Abstract
Background:
The aims were to determine the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) in individuals free from ASCVD (a) in the presence of carotid bifurcation plaques (CBP) < 3-mm thick and ⩾ 3 mm in comparison to a normal vessel wall and (b) the risk modulation in the presence or absence of additional common femoral bifurcations with plaques (CFBP) in a cohort study.
Methods
A total of 1000 subjects aged 58.4 ± 10.5 years, free from ASCVD, were followed up for 15.2 ± 4.9 years (mean ± SD). The primary endpoint was a composite of first time fatal or nonfatal 10-year ASCVD events.
Results
The 10-year risk of ASCVD was 6% in the absence of carotid plaques; 10% in the presence of unilateral and 23% in the presence of bilateral < 3-mm plaques (adjusted hazard ratio [HR] 1.65 [95% CI 1.11–2.47] and 2.03 [95% CI 1.32–3.00], respectively); and 29% for unilateral and 63% for bilateral 3–5 mm plaques (adjusted HR 2.40 [95% CI 1.41–4.09] and 3.78 [95% CI 1.77–8.06], respectively). In those with unilateral or bilateral < 3-mm CBP in the presence of two CFBP, the 10-year risk of ASCVD was 26% and 37% (adjusted HR 3.01 [95% CI 1.38–6.58] and 2.52 [95% CI 1.55–4.10], respectively). The 10-year risk was 2% in those without CBP or CFBP and 26% in those with two CFBP only.
Conclusions
The presence of a < 3-mm CBP may be associated with a significant ASCVD risk, especially if bilateral. This risk is better defined by the additional presence or absence of two CFBPs.
Keywords
Background
Much has been published about the size and texture features of plaques producing ⩾ 50% internal carotid artery stenosis and their relation to future strokes1,2 and atherosclerotic cardiovascular disease (ASCVD).3,4 However, relatively little is known about the association between carotid bifurcation plaques (CBP) producing < 50% stenosis and stroke, although two recent publications suggest that atherosclerotic plaques are more prevalent in the ipsilateral cerebral hemispheric side of patients with embolic stroke of undetermined source.5,6
The recently introduced Reporting and Data System for carotid plaques (Plaque-RADS) applicable to images obtained by ultrasound, computed tomography (CT) angiography, or magnetic resonance imaging (MRI) used maximum plaque thickness (MPT) for distinguishing between Plaque-RADS 2 and Plaque-RADS 3 classes. 7 The Plaque-RADS 1 class was defined as a normal vessel wall with absence of any plaque, the Plaque-RADS 2 class as a plaque with < 3 mm MPT, and the Plaque-RADS 3 class as a plaque with an MPT ⩾ 3 mm.
Other studies in the past have indicated that the presence of common femoral bifurcation plaques (CFBP) is another risk factor with a stronger predictive ability for coronary artery disease than CBP.8,9 We have already demonstrated that the presence of two or more bifurcations with plaque (carotid or common femoral) can up-classify a significant number of the population from the low and moderate risk group based on SCORE2 and SCORE2-OP algorithms into a higher risk group. 10
The primary aims of the study were to determine the risk of future ASCVD in individuals free from ASCVD in (a) the presence of CBP with an MPT < 3 mm and ⩾ 3 mm in comparison to a normal vessel wall and (b) risk modulation in the presence or absence of additional CFBP using data from the cohort of the Cyprus Epidemiological Study on Atherosclerosis (CESA).10 –12 A secondary aim was to determine the relationship between the risk of ischemic cerebrovascular events (ICE) in relation to CBP with an MPT of < 3 mm or ⩾ 3 mm.
Methods
CESA is a prospective study in the Republic of Cyprus with 1102 subjects, aged 40–89 years at baseline, of whom 1000 were initially free of ASCVD. Below we provide a brief description of the study methods. Full details have been presented previously.10 –12
Recruitment
Baseline data were collected from the residents of three randomly selected villages and their relatives who lived in any one of the main towns or villages. The aim was to recruit total populations between the ages of 40 and 89 years. The overall participation rate of those invited was 95%. All participants were Greek Cypriots. At the time of recruitment, access to the north-east part of the island that was under Turkish administration was not possible. The study was approved by the ethics committee of the Cyprus Institute of Neurology and Genetics in 2003. Participants provided written informed consent and gave permission for the data to be used in multiple future scientific studies and publications; recruitment occurred over a 5-year period (2003–2008).
Demographic, clinical, and biochemical data collection
At recruitment, demographics were recorded and a full medical history was taken with emphasis on conventional cardiovascular disease risk factors and cardiovascular symptoms including angina, myocardial infarction, ischemic neurological events, presence of abdominal aortic aneurysm, intermittent claudication, and any revascularization procedures. Subsequently, a physical examination was performed. If any lower-limb pulses were weak or absent, ankle pressures and ankle–brachial index (ABI) were measured. All past and current medications were recorded, as well as height and weight measurements; a sitting blood pressure was measured three times, with the first measurement being discarded. A fasting (8–12 hours) blood sample was obtained for serum total cholesterol, low-density lipoprotein (LDL)-cholesterol, high-density lipoprotein (HDL)-cholesterol, triglycerides, and glucose measurements.
Ultrasonographic data collection and measurements at recruitment
All scans were performed by two experienced vascular ultrasonographers using a Philips HDI 5000 duplex scanner (Seattle, WA, USA). A broad bandwidth linear array transducer 10–15 MHz was used.
