Abstract
Silver nanoparticles (AgNPs) are widely applied in the field of personal protection for their powerful toxic effects on cells, and recently, a new type of vaginal gel with AgNPs is used to protect the female reproductive tract from microbes and viruses. However, a high risk of AgNPs to the fetus and the underlying mechanism of AgNPs to interfere in embryo development still remain unclear. Thus, this study investigated the impact of two drugs of vaginal gel with AgNPs on reproductive capability of the female mouse by animal experiment. Then, kinetics of AgNPs affecting embryo development was investigated by in vitro embryos culturing, and cell membrane potential (CMP) of zygotes was analyzed by DiBAC4(3) staining. Results indicated that one of the drugs of vaginal gel certainly injured embryo development in spite of no apparent histological change found in ovaries and uteruses of drug-treated mice. In vitro embryo culturing discovered that the toxic effect of AgNPs on embryo development presented particle sizes and dose dependent, and AgNP treatment could rapidly trigger depolarization of the cell membrane of zygotes. Moreover, AgNPs changed the gene expression pattern of Oct-4 and Cdx2 in blastocysts. All these findings suggest that AgNPs can interfere with normal cellular status including cell membrane potential, which has not been noticed in previous studies on the impact of AgNPs on mammalian embryos. Thus, findings of this study alarm us the risk of applying vaginal gel with AgNPs in individual caring and protection of the female reproductive system.
Introduction
Nanometer-sized particles of Ag (AgNPs) attract vast interest for their efficiency in daily antibacterial products.1,2 More extensive research has provided solid evidence that AgNPs can damage mammalian reproduction at the level of organ, molecular, and hormone, which had been comprehensively reviewed in detail.3,4 Notably, AgNPs are even regarded as feasible drugs to prevent HIV or to care infection of female reproductive tracts from candidiasis, fungi, bacteria, and viruses,5–9 and more products of vaginal gel with AgNPs are coming out in the market, which gives more chances of AgNPs entering the female reproductive tract. It had been outlined that toxic effects of AgNPs on tissues and organs are tightly decided by dose, shape, and size of particles, and many studies investigated toxicological effects of AgNPs and discovered possible mechanisms of AgNPs involved in interference of RNA polymerase activity, 10 apoptosis by oxidative stress, 11 abnormal glucose metabolism, and disturbing differentiation of stem cells by inactivation of XIST.12,13 However, it remains poor understanding that what is the core mechanism of AgNPs directly disturbing embryo development because the existing data are obtained from the in vivo mouse model prepared by injecting AgNPs into the tail vein or gastric perfusion, which ignores the process and sequence of interactions between AgNPs and embryo cells.
It should be recognized that the probability of AgNP accumulation in the female reproductive system is very rare although AgNPs can impair cells in different manners by passing through the cell wall of almost all tissues because of their unique quality of nanometer size.14,15 However, application of vaginal gel containing AgNPs becomes an essential route that exposes embryos to AgNPs. Thus, the question of the effect of AgNPs on the female reproductive tract needs to be addressed. Today, it is well known that many extracellular factors can intensively impact the cell fate through changing cell membrane potential. For example, G protein-coupled receptors (GPCRs), such as P2Y1 receptors in megakaryocytes, M1 and M2 receptors in xenopus oocytes, and muscarinic receptors in smooth muscle and pancreatic acinar cells, are able to respond to the extracellular signals in a voltage-dependent manner during the process of Ca2+ release and activate GIRK channels. 16 Also, AgNPs were found to be capable of stimulating synaptic transmission in CA1 pyramidal neurons of the hippocampus, 17 and amplitude, amplitude and the frequency of spontaneous excitatory postsynaptic currents happened to change. Until now, we have learned that Ca2+ signals plays a key role in the development of mammalian embryos, 18 and ion channels in the cell membrane controls the level of cytosolic calcium during fertilization or interaction of embryos with the environment.19–21 Thus, it is reasonable to presume that the CMP of early embryos is probably affected by AgNPs, and thus AgNPs play a vital role in deteriorating embryo development besides their negative effects of inducing intracellular reactive oxygen species (ROS) on embryonic cells. Thus, the aim of this study is to explore the effects of AgNPs in vaginal gel on early embryo development and the impact of AgNPs on cell membrane potential.
Here, we explored the process of AgNPs affecting mouse embryo development in detail by using in vitro embryo culturing after analyzing the reproductive achievement of adult female mice treated with two drugs of vaginal gel with AgNPs for 7 days. Results suggested that vaginal gel with AgNPs will cause a higher risk to normal development of mouse embryos. This study also shows that a short-time exposure of embryos to AgNPs should not be ignored.
