Abstract
Background
Pharmacokinetics and safety of JNJ-64530440, a hepatitis B virus capsid assembly modulator producing normal empty capsids (CAM-N), in healthy volunteers were evaluated.
Methods
This Phase I study (NCT03439488) was a double-blind, randomised, placebo-controlled study. Adults (
Results
Less than dose-proportional increases in maximum plasma concentrations (Cmax) and area under the plasma concentration–time curves (AUCs) were observed across the doses. Mean plasma half-lives ranged from 9.3 to 14.5 h. Cmax and AUC were ∼two fold higher under fed versus fasting conditions and slightly higher in Asians versus Caucasians. JNJ-64530440 doses ≥750 mg achieved plasma levels higher than protein-binding adjusted concentrations demonstrating
Conclusions
Single (up to 4000 mg) and multiple doses (up to 2000 mg for 7 days) of JNJ-64530440 were well tolerated in healthy volunteers. Multiple doses ≥750 mg/day achieved plasma concentrations expected to have antiviral activity that may lower hepatitis B surface antigen. No clinically relevant differences in tolerability or pharmacokinetic parameters were seen between Asians versus Caucasians.
Introduction
Infection with the hepatitis B virus (HBV) remains a significant health concern worldwide as ∼3.9% of the world’s population (∼292 million people) are chronically infected with HBV [1]. Globally, more than 80% of the total number of HBsAg-positive infections in the general population come from 21 countries, with China, India, Nigeria, Indonesia and the Philippines accounting for over half of all HBsAg-positive infections [1]. Moreover, ∼20–30% of people with chronic hepatitis B (CHB) will eventually develop serious, life-threatening complications such as cirrhosis, liver failure or hepatocellular carcinoma [2].
Current CHB treatments include life-long treatment with a nucleos(t)ide analogue (NA, e.g. tenofovir, or entecavir) or with pegylated interferon (pegIFN), typically for 1 year [3,4]. A key treatment goal is functional cure, defined as the sustained loss of hepatitis B surface antigen (HBsAg) and undetectable HBV DNA in serum off treatment ≥6 months, with or without HBsAg seroconversion [5]. However, functional cure rates achieved with NAs and pegIFN are low [3,4]. Thus, new agents with different mechanisms of action (MOA), given either alone or in combination with other agents are needed to increase HBV functional cure rates with finite treatment options and reduce the long-term consequences of chronic infection.
A critical step in the HBV life cycle is HBV capsid assembly. Capsid assembly modulators (CAMs) bind to the core dimer, accelerate capsid assembly kinetics and prevent encapsidation of the polymerase-pregenomic RNA complex, thereby blocking HBV replication [6,7]. In addition, CAMs interfere with covalently closed circular DNA (cccDNA) transcription/de novo formation [8-10] and restore innate signalling [11]. At least two classes of CAMs are being investigated. CAM-N compounds induce the formation of empty, morphologically intact, non-functional capsids (N = normal structure), and CAM-A agents result in the formation of pleiomorphic noncapsid structures (A = aberrant particle). Several compounds in both classes of CAMs have undergone early clinical trials and have demonstrated evidence of antiviral activity, for example, CAM-Ns: JNJ-56136379 [12, 13], vebicorvir (ABI-H0731) [14] and NVR3-778 [15]; CAM-As: RO7049389 [16] and GLS4/ritonavir [17].
The CAM-N JNJ-64530440 has demonstrated more potent antiviral activity than JNJ-56136379
Methods
Study design and volunteers
This study (NCT03439488) was a multi-part, double-blind, randomised, placebo-controlled, first-in-human, Phase I trial evaluating the pharmacokinetics, safety and tolerability of JNJ-64530440. Parts 1 and 2 of this study investigated the effects of SAD and MAD of JNJ-64530440, respectively, in healthy volunteers (Supplemental Material 1). Part 3 results (MAD phase in CHB patients including antiviral assessments) are reported separately [19].
Key inclusion criteria for parts 1 and 2 were healthy Asian or Caucasian males and females aged 18–55 years and a body mass index 18–30 kg/m2. Key exclusion criteria for parts 1 and 2 included a history of cardiac arrhythmias, significant/unstable cardiac disease or any clinically significant electrocardiogram (ECG) abnormalities; any current/previous illness or condition that might confound the results or pose a risk for study drug administration (e.g. renal dysfunction and previous gastrointestinal surgery); use of alcohol and/or recreational drugs; current hepatitis or HIV infection and any history of allergy or skin disease.
The study was conducted at a single clinical pharmacology unit (Auckland Clinical Studies, New Zealand) between 19 March 2018 and 17 May 2019. This study was conducted in accordance with the Declaration of Helsinki, Good Clinical Practice and applicable regulatory requirements.
The study protocol and amendments were approved by the Health and Disability Ethics Committee (New Zealand). All volunteers provided informed written consent to participate in the study.
JNJ-64530440 administration
For both SAD and MAD phases, five Asian and five Caucasian healthy volunteers were randomised 1:4 to placebo and JNJ-64530440 groups in each cohort. Volunteers were screened within a month and checked into the relevant clinic the day before the study start, where they remained for 4 days (SAD) or 10 days (MAD).
JNJ-64530440 intake, with water, was at approximately the same time each day. Single-ascending doses of oral JNJ-64530440 50, 150, 300 and 900 mg were administered under fasted conditions, and 750, 1000, 2000 and 4000 mg SAD doses and all MAD doses were administered under fed conditions, that is, a standard meal containing ∼21 g fat/533 kcal was consumed within 30 min prior to dosing. Doses were given after breakfast for once daily dosing (QD) and after breakfast and dinner for twice daily dosing (BID).