All participants were examined in the supine position. Each bifurcation was examined transversely first, and then longitudinally. An arterial bifurcation (carotid or common femoral) was considered as being affected by plaque if there was a focal thickening greater than 1.2 mm. The MPT was measured in both longitudinal and transverse sections. Transverse sections also provided the local % diameter stenosis, which was calculated from measurements of the vessel diameter in addition to MPT whenever it appeared to be close to or greater than 50%. In addition, peak systolic velocity (PSV) and end diastolic velocity (EDV) were measured at the site of maximum stenosis and in the common carotid artery if a local stenosis appeared to be close to 50% or more on diameter measurements. These velocity measurements and their ratios were used to calculate the degree of stenoses ⩾ 50% in relation to the distal internal carotid artery.1,2
Information given to participants and their family doctor
After recruitment and on completion of the investigations, all participants were given an explanation of the findings and a letter for their family doctor outlining the CESA project. In addition, conventional risk factors were indicated if present, as well as the estimated risk for future ASCVD (low, moderate, or high). There was also a recommendation for further assessment with the view to initiate or update prophylactic measures in those at moderate or high risk.
Primary outcome
The primary outcome of ASCVD events was a composite of fatal or nonfatal myocardial infarction, onset of angina, coronary artery revascularization (angioplasty with or without stenting, coronary artery bypass), ICE (stroke, transient ischemic attack [TIA], or amaurosis fugax [AF]), onset of claudication, and chronic limb-threatening ischemia (CLTI).
Myocardial infarction was based on history and hospital documentation, including information on coronary artery intervention. The diagnosis of angina was based on self-reported personal history and confirmatory hospital investigations including an electrocardiogram (ECG) stress test or thallium scintigraphy. The diagnosis of ICE was based on the patient’s self-reported history, hospital records, CT- or MRI-brain scans, and reports from neurologists who saw the patient at the time of the event. Claudication was based on history of recurrent pain on walking at a fixed distance in the presence of weak or absent pulses and an ankle/brachial systolic blood pressure index less than 0.9. Cause of death was determined using death certificates, hospital records, and information from the family doctor.
Follow up
Annual contact was made initially with telephone calls to individuals and their relatives to ascertain the incidence of ASCVD events included in the primary outcome. If any health-related events were reported, the family doctor or hospital were contacted with permission. The last annual follow up was in 2023.
Changes in prophylactic therapy
In addition to the annual contact for all the participants in the cohort, 375 (37.5%) randomly selected subjects (mean follow up of 6.4 ± 2.3 years) were invited for a secondary project on plaque progression where we were able to determine changes in blood pressure and prophylactic therapy.
Study exit points
Follow up ceased with the first occurrence of any of the following: the first primary outcome measure, death from causes other than ASCVE, or lost to follow up. Individuals lost to follow up were included in the analysis for all years up to the time of loss.
Statistical methods
Initially, subjects were allocated into six classes (0–5) according to the MPT and whether carotid plaques were unilateral or bilateral (see Table 1).
Baseline clinical, biochemical, and ultrasonographic features in 1000 individuals of CESA aged 40–89 years free of ASCVD at recruitment. Subjects are also stratified by presence or absence of carotid plaques.
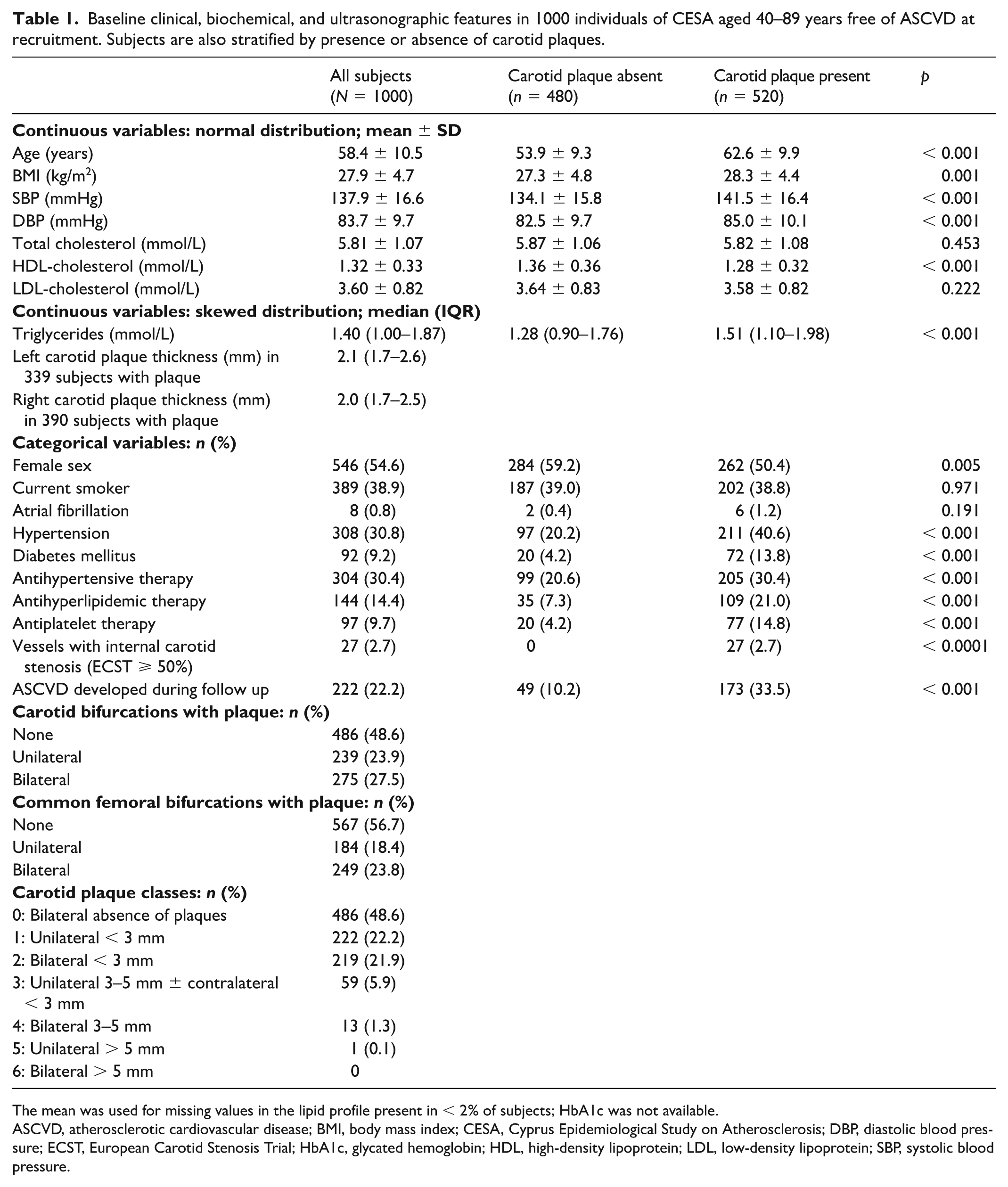
The mean was used for missing values in the lipid profile present in < 2% of subjects; HbA1c was not available.
