Abstract
Background:
We aimed to investigate metabolomic alterations in peripheral artery disease (PAD) by analyzing blood from the femoral artery and vein, assessing metabolite release/uptake in chronically ischemic lower limbs (PAD group) and nonischemic limbs (controls), and evaluating the representativeness of upper-limb venous samples.
Methods:
In this exploratory case–control study, 24 patients with PAD (Fontaine IIb and III) and 18 control subjects with stable angina were enrolled. Blood was drawn from the femoral artery and vein, and the antecubital fossa. Metabolomic profiling of serum samples was performed using liquid chromatography and tandem mass spectrometry. We analyzed 560 metabolites, including amino acids and derivatives, acylcarnitines, ceramides, phosphatidylcholines, tri- and diglycerides, cholesteryl esters, sphingomyelins, and fatty acids, as well as 52 metabolic ratios/sums across various biochemical classes.
Results:
Femoral arteriovenous (AV) concentration differences with in-group significance in at least one group and between-group significance was observed for 47 metabolites and five metabolic sums. Among these, eight metabolic variables in antecubital vein samples were significant. Correlation between AV differences and antecubital vein samples was weak.
Conclusion:
Using the femoral AV concentration differences of metabolites, we identified significant local metabolomic shifts in the PAD group. Most of these changes were not revealed using traditional upper-limb venous blood sampling. Further study of AV differences could improve the understanding of the pathophysiology of PAD.
Keywords
Background
Peripheral artery disease (PAD) is a malignant vascular condition resulting from systemic atherosclerosis. It adversely affects patients’ quality of life and is associated with a high risk of major cardiovascular (CV) and limb events. 1 Available medications provide only modest efficacy in functional improvement, and the success of limb salvage in patients does not always align with the extent of large vessel revascularization. 2 This emphasizes the need for novel approaches to PAD therapy.
Chronic ischemia of the lower extremities leads to a range of metabolic alterations and tissue damage that contribute to the pathophysiology of the disease. 3 Understanding these metabolic effects can reveal new therapeutic targets and biomarkers of disease progression. Large-scale detection and quantification of metabolites in biological samples—a study known as metabolomics—is gaining prominence in vascular research.4,5 Unlike other ‘omics’ strategies, metabolomics aligns more closely with phenotypic changes and serves as a translational bridge between clinical practice and basic science.
The traditional method for studying circulating molecules in PAD involves upper-limb venous samples, with findings then extrapolated to the ischemic lower limb. Alterations in the circulatory levels of amino acids, acylcarnitines, glycerophospholipids, and intermediates of the tricarboxylic acid (TCA) cycle associated with PAD have been described previously.6–8 Yet, data regarding the local release or uptake of metabolites in the PAD circulation remain elusive.
In this study, our goal was to uncover local (in situ) metabolomic changes in patients with PAD and chronic ischemia by examining blood from the femoral artery and vein, thereby isolating the metabolic activity of the lower extremity. We used relative arteriovenous (AV) concentration differences to gauge metabolite release or uptake in the chronically ischemic lower limb. By comparing the ischemic lower limb (PAD group) with the control group, we aimed to reveal metabolomic patterns associated with PAD. Additionally, we evaluated the efficacy of traditional upper-limb blood samples in capturing these changes.
Methods
Eligibility and study groups
We enrolled 24 patients with PAD who had chronically ischemic lower limbs, and 18 patients with stable angina pectoris as the control group. The participants were recruited from Tartu University Hospital’s Surgery Clinic and Heart Clinic before their elective revascularization procedures. We based our sample size on previous AV concentration difference studies of metabolites in other anatomical sites, given the absence of comparable studies on PAD.9,10 The inclusion criteria for the PAD group included either a pain-free walking distance of less than 100 meters on a treadmill or pain at rest, with tissue necrosis, wounds, and acute ischemia being excluded. Approximately 50% of patients had distal infrainguinal lesions, 20% had proximal suprainguinal lesions, and 30% had combined disease affecting both levels. For the control group, the absence of chronic lower-limb ischemia was confirmed by lack of symptoms and by manual ankle–brachial index (ABI) measurements with Doppler between 0.9 and 1.3.
The exclusion criteria for both groups included recent myocardial infarction, revascularization, or stroke (all ⩽ 6 months), acute or chronic inflammatory conditions, severe heart or kidney insufficiency (estimated glomerular filtration rate [eGFR] < 30 mL/min/1.73 m2), oncological disease, inability to give informed consent, or inability to understand the study protocol.
Study protocol
For the PAD group, the claudication distance was measured on a treadmill at 3.2 km/h with 2° incline increases every 2 minutes until the patient first reported typical ischemic pain. For the ABI assessment, manual measurements were performed using the Doppler method with a 5-MHz probe (Dopplex DMX, Huntleigh Healthcare Ltd, Cardiff, UK).
The following day, blood samples were collected from the femoral artery, femoral vein, and antecubital fossa between 07:00 and 14:00 after overnight fasting and abstention from alcohol and tobacco. For the control group, blood sampling and ABI measurements were conducted on the same day.
The study was carried out in accordance with the Declaration of Helsinki, was approved by the Ethics Committee on Human Research of the University of Tartu (approval number 295/T-10), and written informed consent was obtained from all participants.