The effect of food on JNJ-64530440 pharmacokinetics was assessed for the 300-mg dose in the SAD phase. One cohort of volunteers received a single 300-mg dose under fasted conditions, i.e. these volunteers did not eat for at least 10 h overnight until 4 h post-dose. After a washout period of at least 7 days, the same cohort of volunteers also received a single 300-mg dose with a high-fat breakfast (800–1000 kcal, with fat as 50–75% of the calories).
Lunch (∼4 h post-dose) and dinner (∼12 h post-dose) were of the same composition for all volunteers in all cohorts on days of intensive pharmacokinetic sampling.
Two tablet formulations of JNJ-64530440 were used in this study. Both were spray dried dispersions of JNJ-64530440, with different excipients and drug load. The first-generation formulation was available as 25- and 100-mg tablets and was used for QD dosing at 50, 150, 300 (fasted and fed), 900 and 1000 mg in the SAD phase. The second-generation formulation was available as 100- and 250-mg tablets, and was used for QD dosing at 750, 2000 and 4000 mg in the SAD phase, and for all dosing regimens in the MAD phase. Placebo tablets matched the JNJ-64530440 tablets used in the study.
SAD phase
In part 1, ten cohorts were planned and volunteers in nine cohorts were evaluated (one cohort was dosed with 300 mg under fed and fasted conditions). The final cohort, to evaluate a third formulation of JNJ-64530440, was not required.
The starting dose of JNJ-64530440 of 50 mg was predicted to give antiviral activity (primary MOA). JNJ-64530440 dose escalation was only conducted after reviewing the pharmacokinetics and safety data from the previous cohort. The single JNJ-64530440 doses evaluated were 50, 150, 300 and 900 mg (fasted conditions), and 300, 750, 1,000, 2000 and 4000 mg (fed conditions).
MAD phase
In part 2, volunteers received either placebo or JNJ-64530440 QD (750 or 2000 mg) or BID (750 mg, i.e. 1500 mg daily) under fed conditions for 7 days. The higher JNJ-64530440 doses were only administered after the pharmacokinetic and safety data from the previous cohort had been reviewed.
Pharmacokinetic assessments
In the SAD phase, blood samples were collected pre-dose and at 0.5, 1, 2, 3, 4, 6, 8, 10, 12, 18, 24, 36, 48, 72, 96 and 120 h post-dosing. For the 1000 and 4000 mg cohorts, the complete urinary output was collected during the intervals: −12–0, 0–4, 4–8, 8–12, 12–18, 18–24, 24–48 and 48–72 h post-dose.
In the MAD phase, blood samples were collected on Day 1 (pre-dose and at 0.5, 1, 2, 4, 6, 8, 10, 12 and 18 h post-dosing), pre-dose on days 2, 3, 5, and 6 and on Day 7 (pre-dose and at 0.5, 1, 2, 4, 6, 8, 10, 12, 18, 24 and 48 h post-dosing). For the 2000 mg QD cohort, the complete urinary output was collected on Day 1 (intervals: −12–0, 0–6, 6–12 and 12–24 h post-dose) and on Day 7 (intervals: −12–0, 0–6, 6–12, 12–24, 24–48 and 48–72 h post-dose).
Plasma and urine samples were stored frozen until analysed. Upon analysis, JNJ-64530440 and internal standard were extracted from 50 μL or 100 μL of plasma or urine, respectively, by a protein precipitation extraction procedure. JNJ-64530440 concentrations were analysed using a fully validated liquid chromatography with tandem mass spectrometry assay in negative ion mode on an MDS Sciex API 4000 equipped with a Turbo Ionspray® interface. The lower limit of quantification in plasma was 1.00 ng/mL and in urine was 5.00 ng/mL (Syneos Health, Princeton, NJ, USA).
Safety
All volunteers underwent physical examination, 12-lead ECG assessment, a vital signs check, standard laboratory evaluations (clinical chemistry, haematology and urinalysis) at screening, at randomisation and daily for both phases. Safety and tolerability were assessed continuously throughout the study, from consent to check-out from the clinic, by monitoring all adverse events (AEs), which were graded using the Division of Acquired Immunodeficiency Syndrome toxicity grading scale [20].
Data analyses
No formal sample size calculation was performed as this study was exploratory. All demographic, baseline characteristics and safety data (including AEs) were descriptively summarised.
Plasma concentration–time data were plotted and used to calculate JNJ-64530440 pharmacokinetic parameters using non-compartmental analyses (WinNonlin Professional; Certara LP, Princeton, NJ, USA). Key pharmacokinetic parameters included maximum plasma concentration (Cmax); concentration at the end of the dosing period (Ctau); time to Cmax (tmax) and area under the plasma concentration–time curve (AUC), that is, from zero to the last measurable concentration (AUClast), to infinity (AUCinf), to the end of the dosing period (AUCtau). In addition, the terminal elimination half-life (t1/2) was calculated. Descriptive statistics were used to summarise plasma concentrations and derived pharmacokinetic parameters of JNJ-64530440. Descriptive statistics were used to assess the effect of race on JNJ-64530440 pharmacokinetic parameters.
Statistical analyses on log-transformed JNJ-64530440 Cmax and AUClast were performed to assess the effect of food (fed conditions as test and fasted conditions as reference) and the effect of formulation (second-generation tablet, 750 mg versus first-generation tablet, 1000 mg). All observations for test and reference, paired and unpaired, were included in the statistical analysis. The least squares mean (LSM) of the primary parameters for each treatment group was estimated with a linear mixed effects model, controlling for treatment (fed or fasted) as fixed effect and volunteer as a random effect. A 90% confidence interval (CI) was constructed around the difference between the LSM of test and reference. Both the difference between the LSM and the 90% CIs were retransformed to the original scale.