ASCVD, atherosclerotic cardiovascular disease; BMI, body mass index; CESA, Cyprus Epidemiological Study on Atherosclerosis; DBP, diastolic blood pressure; ECST, European Carotid Stenosis Trial; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SBP, systolic blood pressure.
Lifetable analysis was used to demonstrate the cumulative ASCVD rate in the five classes of subjects (0–4) with 10+ subjects (see Table 2). The 10-year ASCVD rates for the different classes were obtained from the lifetable analysis. Hazard ratios (HR) of the four carotid plaque classes (1–4), in reference to no plaques (class 0), with respect to the primary endpoint during follow up were calculated using a Cox model, unadjusted and adjusted for conventional risk factors as well as antihypertensive, antihyperlipidemic, and antiplatelet therapies.
The 10-year risk of ASCVE and hazard ratios in 999 individuals across five classes of carotid plaque distribution.
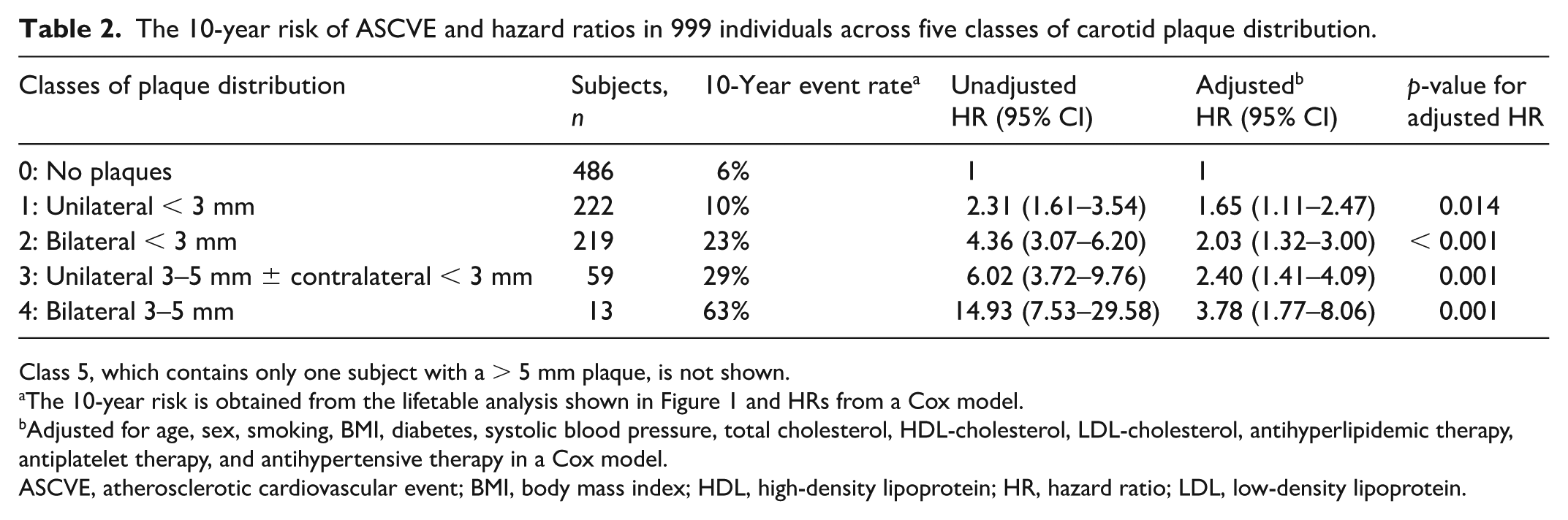
Class 5, which contains only one subject with a > 5 mm plaque, is not shown.
The 10-year risk is obtained from the lifetable analysis shown in Figure 1 and HRs from a Cox model.
Adjusted for age, sex, smoking, BMI, diabetes, systolic blood pressure, total cholesterol, HDL-cholesterol, LDL-cholesterol, antihyperlipidemic therapy, antiplatelet therapy, and antihypertensive therapy in a Cox model.
ASCVE, atherosclerotic cardiovascular event; BMI, body mass index; HDL, high-density lipoprotein; HR, hazard ratio; LDL, low-density lipoprotein.