Biochemical blood analyses
Blood samples from the antecubital fossa for clinical biochemistry analysis and complete blood count were collected using serum separator tubes (BD Vacutainer SST II Advance Tubes; Fisher Scientific, Loughborough, UK) and EDTA tubes (BD Vacutainer EDTA Tubes; Fisher Scientific), respectively. A local clinical laboratory measured common laboratory characteristics using automated analyzers and standardized procedures.
Targeted identification and quantification of serum metabolites
Blood samples intended for metabolite detection were drawn from the antecubital fossa vein, as well as from the femoral artery and vein, using plain tubes (Plain BD Vacutainer Tubes, Fisher Scientific). After centrifugation at room temperature (3000 rpm for 10 minutes), serum was transferred to 0.5 mL tubes and stored at −80°C until analysis.
The MxP Quant 500 Kit (Biocrates Life Sciences AG, Innsbruck, Austria) was employed to measure 628 metabolites across 26 biochemical classes. For 13 compound classes of small molecules (alkaloids, amine oxides, amino acids, bile acids, biogenic amines, carboxylic acids, cresols, fatty acids, hormones, indoles and derivatives, nucleobases, and vitamins), liquid chromatography with tandem mass spectrometry was used. Flow injection analysis with tandem mass spectrometry was employed for 12 lipid classes (acylcarnitines, phosphatidylcholines (PCs), lysophosphatidylcholines (lysoPCs), cholesteryl esters, sphingomyelins, ceramides, dihydroceramides, hexosylceramides, dihexosylceramides, trihexosylceramides, diglyceridess (DGs), triglycerides [TGs]) and hexoses.
The structure of lipid side chains can be denoted as Cx:y, where x indicates the number of carbon atoms in the side chain and y represents the number of double bonds. PCs and lysoPCs are further classified based on the presence of ester and ether bonds within the glycerol moiety. Double letters (aa = diacyl, ae = acyl-alkyl) indicate that two glycerol positions are connected to a fatty acid residue, and a single letter (a = acyl, e = alkyl) signifies a bond with only one fatty acid residue.
Measurements were performed as described in the manufacturer’s manual on the Xevo TQ-XS mass spectrometer using ACQUITY ultra-performance liquid chromatography (Waters, Milford, MA, USA). Metabolites were identified and quantified using multiple reaction monitoring along with internal standards. The MetIDQ software package, an integral part of the MxP Quant 500 kit (Biocrates), automatically calculated the concentrations of all metabolites in μM. Additionally, the software computed 235 predefined metabolic sums and ratios, of which 52 relevant to our research were selected. The quality control serum with known concentrations of metabolites was supplied by the kit vendor and one quality control was analyzed after every 15 samples. The median coefficient of variation was 21.4%. If excluding DGs, TGs, and ceramides, the median coefficient of variation was 9.9%. Accuracy was found to be 70–114%. In general, the quality controls indicate similar performance as reported by others for this metabolomics kit. 11
Based on femoral blood samples, AV concentration differences of metabolites were calculated by dividing the femoral vein value by the femoral artery value. Because limb blood flow was not measured, we define the AV difference here purely as the femoral vein-to-artery concentration ratio of each metabolite. A ratio > 1 indicates net release from the limb; a ratio < 1 indicates net uptake. These concentration-based differences represent relative concentration changes rather than absolute metabolic fluxes.
Statistical analysis
After excluding variables with less than 66% valid measurements above the detection limit, we analyzed 560 metabolites and 52 metabolic ratios/sums. Missing data were imputed using the random forest method. Analyses were carried out using IBM SPSS Statistics, version 25.0 (IBM Corp.), with figures crafted using SPSS, and Python libraries Pandas, Seaborn, and Matplotlib.12–14 We employed nonparametric tests: Mann–Whitney U-test for PAD versus controls comparisons, Wilcoxon signed-rank test for in-group AV concentration difference significance, and Spearman correlations for AV concentration differences versus antecubital fossa venous samples. A chi-squared test was utilized to assess differences in medication use and comorbidities. A p-value below 0.05 was considered significant. For a meaningful difference in the AV concentration differences between the groups, at least one group needed a statistically significant in-group concentration difference, coupled with a significant difference between the groups.
Results
Table 1 shows the characteristics of the study groups. There were no statistically significant differences in clinical attributes, such as age, sex, body mass index, high-sensitivity C-reactive protein, and lipid profile, between the PAD and control groups. The PAD group showed significantly higher peripheral systolic blood pressure, lower cardiac troponin T, and reduced ABI compared to the control group. The PAD group had significantly shorter fasting durations compared to controls, with no overlap between groups.
Clinical and laboratory characteristics of the peripheral artery disease and control groups.