Results
Volunteer disposition and demographics
Overall, 207 healthy volunteers were screened and 97 were deemed screen failures. Eighty volunteers were randomised and received a single dose of JNJ-64530440 (
Baseline characteristics: Parts 1 (SAD) and 2 (MAD).
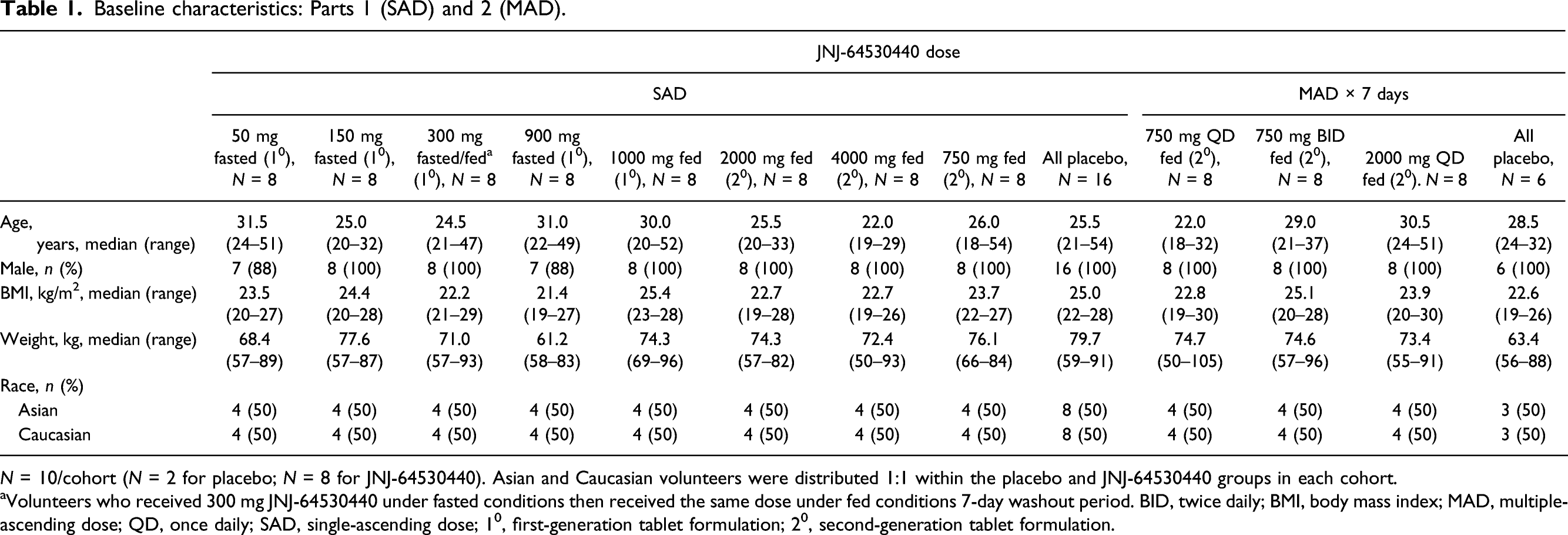
aVolunteers who received 300 mg JNJ-64530440 under fasted conditions then received the same dose under fed conditions 7-day washout period. BID, twice daily; BMI, body mass index; MAD, multiple-ascending dose; QD, once daily; SAD, single-ascending dose; 10, first-generation tablet formulation; 20, second-generation tablet formulation.
Pharmacokinetics of JNJ-64530440
SAD phase
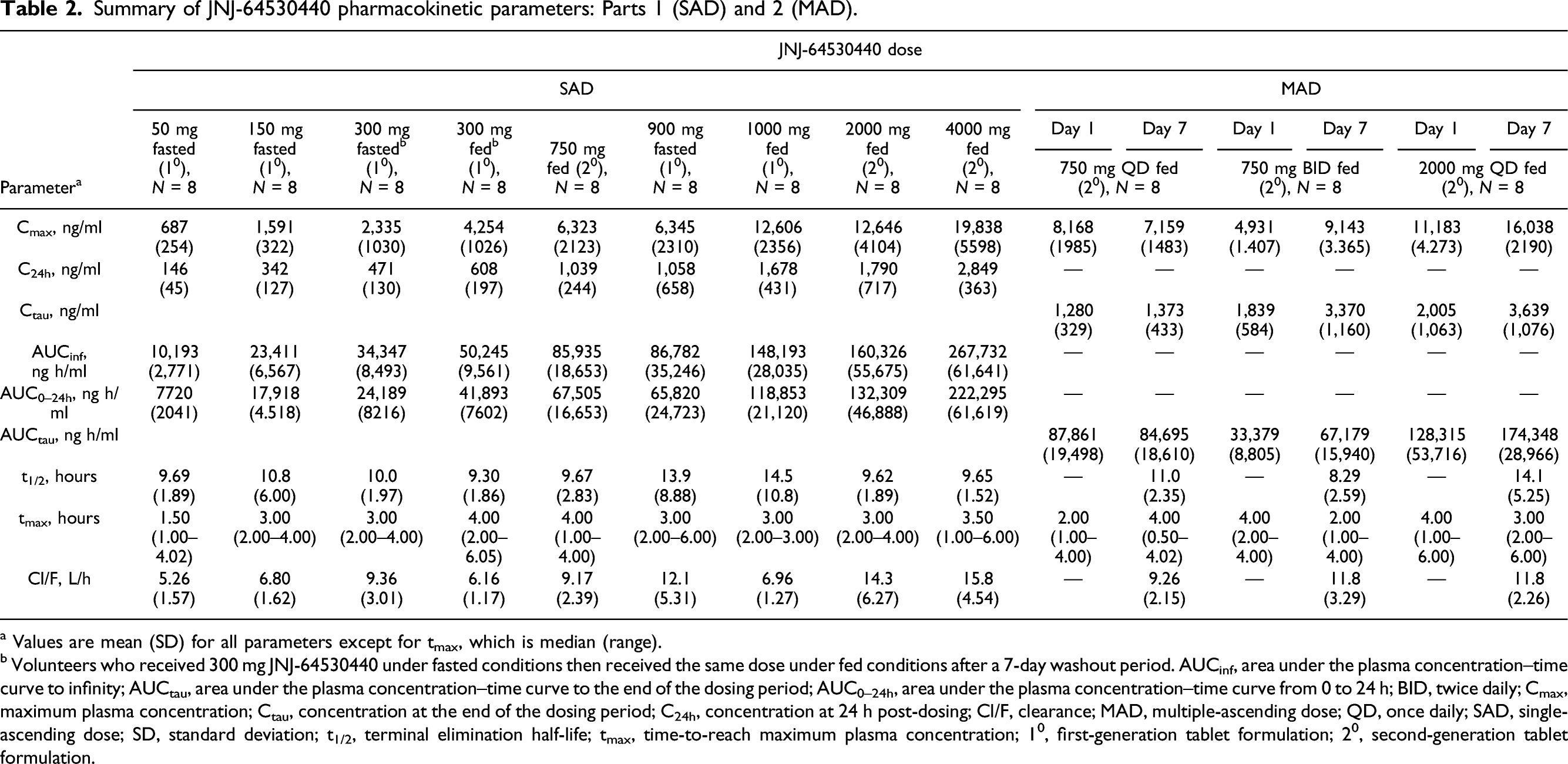
Plasma concentrations of JNJ-64530440 are shown in Figure 1A. After administration of single oral doses of JNJ-64530440 ranging from 50 to 900 mg under fasted conditions, JNJ-64530440 Cmax and AUCinf increased with dose (Table 2), although these increases were not dose proportional. Under fed conditions, less than dose-proportional increases in JNJ-64530440 Cmax and AUCinf occurred following single oral doses of JNJ-64530440 ranging from 300 to 4000 mg (Table 2). JNJ-64530440 plasma concentrations following (A) single doses and (B) multiple doses (on days 1 and 7, blood samples were collected over 24 h); 10, first-generation tablet formulation; 20, second-generation