Abstract
Background
Lopinavir/ritonavir (LPV/r) and hydroxychloroquine (HCQ) are both being used to treat coronavirus disease 2019 (COVID-19), but their relative effectiveness is unknown. The purpose of this study was to compare the clinical outcomes of patients treated for COVID-19 with LPV/r or HCQ.
Methods
A retrospective observational study was conducted at 2 hospitals in Busan, South Korea, where approximately 90% of COVID-19 patients were hospitalised during February/March 2020. All patients aged ≥15 years that were hospitalised with mild or moderately severe COVID-19 received LPV/r or HCQ as their initial treatment and were included in the analysis.
Results
Among the 72 patients with mild-to-moderate disease severity on admission, 45 received LPV/r and 27 received HCQ as their initial therapy. A higher proportion of the LPV/r group had pneumonia on admission (LPV/r, 49% vs HCQ, 15%), but there were no other significant differences in the demographic or clinical characteristics between groups. Switching therapy due to clinical failure was significantly more common in the HCQ group than in the LPV/r group (41% [11/27] and 2% [1/45], respectively, P = .001). Disease progression was also significantly more common in the HCQ group than in the LPV/r group (44% [12/27] and 18% [8/45], respectively, P = .030).
Conclusion
Based on our study results, HCQ shows no apparent advantage compared to LPV/r for preventing progression to severe disease in patients with COVID-19.
Introduction
Coronavirus disease 2019 is an emerging, highly infectious disease caused by a novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). It first appeared in Wuhan, Hubei Province, China, in December 2019 and rapidly spread globally, and on March 11, 2020, the World Health Organization (WHO) declared COVID-19 a pandemic.1-5
An analysis of 72 314 cases from China revealed that most patients experienced mild disease, with 14% developing severe disease and 5% developing critical disease, and the case fatality rate was 2.3%. 6 About 50% of patients with critical disease died, and the mortality rate was high in patients over the age of 70 years and those with pre-existing comorbidities. In other study, of 52 critically ill patients with SARS-CoV-2 infection, 62% died within 28 days. 7 Given the high mortality rate in critically ill patients, effective antiviral treatment, especially when administered early to patients at risk of developing severe disease, may improve the clinical outcomes of COVID-19.
There is currently no standard antiviral therapy for COVID-19, so treatment is supportive. Existing antiviral agents have been repurposed and prescribed for the treatment of COVID-19 based on therapeutic experience with 2 other infections caused by human coronaviruses: severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV). 8
Lopinavir/ritonavir (LPV/r) is a protease inhibitor that is used to treat human immunodeficiency virus (HIV) infection. It has been hypothesised that this combination drug may inhibit the protease of MERS-CoV and SARS-CoV, thus blocking the processing of the viral replicase polyprotein and preventing the replication of viral RNA.8,9 However, it is controversial whether HIV protease inhibitors could effectively inhibit the protease of SARS-CoV-2. 8 A non-randomised, open-label trial revealed that among patients with SARS, patients treated with LPV/r (n = 41) experienced fewer adverse clinical outcomes (acute respiratory distress syndrome [ARDS] or death) than those treated with ribavirin alone (n = 111) (2.4% versus 28.8%, P < .001). 10 In previous MERS outbreak, LPV was reported to have antiviral activity in vitro and in vivo, and Chan et al. 11 demonstrated that treatment of animals infected with MERS-CoV with LPV/r improved the clinical outcomes and reduced viral loads in lung and most extra-pulmonary tissue. However, another in vivo study showed that therapeutic effect of LPV/r and interferon beta did not reduce viral replication. 12 A recent case report demonstrated that the treatment of SARS-CoV-2-infected patients with LPV/r was associated with a reduction in the viral load and improved clinical symptoms. However, there have been no clinical studies to date that have provided evidence to support the use of LPV/r as treatment for SARS-CoV-2. 13
Chloroquine/hydroxychloroquine (HCQ), a drug that is widely used as an anti-malarial and for the treatment of autoimmune disease, exerts direct antiviral effects, inhibiting pH-dependent steps of the replication of several viruses, including coronaviruses. 14 Moreover, chloroquine has immunomodulatory effects and reduces the production/release of tumour necrosis factor alpha and interleukin-6, which mediate the inflammatory complication of viral disease. 14 In an in vitro study, chloroquine was found to have antiviral activity against SARS-CoV and MERS-CoV.15-17 Recently, chloroquine/HCQ has been shown to inhibit the growth of SARS-CoV-2 in vitro, and clinical studies are in progress.18,19
Both LPV/r and HCQ have been used, long-term, and their pharmacokinetics, pharmacodynamics, and side effects are well known. Therefore, their prescription can be considered for patients with early, mild COVID-19. In HIV patients with long-term LPV/r administration, drug discontinuation rate due to adverse effects was 5–10%.20,21 In systemic lupus erythematosus patients with long-term HCQ administration, drug discontinuation rate due to adverse effects was 11%. 22 In a series of studies, both drugs are known to be well tolerable. There is a question of whether an early administration of antiviral agent can reduce the progression to severe COVID-19 infection or improve the overall course of treatment, but there have been no studies to date. The aim of this study was to evaluate the clinical outcomes of early antiviral treatment and to compare LPV/r and HCQ as treatments of mild COVID-19.