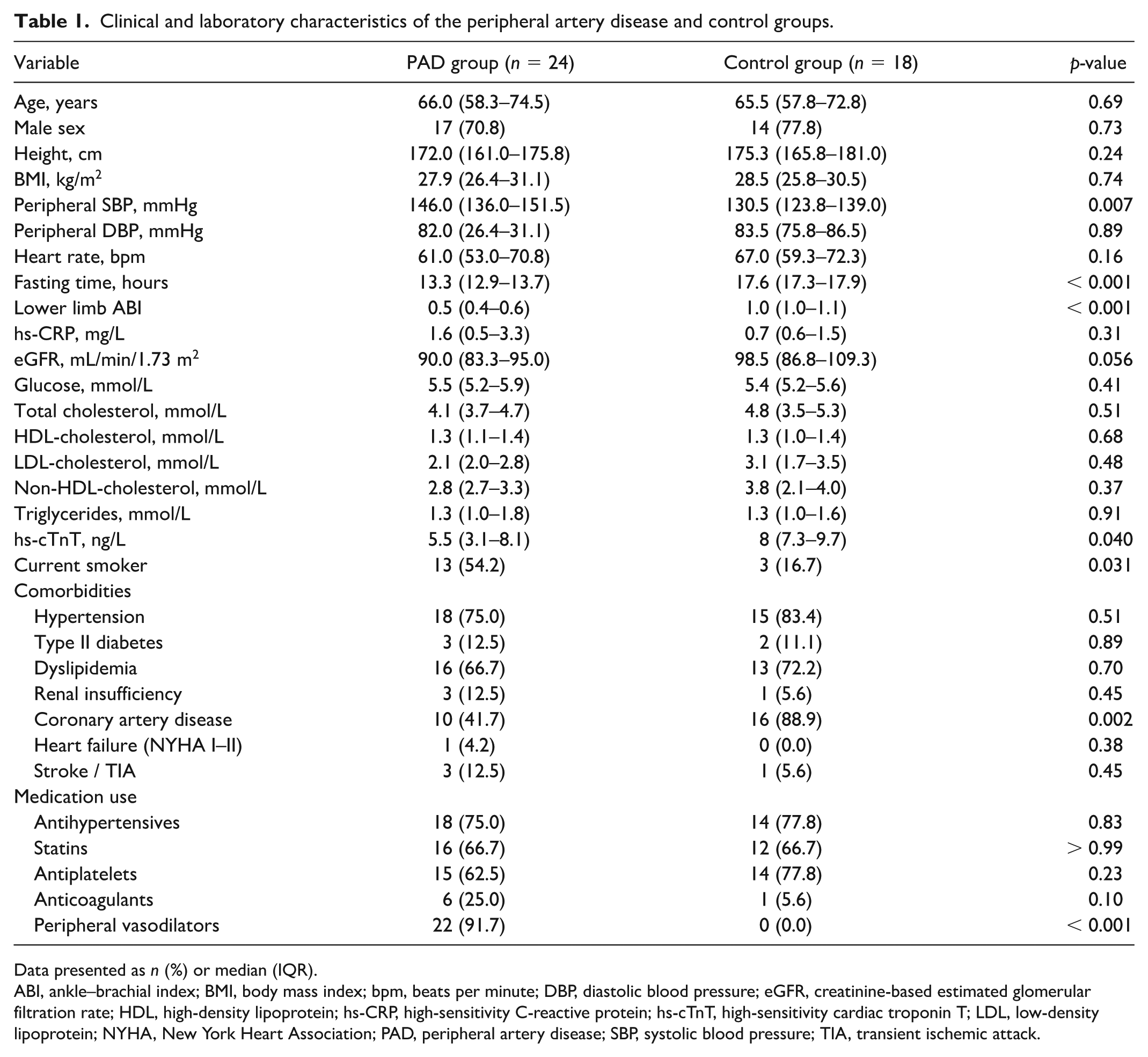
Data presented as n (%) or median (IQR).
ABI, ankle–brachial index; BMI, body mass index; bpm, beats per minute; DBP, diastolic blood pressure; eGFR, creatinine-based estimated glomerular filtration rate; HDL, high-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein; hs-cTnT, high-sensitivity cardiac troponin T; LDL, low-density lipoprotein; NYHA, New York Heart Association; PAD, peripheral artery disease; SBP, systolic blood pressure; TIA, transient ischemic attack.
Additionally, no significant differences were observed in the prevalence of hypertension, type II diabetes, dyslipidemia, renal insufficiency, heart failure, stroke, or in the usage of antihypertensive, antidyslipidemic, antiplatelet, and anticoagulant medications (Table 1). However, statistically significant differences were identified, with the PAD group showing higher use of peripheral vasodilators (92% PAD vs 0% controls, p < 0.001) and a greater prevalence of current smoking (54% PAD vs 17% controls, p = 0.031).
In the PAD group, 33% were classified as Fontaine stage III, and 66% as Fontaine IIb. For those in Fontaine IIb, the median pain-free claudication distance measured on a treadmill was 38.5 meters.
Femoral arteriovenous (AV) concentration differences of serum metabolites
A statistically significant in-group femoral AV concentration difference was found for 70 individual metabolites and four metabolic ratios/sums (74 variables in total) in the PAD group and 62 individual metabolites and 11 metabolic ratios/sums (73 variables in total) in the control group. Only 14 metabolic variables were found to have a significant concentration difference in both groups (Figure 1 and Supplemental Table S1). Medium chain acylcarnitines and amino acids contributed the most to these metabolites. Their concentration difference was also in the same direction in both the PAD and the control groups. PC aa C32:2 and taurodeoxycholic acid were exceptional as both had a significantly increasing concentration difference in the PAD group and a significantly reducing concentration difference in the control group.