tablet formulation; BID, twice daily; QD, once daily; SD, standard deviation. Summary of JNJ-64530440 pharmacokinetic parameters: Parts 1 (SAD) and 2 (MAD). a Values are mean (SD) for all parameters except for tmax, which is median (range). b Volunteers who received 300 mg JNJ-64530440 under fasted conditions then received the same dose under fed conditions after a 7-day washout period. AUCinf, area under the plasma concentration–time curve to infinity; AUCtau, area under the plasma concentration–time curve to the end of the dosing period; AUC0–24h, area under the plasma concentration–time curve from 0 to 24 h; BID, twice daily; Cmax, maximum plasma concentration; Ctau, concentration at the end of the dosing period; C24h, concentration at 24 h post-dosing; Cl/F, clearance; MAD, multiple-ascending dose; QD, once daily; SAD, single-ascending dose; SD, standard deviation; t1/2, terminal elimination half-life; tmax, time-to-reach maximum plasma concentration; 10, first-generation tablet formulation; 20, second-generation tablet formulation.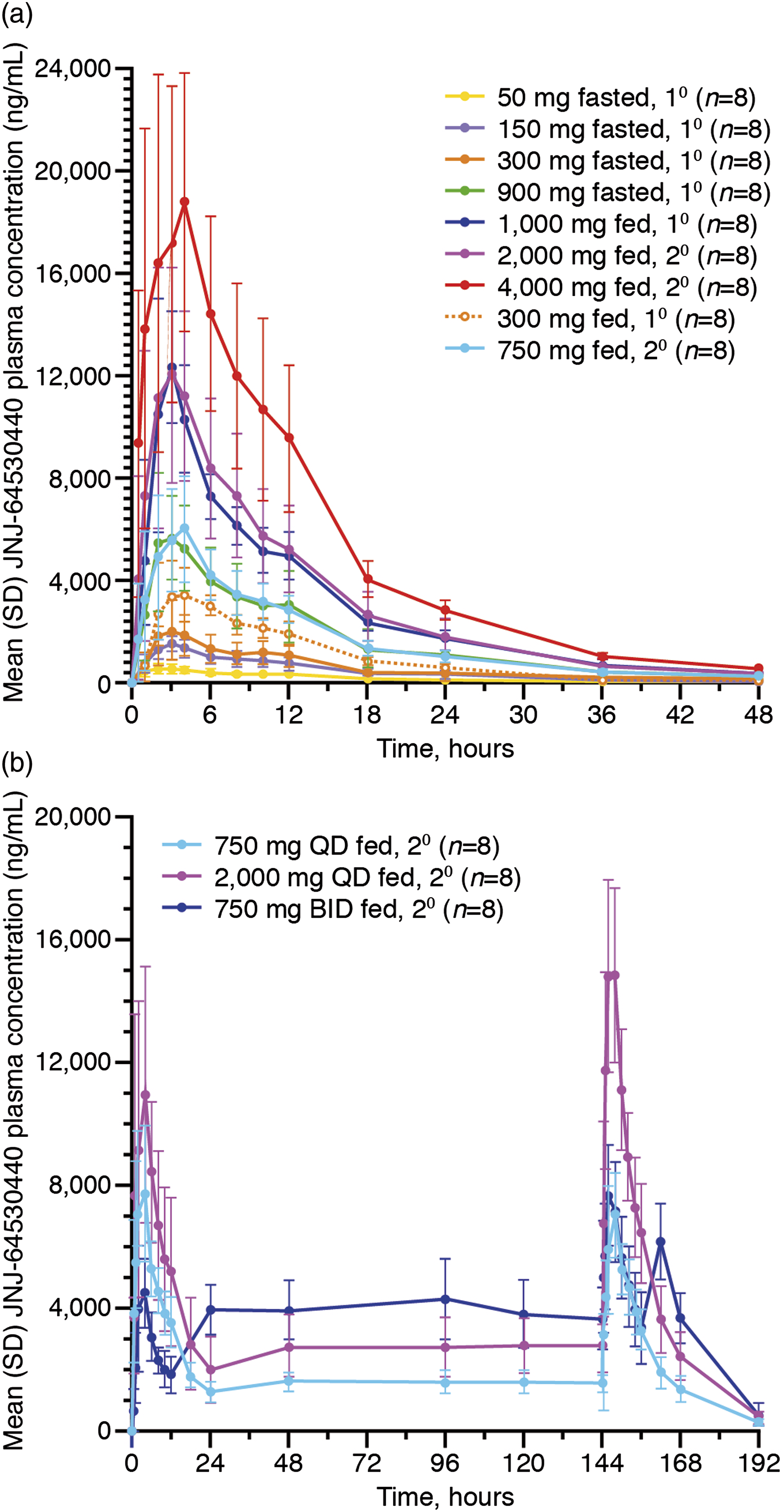
JNJ-64530440 was excreted into the urine following single oral doses of 1000 and 4000 mg (Supplemental Material 4). The mean (range) percentage of the dose excreted unchanged in the urine 0–72 h post-dosing was 21% (12–31%) and 9% (4–14%) for the 1000 and 4000 mg doses, respectively.