Materials and methods
Study design, setting, and participants
We conducted a retrospective observational study at Pusan National University Hospital (PNUH) and Busan Medical Centre (BMC) in Busan, South Korea. Busan is a metropolitan city with a population of more than 3 million people. PNUH and BMC are the only hospitals in Busan designated to treat patients with COVID-19. A COVID-19 outbreak was detected in Busan on February 21, 2020. The Busan health authority referred most patients with confirmed COVID-19 to PNUH and BMC on a non-selective basis. We enrolled patients who were admitted to 2 hospitals from February 21, 2020, to March 21, 2020, and observed patients until April 3, 2020. During the 3-week study period, about 48 000 confirmed tests of COVID-19 were performed in Busan and 92 patients (≥15 years) were diagnosed with COVID-19. All the patients were hospitalised throughout their entire disease period from diagnosis to recovery and discharge, and their clinical progress was monitored. In PNUH, LPV/r was administered to almost all the patients regardless of the severity of disease. In BMC, HCQ was administered to patients with mild illness, without comorbidities, as the primary antiviral agent, and LPV/r was administered to people with comorbidities or pneumonia at presentation. Generally, patients treated with HCQ were switched to LPV/r if their clinical condition worsened (Figure 1). Distribution of patient and treatment assignment of COVID-19 patient in Busan.
Patients (≥15 years) with confirmed COVID-19 who received LPV/r (400/100 mg orally every 12 hours) or HCQ (400 mg orally every 24 hours) as the initial antiviral treatment were enrolled in this study. We excluded patients who had severe disease who required oxygen supplementation or mechanical ventilation at the time of diagnosis and patients who did not live in Busan.
Data collection procedure
We extracted epidemiological, demographic, clinical, laboratory, and outcome data from each patient’s electronic medical records using a standardised data collection form. All data were checked by 2 physicians (S Lee and J Heo) and a third physician (SO Lee) adjudicated any difference in interpretation.
Patients that were included in this study had a documented positive oropharyngeal or nasopharyngeal swab or sputum test for SARS-CoV-2 performed at public health centres or laboratory centres certified by the Korean Centres for Disease Control and Prevention before admission. The definition for cure was 48 hours after all symptoms disappeared, and 2 oropharyngeal/nasopharyngeal swab and sputum samples, obtained at least 24 hours apart, were negative for SARS-CoV-2 RNA. Additionally, patients underwent routine blood testing (including complete blood count, coagulation profile, serum biochemical test, myocardial enzymes, C-reactive protein, and procalcitonin) and chest X-rays or computer tomography. The frequency and timing of the tests was determined by the attending physicians.
Outcomes
Patients were assigned to the LPV/r group or the HCQ group based on the initial antiviral agents with which they were treated. The primary objective of this study was to compare the early clinical outcomes of treatment for mild-to-moderate COVID-19 with LPV/r and HCQ, and the endpoint was disease progression. The progression of clinical disease was defined as exacerbation of any category of clinical syndrome associated with COVID-19, as described in the WHO interim guidance. 23 The 6 clinical syndrome categories, in order of increasing severity, were (1) uncomplicated illness, (2) mild pneumonia, (3) severe pneumonia, (4) ARDS, (5) sepsis, and (6) septic shock. We also compared the development of pneumonia and progression of disease that required mechanical ventilation between the 2 groups as the secondary outcomes.