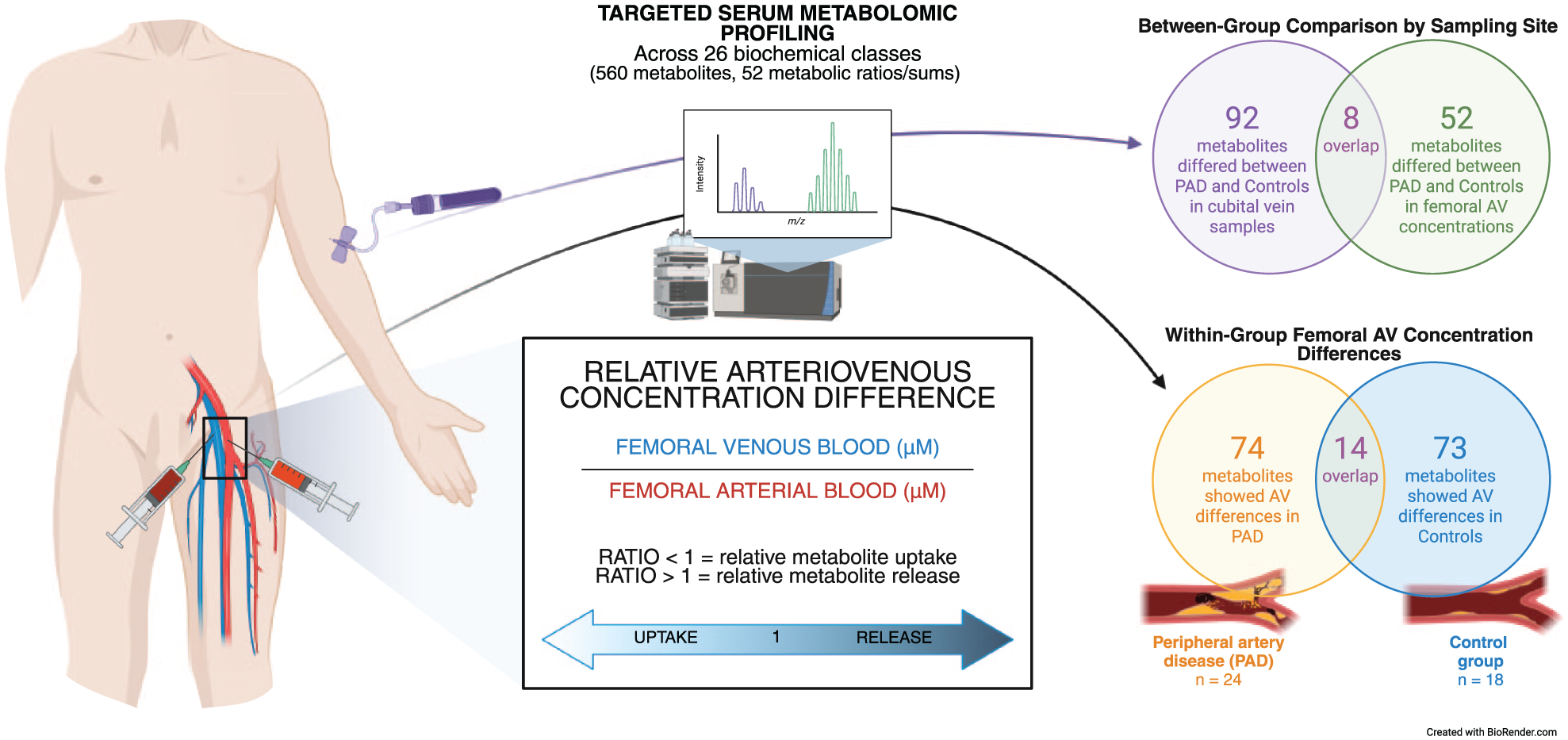
Study design and key findings.
Applying the study criteria for meaningful changes in the AV concentrations, we observed femoral AV concentration differences with in-group significance in at least one group and between-group significance for 47 distinct metabolites and five metabolic sums (52 variables in total). These metabolites exhibited either a relative release or uptake in the limb, coupled with a significant difference between the groups (Figures 1 and 2, Table S1, and Figures S1 and S2).