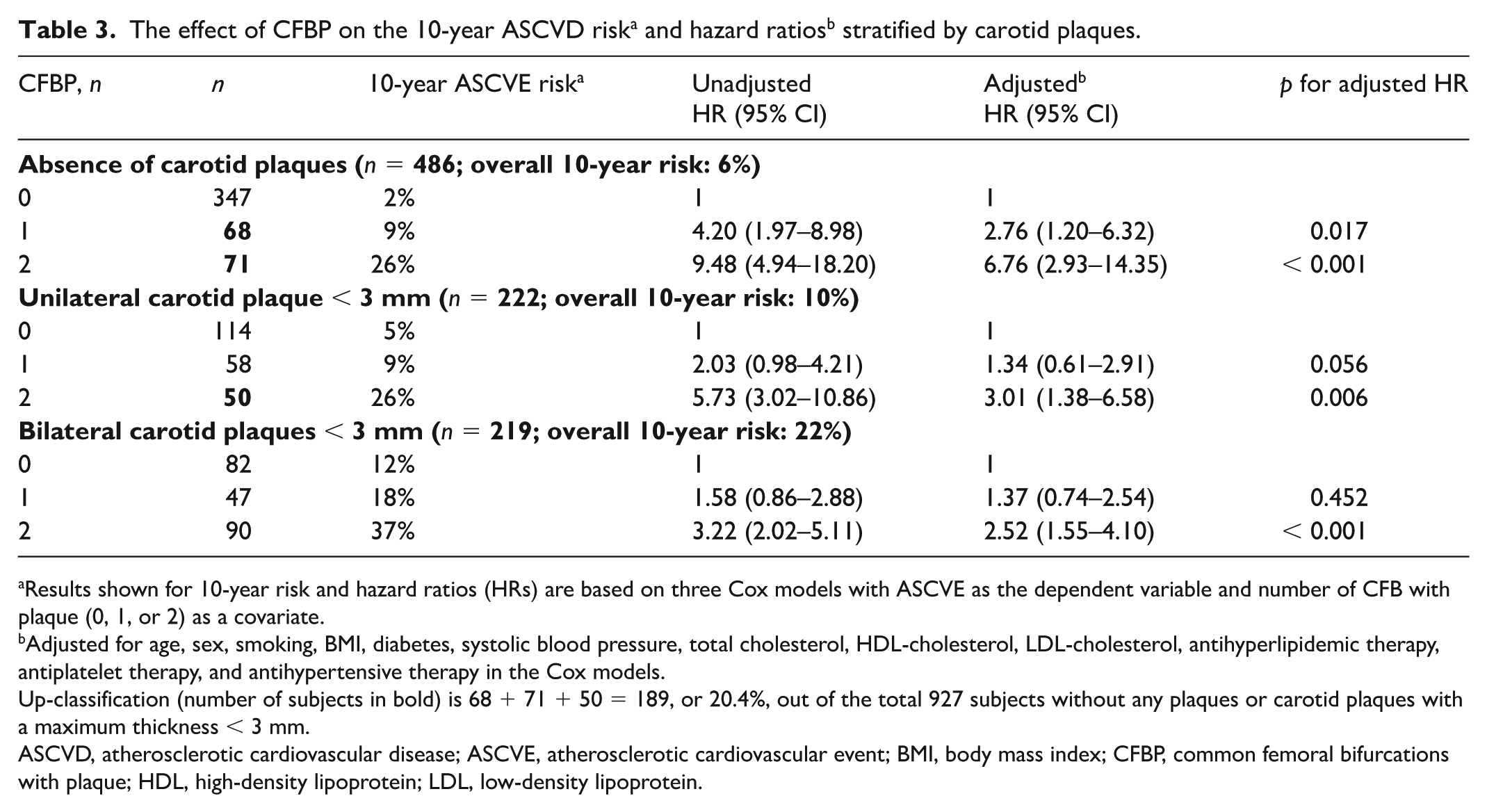
The effect of the number of CFBP (0–2) on the 10-year ASCVE risk was examined using Cox models for the subgroups with (a) the absence of carotid plaques, (b) the presence of < 3-mm unilateral carotid plaques, and (c) the presence of < 3-mm bilateral carotid plaques (see Table 3).
Results shown for 10-year risk and hazard ratios (HRs) are based on three Cox models with ASCVE as the dependent variable and number of CFB with plaque (0, 1, or 2) as a covariate.
Adjusted for age, sex, smoking, BMI, diabetes, systolic blood pressure, total cholesterol, HDL-cholesterol, LDL-cholesterol, antihyperlipidemic therapy, antiplatelet therapy, and antihypertensive therapy in the Cox models.
Up-classification (number of subjects in bold) is 68 + 71 + 50 = 189, or 20.4%, out of the total 927 subjects without any plaques or carotid plaques with a maximum thickness < 3 mm.