Statistical analysis
R version 3.3.2 (R Foundation for Statistical Computing, Vienna, Austria) was used for all statistical analyses. Categorical variables were compared using Pearson’s chi-square test or Fisher’s exact test, and continuous variables were tested using the Kruskal–Wallis test. All tests of significance were 2-tailed; P < .05 was considered to be significant.
Ethics
The study protocol was reviewed and approved by the Institutional Review Board of PNUH (IRB No. H 2003-015-089). The requirement for consent was waived by the board because of the retrospective nature of the study.
Results
COVID-19 epidemics in Busan
Among the 92 confirmed COVID-19 patients, 81 were hospitalised in PNUH or BMC. Their median age was 35 years (interquartile range [IQR]: 24–55 years) and 37 (46%) were male. Seven patients (10%) were asymptomatic at the time of diagnosis. Among the symptomatic patients, 85% were diagnosed within 5 days of symptom onset; and the median time from onset to diagnosis was 3 days (IQR: 1–5 days). At the time of diagnosis, 54 patients (67%) had uncomplicated illness (WHO class 1), 26 (32%) had mild pneumonia (WHO class 2), and 1 (1%) had ARDS (WHO class 4).
Clinical characteristics
Clinical feature of patients with SARS-CoV-2 who treated with LPV/r or hydroxychloroquine.

Abbreviations: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; LPV/r, lopinavir/ritonavir; IQR, interquartile range; WBC, white blood cells; LDH, lactate dehydrogenase; WHO, World Health Organization.
Early antiviral treatment outcomes of COVID-19
Comparative outcome of early antiviral treatments between lopinavir/ritonavir versus hydroxychloroquine.
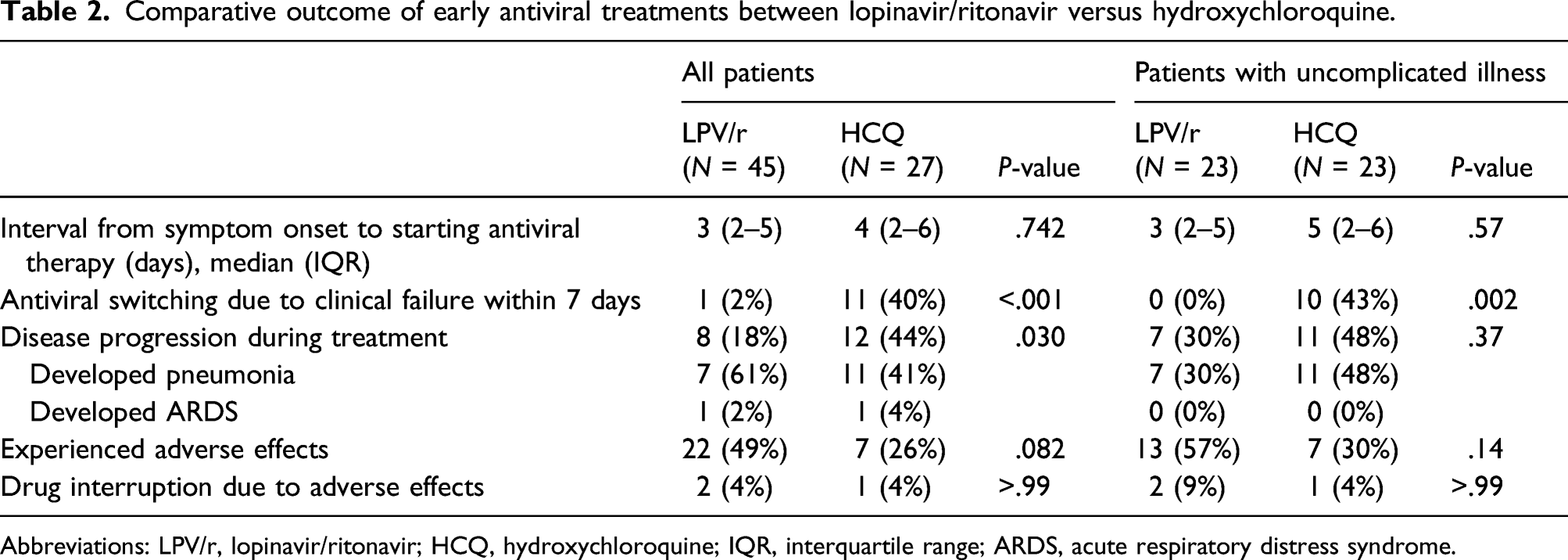
Abbreviations: LPV/r, lopinavir/ritonavir; HCQ, hydroxychloroquine; IQR, interquartile range; ARDS, acute respiratory distress syndrome.