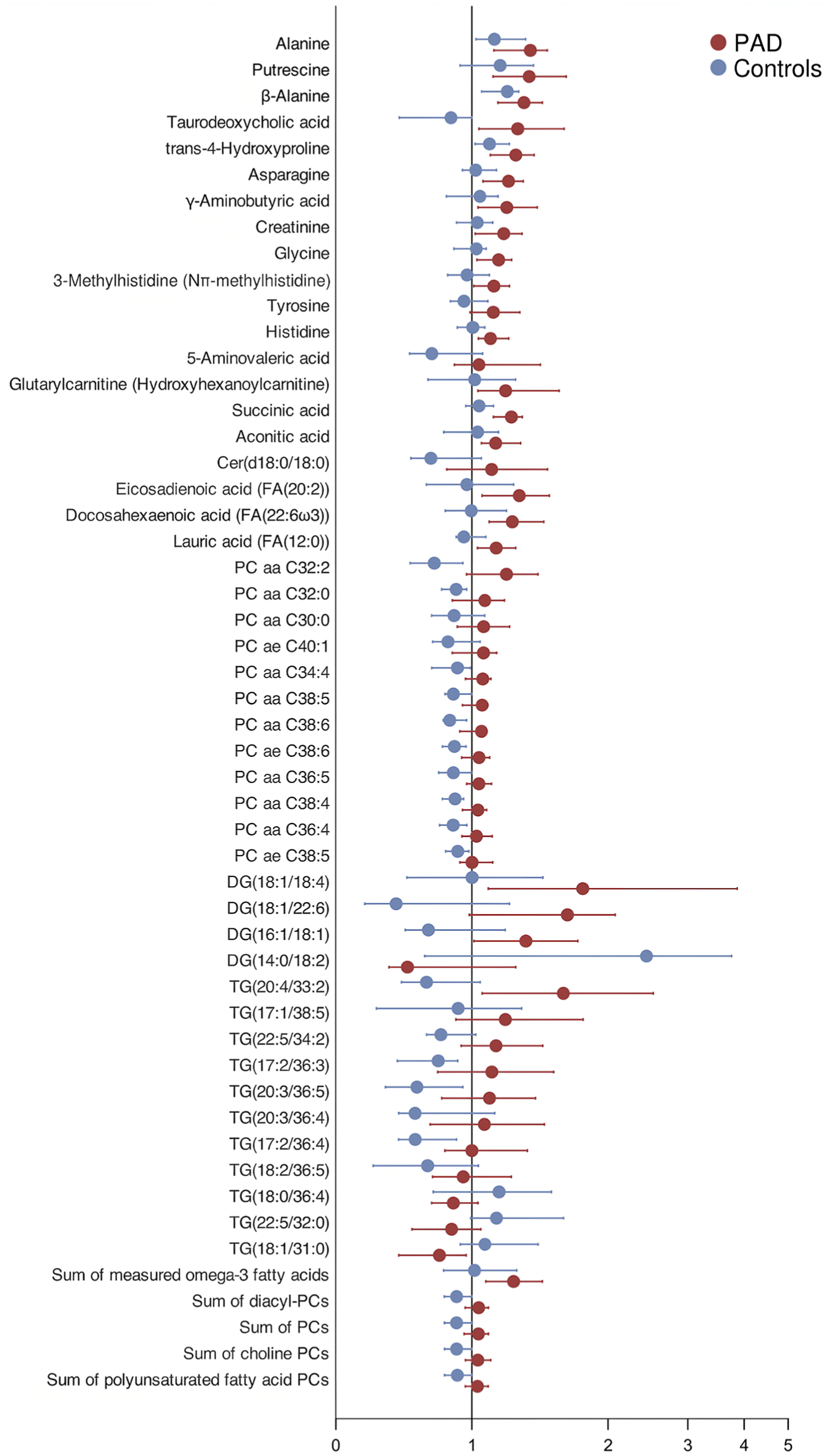
Medians of femoral arteriovenous metabolite concentration differences presented on a logarithmic scale displaying statistical significance (p < 0.05) between the peripheral artery disease (PAD) and control groups with 95% CIs.
Serum metabolite levels in antecubital venous blood in comparison to femoral AV concentration differences
Out of 52 metabolites or metabolite groups with statistically significant femoral AV concentration differences, the absolute values of only eight showed further statistical significance in forearm blood samples between the PAD and control groups: tyrosine, docosahexaenoic acid (FA[22:6ω3]), PC ae C40:1, PC ae C38:6, PC aa C38:5, DG(18:1/18:4), TG(18:0/36:4), and the sum of measured omega-3 fatty acids (Figure 1).
Furthermore, the AV concentration differences demonstrated limited correlation with the samples obtained from the upper extremity, revealing only a small number of statistically significant relationships. This observation applies to both PAD and controls, as depicted in Figure 3.
