Abstract
Tartrazine (TTZ) is an azo dye used as a colorant in food products, drugs, and cosmetics. The present study evaluates the impacts of TTZ on embryonic development of zebrafish (Danio rerio). Laboratory-raised D. rerio embryos (n = 20/concentration) were exposed to graded dilutions of TTZ (0, 0.1, 1, 2, 3, 4, 5, 10, 20, 30, 40, 50, 75, and 100 mM) from gastrulation stage (5.25 hours postfertilization [hpf]) until hatching and developmental trajectory was traced up to day 7. The no observed effect concentration (NOEC), median lethal concentration (LC50), median effective concentration (EC50), and teratogenic index (TI) were calculated. Exposure of embryos to < 10 mM TTZ had no effects; 20 to 30 mM TTZ caused tail bending, cardiac and yolk sac edema in 50% of larvae; in 30 to 50 mM TTZ-exposed embryos the heart rates declined along with the above mentioned deformities, causing mortality within 96 to 144 hpf; development ceased completely at 75 to 100 mM concentration. The NOEC and LC50 were recorded at 5 and 29.4 mM dose, respectively. The EC50 values for heart rate, cardiac edema, tail bending, and hatching success were at 59.60, 53.81, 98.28, and 58.97 mM with TI quotient 0.49, 0.54, 0.29, and 0.49, respectively. We conclude that TTZ is not embryo toxic/teratogenic for zebrafish embryos up to a dose level of 10 mM concentration.
Introduction
Natural and artificial azo dyes (bearing azo group as part of their structure) are widely used as coloring agents for foodstuffs, drugs, and cosmetics. Tartrazine (TTZ) is a mono azopyrazolone dye used as a colorant in bakery products (decoration and coatings), flavored cheese, carbonated drinks, juices, toothpaste, drugs, and cosmetics. 1 In addition, this dye is also used as a substitute for saffron in tropical cuisine. 2 Although the acceptable daily intake (ADI) of TTZ for humans is 7.5 mg/kg body weight, 3 prolonged use of this dye at higher concentrations is reported to increase the number of lymphocytes and eosinophils in gastric mucosal cells, produce abnormal reproductive cells, cause hepato/renal toxicity, and neurobehavioral alterations in laboratory rodents. 4 –7 Studies on safety limits of this food additive (FA) for developing embryos/gestating woman are not well-documented.
In recent years, zebrafish (Danio rerio) has emerged as a versatile vertebrate model to study mechanisms of development. 8 Easy maintenance, tiny size, shorter life cycle, ex utero development, and transparency of embryo have made this fish an attractive research model in genetics and development. 9 Consequently, zebrafish embryos are widely employed in alternative protocols for assessing developmental toxicity, as a predictive model of mammalian toxicity. 10,11 The present study aims to set a developmental toxicity assay for testing the embryo toxic/teratogenic potentials of TTZ using laboratory-raised zebrafish embryos as model system.
Materials and Methods
Procurement and Maintenance of Zebrafish in Laboratory
Adult farm-bred wild-type zebrafish (Danio rerio) were obtained from the wholesale distributors Pet-Bonding, Bangalore, India, and were housed in aquaria containing conditioned water with the prescribed range of physicochemical parameters defined for maintenance of zebrafish in laboratory. 12 They were acclimated to laboratory conditions (temperature 27°C ± 1°C; photoperiod of 14 hours light and 10 hours of darkness). Fish were fed twice a day with freeze-dried tubifex worms with stabilized vitamin C purchased from Taiyo Feed Mill Pvt Ltd, Chennai, India. Food was also supplemented with live brine shrimps (Artemia nauplii) ad libitum twice a week. 13 Permission to work on zebrafish was obtained from the Animal Ethical Committee, CPCSEA (Committe for the Purpose of Control And Supervision of Experiments on Animals), India, under Institutional Registration #639/GO/02/a/ CPCSEA.