ASCVD, atherosclerotic cardiovascular disease; ASCVE, atherosclerotic cardiovascular event; BMI, body mass index; CFBP, common femoral bifurcations with plaque; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
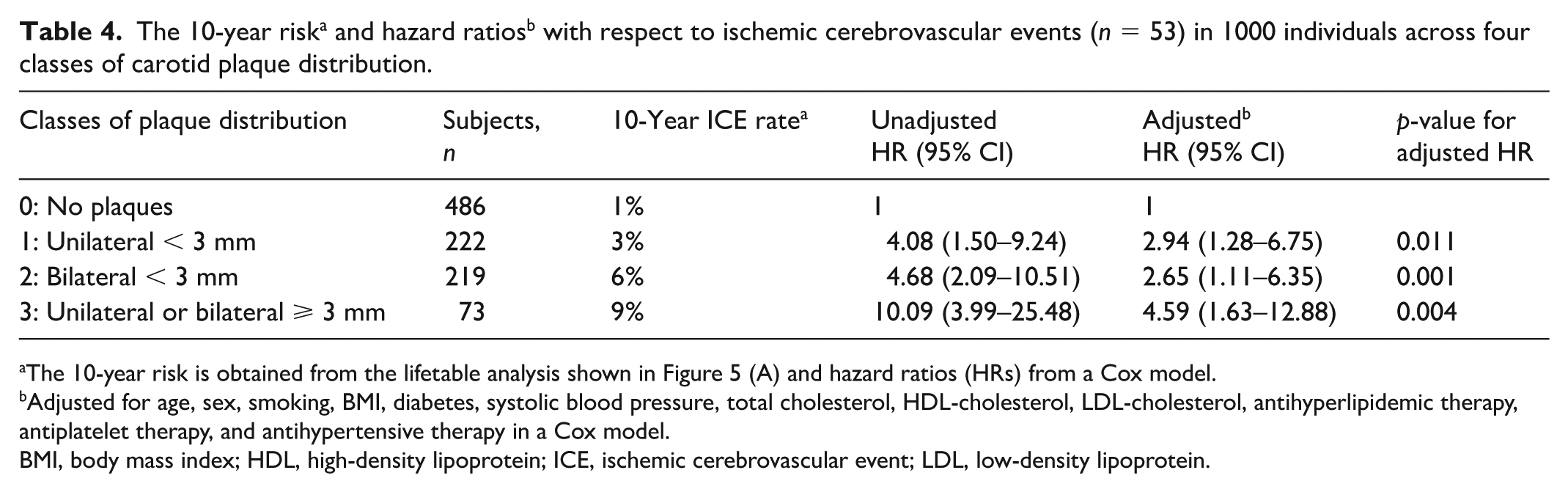
Subsequently, the analysis focused on cerebrovascular events. Lifetable analysis was used to determine the 10-year ICE rate in the absence of carotid plaques, in individuals with unilateral < 3-mm plaques, those with bilateral < 3-mm plaques, and those with unilateral or bilateral ⩾ 3-mm plaques. Unadjusted and adjusted HRs were obtained using a Cox model (see Table 4).
The 10-year risk is obtained from the lifetable analysis shown in Figure 5 (A) and hazard ratios (HRs) from a Cox model.
Adjusted for age, sex, smoking, BMI, diabetes, systolic blood pressure, total cholesterol, HDL-cholesterol, LDL-cholesterol, antihyperlipidemic therapy, antiplatelet therapy, and antihypertensive therapy in a Cox model.
BMI, body mass index; HDL, high-density lipoprotein; ICE, ischemic cerebrovascular event; LDL, low-density lipoprotein.
Finally, lifetable analysis was used in the subgroup of individuals with carotid plaques ⩾ 3 mm to determine the 10-year ICE rate in those who had a diameter stenosis < 50% or ⩾ 50%.
All statistical analyses were performed using IBM SPSS statistics, version 19.0 (IBM Corp.). A p-value less than 0.05 (two-sided) was considered statistically significant.
Results
A total of 1000 individuals without ASCVD aged 40–89 years (mean age: 58.4 ± 10.5 years; 55% women) at recruitment were included in the analysis. Eight (0.8%) participants were lost to follow up at different times but were included in the analysis as right censored observations. Follow up was for a mean of 15.2 ± 4.9 years (range 1–20). The baseline clinical, biochemical, and ultrasonographic features at recruitment are shown in Table 1.
Primary and other outcomes
First ASCVD events (98 fatal and 121 nonfatal) occurred in 219 subjects: myocardial infarction (n = 99), onset angina (n = 21), coronary angioplasty/stenting (n = 20), coronary artery bypass (n = 20), ICE (n = 53) (stroke in 47, TIA in five, and AF in one), onset of claudication (n = 4), and CLTI (n = 2). The mean length of time between recruitment and event was 10.2 ± 5.3 years. There were 122 additional deaths not related to ASCVD (six from hemorrhagic stroke, 39 from malignancy, 62 from other non-ASCVD events, and 15 from unknown causes).
Changes in prophylactic therapy
At the follow-up visit of 375 (37.5%) subjects aged 58.2 ± 8.9 years (58.4% women), it was determined that the prevalence of lipid-lowering therapy increased from 13.1% to 60.0% (p < 0.0001), antiplatelet therapy from 11.2% to 25.1% (p < 0.0001), and antihypertensive therapy from 34.2% to 60.0% (p < 0.0001). Systolic blood pressure also changed from 138.8 ± 16.4 mmHg to 135.7 ± 17.7 mmHg (p = 0.002) and diastolic blood pressure from 84.9 ± 9.4 mmHg to 79.5 ± 10.8 mmHg (p < 0.001).
Association between carotid bifurcation plaque (CBP) classes and atherosclerotic cardiovascular disease (ASCVD)
All CBP except one had an MPT of 5 mm or less (Table 1). Thus, six classes (0–5) of subjects were identified based on the cut-off points of 3-mm and 5-mm MPT and whether carotid plaques were unilateral or bilateral. Thus, only one subject was in class 5. The cumulative ASCVD rates in the five classes of carotid plaque with 10+ subjects (0–4), derived from the lifetable analysis, are shown in Figure 1 and listed together with the HRs with respect to the primary endpoint in Table 2. Compared with a normal vessel, there were significant increases in the HR for all 1–4 classes, and these remained significant after adjusting for conventional risk factors as well as antihypertensive, antihyperlipidemic, and antiplatelet therapies in a Cox model (Table 2).
