivudine/dolutegravir. This data gap is currently being explored in the D2EFT and NADIA studies [6,7]. The primary outcome of the NADIA trial recently demonstrated high and non-inferior efficacy of dolutegravir + 2 NRTIs versus ritonavir-boosted darunavir + 2 NRTIs; at Week 48, viral load was <400 copies/ml in 90% (212/235) of participants in the dolutegravir group and 92% (210/229) in the darunavir group (difference, −1.5; 95% CI: −6.7%, 3.7%; P = 0.58) [8]. In subgroup analyses, similarly high response rates were seen among participants with no predicted active NRTIs in the prescribed regimen and those who maintained TDF plus lamivudine in second-line.
WHO treatment guidelines for first- and second-line antiretroviral therapy now include dolutegravir with an optimised NRTI backbone as a preferred second-line treatment option for patients failing an NNRTI- or PI-based first-line antiretroviral regimen [4]. The DAWNING study and this analysis support this recommendation.
Supplemental Material
sj-pdf-1-avt-10.1177_13596535221077487 – Supplemental Material for Efficacy of Second-line Dolutegravir Plus 2 NRTIs by Baseline NRTI Resistance and NRTI Use in the DAWNING Study
Supplemental Material, sj-pdf-1-avt-10.1177_13596535221077487 for Efficacy of Second-line Dolutegravir Plus 2 NRTIs by Baseline NRTI Resistance and NRTI Use in the DAWNING Study by Dannae Brown, Richard Kaplan, Marcelo Losso, Carlos Brites, Ruolan Wang, Mark Underwood, Judy Hopking, Michael Aboud, and Jörg Sievers in Antiviral Therapy
Footnotes
Acknowledgements
We thank everyone who has contributed to the success of this study, including all study participants and their families, the independent data monitoring committee, the clinical investigators and their staff, and the GlaxoSmithKline and ViiV Healthcare study teams. Editorial assistance was provided under the direction of the authors by MedThink SciCom, Cary, NC, USA, and was funded by ViiV Healthcare. These data have previously been presented in full at the Conference on Retroviruses and Opportunistic Infections; March 4–7, 2019; Seattle, WA; Slides 144.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dannae Brown, Ruolan Wang, Mark Underwood, Michael Aboud, and Jörg Sievers are employees of ViiV Healthcare and own stock in GlaxoSmithKline (GSK). Marcelo Losso has received grants from ViiV Healthcare. Carlos Brites has served as a speaker and advisory board member for GSK, Gilead, Janssen and Merck. Judy Hopking is an employee of GSK and owns stock in GSK. Richard Kaplan has nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by ViiV Healthcare.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
