Abstract
Introduction:
Extracoronary vascular abnormalities (EVAs) are common in patients with spontaneous coronary artery dissection (SCAD), and expert consensus statements recommend evaluation with head-to-pelvis cross-sectional imaging. Lack of a universal imaging protocol remains a barrier to consistent implementation of this recommendation. We sought to implement a high-fidelity, single-session, head-to-pelvis computed tomography angiography (CTA) protocol that minimizes radiation and contrast exposure compared to multisession CTA.
Methods:
A total of 97 consecutive patients with SCAD were evaluated with a single-session CTA (SS-CTA protocol). The comparison group consisted of 63 consecutive patients who underwent asynchronous CTA of different body segments (MS-CTA). The primary outcome was total radiation dose and contrast volume in each imaging protocol. The secondary outcome was prevalence of EVAs.
Results:
There was no difference in baseline characteristics, SCAD presentation, or management between groups. The SS-CTA protocol had a 24% lower radiation dose (1873 ± 676 mGy-cm vs 2452 ± 1099 mGy-cm, p < 0.001) and a 21% lower contrast volume (161.2 ± 48.7 mL vs 203.8 ± 68.4 mL, p < 0.001) compared to the MS-CTA protocol. There was no significant difference in the prevalence of EVAs detected between imaging groups (55.7% vs 57.1%, p = 0.854).
Conclusion:
This study demonstrates the effectiveness of a single-acquisition CTA protocol for EVA detection in patients with SCAD. The SS-CTA protocol minimized radiation and contrast exposure for a patient population that may require frequent imaging over the lifespan. Further studies are needed to determine whether expanded availability of this protocol would improve adherence to expert consensus recommendations.
Keywords
Introduction
Spontaneous coronary artery dissection (SCAD) is frequently associated with extracoronary vascular abnormalities (EVAs).1 –6 These may include imaging evidence of fibromuscular dysplasia (FMD), aneurysm, pseudoaneurysm, and/or dissection. Cohort studies have demonstrated a > 50% prevalence of EVAs in patients with SCAD.7 –12 SCAD may also be the initial manifestation of heritable connective tissue disorders, such as vascular Ehlers-Danlos syndrome. Therefore, detection of EVAs is essential to identify vascular segments that warrant surveillance or intervention and to potentially diagnose an underlying heritable arteriopathy. Expert consensus advises head-to-pelvis screening for EVAs following SCAD, and computed tomography angiography (CTA) is favored as the initial imaging modality of choice, particularly for the detection of FMD.2,4,13,14
Despite these recommendations, comprehensive imaging occurs in less than 20% of cases. 15 This may be in part due to a lack of consistent imaging protocols, logistical challenges in imaging multiple body segments, variability in vascular imaging expertise, and radiation and contrast concerns. 16 Prior studies have proposed single-session head-to-pelvis imaging protocols. However, these studies have not assessed whether these protocols reduce radiation or contrast exposure, 17 or are effective in identifying EVAs.17,18
To address this care gap, we developed a single-session CTA (SS-CTA) protocol that allows assessment of all major vascular territories from head to pelvis. We deployed this protocol for patients receiving care in a dedicated arteriopathy clinic and compared contrast exposure, radiation exposure, and diagnostic yield to alternative multiacquisition protocols at our own and other institutes.
Methods
Study design and population
This is a retrospective, observational cohort study designed to evaluate radiation exposure, contrast volume, and diagnostic yield of an institutional CTA protocol compared to usual care. The study population included all consecutive patients with a clinical history of SCAD who presented to the Vanderbilt University Medical Center (VUMC) Arteriopathy Clinic from June 2016 to January 2022. All patients provided informed consent for participation in an institutional SCAD registry. Study approval was granted through the Institutional Review Board at VUMC.
Clinical details
Baseline characteristics and past medical history were extracted from patient report and the electronic medical record. Electrocardiograms, laboratory data, and hospital cardiology notes were reviewed to determine the presentation of SCAD, which included ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), sudden cardiac death, or shock. Original coronary angiograms were independently reviewed by a vascular cardiologist (ESHK) to confirm SCAD diagnosis, vessel involvement, and type. Details of medical, percutaneous, and surgical management were also recorded from the medical record at VUMC and/or based on comprehensive review of medical records from the facility where the event was treated.