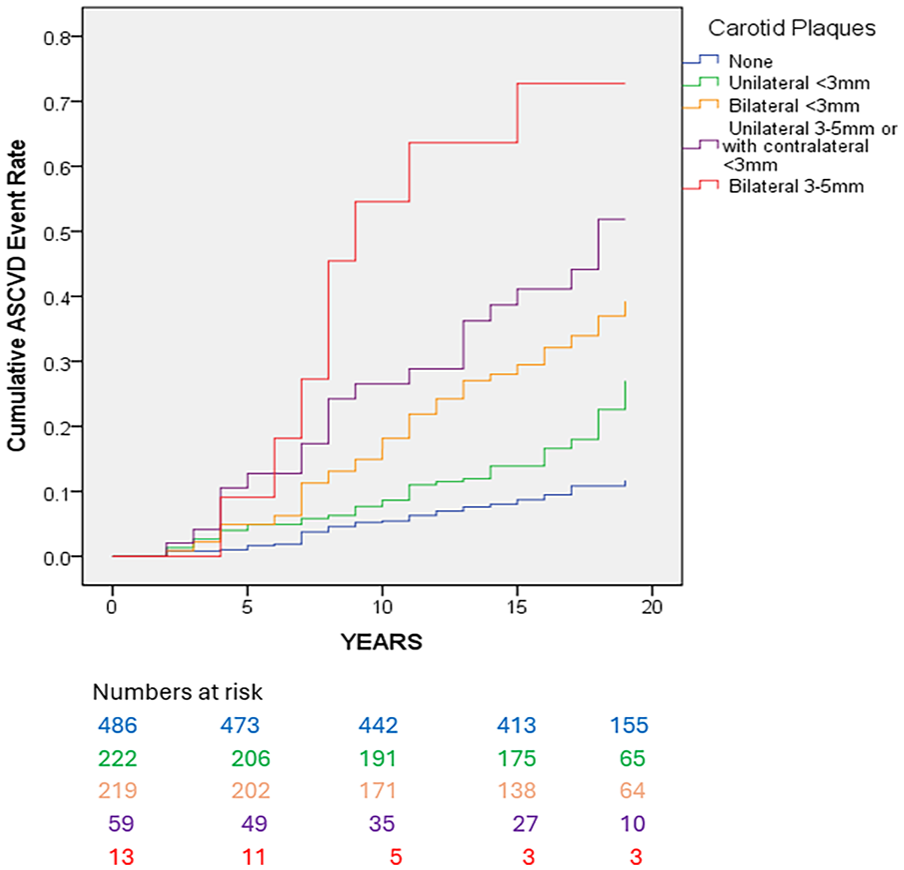
Cumulative atherosclerotic cardiovascular disease (ASCVD) event rate in the five classes of carotid plaque distribution based on maximum plaque thickness and number of sides involved. Overall comparison (Wilcoxon–Gehan statistic)p < 0.001.
Effect of presence or absence of common femoral bifurcations with plaques (CFBP)
The effect of the number of CFBP (0, 1, or 2) on the 10-year ASCVE rates in the subgroups of (a) absence of CBP, (b) presence of unilateral CBP < 3 mm plaques, and (c) presence of bilateral CBP < 3 mm plaques is shown in Figure 2 and Table 3.
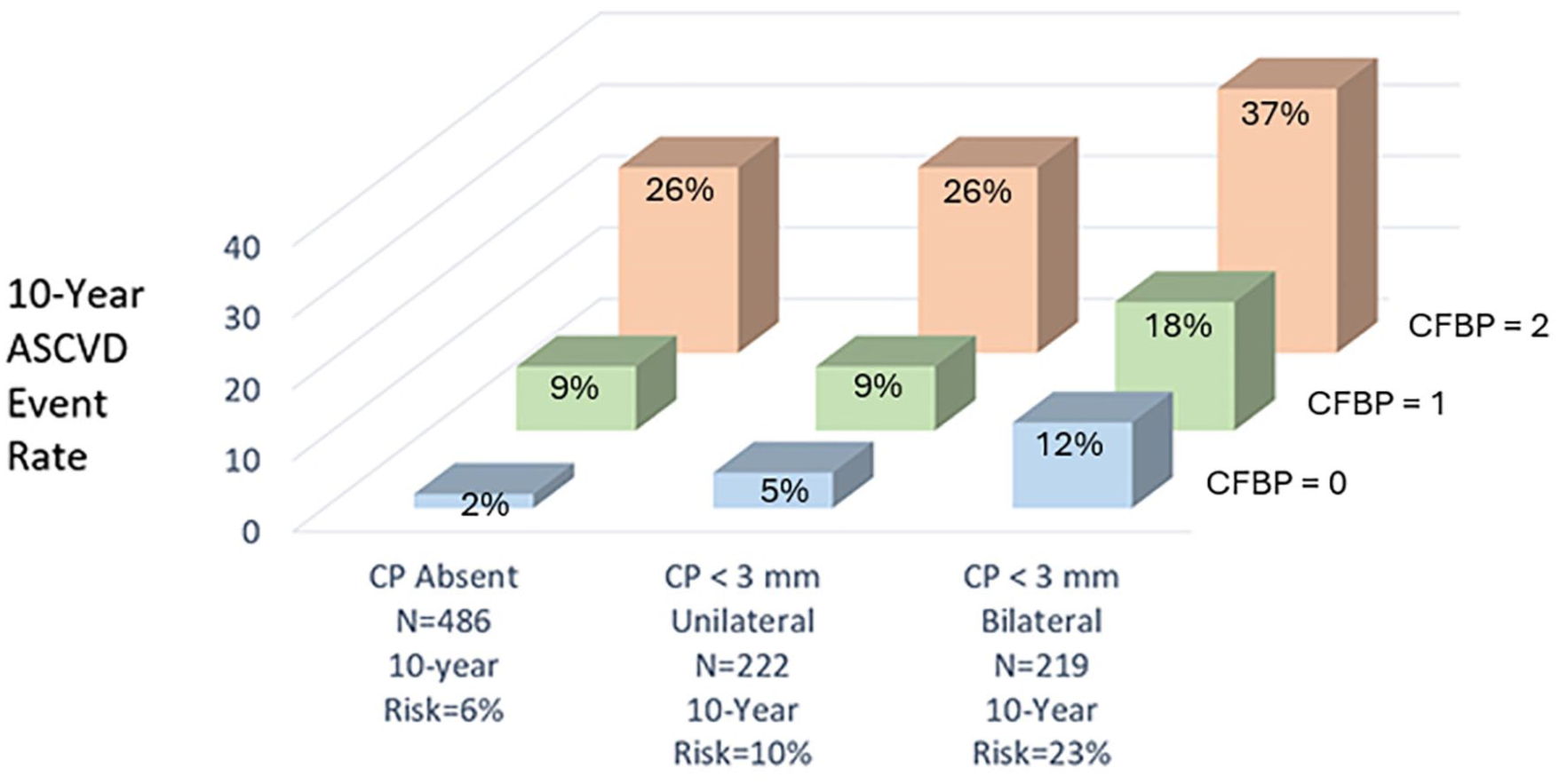
The effect of the number of common femoral bifurcations with plaque (CFBP) on the 10-year atherosclerotic cardiovascular disease (ASCVD) event rate in subjects with carotid plaques (CP) having a maximum plaque thickness < 3 mm.