In a subgroup analysis of WHO class 1 patients, 44% (10/23) of patients who initially received HCQ, and none of the patients who initially received LPV/r had their antiviral therapy modified by the attending physician (P = .002). Eleven patients in the LPV/r group (48%) and 7 patients in HCQ group (30%) developed pneumonia during therapy (P = .37).
Comparison of outcomes of antiviral therapy by hospital
Comparison of early outcomes of antiviral therapy by hospital, and subgroup analysis, comparing the outcomes of LPV/r vs HCQ in BMC.
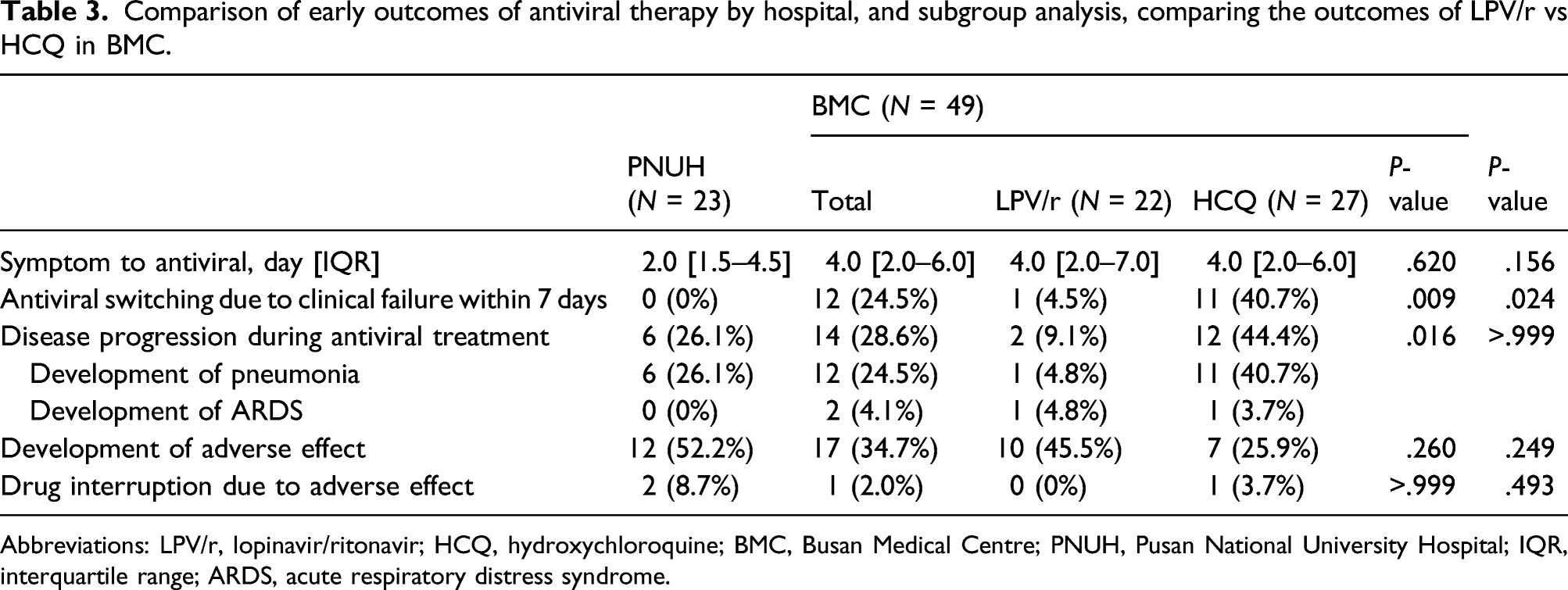
Abbreviations: LPV/r, lopinavir/ritonavir; HCQ, hydroxychloroquine; BMC, Busan Medical Centre; PNUH, Pusan National University Hospital; IQR, interquartile range; ARDS, acute respiratory distress syndrome.