Breeding and Raising Embryos
Following acclimation, gravid females and males in the ratio 1:2, respectively, were placed in a breeding trap made out of nylon mesh with a pore size of 3 mm (that allowed oviposited eggs to reach the bottom of the aquarium). One hour after the last feeding of the day, the breeding trap was immersed in a glass aquarium filled with conditioned water. Fish were left undisturbed overnight. On the next morning daybreak, numerous fertilized eggs were found collected at the bottom of the aquarium, which were carefully siphoned using a Pasteur pipette into the Petri dishes and washed twice or thrice thoroughly with distilled water to get rid of dirt/waste particles. 14,15 They were then transferred to a Petri dish containing freshly prepared embryo (E3) medium. 16
Embryo and Exposure Media
Tartrazine was purchased from Sigma-Aldrich, St. Louis, MO, USA, and other chemicals required to prepare embryo medium were purchased from Hi-media, Mumbai, Maharashtra, India. The E3 embryo medium was prepared using a standard protocol. 16 All reagents were prepared freshly. Tartrazine (water soluble) was dissolved in E3 medium just prior to exposure.
Experimental Design
The fertilized eggs were carefully sorted from unfertilized eggs (coagulated, damaged, asymmetric, and nontransparent) under a stereomicroscope (Leica MZ6, Leica Microsystems, Bangalore, Karnataka, India) and placed in a Petri dish containing embryo medium (E3) as per Organization for Economic Cooperation and Development regulation (2013). Zebrafish embryos (n = 20/concentration) were exposed to 0, 0.1, 1, 2, 3, 4, 5, 10, 20, 30, 40, 50, 75, and 100 mM dilutions of TTZ. The experiments were carried out in triplicate sets each time and repeated thrice. Developmental anomalies like pericardial edema and heart rate (beats/minute), yolk sac edema, tail distortion, hatching, and mortality rates were recorded. 17 Observations were made under Olympus bright field microscope (Olympus BX51, Japan) at 24, 48, 72, 96, 120, 144, and 168 hpf and photographed using ProgRess 3 camera (Jenoptik Germany).
Calculation of Median Lethal Concentration, Median Effective Concentration, and Teratogenic Index