Imaging evaluation
All patients with a history of SCAD were referred for head-to-pelvis CTA for evaluation of EVAs. Of the 197 patients enrolled in the study, 160 patients (81.2%) underwent complete imaging. Only those who completed the recommended imaging were included in this analysis. For patients in the SS-CTA protocol group, head-to-pelvis cross-sectional imaging was performed at our center in a single sequence. The multisession imaging group (MS-CTA) underwent asynchronous EVA screening, often having head-and-neck imaging performed separately from chest, abdomen, and pelvis imaging. The MS-CTA group included patients that received all images at outside facilities or patients that had a combination of VUMC and outside images. All post-SCAD CTA images and reports from outside institutions were collected. The site of imaging, radiation dosage, contrast volume, and presence of EVAs by radiology interpretation were recorded for all study subjects. All arterial imaging was independently re-analyzed by a vascular cardiologist (ESHK) for confirmation of EVAs. EVAs were defined as beading typical of multifocal FMD, aneurysm, pseudoaneurysm, and/or dissection in the following arterial beds: intracranial, extracranial carotid, extracranial vertebral, aorta, renal, mesenteric, and iliac.
Study outcome
The primary outcomes were radiation dose and contrast volume, which were ascertained from the CT radiation dose sheet accompanying all studies. Radiation exposure was estimated by dose length product (mGy-cm), generated by the scanner at the time of imaging. Contrast volume was reported in milliliters. The secondary outcome was the prevalence of EVAs.
Imaging technique
The VUMC SS-CTA SCAD protocol includes CTA of the brain, neck, chest, abdomen, and pelvis in a single session. The protocol utilizes dual-source CT and rapid sequential acquisition with initial scanning of the head and neck immediately followed by a scan of the chest, abdomen, and pelvis. The dual-energy head/neck protocol was performed with head-first, supine positioning, with the head and neck positioned and centered in a head holder and patients’ arms by their sides and scanned in the craniocaudal direction. A total of 60 mL of Omnipaque 350 (GE Healthcare, Chicago, IL, USA) was used at an injection rate of 4 mL/sec. Contrast was preceded by a test saline flush of 40 mL, and a postcontrast saline flush of 20 mL was administered. An arterial phase only of the region of interest was achieved by using a region of interest trigger placed in the ascending aorta with an auto-trigger set at 100 Hounsfield units. Pitch was set at 1, kV was set at 90, and quality reference mAs was set at 220 with an effective mAs of tube A at 90 and an effective mAs of tube B at 69.
The scan of the chest, abdomen, and pelvis was performed immediately thereafter with the scan in the craniocaudal direction during inspiration in the arms-up position. Contrast consisted of 100 mL of Omnipaque 350 at an injection rate of 4–5 mL/sec. A similar pre- and postcontrast saline flush was administered. An arterial phase only of the region of interest was achieved by using a region of interest trigger placed in the ascending aorta with an auto-trigger set at 150 HU. Pitch was set at 1.5, reference kV was set at 100, and reference mAs was set at 120. The protocols used a slice thickness of 3 mm and acquisition of 0.6 mm. CARE Dose4D (Siemens Healthcare, Erlangen, Germany) was used for all imaging studies.
Statistical analysis
Patients were stratified into two groups: SS-CTA and MS-CTA. We reported descriptive statistics on demographic and clinical characteristics, including mean (SD) and n (%). We evaluated differences between imaging groups using chi-squared tests (or Fisher’s exact tests) for categorical variables and Wilcoxon rank-sum tests for continuous variables.
Results
Baseline characteristics
All patients had complete head-to-pelvis CTA imaging. A total of 97 patients underwent the SS-CTA protocol and 63 patients underwent the MS-CTA protocol. The patients in the MS-CTA group underwent an average of 2.4 scans. Among the 160 patients, the mean age at SCAD event was 47.2 ± 10.2 years, 151 (94.4%) were women, and 143 (93.5%) were White (Table 1). Most patients had few or no traditional coronary artery disease risk factors, such as hypertension (35.0%), hyperlipidemia (31.2%), diabetes (5.0%), or smoking (24.4%). There were no significant between-group differences in baseline characteristics, including age, sex, or medical history. The SS-CTA group had a lower prevalence of family history of aneurysm (17.6% vs 33.3%, p = 0.026).