Volunteers receiving single JNJ-64530440 doses of ≥750 mg in the fed state had 24-h post-dose plasma concentrations (ranges: 750 mg 725–1440 ng/mL; 900 mg 559–2180 ng/mL; 1000 mg 1430–2630 ng/mL; 2000 mg 778–2540 ng/mL).
Across the JNJ-64530440 doses, mean t1/2 was ∼9.3–14.5 h and was comparable between fasted (10.0 h) and fed (9.3 h) volunteers for the 300-mg JNJ-64530440 single-dose cohort (Table 2). The median tmax for JNJ-64530440 ranged from 1.5 to 3.0 h for fasted conditions and 3.0 to 4.0 h under fed conditions (Table 2).
For the volunteers receiving a single 300-mg dose of JNJ-64530440 in the fed state, both Cmax and AUC of JNJ-64530440 were increased by ∼two fold versus the fasted state (Table 2). For Cmax, the point estimate (90% CI) of the LSM ratio was 199.40 (94.27–421.74) for the fed versus fasted state. For AUClast, the point estimate (90% CI) of the LSM ratio was 148.60 (114.54–193.00) for the fed versus fasted state.
For JNJ-64530440 Cmax, the point estimate (90% CI) of the LSM ratio was 64.12 (41.07–100.12) for the second-generation 750 mg tablet versus the first-generation 1000 mg tablet. For AUClast, the point estimate (90% CI) of the LSM ratio was 76.94 (56.15–105.44) for the second-generation versus the first-generation formulation.
MAD phase
Plasma concentrations of JNJ-64530440 multiple dosing are shown in Figure 1B. Administration of JNJ-64530440 at 750 mg QD or BID or 2000 mg QD under fed conditions for 7 days resulted in steady-state conditions as mean plasma concentrations were comparable through days 1–7 (pre-dose) (Figure 1B); mean plasma trough concentrations were also similar on each day of dosing.
With QD dosing, JNJ-64530440 Cmax and AUCtau increased with dose, although this increase was not dose proportional (Table 2). After 7 days of dosing, these volunteers had 24-h post-dose concentrations (ranges: 750 mg 832–2010 ng/mL and 2000 mg 1230–3490 ng/mL). The mean t1/2 was approximately 11.0–14.1 h with QD dosing (Table 2). Exposure to JNJ-64530440 was higher with BID dosing versus QD dosing (Figure 1B and Table 2). With 750 mg BID dosing, the 12-h post-last dose plasma concentrations ranged from 3280 to 4920 ng/mL (Day 1) and 2470 to 4530 ng/mL (Day 7). With both QD and BID dosing, these plasma concentrations are expected to be therapeutic based on the protein-binding adjusted 90% effective concentration of JNJ-64530440
After the first 2000-mg JNJ-64530440 QD dose, the mean (range) percentage of the dose excreted unchanged into the urine was 10% (5–16%) (Supplemental Material 4). After 7 days of QD dosing, the mean (range) percentage of the JNJ-64530440 dose excreted unchanged in the urine 0–72 h post-dosing on Day 7 was 17% (9–25%) (Supplemental Material 4).
JNJ-64530440 pharmacokinetic profile in Asian and Caucasian volunteers
In the SAD phase, mean Cmax was higher in Asian versus Caucasian volunteers at both the 750 mg (7110 [standard deviation 1967] versus 5535 [2234] ng/mL) and 2000-mg QD doses (14,646 [3361] versus 10,645 [4161] ng/mL). Mean AUCinf in the SAD phase was also slightly higher in Asian versus Caucasian volunteers with 750 mg JNJ-64530440 (73,980 [17,585] versus 61,031 [15,035] ng.h/mL) and higher with 2000 mg (149,003 [37,489] versus 115,614 [54,600] ng.h/mL) (Supplemental Material 5). In the MAD phase, JNJ-64530440 concentrations and AUCtau on Day 7 were generally comparable between Asian and Caucasian volunteers following 7 days of JNJ-64530440 QD dosing at 750 mg or 2000 mg (Supplemental Material 6). With BID dosing (750 mg) for 7 days, exposure to JNJ-64530440 was slightly higher in Asians versus Caucasians (Supplemental Material 6).