For the calculation of median effective concentration (EC50) and median lethal concentration (LC50), linear regression was used. 18 In order to estimate the teratogenic potential of the test substance, teratogenic index (TI), the quotient of LC50 and EC50 were calculated for each teratogenic end point.
Statistical Analysis
The data were analyzed using 1-way analysis of variance and the differences between the means were assessed using Tukey multiple comparable test (SPSS version 16.0).
Results
Controls
All the embryos of the control group exposed to E3 medium alone, underwent normal development and hatched around 72 hpf (Figure 1). The newly hatched larvae had a straight tail detached from yolk sac, normal heart, and heart rate of 140 to 156 beats per min. Larvae belonging to this group were active, healthy, and survived for several weeks after hatching.
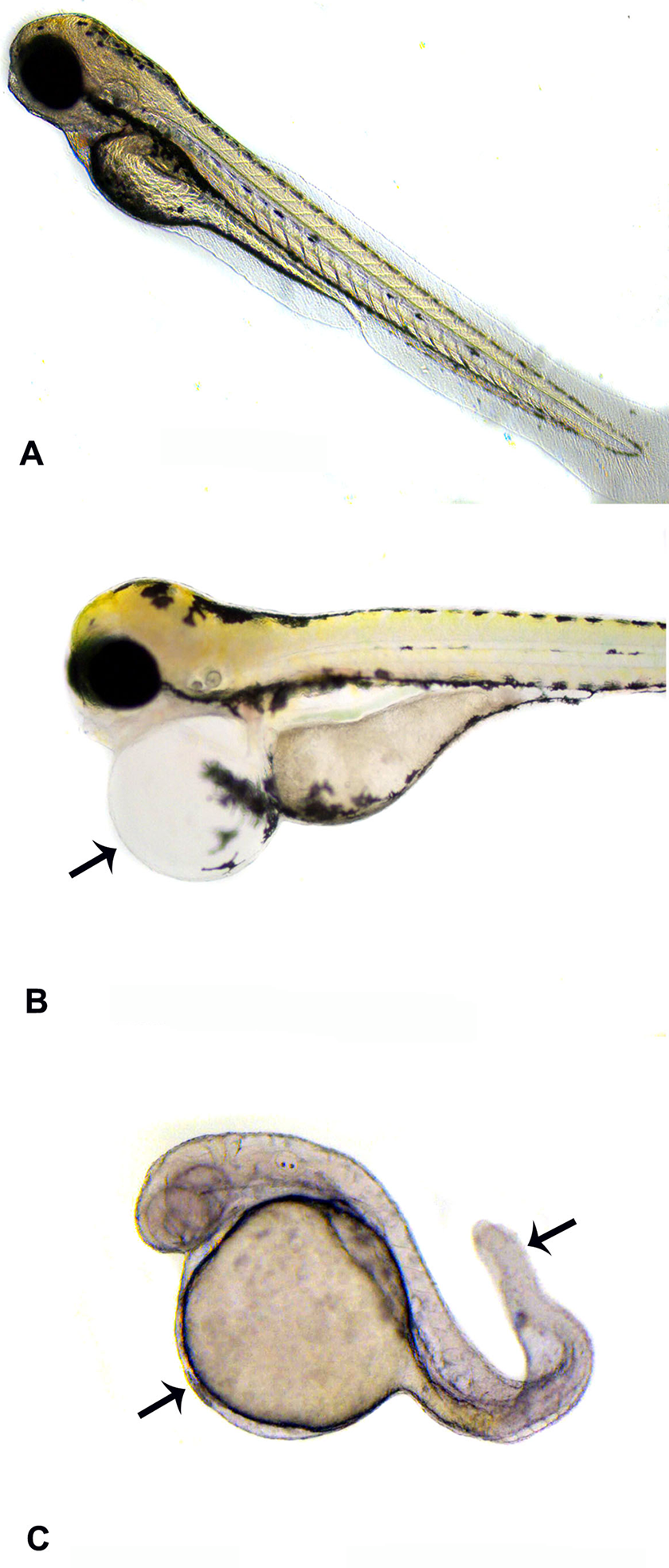
A-C, Larvae of embryos, belonging to control group (A); exposed to 30 mM TTZ showing cardiac edema (arrow) at 96 hpf (B); exposed to 40 mM TTZ showing yolk sac edema and tail distortion at 96 hpf (C). TTZ indicates tartrazine; hpf, hours postfertilization.