In a subgroup analysis comparing the outcome of LPV/r vs HCQ in BMC, 40.7% (11/27) of patients who initially received HCQ and 4.5% (1/22) of patients who initially received LPV/r had their antiviral therapy modified (P = .009). Two patients in the LPV/r group (9.1%) and 12 patients in the HCQ group (44.4%) experienced clinical progression (P = .016).
Outcomes of LPV/r in patients with COVID-19 pneumonia
Forty-five (63%) of the patients hospitalised with COVID-19 developed pneumonia. Twenty-seven patients (38%) were diagnosed with pneumonia at the time of presentation and 18 patients (25%) were diagnosed with pneumonia during the course of antiviral therapy. Among the 45 patients with COVID-19 pneumonia, 41 (91%) were treated with LPV/r; 30 (67%) as primary therapy; and 11 (25%) as salvage therapy after the primary HCQ therapy. Among the patients who received LPV/r, 9 (22%) were over 70 years old. Thirty-two patients (78%) had multifocal lesions, 28 (68%) had bilateral lesions, and 4 (10%) had diffuse bilateral pneumonia.
Among the 45 patients with COVID-19 pneumonia who received LPV/r, 38 (93%) recovered without oxygen supplementation or mechanical ventilator therapy. Three patients needed mechanical ventilation. Two patients with WHO classes 3 and 4 severity at initiation of LPV/r and 1 patient in whom LPV/r was initiated for mild COVID-19 developed severe disease requiring ventilator therapy. Of the 3 patients treated using a mechanical ventilator, one was cured but the other 2 died due to the exacerbation of COVID-19 pneumonia.
Adverse drug events
Adverse events associated with using LPV/r or HCQ as treatment of COVID-19.
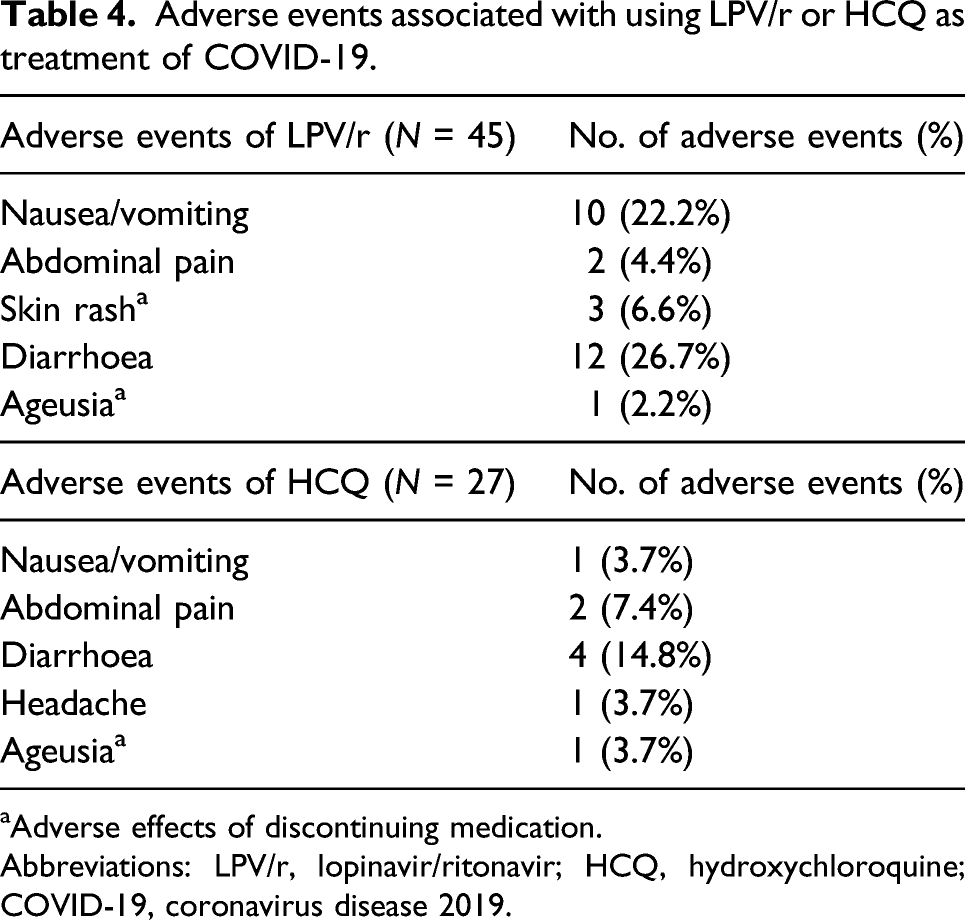
aAdverse effects of discontinuing medication.