Materials and methods
Unless otherwise stated, all chemicals used in this study were purchased from Sigma (St Louis, MO, USA). Two drugs of vaginal gel with AgNPs obtained from market were named Drug A and Drug B. The concentration of AgNPs in Drug A was 1000 μg/mL and in drug B 350 μg/mL. To find the size of AgNPs in Drugs A and B, we analyzed Drugs A and B by high-resolution TEM. Results showed that the size of AgNPs is about 20–30 nm in Drug A and about 3–5 nm in Drug B. The corresponding figure is presented in Supplementary Material.
Animals and ethics statement
Virgin 6–8-week old ICR male and female mice were bought from Anhui Medical University and were housed in cages maintained under a constant 12 h light–dark cycle at 22–26°C with standard feed. This study was approved by the Animal Care and Use Committee of Fuyang Normal University. All animal experiments were carried out according to the guidelines of the Animal Care and Use Committee of Fuyang Normal University.
Experimental design
The overall procedure of this research is shown in Figure 1. First, to analyze the effect of vaginal gel with AgNPs on birth, the drug was put into the vagina of 8-week-old female mice according to the ratio of drug dosage applied in an adult woman. After that, these treated female mice were mated with male by 1:1, and mice with vaginal plug were picked out after 12 h. Twenty days later, the number of newborns was counted. In this test, each group consisted of 15 mice, and all the tests were repeated three times. Besides, ovaries and uteruses of mice were obtained 1 day after the last drug treatment and were checked with hematoxylin eosin staining. Then, we investigated the dose and size effect of AgNPs in embryo culture medium KSOM (simplex optimized medium) on embryo development.
22
A suitable dose and size of AgNPs were defined through finding out complete inhibition of blastocyst formation on day 4, when mouse eggs were activated on day 0. Then, in vitro fertilization (IVF) embryos were assigned to be cultured in the medium with or without AgNPs. Then, all embryos were used for subsequent study. Scheduling of the experiment.
Observation on tissues of mouse ovary and uterus
Ovaries and uteruses from mice of the control group and drug-treated group were fixed in 4% paraformaldehyde at 4°C until further steps. The sample was embedded in paraffin, sectioned, and stained with hematoxylin eosin as described in earlier studies. For the uterus, the middle segment of each uterus was used to examination.
Preparation and characterization of AgNPs
AgNPs used in this study were purchased from nanoComposix and stored in dark at room temperature. The preparation of AgNPs solution was according to the early index as described in Wang’s work. 10 In brief, the sample of AgNPs was suspended in deionized H2O after centrifugation to prevent Ag ions into a culture medium and then diluted with KSOM medium to working concentration for embryo culture according to the requirement of the experiment. The size of AgNPs was characterized by transmission electron microscopy (TEM, Hitachi H-7500, Japan). The hydrodynamic diameter and zeta potential were determined using a Zetasizer (Malvern Nano series, Malvern, UK), and the absorption spectrum was measured by UV–vis spectrometry (Beckman Coulter, USA).
AuNPs of 5–10 nm were obtained by the method as described in the earlier study. 23 In brief, 0.05% polyethyleneimine (PEI) solution with a molecular weights of 25,000 and 1800 was mixed in an equal volume ratio, then 700 μL of fresh 2% HAuCl4 solution was added into 16 mL of freshly prepared PEI solution, and the final volume was made to 22 mL with ultrapure water. Then, at room temperature, the mixture was stirred at 800 rpm/min for 48 h, filtered with a membrane of 0.22 μm pore size, and stored at 4°C.
Preparation of mouse oocytes, IVF, and preimplantation embryo culturing
For the more details of the experiment, refer to previous work.24,25 MII oocytes collected from the oviducts 14 h after human chorionic gonadotropin (HCG) 5–10 IU injection were transferred into the human tubal fluid (HTF) medium containing 4 mg/mL bovine serum albumin (BSA fraction V), and then IVF was carried out as follows: each cauda epididymis of male mice was obtained, and sperms were released into the HTF medium for capacitation for 2 h. Capacitated sperms were transferred into the HTF medium containing MII oocytes for about 1.5 h, and then they were co-cultured for 4–6 h in a cell incubator with 5% CO2 at 37°C. Fertilized eggs possessing two pronuclei were cultured for 4 days until blastocysts were formed normally.