Effects of Exposure to TTZ
Eggs exposed to 0.1 to 5 mM TTZ underwent normal development comparable to controls and hatched into larvae at 72 hours that survived 7 to 10 dpf. In the larvae of 10 mM TTZ-exposed embryos, no significant changes were noticed compared to controls. Hatching was advanced (< 48 hpf) and 50% of early hatched larvae exhibited tail distortion, yolk sac edema, and cardiac edema in 20 to 30 mM TTZ-exposed groups (Figures 1 –3). In 40 to 50 mM TTZ-exposed embryos in addition to the above deformities, the larvae exhibited a decline in heart rates followed by mortality within 96 to 144 hpf (Figures 1 –3). Development was hampered and >50% embryos died in 50 mM TTZ-exposed group, while in 75 to 100 mM TTZ-exposed embryos, development ceased completely by 48 hours, resulting in mortality (Figure 4).
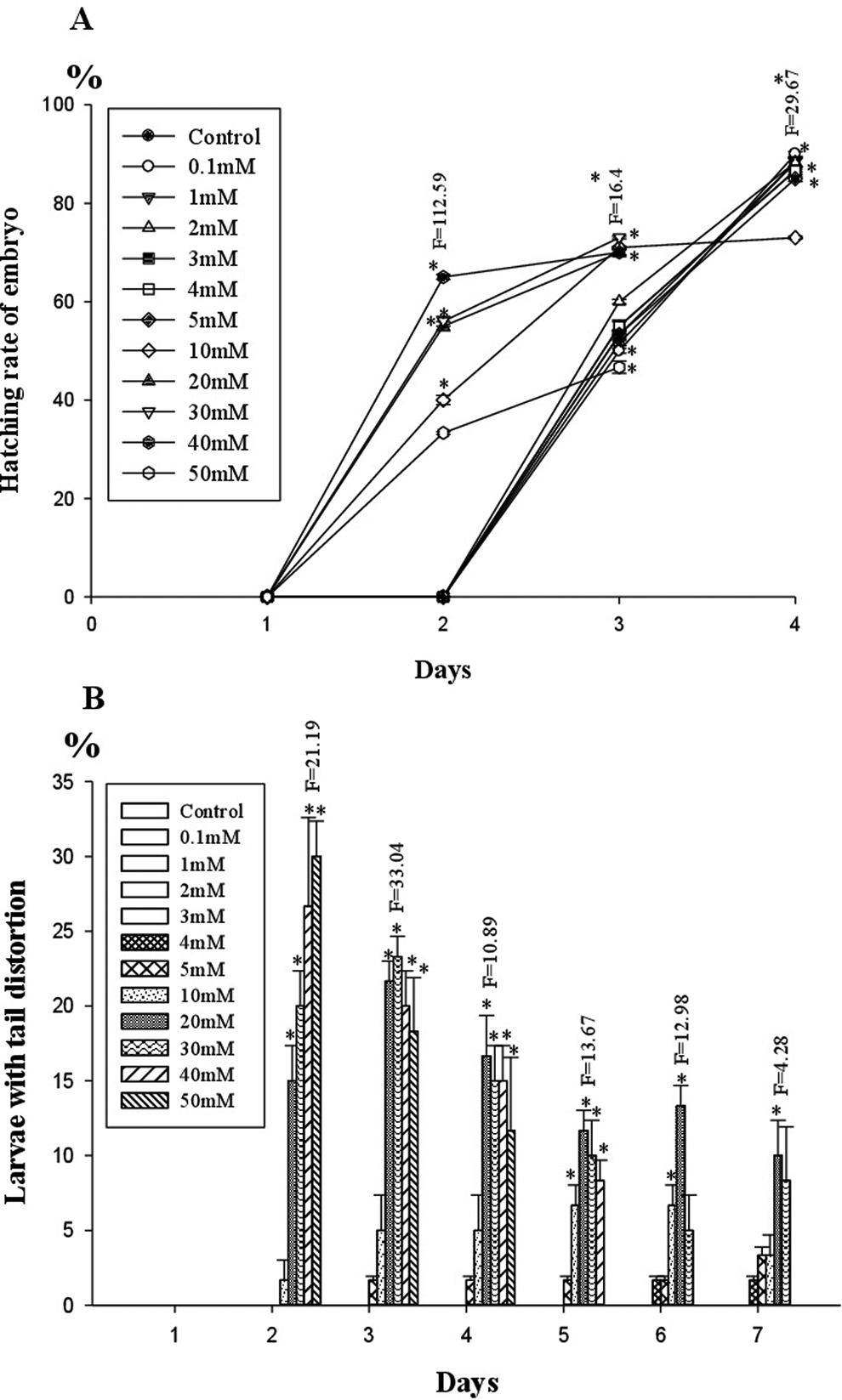
A-B, Hatching rate and tail distortion in larvae exposed to TTZ. *Significant at 5% level. TTZ indicates tartrazine.
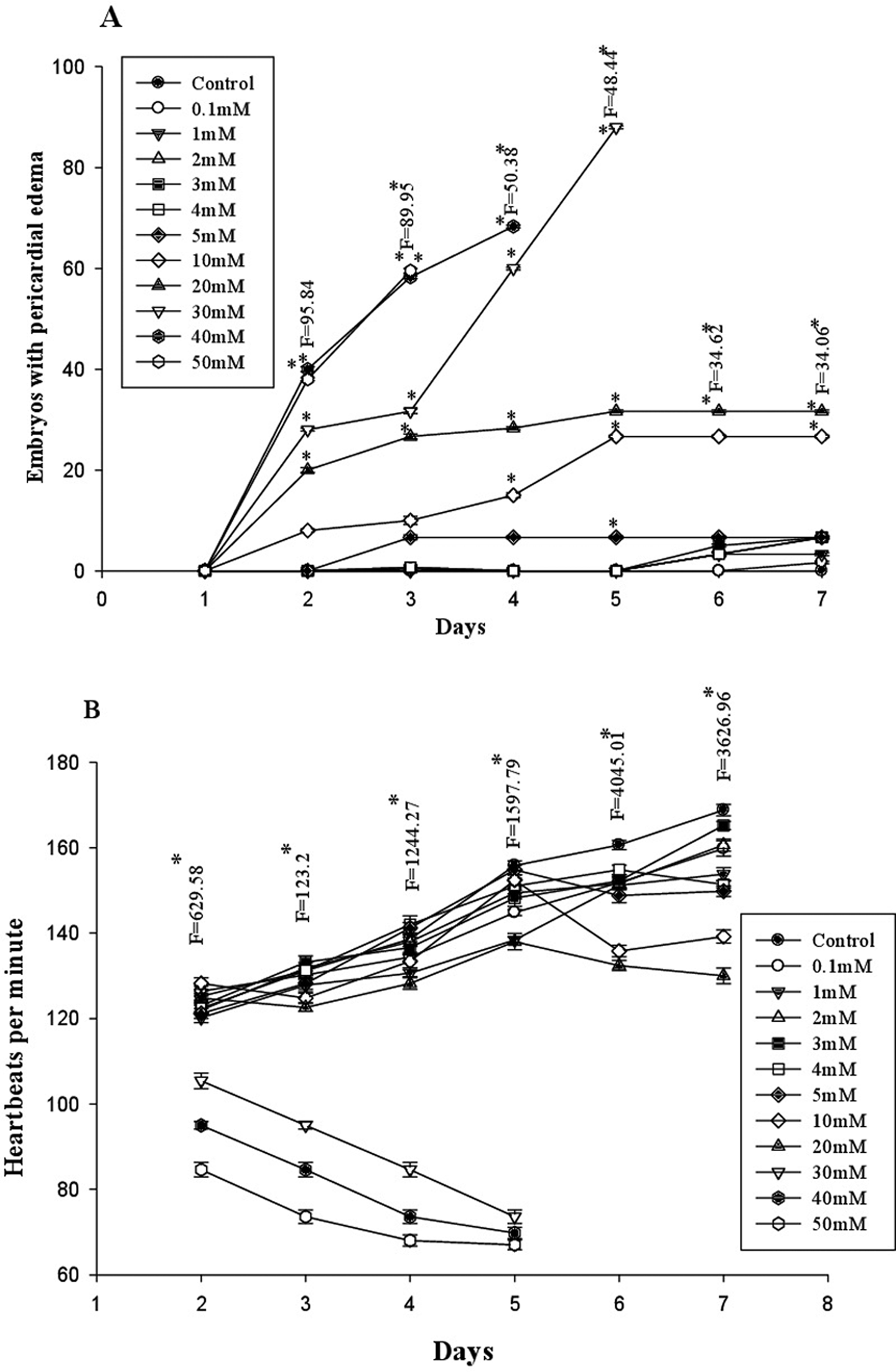