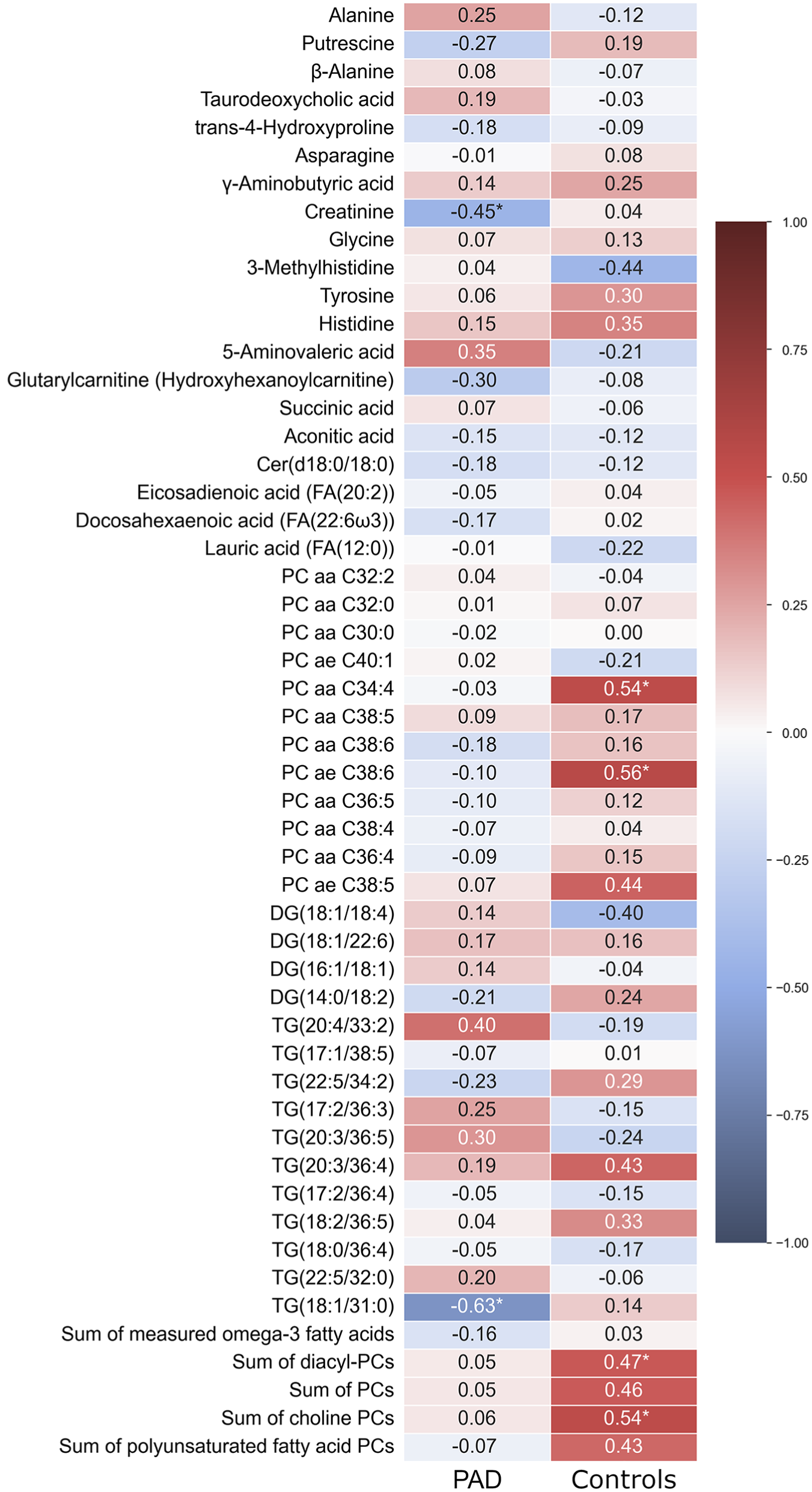
Heatmap illustrating the Spearman correlations between femoral arteriovenous metabolite concentration differences and the absolute values obtained from antecubital vein samples in both the peripheral artery disease (PAD) and the control groups.
Discussion
To our knowledge, this is the first study employing an exploratory targeted metabolomic approach to detect variations in the relative release and uptake of metabolites in the chronically ischemic lower limb of patients with PAD. We examined metabolomic shifts and limb–systemic circulation relationships using relative femoral AV concentration difference measurements. The distinct metabolomic shifts observed in the femoral AV concentration differences compared to systemic measurements suggest that standard upper-limb venous blood sampling may not adequately reflect localized pathophysiological changes in the lower extremity.
Altered energy metabolism in the ischemic limb
Several metabolites involved in glycolysis and amino acid metabolism showed net release from the ischemic limb in PAD, including lactate, alanine, histidine, 3-methylhistidine (Nπ-methylhistidine), and beta-alanine (Figure 2). Anaerobic glycolysis and amino acid catabolism are prominent features of ischemic metabolism. Elevated femoral AV concentration differences of lactate and alanine in PAD indicate increased reliance on glycolysis and amino acid catabolism under hypoxic conditions. Lactate concentration differences were elevated in both groups without significant between-group differences, whereas alanine showed significant release in both groups but was markedly greater in PAD, resulting in a significant between-group difference and potentially reflecting a more distinct metabolic signal of chronic ischemic stress. This is consistent with its formation from pyruvate via transamination, which relies on amino groups from catabolized amino acids, linking alanine metabolism to both energy and nitrogen handling. Within the PAD group, elevated AV concentration differences of additional amino acids and derivatives were observed, suggesting broader metabolic adaptations to ischemia. Notably, increased concentrations of histidine, 3-methylhistidine (Nπ-methylhistidine), and beta-alanine—all linked to carnosine and anserine metabolism—may reflect metabolic adaptations to ischemia, including altered pH buffering capacity and increased amino acid turnover. Although Nπ-methylhistidine is not a direct marker of muscle protein breakdown, its increased release may indicate shifts in muscle protein metabolism, particularly through anserine turnover. Histidine itself can originate from both dietary intake and protein catabolism, and beta-alanine is a known product of carnosine degradation.15–17
Succinate and aconitic acid, two key TCA cycle intermediates, showed increased AV concentration differences indicating net release from the ischemic limb in PAD, suggesting mitochondrial stress and impaired aerobic metabolism (Figure 2). Our findings are consistent with prior research indicating that succinate accumulation during ischemia results from TCA cycle slowdown and contributes to reperfusion injury via mitochondrial reactive oxygen species.18,19 The potential clinical significance of an elevated cardiac AV concentration difference of succinate was underscored in a previous study by its correlation with the extent of ischemia in patients with acute myocardial infarction. 20 Aconitic acid is a TCA cycle intermediate positioned upstream of two key dehydrogenases that irreversibly convert downstream intermediates to succinyl-CoA and succinate. Though this irreversibility suggests that aconitic acid accumulation may result from inhibition of these enzymes, it does not conclusively prove it. A redox imbalance during ischemia could impair NAD+-dependent dehydrogenases as well as aconitase, further contributing to the buildup of aconitic acid. 21
Medium-chain acylcarnitines, including octanoyl-, decanoyl-, and dodecanoylcarnitine, showed significant AV concentration differences in both groups without consistent patterns of net release or uptake, indicating ongoing fatty acid beta-oxidation under resting conditions (Table S1). 22 The lack of significantly greater release in PAD compared to controls may suggest that mitochondrial fatty acid metabolism is relatively preserved at rest in chronic ischemia.