Detection of the expression of Cdx2 and Oct-4 in blastocysts
The procedures for indirect immunofluorescence were performed as described in a previous study. 24 The blastocysts were fixed with 4% paraformaldehyde after being washed three times with phosphate-buffered saline (PBS). The fixed embryos were permeabilized with 0.5% Triton X-100 at room temperature for 30 min. After washing several times, the embryos were blocked with PBS (containing 1% bovine serum albumin) at room temperature for 1 h. Then, the blastocysts were stained with monoclonal anti-Cdx2 antibody (BioGenex MU392A5UC) and anti-Oct-4 antibody (Abcam Ab27985) at 4°C overnight. After washing three times, they were incubated with donkey anti-goat antibody (Thermo Fisher, A32814) and donkey anti-mouse antibody (Thermo Fisher, A31571). Finally, the chromatin was stained with Hoechst33342 (Sigma B2261) at 37°C for 15 min, followed by washing several times. The embryos were mounted on slides and were observations as shown in an previous study. 25
Monitoring cell membrane potential (CMP) of embryos by DiBAC4(3) staining
Analyzing the change in the CMP of embryos in this study was mainly referred to previous studies and carried out as follows.26–28 DiBAC4(3) (Sigma, D8189) was dissolved in DMSO at 5 mM and stored at −20°C, and the embryos were transferred into the M2 medium containing DiBAC4(3) of 5 μM for 30min and then transferred into the M2 medium containing AgNPs with different concentrations, or M2 medium supplemented with KCl at 150 mM as a positive control. All samples were observed with fluorescence at an excitation wavelength of 488 nm. The fluorescence intensity was surveyed every 1 minute continuously. The regent H2O2 with a final concentration of 500 mM in the M2 medium was used for analysis of difference between AgNPs and H2O2.
Statistical analysis
All data were analyzed by one-way analysis of variance (ANOVA) for assessing any significant difference among the different groups. Only probabilities lower than 0.05 were considered significant, and lower than 0.01 were considered extremely significant.
Results
The drug of vaginal gel with AgNPs reduced the birth number of pups, but did not make any abnormal histology of the ovary and uterus
Effect of vaginal gel with AgNPs on reproductive capability of female mice.

aGroup A represents Drug A-treated mice, group B represents Drug B-treated mice, and group C represents untreated mice.
bAnalysis of significant difference between the drug treatment groups and the control group.
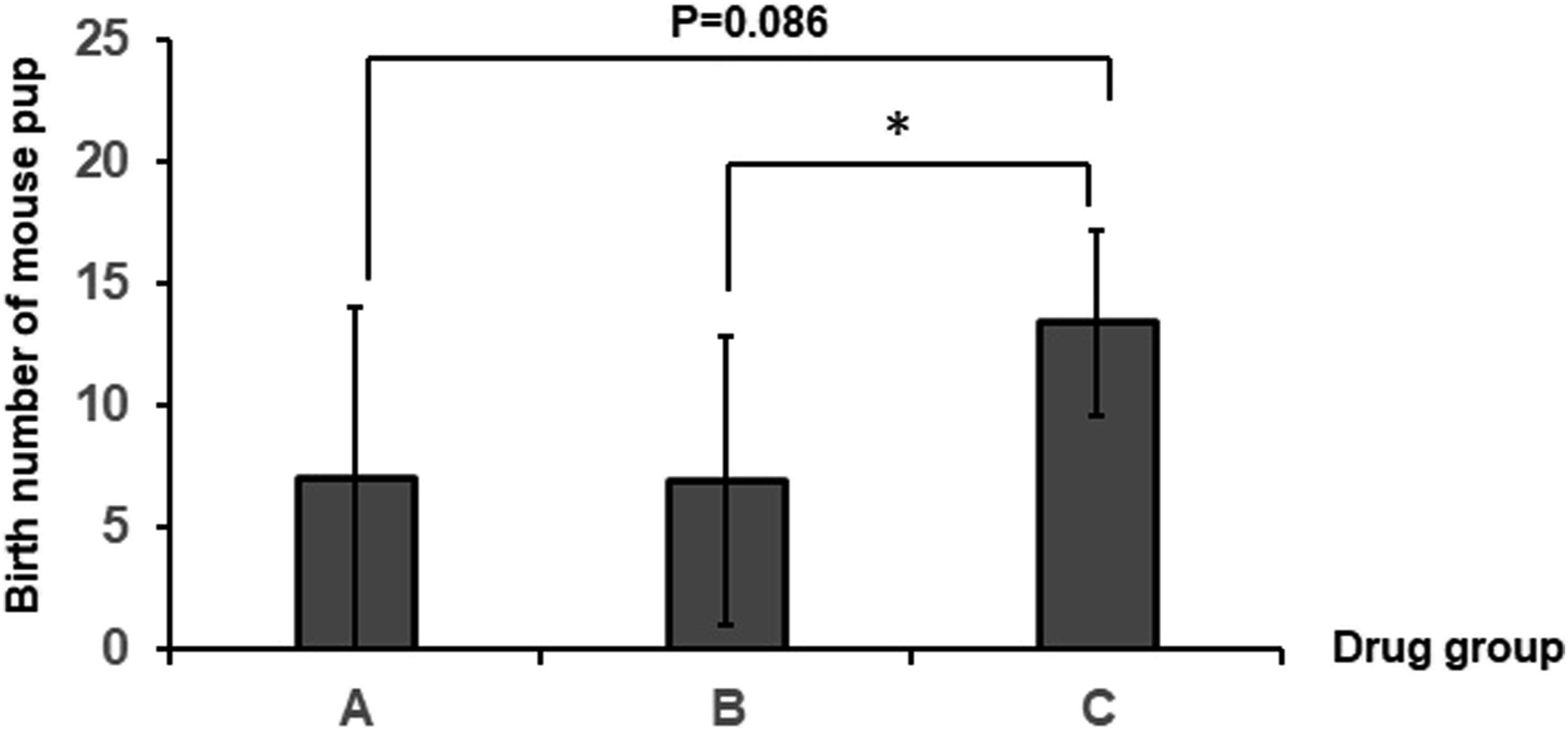
Comparison of reproductive capability among Drug A- and Drug B-treated mice and control groups. Obvious reduction in the birth number of pups occurs in the Drug B group, but does not occur in the Drug A group.