A-B, Pericardial edema and heartbeats per minute in larvae exposed to different concentrations of TTZ. * Significant at 5% level. TTZ indicates tartrazine.
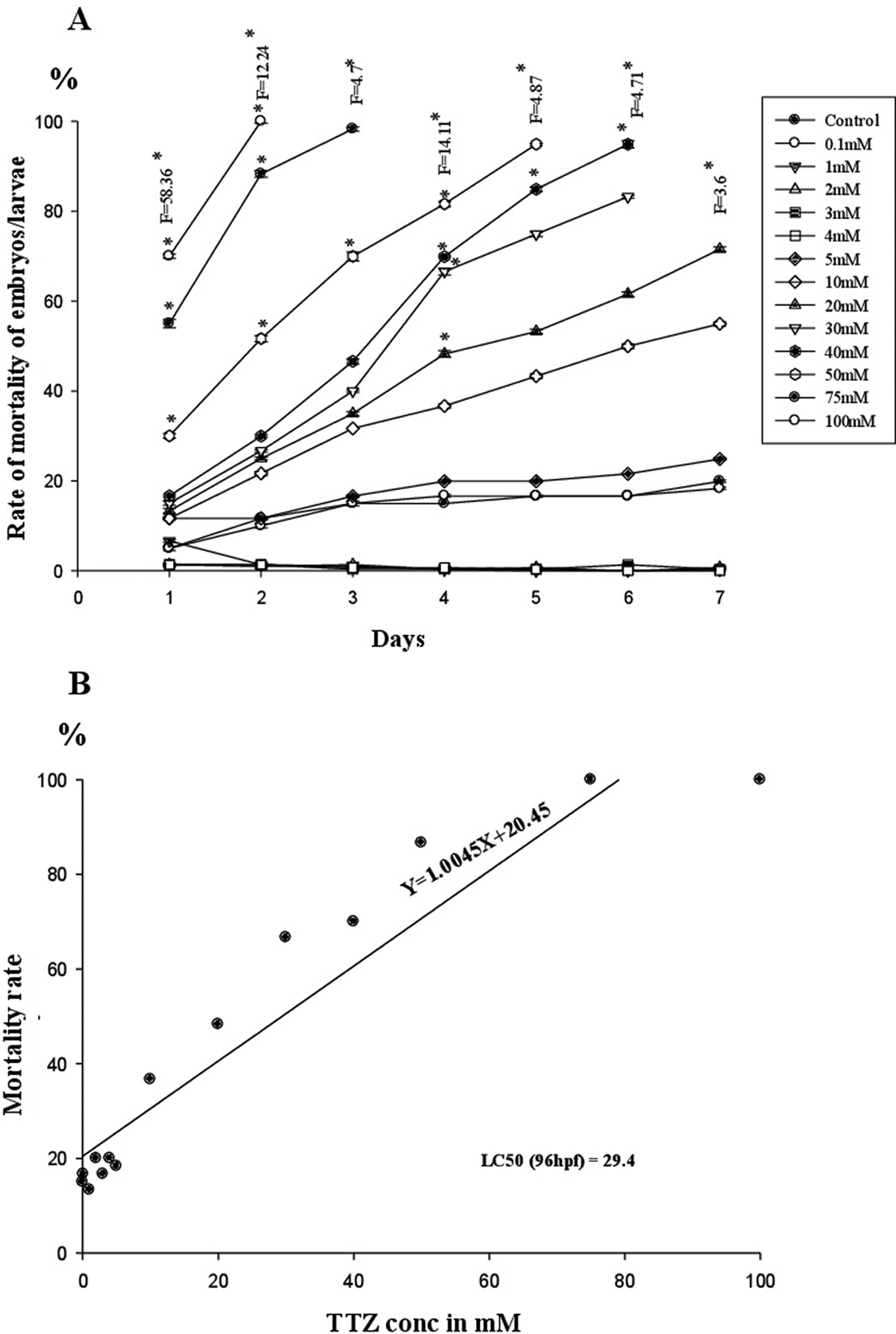
A-B, Mortality rate and LC50 (48 hpf) in TTZ exposed to zebrafish embryo/larvae. hpf indicates hours postfertilization; LC50, median lethal concentration; TTZ, tartrazine.
No Observed Effect Concentration, LC50, EC50, and TI
On the TTZ-induced mortality curve of D. rerio embryos, the no observed effect concentration (NOEC) was recorded at 5 mM (Figure 4). The LC50 estimated at 96 hpf was 29.4 mM (Figure 4). EC50 calculated for heart rate, cardiac edema, tail bending, and hatching success was 59.6, 53.81, 98.28, and 58.97 mM, respectively. The TI quotients calculated for heartbeat rate, cardiac edema, tail bending, and hatching success were 0.49, 0.54, 0.29, and 0.49, respectively (Table 1).
Shows LC50, EC50, and TI at 96 hpf of Zebrafish Larvae Exposed to TTZ.
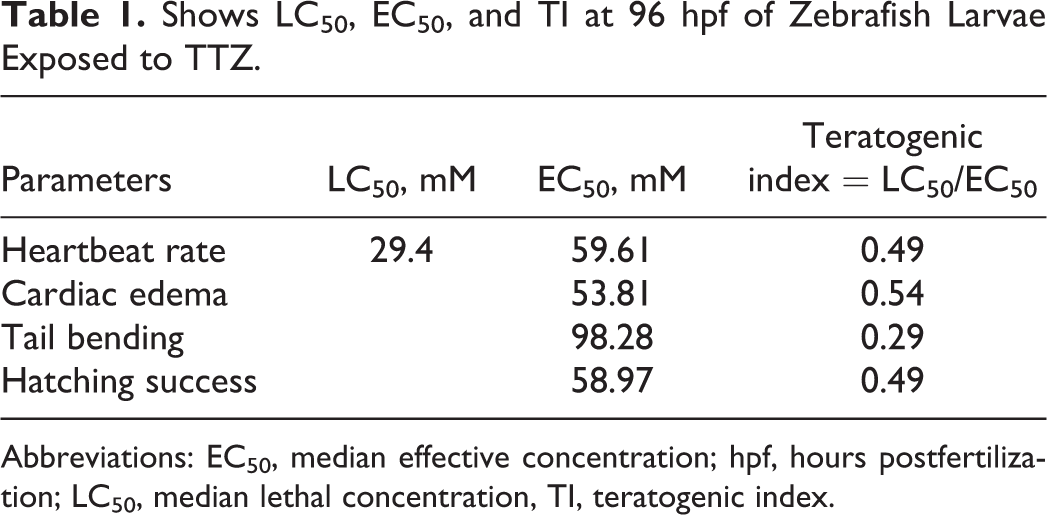
Abbreviations: EC50, median effective concentration; hpf, hours postfertilization; LC50, median lethal concentration, TI, teratogenic index.