Baseline characteristics.
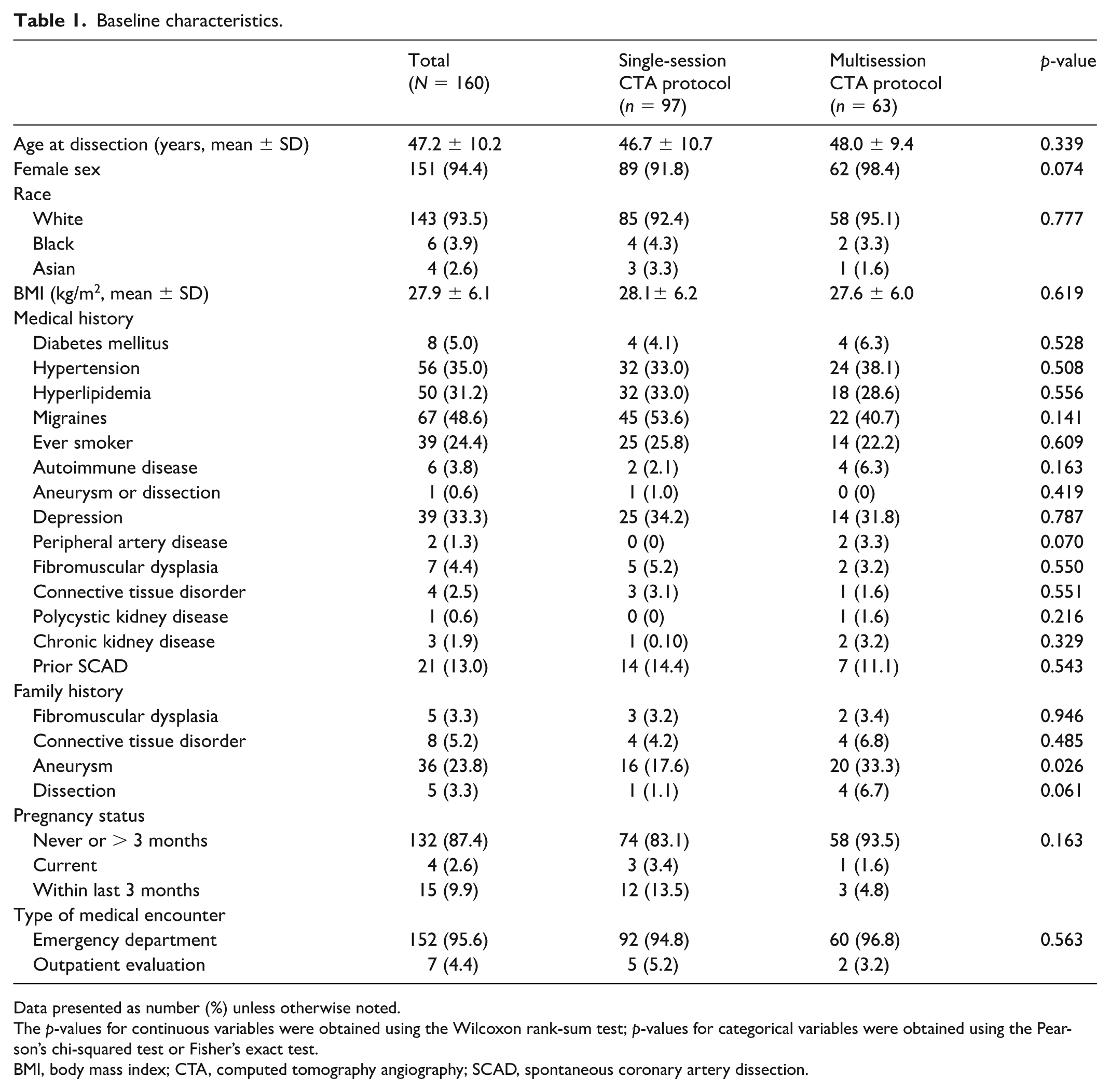
Data presented as number (%) unless otherwise noted.
The p-values for continuous variables were obtained using the Wilcoxon rank-sum test; p-values for categorical variables were obtained using the Pearson’s chi-squared test or Fisher’s exact test.