Effects of AgNPs on the development of mouse early embryos are size and dose dependent
To make a standard assessment of toxic effects of AgNPs on early embryo development, the impact of AgNPs with different sizes and concentrations on fertilized eggs was studied. Zygotes after 6 hours of IVF were transferred into the KSOM medium supplemented with AgNPs and cultured for 96 h until the blastocyst stage, and then the whole process of embryos culturing was regularly observed and noted. The preimplantation development of embryos treated with 20 nm AgNPs with different concentration is shown in Figure 3(a), and embryos treated with 10 nm AuNPs at 5 μg/mL were taken as a parallel control. Results showed that AgNPs obviously inhibited embryo development at 0.5 μg/mL and above, but blastocysts formation was hardly blocked in the 5 μg/mL AuNP treatment group. Effect of AgNPs with different sizes and concentrations on early embryo development. 
Then, the effects of size and concentration of AgNPs on early embryo development were investigated. Data showed that development of early embryos in all AgNPs-treated groups presented an obviously inhibitory trend, a result similar to that of previous studies of toxic effects of AgNPs on cells (Figure 3(b)–(e)). It was found that 5 nm AgNPs have the most serious impact on embryo development and even completely inhibited blastocyst formation at a concentration of 300 ng/mL. However, 80 nm AgNPs could not completely block blastocyst formation until its concentration reached 2 μg/mL. All these suggest that the inhibitory effect of AgNPs on embryo development should be size and dose dependent, and when size of AgNPs is 40 nm or less, AgNPs can inhibit embryo development even at a concentration of 0.5 μg/mL. In addition, an obvious character is that inhibition of embryo development by AgNPs mainly occurs at the 2-cell stage during the whole preimplantation stage, which means AgNPs damage embryos before the 2-cell stage. On the contrary, no visible injury by AuNPs in embryo development was observed. Therefore, to clarify the effects of AgNPs on embryo development, AgNPs with a size of 20 nm were used in the subsequent work. One question that how long embryos should be exposed to AgNPs to influence embryo development needs to be answered. Thus, the minimum exposure time of AgNPs with different concentrations in the medium to block blastocysts formation was investigated by culturing zygotes in the KSOM medium containing 20 nm AgNPs for different culturing durations. Data showed that blastocyst formation was obviously blocked by AgNPs at concentrations of 0.5 μg/mL for 12–24 h and 2 μg/mL and 5 μg/mL for 3–6 h (see Supplementary Material). These suggest that a shorter time exposure of AgNPs to early embryos would cause a significant negative impact on long-term development of embryos.
To uncover if silver ion possibly results from AgNPs and affects development of mouse early embryos, we obtained the supernatant of the KSOM medium with AgNP-cultured early embryos and then analyzed concentration of silver ions by ICP-MS (a universal method to test AgNPs). Results indicated that concentration of silver ion in the medium KSOM only reached 0.196 μM when 20 nm AgNPs were co-incubated with embryos for 4 days at 0.5 μg/mL. Meanwhile, the effect of silver ions on development of mouse early embryos was analyzed through a serial dilution of AgNO3 in the KSOM medium to culture mouse early embryos. Four concentrations of silver ions in KSOM were checked, and the results showed that silver ions can effectively block embryo development at a concentration of 10 μM and above (see Supplementary Material). Thus, comparatively, silver ion concentration is too low in the KSOM medium containing AgNPs to effectively affect development of mouse early embryos.