Safety
Overall population
Summary of treatment-emergent AEs following administration of JNJ-64530440: Parts 1 (SAD) and 2 (MAD).
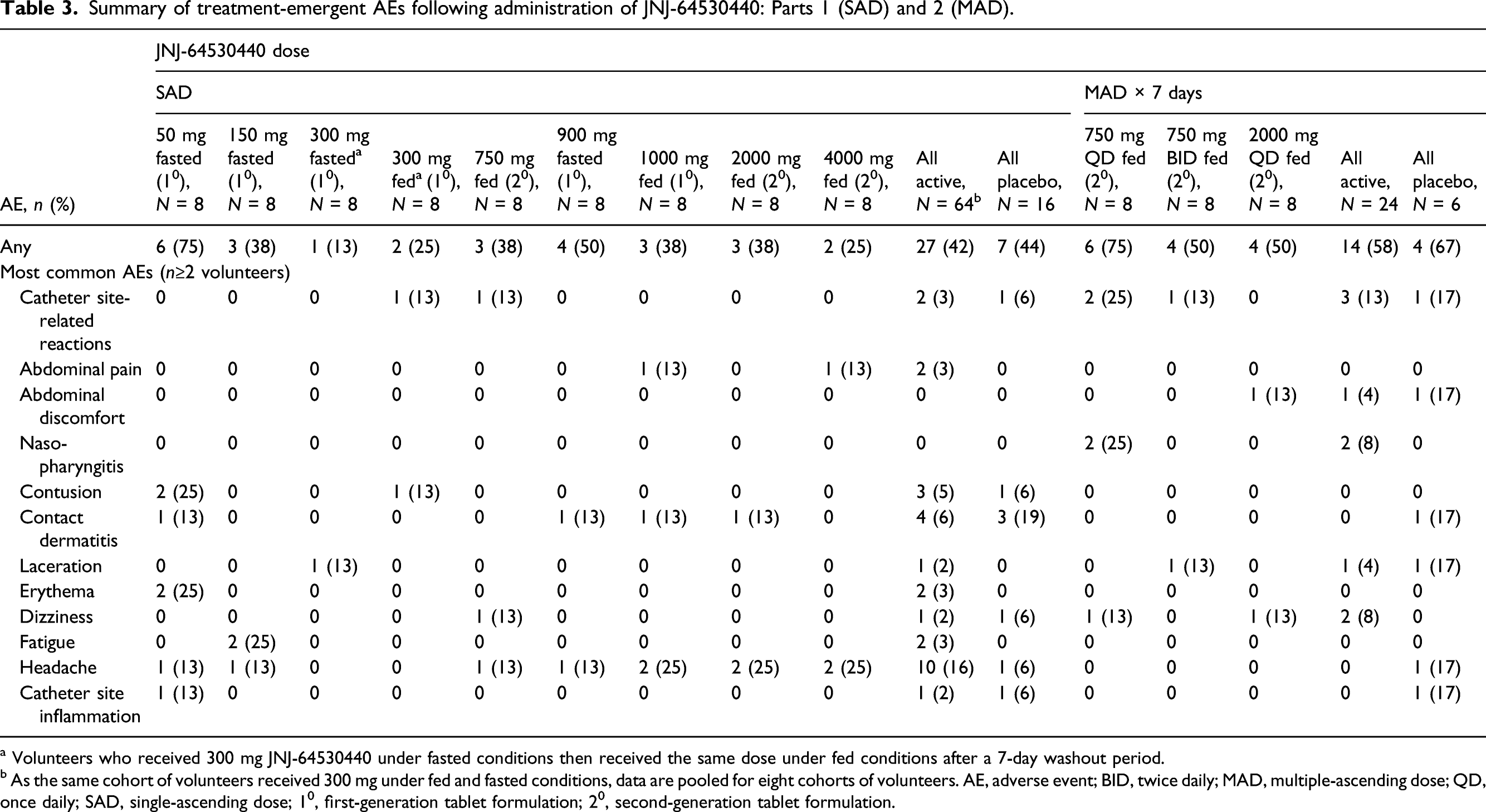
a Volunteers who received 300 mg JNJ-64530440 under fasted conditions then received the same dose under fed conditions after a 7-day washout period.
b As the same cohort of volunteers received 300 mg under fed and fasted conditions, data are pooled for eight cohorts of volunteers. AE, adverse event; BID, twice daily; MAD, multiple-ascending dose; QD, once daily; SAD, single-ascending dose; 10, first-generation tablet formulation; 20, second-generation tablet formulation.
In the SAD phase, treatment-related AEs were reported in the following cohorts: placebo (
Incidence of grade ≥2 treatment-emergent laboratory abnormalities following administration of JNJ-64530440: Parts 1 (SAD) and 2 (MAD).