BMI, body mass index; CTA, computed tomography angiography; SCAD, spontaneous coronary artery dissection.
SCAD presentation and management
There were no differences in SCAD precipitating factors, presentation, management, procedural complications, and discharge medications between imaging groups (Table 2). The majority of patients (98, 61.3%) were managed without coronary revascularization. The SS-CTA protocol group had a greater prevalence of type 4 dissection, defined as complete vessel occlusion mimicking atherosclerotic thrombi (7.2% vs 0%, p = 0.029). Most patients were discharged on aspirin (89.4%), another antiplatelet agent (75.6%), and a beta blocker (83.8%).
Initial presentation and management.
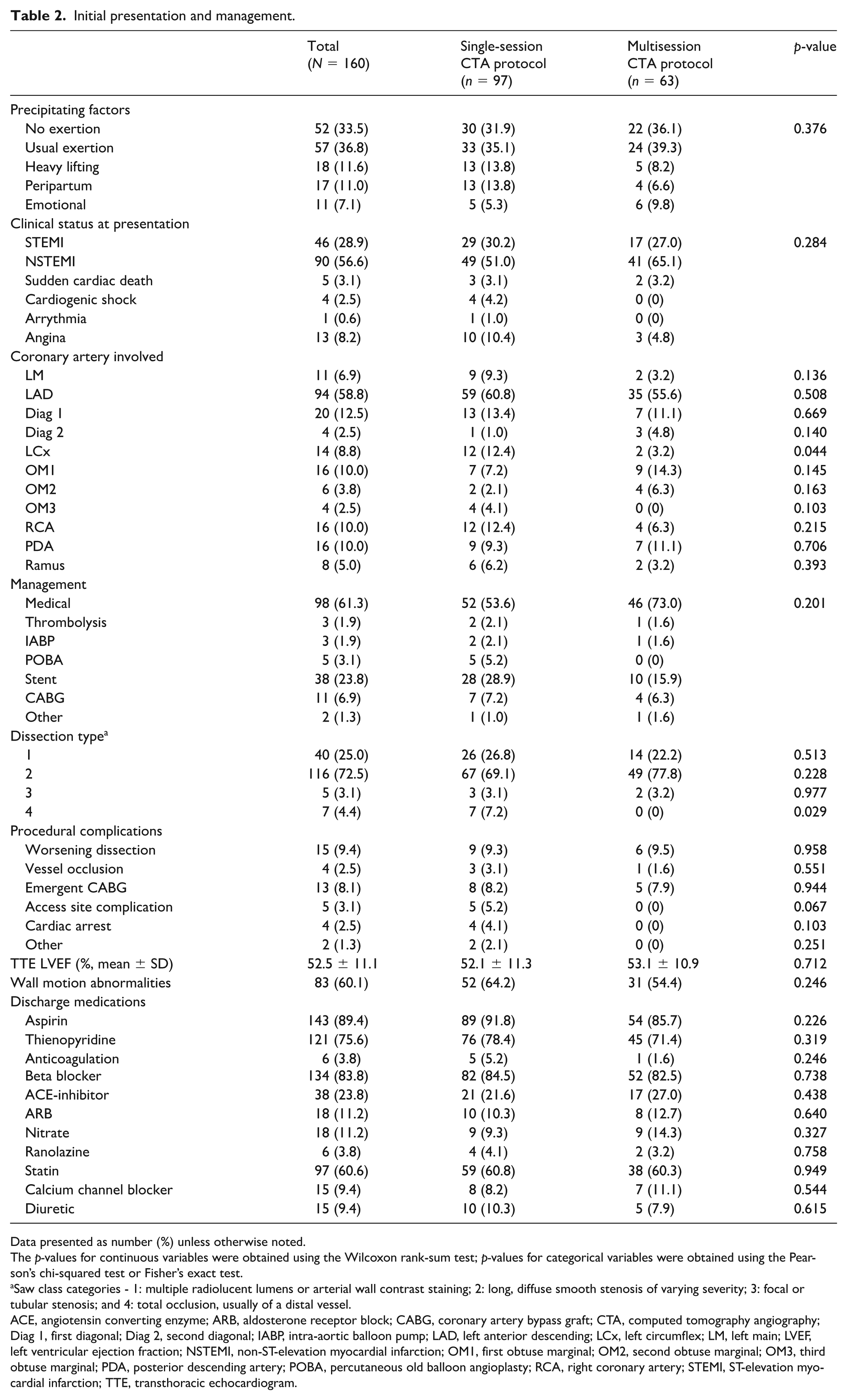
Data presented as number (%) unless otherwise noted.