AgNP treatment caused abnormal cell differentiation in blastocysts
From the aforementioned results, it was confirmed that AgNPs in the medium indeed influence embryo development. But, a majority of zygotes in AgNP treatment groups presented normal morphology and no obvious difference as compared to the control group (Figure 4(a)). Even to survived blastocysts of AgNP treatment groups, no obvious morphological change was found (Figure 3(a)). All these indicate that the morphology of embryos is unsuitable to gauge embryo developmental status. To discover the true effect of AgNPs on survived blastocysts, the expression pattern of Cdx2 and Oct-4 in blastocysts was analyzed in that they are key marker genes of trophoblast ectoderm (TE) and inner cell mass (ICM), respectively, in blastocysts for cell differentiation, and AgNPs of 0.5 μg/mL were adopted here. By using immunofluorescence detection with anti-Oct-4 and anti-Cdx2 antibody, the profile of cell differentiation can be analyzed easily. Results showed that expression level of Oct-4 was significantly higher in AgNP-treated embryos than in the control group, but no differences were found in Cdx2 between the AgNP-treated group and the control group (Figure 4(b)–(d)). These suggest that AgNPs definitely affect early embryo development by interfering differentiation of cells in embryos. Expression of Oct-4 and CDX2 in blastocysts from AgNP treatment. (a) Morphological comparison of zygotes between treatment by 20 nm AgNPs at 5 μg/mL and the control group. No obvious difference can be found under bright field of the optical microscope. (b) Expression pattern of CDX2 and Oct-4 in blastocysts resulted from AgNPs treatment and control groups. (c) Expression of Oct-4 in 20 nm AgNPs at the 0.5 μg/mL treatment group is significantly higher than in the control group (p < 0.05), and (d) no difference in CDX2 expression between the two groups.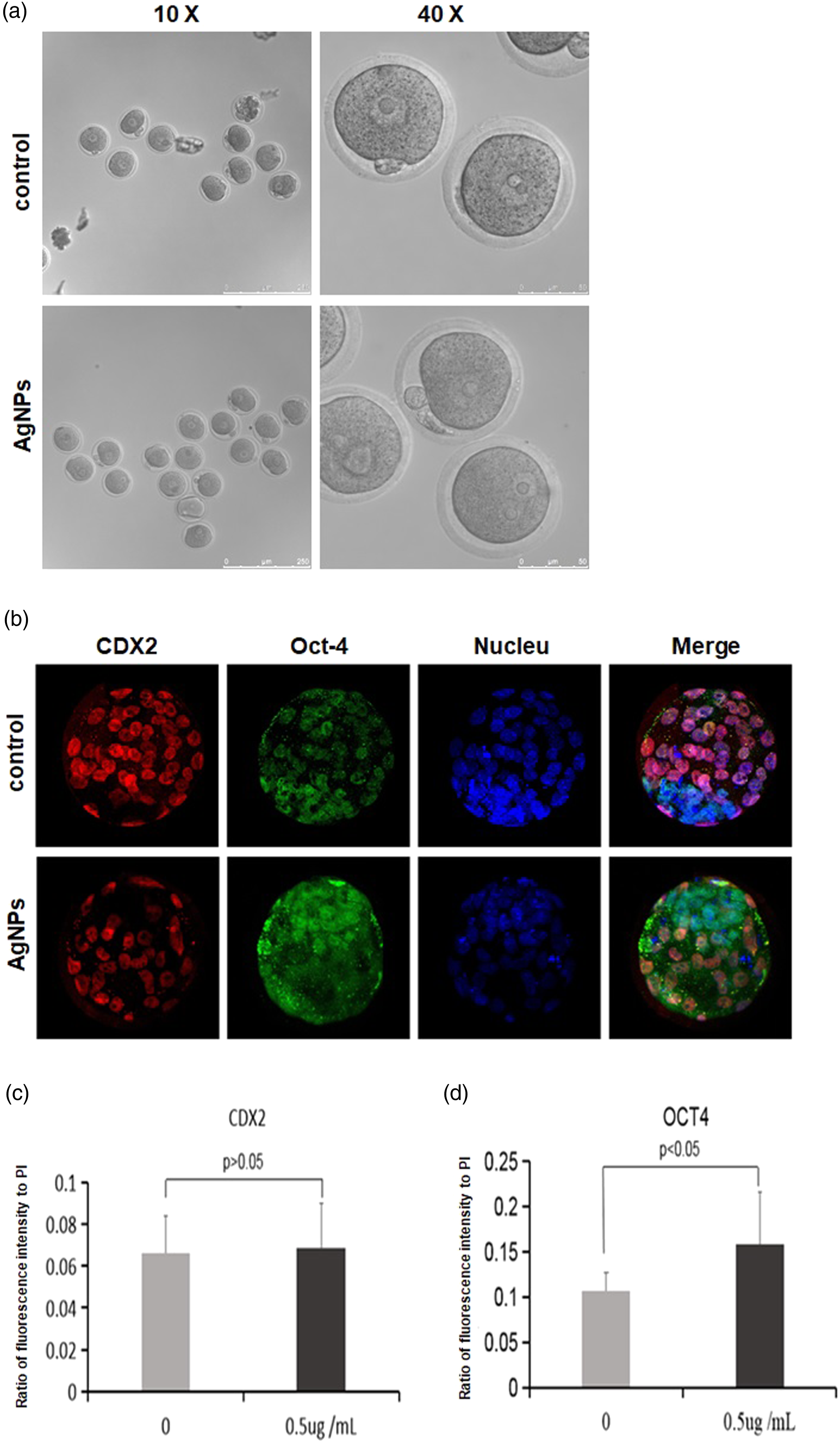
AgNPs rapidly made cell membrane depolarization in embryonic cells
Early research had suggested that AgNPs can induce a higher level of intracellular ROS in embryos, and the same result was obtained in our study. Thus, a higher level of ROS by AgNPs certainly plays an important role in impairing embryo development, particularly while embryos are exposed to toxic substances for a long time. However, another possible damage of AgNPs to embryos may be achieved through a rapid interaction between AgNPs and cell membranes to change cell membrane potential (CMP) of embryonic cells, which