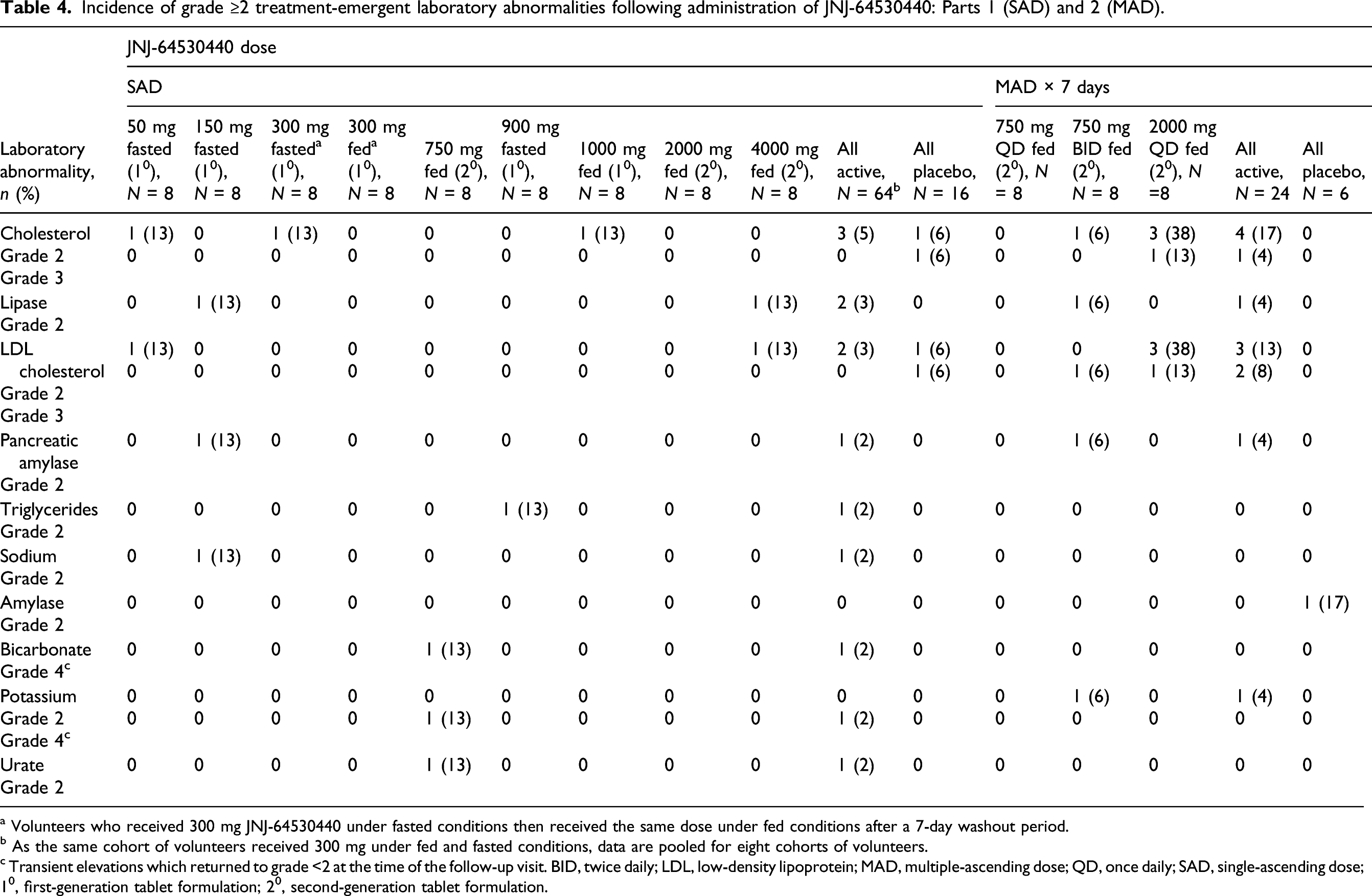
a Volunteers who received 300 mg JNJ-64530440 under fasted conditions then received the same dose under fed conditions after a 7-day washout period.
b As the same cohort of volunteers received 300 mg under fed and fasted conditions, data are pooled for eight cohorts of volunteers.
c Transient elevations which returned to grade <2 at the time of the follow-up visit. BID, twice daily; LDL, low-density lipoprotein; MAD, multiple-ascending dose; QD, once daily; SAD, single-ascending dose; 10, first-generation tablet formulation; 20, second-generation tablet formulation.
Asians versus Caucasians
Overall, there was no apparent difference in the incidence of treatment-emergent AEs between Asian and Caucasian volunteers with SAD (Supplemental Material 7) or MAD (Supplemental Material 8), although the small sample sizes for placebo and active treatment volunteers within cohorts make comparisons difficult. There were no clinically significant changes in physical examination results, vital signs, ECGs, haematology, laboratory parameters and urinalysis results (data not shown) between the races.
Discussion
First-in-human studies provide an opportunity to evaluate multiple factors related to pharmacokinetics such as relative bioavailability of different formulations, food effect, drug interactions and/or covariates (e.g. sex and race) [21]. Parts 1 and 2 of this first-in-human study in adult healthy volunteers demonstrated that SAD (at 50–4000 mg) and MAD doses (750 QD or BID or 2000 mg QD for 7 days) of JNJ-64530440 resulted in increasing plasma concentrations, although there was a less than a dose-proportional increase in plasma drug exposure. There were no major differences in exposure between Asian and Caucasian volunteers. Food intake approximately doubled the exposure to JNJ-64530440 compared with fasting conditions. In healthy participants, JNJ-64530440 was well tolerated at the doses tested and over the dosing periods evaluated.
The observed mean t1/2 of 9–14.5 h supports QD or BID administration. The clear impact of food intake resulting in an approximately twofold increase in exposure to JNJ-64530440 versus fasted conditions supports administration of this agent under fed conditions in future studies, including part 3 of the current study [19]. Renal excretion of JNJ-64530440 was low at 9–21%.
Two formulations of JNJ-64530440 were used in this study. Second-generation tablets were developed during the study to support higher doses with less pill burden, and this formulation had a higher drug load (100- and 200-mg strengths) compared with the first-generation tablets (25- and 50-mg strengths). When comparing the bioavailability of the two formulations, 750 mg (3 × 250-mg second-generation tablets) was compared with 1000 mg (10 × 100-mg first-generation tablets) both under fed conditions in separate cohorts of volunteers. After adjusting for the difference in dose, Cmax was slighter lower for the second-generation formulation and the AUC was comparable.