The p-values for continuous variables were obtained using the Wilcoxon rank-sum test; p-values for categorical variables were obtained using the Pearson’s chi-squared test or Fisher’s exact test.
Saw class categories - 1: multiple radiolucent lumens or arterial wall contrast staining; 2: long, diffuse smooth stenosis of varying severity; 3: focal or tubular stenosis; and 4: total occlusion, usually of a distal vessel.
ACE, angiotensin converting enzyme; ARB, aldosterone receptor block; CABG, coronary artery bypass graft; CTA, computed tomography angiography; Diag 1, first diagonal; Diag 2, second diagonal; IABP, intra-aortic balloon pump; LAD, left anterior descending; LCx, left circumflex; LM, left main; LVEF, left ventricular ejection fraction; NSTEMI, non-ST-elevation myocardial infarction; OM1, first obtuse marginal; OM2, second obtuse marginal; OM3, third obtuse marginal; PDA, posterior descending artery; POBA, percutaneous old balloon angioplasty; RCA, right coronary artery; STEMI, ST-elevation myocardial infarction; TTE, transthoracic echocardiogram.
Radiation dose and contrast volume
The SS-CTA protocol had a 24% lower radiation dose (1873 ± 676 mGy-cm vs 2452 ± 1099 mGy-cm, p < 0.001) compared to the MS-CTA protocol (Table 3). Additionally, the SS-CTA protocol had a 21% lower contrast volume (161.2 ± 48.7 mL vs 203.8 ± 68.4 mL, p < 0.001). Baseline creatine at index scan was equal between imaging groups (0.8 vs 0.8 mg/dL, p = 0.376).
Radiation exposure and contrast volume by imaging protocol.

The p-values were obtained using the Wilcoxon rank-sum test.
CTA, computed tomography angiography.
Prevalence of extracoronary vascular abnormalities (EVAs)
EVAs were common in both groups (55.7% vs 57.1%, p = 0.854) (Table 4). Carotid (40.0%) and renal (33.8%) involvement were the most common, and less frequent were intracranial EVAs (8.1%). There was no difference in the detected prevalence of FMD, aneurysm, dissection, and pseudoaneurysm between the protocols.
Detection of extracoronary vascular abnormalities by imaging protocol.
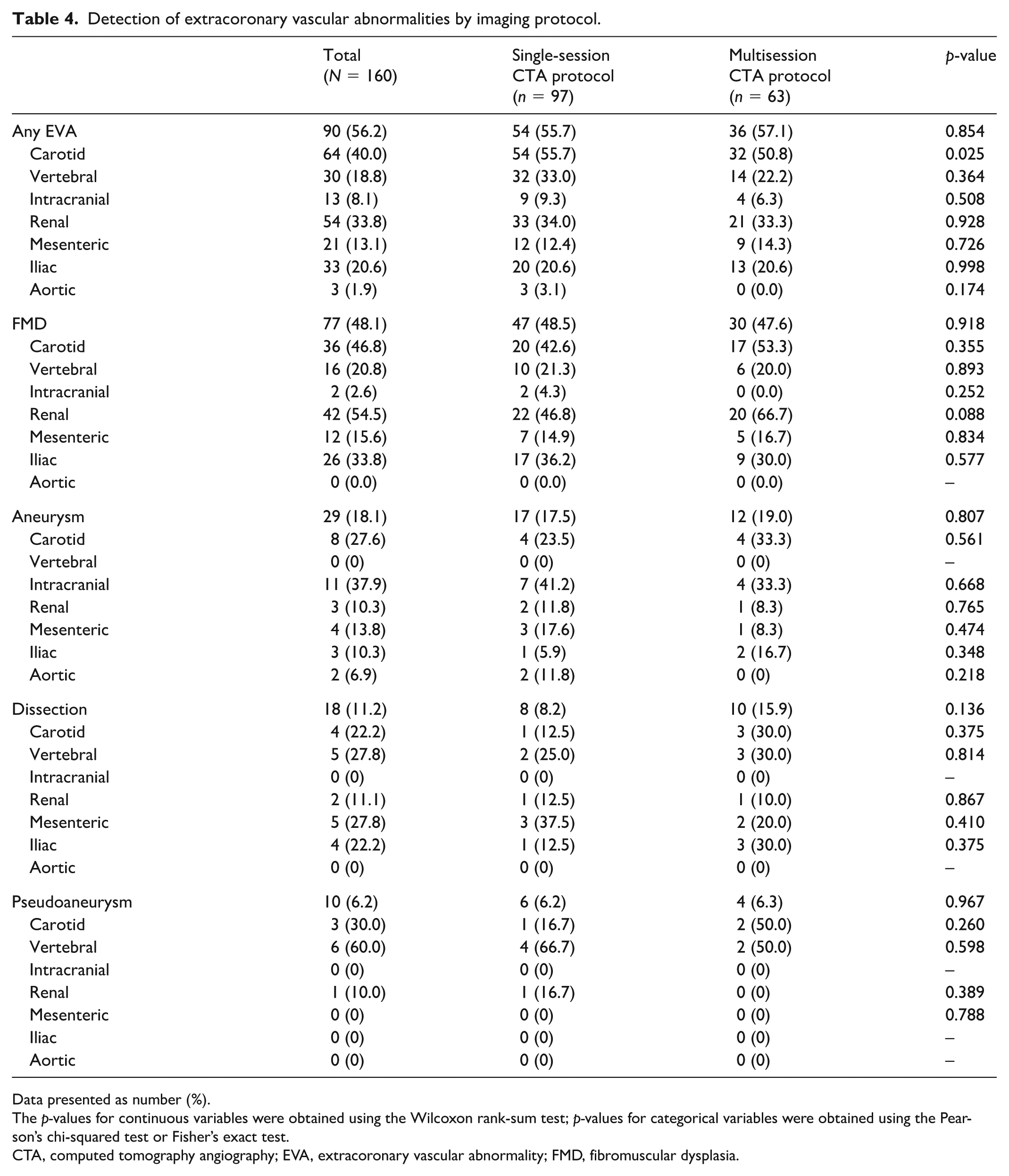
Data presented as number (%).
The p-values for continuous variables were obtained using the Wilcoxon rank-sum test; p-values for categorical variables were obtained using the Pearson’s chi-squared test or Fisher’s exact test.