subsequently triggers the activation of signaling pathways involved in apoptosis and others. Thus, we surveyed if AgNPs could rapidly influence CMP of zygotes by using DiBAC4(3) staining, in which three different concentrations of AgNPs at 0.5 μg/mL, 2 μg/mL, and 5 μg/mL were adopted. Meanwhile, KCl (150 μM) and H2O2 (500 μM) were used to align the authenticity of data for KCl as being a positive reagent to depolarization of CMP. Results (in Figure 5(c) and (d)) exhibited that an obvious change of CMP lasted for 5 min in zygotes treated with AgNPs, and then the changed potential gradually restored to the normal status after treatment for about 15 min. From Figure 5(a) and (b), it could be noticed that H2O2 also greatly changes CMP possibly because of H2O2 being a stronger oxide to denature proteins and destructing cell membranes. All these suggested the value of exploring the role of CMP in the damage caused by AgNPs to embryos (Figure 5). Change in the cell membrane potential (CMP) of zygote treated with AgNPs with different concentrations. (a, b) Effect of KCl and H2O2 on CMP of zygotes as being positive control to AgNPs. (c, d) Obvious difference of changed CMP by AgNPs with three concentrations: 0.5 μg/mL, 2 μg/mL, and 5 μg/mL. Values of fluorescence intensity with DiBAC4(3) staining were recorded at regular intervals after embryos were transferred into the medium containing AgNPs. An obviously correlated change could be observed in concentrations of AgNPs in the medium. Comparison of CMP changing among all four groups. It could be found that depolarization of the cell membrane by KCl is stronger than by AgNPs and last for longer time.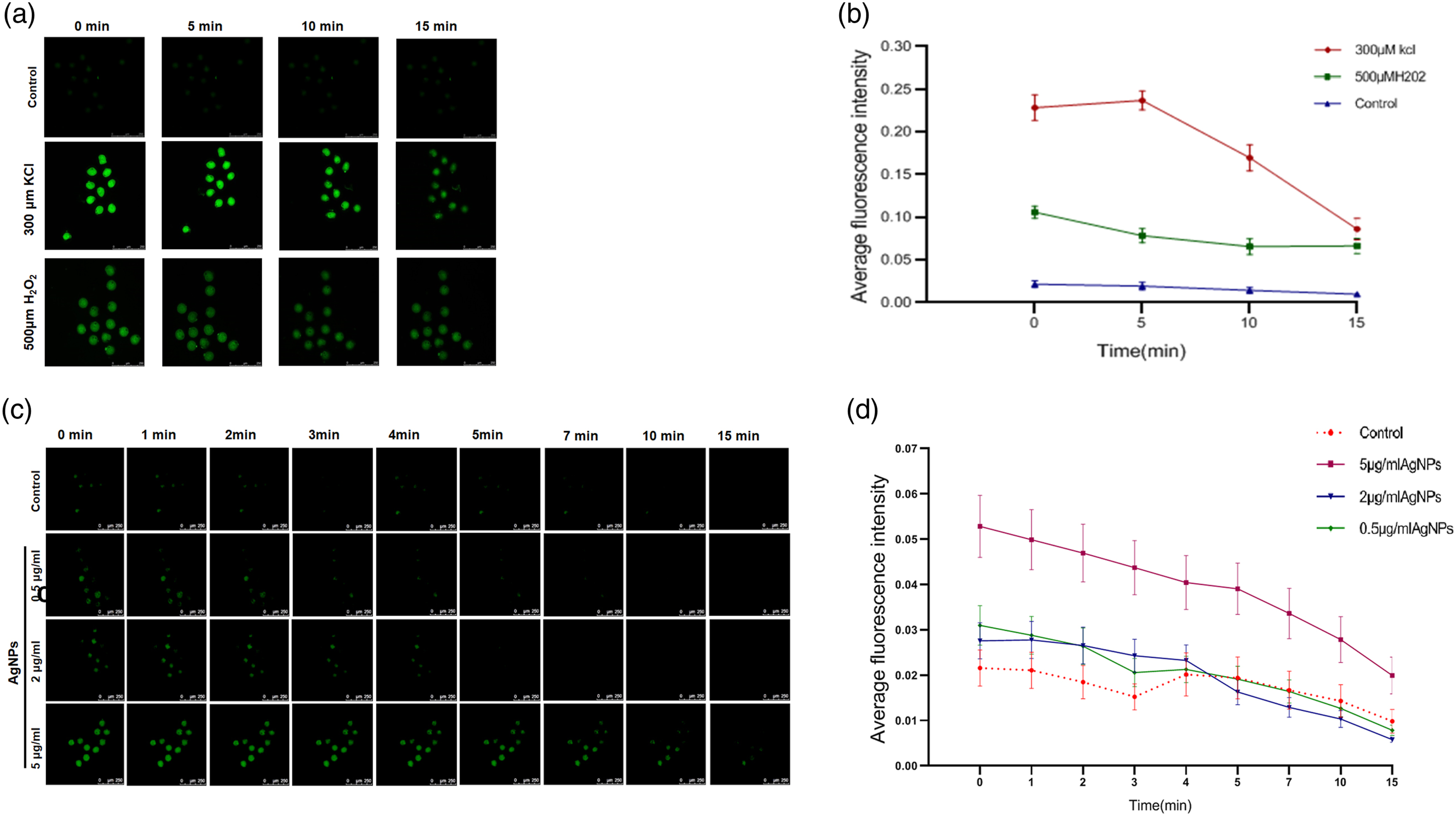
Discussion
A lot of studies had explored the effects of AgNPs on almost all of animal tissues and physiological activities including the liver, lung, kidney, brain, and even reproductive organs. However, a key question that what is the immediate effect of AgNPs on embryos remains unclear because most of research studies used the model of gastric perfusion or tail vein injection. Here, our findings in this study are as follows: (1) vaginal gel with AgNPs is harmful to the female reproductive system; (2) AgNPs can deteriorate embryo development without requirement of a long-time exposure of embryos to them; and (3) extracellular AgNPs can effectively and rapidly change CMP of embryos.