Overall, these metabolic shifts appear to be limb-specific and were largely undetectable in antecubital vein samples. Systemic venous comparisons failed to capture key differences in glycolytic, amino acid, and TCA cycle intermediates—including alanine, histidine, succinate, and aconitate.
Lipid remodeling and membrane turnover
Analysis of lipid components revealed significant changes in PCs, which are essential and predominant components of cellular membrane structures. Their fatty acid composition and variability can be indicative of certain pathological conditions. 23 Prior literature indicates correlation between vascular damage and circulatory PC levels.24,25 Interestingly, analysis of femoral AV concentrations did not reveal a significant decrease in these PCs within ischemic extremities, but instead suggested either no net change or a trend toward release in the PAD group, whereas a clear uptake pattern was observed in the control group (Figure 2, Figure S2). Concentration differences of summed PC classes, including saturated PCs, monounsaturated fatty acid (MUFA), and polyunsaturated fatty acid (PUFA) PCs, were decreased in the control group. Intriguingly, no consistent AV shift in lysoPC levels was observed, implying no major change in phospholipase activity. These AV concentration differences were not mirrored in antecubital venous samples, where systemic PC concentrations were lower in the PAD group—similar to our earlier findings that patients with PAD have reduced PC levels compared to those with coronary artery disease and healthy controls. 26
In contrast, the concentration difference for total MUFAs was increased in the PAD group, whereas the PUFA concentration difference showed a trend toward elevation. Consistent with this trend, docosahexaenoic acid (DHA), a key PUFA, also demonstrated enhanced release in ischemic limbs (Figure 2). PUFAs are precursors for oxylipins (e.g., prostaglandins), which mediate both inflammatory responses and protective mechanisms against ischemic damage. Ischemic preconditioning is known to upregulate COX-2, the enzyme responsible for converting PUFAs into oxylipins. However, this process requires oxygen as a co-substrate and may therefore be ineffective during ischemia.27,28
Our study also made use of AV concentration differences to measure specific DGs and TGs, providing a more detailed insight than traditional cumulative measurements. Although we identified differences in several DG and TG AV concentrations across study groups, the data did not show consistent patterns (Figure 2, Table S1). Ischemic events, known to disrupt both metabolic and signaling pathways, can influence DG and TG processing. 29 This, combined with diet, fasting variation, inflammation and oxidative stress, adds layers of metabolic complexity. 30 Additionally, we observed no distinct release or uptake patterns in ceramides, sphingomyelins, or cholesteryl esters (Table S1).
Altogether, most AV concentration differences that were significantly different between the PAD and control groups showed higher values in PAD, indicating increased release of metabolites from tissue into the bloodstream in the ischemic limb (Figure 2). We believe that some of these increases reflect cellular damage, whereas others may result from regulated metabolic processes. Nonetheless, even if these metabolites are released secondary to damage or adaptive responses, their presence in the circulation could still influence ongoing injury or repair mechanisms and provide additional insight into the local pathophysiological state of the ischemic limb. Additionally, variation in fasting duration between groups may have influenced circulating levels of certain lipid species and other nutrient-sensitive metabolites, which should be considered when interpreting group-level differences.
Differences in circulating bile acid levels
One of the most unexpected findings in this exploratory analysis was the considerably higher levels of various bile acids (including total, conjugated, primary, secondary, and glycine-conjugated bile acids) in the antecubital venous blood of PAD compared to control subjects. Differences between femoral AV concentrations of bile acids were overall nonsignificant, with the exception of taurodeoxycholic acid, which appeared to be released from the ischemic limb but absorbed under nonischemic conditions. Although bile acids are synthesized in the liver and intestine, their bidirectional transport in skeletal muscle—via uptake carriers and receptors such as farnesoid X receptor (FXR) and Takeda G protein-coupled receptor 5 (TGR5) 31 —can shape femoral AV concentration differences in ischemic limbs. Though this finding could also be partially influenced by moderately different fasting times, we observed a similar pattern in a separate cohort of patients with Fontaine IIa PAD compared to healthy individuals (data not yet published). It is beyond the scope of this study to clarify the underlying mechanisms, but bile acids have increasingly been recognized as potential contributors to cardiovascular disease processes. 31 Notably, ursodeoxycholic acid (UDCA) has shown promise as a protective marker; 32 though UDCA itself was not measured here, its conjugated form, glycolursodeoxycholic acid (GUDCA), was elevated in the systemic circulation of patients with PAD. The relevance of bile acid levels and their AV concentration differences in the femoral circulation warrants further investigation.