CTA, computed tomography angiography; EVA, extracoronary vascular abnormality; FMD, fibromuscular dysplasia.
Discussion
In a single-center cohort of 160 patients with SCAD treated in a dedicated arteriopathy clinic, we showed that our single-acquisition CTA imaging protocol led to significant reductions in radiation exposure and contrast volume for head-to-pelvis imaging compared to alternative usual care protocols (Supplemental Figure S1). Though some prior studies have documented the importance of EVA screening in patients with arteriopathies, our study builds upon this literature by deploying a rapid, single-acquisition imaging protocol instead of relying on separate imaging studies to complete screening. 6 This study is further distinct from previously published protocols as it includes intracranial vascular assessment 17 and was deployed in a population with confirmed SCAD rather than FMD. 18 Despite less radiation and contrast, diagnostic yield in terms of EVA detection was no different, including detection of FMD, aneurysm, or dissection. Our findings provide an important tool to help facilitate appropriate diagnostic and surveillance imaging for this patient population in which such imaging is not consistently performed.
Many patients with SCAD present at an early age, and lifetime radiation exposure is of particular concern. This must be balanced by the need for high-resolution axial imaging to help identify EVAs that may warrant intervention or surveillance. Prior studies have found that the lifetime attributable risk of cancer in 40-year-old women can be one in 270 from a single coronary angiography CT scan. 19 Based on current practices, 5% of new annual cancer diagnoses are projected to be CT-associated cancer. 20 A recent systematic review and dose–response meta-analysis demonstrated an inordinate increase in cancer risk from CT scans, with the linear increase based on radiation dose and multiple CT scans. 21 Young age and female sex, as typified in the SCAD population, are risk factors for increased tissue sensitivity to radiation. 22 Clinicians have a responsibility to minimize patient exposure through judicious imaging modality selection and protocol optimization.22,23 The SS-CTA protocol effectively decreased the radiation dose while still yielding sufficient imaging for diagnosis.