Abbreviations: LPV/r, lopinavir/ritonavir; HCQ, hydroxychloroquine; COVID-19, coronavirus disease 2019.
A total of 3 patients stopped the drugs early. One patient in the LPV/r group stopped the drug due to rashes, while 2 patients (one each in the LPV/r and HCQ groups, respectively) stopped the drug due to loss of taste. No life-threatening adverse effects have been identified in these patients.
Discussion
The incidence of COVID-19 is high in many countries globally, and the number of patients with severe disease requiring mechanical ventilation is also rapidly increasing. The number of deaths is increasing due to a lack of sufficient critical care facilities and equipment such as mechanical ventilators.
Conducting clinical trials to determine the efficacy of new drugs and licensing new drugs takes time. Even drugs that are known to have therapeutic effects require comparative studies on the therapeutic effects in various clinical situations. Patient with mild-to-moderate COVID-19 severity at presentation may be cured without complications or may progress to severe and debilitating disease. Clinical information is needed to determine whether administration of antiviral agents early in the course of disease can reduce the incidence of severe cases requiring oxygen supplementation or mechanical ventilation. However, most of the information currently available is based on studies of patients who have already developed severe disease on admission. 24 The results of this study provide some useful information regarding early antiviral administration of LPV/r or HCQ for the treatment of mild COVID-19.
First, our study suggests that early administration of antiviral agent, especially LPV/r, to the COVID-19 patients with mild-to-moderate severity can reduce the exacerbation of the disease to severe cases needing oxygen supplementation and mechanical ventilator therapy. One of the largest epidemiologic studies in China reported that among 72,314 COVID-19 cases, 14% were severe and 5% were critical. 6 Guan et al. 25 reported that the proportion of severe disease was 15·7%.
In the clinical progression to severe in COVID-19 infection, the median duration from symptoms to dyspnoea was 8 days, to ARDS was 9 days, and to intensive care unit admission and mechanical ventilation was 10.5 days. 2 In our study, about 90% of patients with COVID-19 in Busan were hospitalised in the 2 study hospitals, and most of them initiated antiviral drugs early before exacerbation of their COVID-19; that is, from symptom onset to antiviral administration was 3 days. Considering the clinical course of COVID-19, antiviral drugs were administered relatively early in our study and 96% of patients did not need oxygen supplementation or mechanical ventilation. Only 4% of patients required mechanical ventilation. Compared to the result of the study from China, the proportion of severe patients in our study was low although our study included a considerable number of elderly patients and patients with significant extent of pneumonia. 6 Therefore, our findings suggest that early diagnosis and early administration of antiviral agents for COVID-19 may reduce the incidence of progression to severe disease and may be associated with better outcomes than if administration of antiviral agents is reserved for patients with severe and complicated disease.
Second, our study demonstrated that patients who received LPV/r were less likely to experience disease progression than those who received HCQ when the antiviral agents were administered early to patients with uncomplicated illness or mild pneumonia, and that these patients were rarely switched to a secondary drug. Conversely, 40% of the patients who were initially treated with HCQ were switched to LPV/r as salvage therapy due to the attending physician judging that the patient had treatment failure. Although not statistically significant, newly developed pneumonia was also less frequent in patients who received LPV/r than those who received HCQ. It is also noteworthy that the patients receiving LPV/r as the initial antiviral agent had relatively worse severity of disease and higher number of comorbidities than the patients who were treated with HCQ, suggesting that the physicians had a tendency to select LPV/r for patients who had poorer clinical condition. This might have masked the possible benefits of LPV/r treatment.