Results in this study showed that vaginal gel with AgNPs can reduce the birth number of female mice, although no abnormality is found in the ovary and uterus of drug-treated mice. We did not investigate the contribution of silver ions versus AgNP size, and we found that the concentration of silver ions is low in the embryo culture medium KSOM with AgNPs relative to the concentration of AgNO3 dilution of the KSOM medium, in which mouse early embryos can normally develop to blastocysts (see Supplementary Material). Thus, it can be reasonably speculated that the effect of AgNP size afford decisive roles in their damage to embryo development. In this regard, other studies on the toxicity of AgNPs on a cell had yielded similar results. Previous studies on the toxic effect of AgNPs on the animal cell had discussed this problem and defined that the toxicity of AgNPs mainly depends on their size; although silver ions also play a certain role, “particle-specific” effects should be predominant mediators in their effect on cells under low dose of AgNPs. 10
With regard to the mechanism, it is accepted that a higher level of ROS exacerbates cellular physiological activity including mitochondrion, membrane structure at subcellular level, metabolism of the one-carbon unit cycle, normal function of RNA polymerase, and DNA structure. But, an unexpected effect of AgNPs on CMP was discovered, which provides a new insight to explore the immediate effect of AgNPs on embryo development. It prompts that a short-time exposure of early embryos to AgNPs probably leads to a serious outcome of embryo development, for which studies have suggested that the events in eggs activation such as calcium signaling are capable of influencing the embryo development. The cell membrane is the border and transductor of intracellular environment to recognize an external signal, and cell death or apoptosis can be triggered by a cell membrane-mediated extracellular signal. 29 Existing studies indicate that a change in CMP, a key bioelectrical signal, is involved in modulating multiple important cellular activities including cell differentiation, cell cycle, secretion, cell migration, and tumorigenesis.30–33 Importantly, there is a correlation between CMP and potential of cell proliferation. 34 Like tumor cells, both zygotes and early embryonic cells are depolarized relative to highly differentiated somatic cells. 35 Thus, although research has not yet investigated the impact of AgNPs on CMP of embryonic cells, it is worthy to find out if extracellular AgNPs might alter CMP of zygotes and embryonic cells and further trigger activation of a signaling pathway involved in cell apoptosis or death.