Limited overlap of femoral concentration differences with upper-limb venous samples
The main aim of our study was to show, as a proof of concept, that—similar to the heart or kidney, where AV concentration differences have been shown to reflect organ-specific metabolism—it is possible to assess AV concentration differences in the lower limb, and that this approach provides additional, unique information about metabolism in chronic ischemia. We demonstrated that the metabolomic changes identified through femoral AV concentration differences showed only partial correlation with the absolute venous values obtained from the upper limb (Figure 3). Although a few significant correlations were identified, the overall trend suggests that the systemic metabolomic environment does not consistently reflect the specific alterations observed in the ischemic lower limb. This underscores the potential value of localized measurements in PAD, particularly when assessing ischemia and its metabolic consequences. Additionally, the anatomical distribution of lesions may influence these AV concentration differences, as more proximal disease could affect a larger vascular territory and produce broader metabolic shifts than distal involvement alone.
Limitations
We acknowledge several limitations in this study. First, its cross-sectional design limits the ability to draw causal inferences. Second, the relatively small sample size precluded multivariate analyses, limiting our ability to adjust for additional metabolic influences, including imbalances in smoking status and medication use. Specifically, peripheral vasodilators such as naftidrofuryl and pentoxifylline were used by the vast majority of patients with PAD but not by any controls, which may have influenced metabolite levels. Third, our focus on patients with Fontaine IIb–III PAD may not capture the full spectrum of metabolomic alterations occurring across different stages of PAD progression. Also, lesion distribution varied across participants, which may have influenced femoral AV concentration differences, as proximal disease may affect a larger vascular territory and exert broader metabolic effects than isolated distal lesions; future studies with larger or pooled samples should consider stratification by lesion level. Fourth, using patients with stable angina with a normal ABI as controls may not fully represent a metabolically healthy comparison group. Fifth, factors such as inflammation, metabolic adaptation, collateral circulation, reduced regional blood flow, and subsequent metabolite accumulation complicate the interpretation of AV concentration differences. Additionally, the lack of direct blood flow measurements and information on tissue composition prohibited estimation of metabolic fluxes and differentiation of tissue-specific contributions. Sixth, moderate but systematic differences in fasting duration between groups may have influenced circulating metabolite levels, including those involved in energy metabolism, lipid turnover, and amino acid catabolism, thereby potentially altering AV concentration differences in either direction. Although a subgroup analysis using participants with more closely matched fasting times showed that most findings were retained or remained directionally similar with borderline significance (data not shown), the results were not fully consistent, and the small sample size precludes definitive conclusions; therefore, differences in fasting duration must be considered a potential confounder in the interpretation of our results. Finally, owing to the exploratory nature and limited sample size, our results are not adjusted for multiple comparisons. However, many of the metabolites identified are metabolically related, and the consistent, biologically plausible changes observed across these related compounds reduce the likelihood that the findings are false positives due to random variation.
Conclusion
This proof-of-concept study showed that femoral AV concentration difference analysis can reveal local metabolic changes in the ischemic lower limb that are not captured by systemic sampling. In the future, further investigation of femoral AV concentration differences may offer deeper insights into the pathophysiology of PAD. Such analyses may also improve prognostic accuracy by providing a potential proxy for assessing tissue perfusion success after conduit vessel revascularization.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251370352 – Supplemental material for Femoral arteriovenous concentration differences reveal metabolomic shifts in peripheral artery disease (PAD): A case–control study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251370352 for Femoral arteriovenous concentration differences reveal metabolomic shifts in peripheral artery disease (PAD): A case–control study by Tuljo Ööbik, Jaak Kals, Jaan Eha, Mihkel Zilmer, Kalle Kilk and Kaido Paapstel in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X251370352 – Supplemental material for Femoral arteriovenous concentration differences reveal metabolomic shifts in peripheral artery disease (PAD): A case–control study
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X251370352 for Femoral arteriovenous concentration differences reveal metabolomic shifts in peripheral artery disease (PAD): A case–control study by Tuljo Ööbik, Jaak Kals, Jaan Eha, Mihkel Zilmer, Kalle Kilk and Kaido Paapstel in Vascular Medicine
Footnotes
Acknowledgements
This study was presented at the 37th Annual Meeting of the European Society for Vascular Surgery, Belfast, United Kingdom, September 26–29, 2023. The authors thank Ester Jaigma (Department of Surgery, Institute of Clinical Medicine, University of Tartu, Tartu, Estonia) for linguistic revision of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by personal research grants (PRGs) from the Estonian Research Council (PRG no. 1437 and PRG no. 435).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