A recent randomised controlled trial demonstrated that LPV/r treatment did not significantly accelerate clinical improvement, reduce mortality, or diminish throat viral RNA detectability in patients with severe COVID-19. 24 However, in this study, the median interval from symptom onset to initiation of antiviral therapy was 13 days, and it is possible that the LPV/r treatment was initiated after clinical deterioration. 24 In a study on the use of LPV/r in patients with SARS, LPV/r was associated with reduced steroid use, a reduced need for mechanical ventilation, and lower mortality, when it was administered in the early stage, but did not show a therapeutic effect when the treatment was initiated at a later course of disease. 26 Based on these findings, we hypothesise that early LPV/r treatment for COVID-19 can improve clinical outcomes although LPV/r does not appear to have a clinically significant effect in patients with severe COVID-19.
Some recent studies have shown that HCQ is a potent inhibitor for SARS-CoV-2 in vitro.19,27 However, in our study, HCQ, compared to LPV/r, did not effectively prevent the clinical progression of COVID-19, and about 40% of patients who received HCQ had their antiviral agent switched due to clinically diagnosed failure. To date, no controlled trials of HCQ for SARS-CoV-2 infection have reported clinical outcomes other than a reduction of viral load.
Third, our study suggested that LPV/r could be used without major side effects in mild COVID-19 infection. The safety of LPV/r has been confirmed in individuals with HIV infection, and LPV/r has been used to treat HIV infection for a long time. Known side effects of LPV/r include diarrhoea, abnormal stooling, abdominal pain, nausea, vomiting, and asthenia. 28 In our study, 22 patients (49%) in the LPV/r group experienced adverse effects, and 2 patients (4%) interrupted their drugs due to adverse effects. One patient stopped LPV/r because of skin rash while the other patient stopped LPV/r because of loss of the sense of taste. It is likely that ageusia is a symptom of COVID-19 rather than an adverse effect of the drug.29-31 No life-threatening side effects have been identified in mild patients.
Our study has some limitations. First, the patients who were enrolled in our study were not randomly assigned to LPV/r and HCQ groups because our study was an observational study. Therefore, attending physicians’ prior knowledge or preconceptions of antiviral agents may have influenced their choice of antiviral agent and led them to prescribe LPV/r for more severe cases. Although the patients were not randomly assigned to LPV/r or HCQ groups, the Busan City Government distributed patients with confirmed COVID-19 patients to the 2 hospitals on a non-selective basis, and treatment practices differed by hospital, regardless of severity. This reduced the probability that relatively serious patients would preferentially receive LPV/r because PNUH used LPV/r as the initial antiviral agent in almost all the patients.
Second, there was no uniform protocol in our study because this was not an experimental study. Therefore, the standard of care may have differed between the 2 hospitals. However, the staff at both hospitals had to follow the KCDC and the Busan City Government’s guidelines that all patients with confirmed COVID-19 should be hospitalised in the isolation wards; that their clinical course should be observed during hospitalisation; and that they should use the KCDC criteria to decide when patients were cured and could be released from isolation. Therefore, the entire treatment period was observed in an isolated ward even though some patients had only mild symptoms. This allowed the clinical effects of the 2 drugs to be compared in settings similar to that of an experimental study, despite the lack of a uniform protocol.
In conclusion, our study demonstrated that rapid diagnosis and early administration of antiviral agent might prevent the progression of COVID-19 to severe disease requiring oxygen supplementation or mechanical ventilation. Based on our study results, HCQ shows no apparent advantage compared to LPV/r for preventing progression to severe disease in patients with COVID-19.
Supplemental Material
sj-pdf-1-avt-10.1177_13596535211039595 – Supplemental Material for Transforming growth factor-β: A good early predictor for functional cure in HBeAg-negative chronic hepatitis B patients treated with pegylated interferon
Supplemental Material, sj-pdf-1-avt-10.1177_13596535211039595 for Transforming growth factor-β: A good early predictor for functional cure in HBeAg-negative chronic hepatitis B patients treated with pegylated interferon by Li-Li Wu, Xiao-Yan Li, Hong Deng, Dong-Ying Xie, Yu-Rong Gu, Yan-Hua Bi, Yue-Hua Huang and Zhi-Liang Gao in Antiviral Therapy
Footnotes
Author contributions
Conceptualisation: SL, JEL, and SOL; data collection and curation: SL, JEL, SOL, JH, MRP, and DWK; formal analysis: SL and SOL; supervision: SHL, DKK, K-HK, and HS; writing—original draft: SOL; and writing—review and editing: SL, JEL, and SOL.
Declaration of conflicting interests
The authors declare no competing interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