Iodinated contrast administration for angiographic imaging confers risk of hypersensitivity reaction, contrast extravasation, and kidney injury. Contrast-induced nephropathy (CIN) following administration of iodinated contrast media may increase hospital length of stay and morbidity. 24 The risk of CIN increases with older age, diabetes, chronic renal impairment, and contrast volume greater than 100 mL.25,26 As demonstrated among individuals who underwent MS-CTA imaging, the mean contrast volume administered exceeded 200 mL. Administration of such a volume that exceeds recommendation is reflective of the real-world experience of patients with SCAD and is likely reflective of heterogeneous imaging center routines, image quality subjectivity, and patient-specific parameters. 27 However, national consensus guidelines have demonstrated that the risk of CIN has been conflated with uncontrolled studies that fail to link a causal relationship between contrast media and acute kidney injury. 28 The VUMC SCAD protocol was associated with a 21% reduction in contrast volume. Notably, our patient population was middle-aged with normal baseline renal function and was therefore at low overall risk of CIN. 29
The reported FMD prevalence detected with CTA in patients with SCAD ranges from 18% to 86%.3,9 –11,30 –32 This wide variation is likely the result of small sample sizes, inconsistent protocols, and incomplete imaging. Concomitant FMD was seen in > 50% of patients with SCAD in cohorts with complete head-to-pelvis imaging, which is consistent with our study findings. 13 This demonstrates that our single-acquisition CTA protocol confers numerous benefits, such as reduced radiation exposure and streamlined care, without any decrease in diagnostic yield.
The diagnosis of EVAs could have a significant impact on prognosis, lifestyle counseling, and family screening. The goal of EVA surveillance is not only to solely diagnose conditions such as FMD, but also to detect clinically actionable abnormalities. Common in the FMD population, intracranial aneurysms are high-risk, potentially practice-changing EVAs. 33 In our study population, one in 12 patients had an intracranial abnormality requiring either intervention or surveillance.
There are several limitations to this study. First, this is a single-center, retrospective study of patients referred to a dedicated arteriopathy clinic, thus introducing selection and referral bias which may influence the generalizability of some of our results. Although we analyzed the radiation dose emitted by the CT scanner, it was not possible to measure the precise amount of radiation absorbed by the individual patient. Future studies would be needed to evaluate the effective dose of radiation exposure, accounting for both radiation amount and exposed organ sensitivity. Direct comparison of the SS-CTA protocol and MS-CTA protocol on a per-patient basis was also not possible. Our SS-CTA protocol uses certain imaging technologies, such as dual-energy CT, that may be unavailable at other institutes, and this could slow broad adoption of our protocol. Long-term outcome data related to EVAs detected in this analysis were unavailable. Finally, imaging analysis was unblinded.
Conclusions
This study demonstrates the reliability and feasibility of EVA screening in a SCAD population using a standardized single-acquisition CTA protocol, with a lowering of radiation dose and contrast volume. This adheres to the mantra of radiation exposure protection (‘as low as reasonably possible’) to minimize radiation-induced cancer risk. It is our hope that this will serve as a blueprint for EVA screening in patients with SCAD across institutions.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251383872 – Supplemental material for A single-session CT angiography protocol for extracoronary vascular abnormality screening in spontaneous coronary artery dissection (SCAD)
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251383872 for A single-session CT angiography protocol for extracoronary vascular abnormality screening in spontaneous coronary artery dissection (SCAD) by Jacqueline M Visina, Aaron W Aday, Nowrin Haque, Shi Huang, Thomas A McLaren, Brett R Murdock and Esther SH Kim in Vascular Medicine
Footnotes
Data availability
The data used in these analyses are available upon reasonable request from other investigators, subject to requirements of a data use agreement, by contacting the corresponding author.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr Aday reports receiving consulting fees from Janssen, Merck, and Tactile Medical outside of the current work. The remaining authors have nothing to disclose.
Funding
This work was supported by the National Institutes of Health (Dr. Aday: number K23 HL151871).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