Two key messages are provided by the analysis in Table 3. In the absence of CBP (n = 486) or in the presence of unilateral < 3-mm plaques (n = 222), the presence of two CFBPs assigns subjects to a high 10-year risk of 26%. This occurred in 71 (14.6%) subjects out of 486 who did not have any CBP and in 50 (22.5%) out of 222 subjects who had unilateral < 3-mm carotid plaques (Table 3). Subjects with bilateral < 3-mm plaques were assigned into a very high 10-year risk of 37% by the presence of two CFBPs. The presence of only one CFBP had little effect in terms of reclassifying subjects to a higher-risk class.
Association between maximum plaque thickness (MPT) and degree of local diameter stenosis
None of the carotid plaques with an MPT < 3 mm (n = 701) produced a stenosis ⩾ 50%. Of the plaques with an MPT 3–5 mm (n = 86), 25 (29%) were associated with a local internal carotid diameter stenosis ⩾ 50% in 23 participants (bilateral in two participants). The remaining one plaque had an MPT of 7.0 mm and produced a 95% stenosis in the internal carotid artery. In the 25 internal carotid arteries with ⩾ 50% diameter stenosis, the 3–5-mm plaque was found to either extend distally beyond the carotid bulb (Figure 3) or involve most of the arterial wall circumference (Figure 4).