In the regulation of CMP, TRP and KV are key factors because they decide the status of CMP transduced signals in the subsequent steps. A change in CMP will influence membrane channel proteins to control various ion influxes, calmodulin and its related kinase, MAPK, and so on. Meanwhile, depolarization of CMP possibly activates voltage-dependent calcium ion (Ca2+) influx, which causes an increase in the intracellular Ca2+ concentration and triggers signal transduction through stimulating protein tyrosine phosphorylation or MAPK pathway.36–38 In mammalian reproduction, it had been well known that Ca2+ signaling in fertilization plays a vital role in egg activation and normal development of embryo,24,39 and the inhibitor of ion channel can effectively affect early embryo development.40,41 Therefore, our evidence of AgNPs depolarizing CMP of zygotes strongly suggests that it should not be ignored to explore effects of extracellular AgNPs on CMP of eggs, zygotes, and embryos, and this effect has been partly confirmed by other previous studies including those of carbon nanotubes being ion channel blockers 42 and AgNPs inhibiting action potential in pyramidal neurons. 17 Although our data provided primary evidence of AgNPs influencing CMP of zygotes, further studies should explore why AgNPs can depolarize CMP of embryonic cells, and what the key compound is in the process of AgNPs changing CMP. Application of the patch-clamp technique and inhibitor of ion channels can be useful to discover the underlying mechanism as studies on channel of K+, Ca2+, and Na+ have conducted.
Several studies have suggested a lower level of Caspase-3 expression in mouse preimplantation embryos. Although our work did not answer that if the change in CMP of zygotes might directly activate Caspase-3, numerous studies have indicated that extracellular signals are capable of activating Caspase-3 and triggering apoptosis and cell death by mediation of Caspase-8, and apoptosis can also be induced through cytochrome C released from damaged mitochondrial membranes to activate Caspase-3. 43 Thus, choosing a possible downstream receptor of the CMP regulated pathway is the precondition for exploring the toxic effects of AgNPs on embryo development.
In the field of health care, AgNPs are applied in antibiotic drugs in skin and wound infection, by which the quantity of them absorbed into the human body is extremely rare and incapable of affecting embryos directly. However, an increasing number of productions, especially AgNP vaginal gel, greatly expands the opportunity of human oocytes and embryos to contact with AgNPs. These productions are easily obtained from hospital or pharmacy, despite no safety assessment data on human reproduction. In this study, data show that AgNPs with a size of 20 nm and below can greatly harm embryo development. Thus, it is reasonable to speculate that the accumulative amount of AgNPs in the oviduct and uterus might bring the risk to embryo development. Together with the results here, a serious question should be asked that what is the long-term and potential impact of vaginal gel with AgNPs on development of human embryos when it is being a routine therapy drug to diseases of the reproductive tract. Our study suggests that further studies should investigate if AgNP vaginal gel can diffuse to the oviduct and influence embryo development in living animal models.
In summary, all above suggest that ability of AgNPs to injure embryo development can be illustrated on the general injury of oxidative stress from ROS and the immediate effect on CMP by a non-invasive manner. Although no other study has yet investigated whether AgNPs can affect CMP of embryonic cells as so far, our work provided preliminary but credible proofs. More importantly, a non-invasive interaction between AgNPs and embryonic cells could play a crucial epigenetic effect on long-term embryo development, which needs be avoided for humans in vigorous development of nanotechnology today.
Supplemental Material
sj-pdf-1-het-10.1177_13596535211080235 – Supplemental Material for AgNPs reduce reproductive capability of female mouse for their toxic effects on mouse early embryo development
Supplemental Material, sj-pdf-1-het-10.1177_13596535211080235 for AgNPs reduce reproductive capability of female mouse for their toxic effects on mouse early embryo development by Di Zhang, Fangfang Yu, Huanhuan Li, Qiuyue Wang, Meiya Wang, Hongli Qian, Xiaoqing Wu, Fengrui Wu, Yong Liu, Shuanglin Jiang, Pu Li, Rong Wang and Wenyong Li in Human & Experimental Toxicology
Footnotes
Acknowledgments
The authors thank the National Natural Science Foundation of China for the financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by grants from the Key Program of National Natural Science Foundation of China (No. 21637004), Grant of Anhui Provincial Natural Science Foundation (1908085MC85), Key Grant of the Natural Science Research Program of Anhui Higher Education Institutions of China (Nos. KJ2017A339, KJ2018A0351, KJ2018A0352, and KJ2019A0514), Grant from the Innovation Project for College Students of Anhui Province (S202010371100), and Project of School Grade Research in 2020 (2020PTFY01ZD).
Supplemental material
Supplemental material for this article is available online.