Discussion
Food additives (FAs) are the substances/chemicals supplemented during the processing of food either to enhance the nutritive value or quality or to enhance taste, flavor, freshness, and appearance (decoration/presentation). 19,20 However, the safety limits and postconsumption health hazards associated with these FAs are of apprehension. Therefore, in recent years, there is an increasing concern about the impacts of FAs on human health that necessitates the development/standardization of sensitive-testing protocols for toxicity assessment.
Among the food colorant azo dyes, TTZ is the most widely used FA. Earlier studies on the toxicity of TTZ conducted on laboratory rodents reveal that this compound at a concentration of 500 mg/kg body weight (approximately 200 times greater than ADI for human) is nephrotoxic and hepatotoxic in male rats. 21 At a dose level of 500 mg/kg body weight, TTZ is known to induce a decrease in concentrations of the brain neurotransmitter with a sharp decline in the level of antioxidant biomarkers and elevate the levels of melonaldehyde in the brain of rat pups. 22 Biochemically, TTZ is known to cause a decline in enzyme activities like catalase, glutathione peroxidase, and superoxide dismutase, with a rise in the level of melonaldehyde which causes adverse effects on learning and memory functions in mice and rats. 23 Tartrazine is reported to have negative effects on fertility, reproductive performance, and sperm parameters of male rat at a dose level of 200 to 500 mg/kg body weight. 6 It is also genotoxic for white blood cell (leucocytes) at a dose level of 7.5 mg/kg body weight in rats. 7 Interestingly, administration of TTZ through gavage to gestating rats from 0 to 19 days of pregnancy at a concentration of 60, 100, 200, 400, 600, or 1,000 mg/kg body weight neither caused maternal toxicity nor was found teratogenic or lethal to embryo. 24,25
In the present study, we used a wide range of dilutions (0-100 mM) of TTZ for the exposure of developing zebrafish embryos (body mass: 80-100 μg) to determine no effect dose, safe dose, effective dose, lethal dose, and teratogenic potential of the FA. Each exposure consisted 60 developing embryos, and the experiments were repeated thrice to check the consistency of effects of TTZ. Our results reveal that TTZ at 0 to 5 mM had no effect. A concentration of up to 10 mM (0-26.7 mg) neither was lethal to embryo nor induced any noticeable deformities morphologically. At 20 to 30 mM (53-80 mg) concentrations, TTZ caused anomalies such as cardiac deformities; bradycardia was prominently observed in embryos exposed to a dose of >30 mM (80 mg) concentration followed by impaired heart rates, tail distortion, and yolk sac edema. While at extreme higher (50-100 mM or 133-267 mg) concentrations, development ceased, eventually leading to mortality. Therefore, at the admissible concentration of ADI (7.5 mg/kg body weight) prescribed for humans (i.e. 0.06 ng for zebrafish embryos), TTZ was neither teratogenic nor embryo toxic. The LC50 indicated the median lethal dose was 29.4 mM and EC50 for cardiac edema, heart rate, tail distortion, and hatching rate were 53.81, 59.6, 98.28, and 58.97, respectively. The TI quotients were calculated for cardiac edema (0.54), heart rate (0.49), tail bending (0.29), and hatching success (0.49), indicating that all the values were < 1 suggesting that the substance is not teratogenic. 26
In conclusion, the study directs us to infer that up to a dose level of 10 mM or 26 mg TTZ is neither teratogenic nor embryo toxic for zebrafish embryos.
Footnotes
Authors’ Contributions
V. Joshi contributed to acquisition, analysis, and interpretation, and drafted manuscript. P. Katti contributed to conception and design, drafted manuscript, and critically revised the manuscript. Both authors gave final approval and agree to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by UGC-SAP-DSA-Phase-I Program sanctioned to Department of Zoology, Karnatak University, Dharwad. VJ is grateful to Karnatak University, Dharwad for the award of University Research Studentship Scholarship.