Importantly, the trough plasma concentrations of JNJ-64530440 with doses of ≥750 mg in the fed state following seven daily doses (range 832–3490 ng/mL) were higher than the median 50%/90% effective concentrations for inhibition of capsid assembly (primary MOA), that is, 21 nM/103 nM, respectively, for inhibition of HBV DNA in HBV-infected primary human hepatocytes (PHHs), and for the inhibition of cccDNA formation (secondary MOA), that is, 136 nM/373 nM, respectively, for inhibition of HBV RNA in HBV-infected PHHs [18]. Moreover, these trough JNJ-64530440 plasma concentrations were also higher than the 90% effective concentration for antiviral activity
It is well recognised that metabolism of drugs in Asian populations can differ from other races and can lead to differences in pharmacokinetics [22]. In addition, the prevalence of chronic HBV infection is particularly high in the Asia-Pacific region [23-25]. Thus, it is important to evaluate any potential impact of Asian race on the pharmacokinetics of any new agent for CHB treatment. As such, both Asian and non-Asian volunteers were equally enrolled in the SAD and MAD phases to determine whether pharmacokinetics and safety would differ among these populations. Our comparison of JNJ-64530440 pharmacokinetic parameters between Asians and Caucasians demonstrated slightly higher Cmax and AUCtau values on Day 1 of QD multiple dosing in Asian healthy volunteers, whereas following 7 days of QD or BID dosing, these parameters were generally similar between the two races. Given the wide variability of pharmacokinetic parameters in the overall population as well as within both races, the small differences in JNJ-64530440 pharmacokinetics seen between Asians and Caucasians are considered not to be clinically relevant. Thus, our data support the use of the same dose(s) of JNJ-64530440 in both races.
Our study showed that with short-term dosing, there were no serious AEs, treatment discontinuations or dose-limiting toxicities, and all AEs were mild or moderate in severity following JNJ-64530440 administered to healthy volunteers. There were no dose-related increases in the frequency or severity of AEs at JNJ-64530440 single doses up to 4000 mg and multiple daily doses of up to 2000 mg in either Asian or Caucasian healthy volunteers. The most common AEs observed were headache, contact dermatitis and contusion. Thus, the safety profile of JNJ-64530440 at high doses for a short time (a single dose or multiple doses for 7 days) was favourable, which supported JNJ-64530440 administration to CHB patients in part 3 of this study [19].
Two limitations of this study are the predominantly male study population and the relatively small sample size of volunteers in each dosing cohort, although the number of volunteers per cohort is in keeping with this type of study. However, in the context of being the first-in-human study with JNJ-64530440, the overall sample size of 110 healthy volunteers is sufficient to provide valuable data for subsequent dose selection.
In conclusion, this first-in-human study demonstrated that JNJ-64530440 was well tolerated by Asian and Caucasian healthy volunteers. The pharmacokinetic profile allows QD dosing and provides therapeutic exposures that should cover both MOAs of this CAM-N. The pharmacokinetic data obtained helped to identify two dosing regimens which were subsequently evaluated in patients with CHB for up to 28 days, JNJ-64530440 750 mg QD or BID where antiviral activity was observed (data reported separately [19).
Supplemental Material
sj-pdf-1-avt-10.1177_13596535211044331 – Supplemental Material for Pharmacokinetics, safety and tolerability of single- and multiple-ascending doses of JNJ-64530440, a novel hepatitis B virus capsid assembly modulator, in healthy volunteers
Supplemental Material, sj-pdf-1-avt-10.1177_13596535211044331 for Pharmacokinetics, safety and tolerability of single- and multiple-ascending doses of JNJ-64530440, a novel hepatitis B virus capsid assembly modulator, in healthy volunteers by Thomas N Kakuda, Jeysen Z Yogaratnam, Christopher Westland, Edward J Gane, Christian Schwabe, Jennifer Vuong, Megha Patel, Jan Snoeys, Willem Talloen, Oliver Lenz, John Fry, Sushmita Chanda and Pieter van Remoortere in Antiviral Therapy
Footnotes
Acknowledgements
The authors express their gratitude to the volunteers who participated in this study. The authors thank other Janssen staff members for their contributions to this study, including Nick Khorlin, and Moana Hodari, and also the study co-ordinator, Emily Shearer, at Auckland Clinical Studies, New Zealand. The authors also thank Jackie Phillipson of Ashfield MedComms, Macclesfield, UK, an Ashfield Health company, who provided medical writing support (all drafts, assembling tables and figures, collating author comments, grammatical editing and referencing); this support was funded by Janssen Pharmaceuticals.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Janssen Biopharma.
ClinicalTrials
ClinicalTrials.gov registration number: NCT03439488.
Disclosure statement
TNK, MP, JS, WT, OL and PVR are employees of Janssen Pharmaceuticals and may be Johnson & Johnson stockholders. JZY, CW, JV, JF and SC were former employees of Janssen Pharmaceuticals and are now employees of Aligos Therapeutics and may be stockholders. EJG has been an advisor and/or speaker for AbbVie, Aligos, Gilead, Janssen, Novartis and Roche. CS has provided advice to Johnson & Johnson and Vir Biotechnology. Part 1 (SAD) and Part 2 (MAD) data were presented in part at EASL, The International Liver Congress, Vienna, Austria, April 10–14, 2019; Kakuda TN, Yogaratnam JZ, Westland CW et al. JNJ-64530440, a novel capsid assembly modulator: single- and multiple-ascending dose safety, tolerability and pharmacokinetics in healthy volunteers. J Hepatol 2019; 70 (suppl 1): e469.
Availability of data and material
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.jansen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at http://yoda.yale.edu.
Author contributions
EJG and CS were investigators in the study and reported data for the volunteers. TNK, MP, JS, WT, OL, PVR, JZY, CW, JV, JF and SC were involved in the data analyses. All authors were involved in the development of the primary manuscript and interpretation of data, have read and approved the final version, and have met the criteria for authorship as established by the ICMJE.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