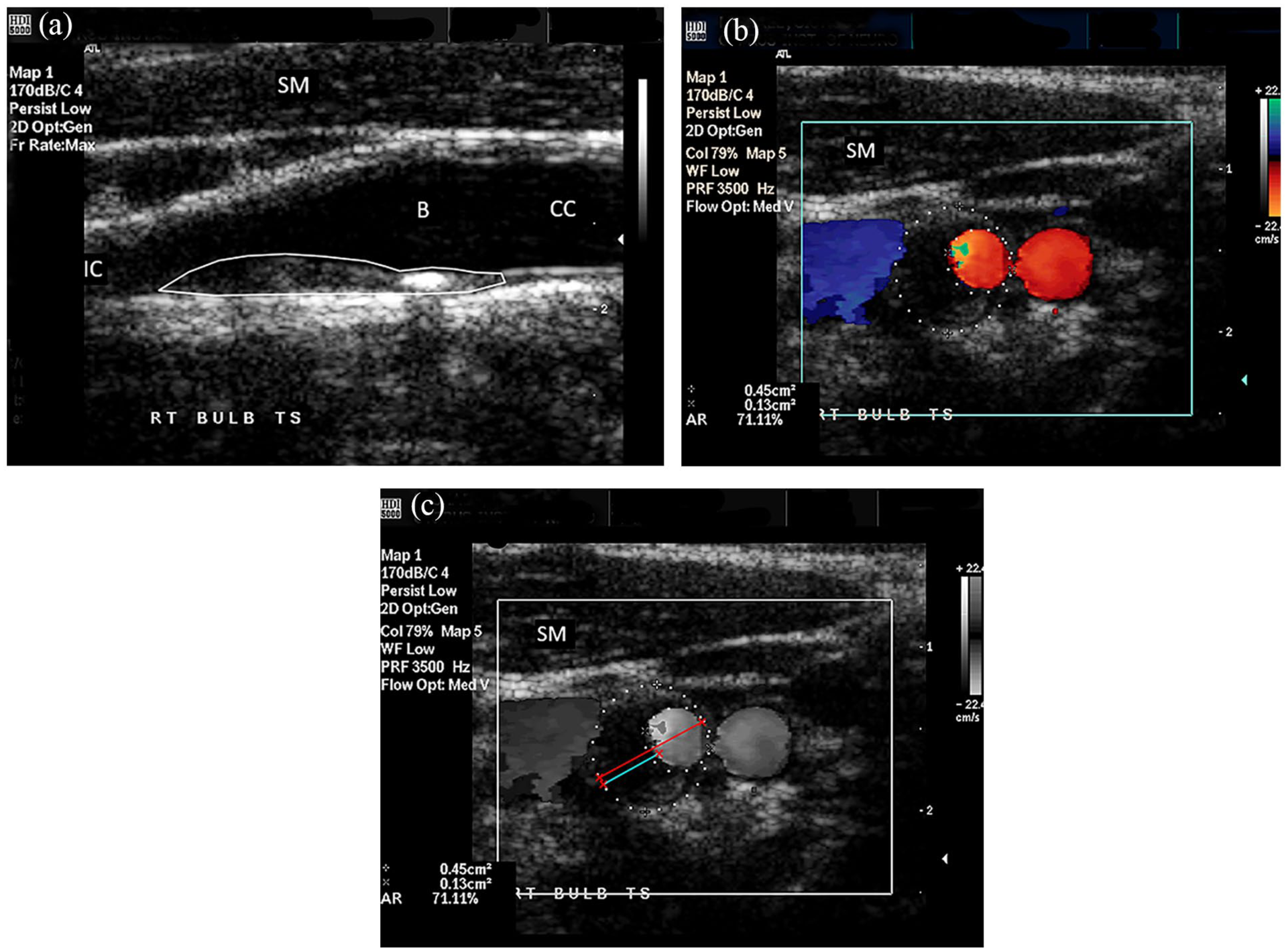
Example of a plaque with maximum thickness of 4.0 mm extending cephalad into the internal carotid artery and producing a 55% diameter stenosis. (
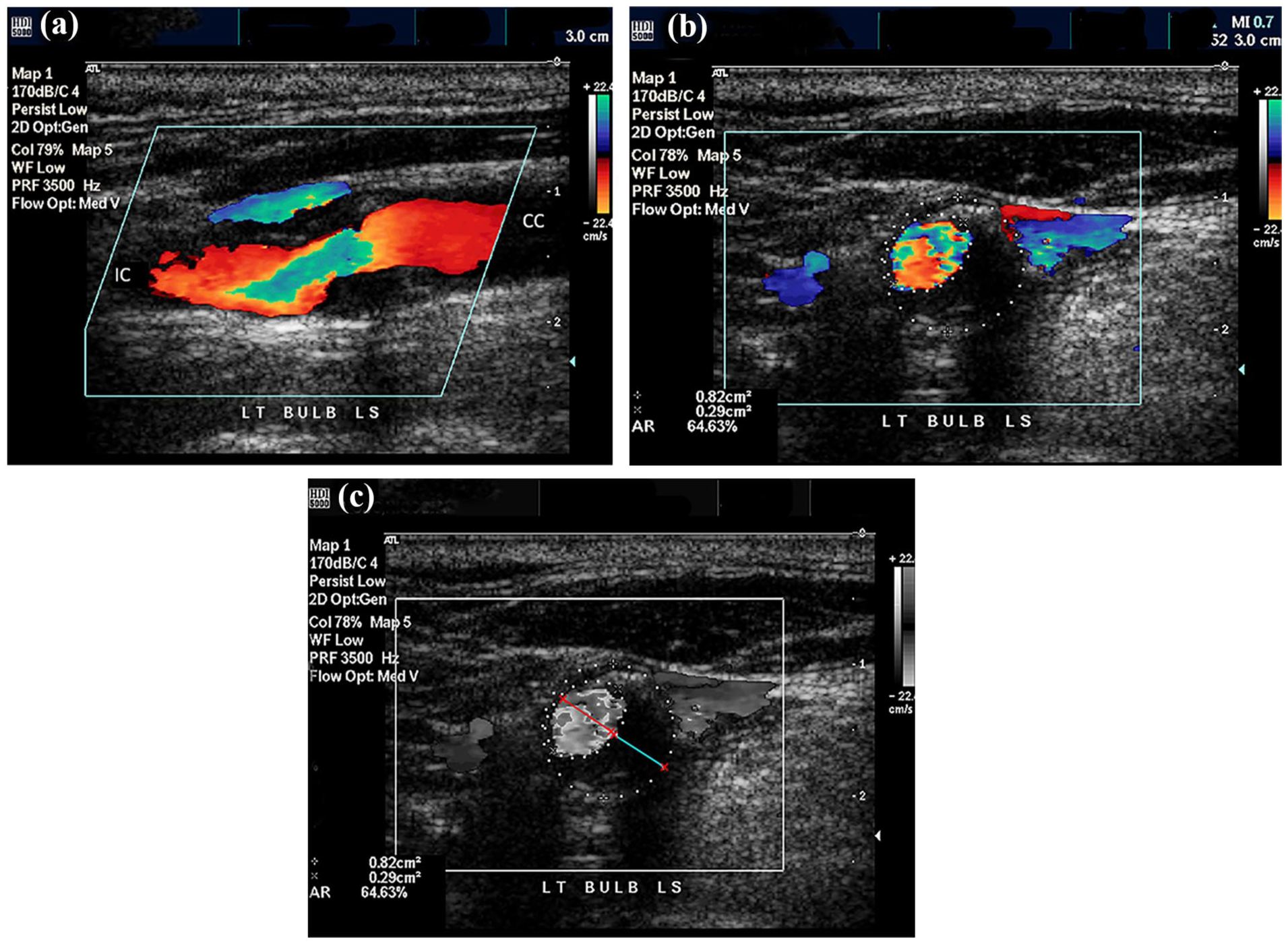
Example of a hypoechoic plaque with maximum thickness of 4.6 mm producing a 51% stenosis in the carotid bulb. (
Association between CBP classes and ischemic cerebrovascular events (ICE)
Fifty-three ICE events (47 ischemic strokes, five TIAs, and one AF) were observed during follow up. The cumulative 10-year ICE rate and HR in relation to plaque thickness and sides involved is shown in Figure 5 and Table 4. There was a progressive increase in the 10-year ICE rate from 1% in the absence of CBP up to 9% in the presence of unilateral or bilateral ⩾ 3-mm plaque (Table 4). The 10-year ICE rate was 3% in the 50 individuals with < 50% diameter stenosis and 22% in the 23 individuals with ⩾ 50% diameter stenosis, with an HR of 8.98 (95% CI 1.56–51.54) after adjustment for conventional risk factors (Figure 5).
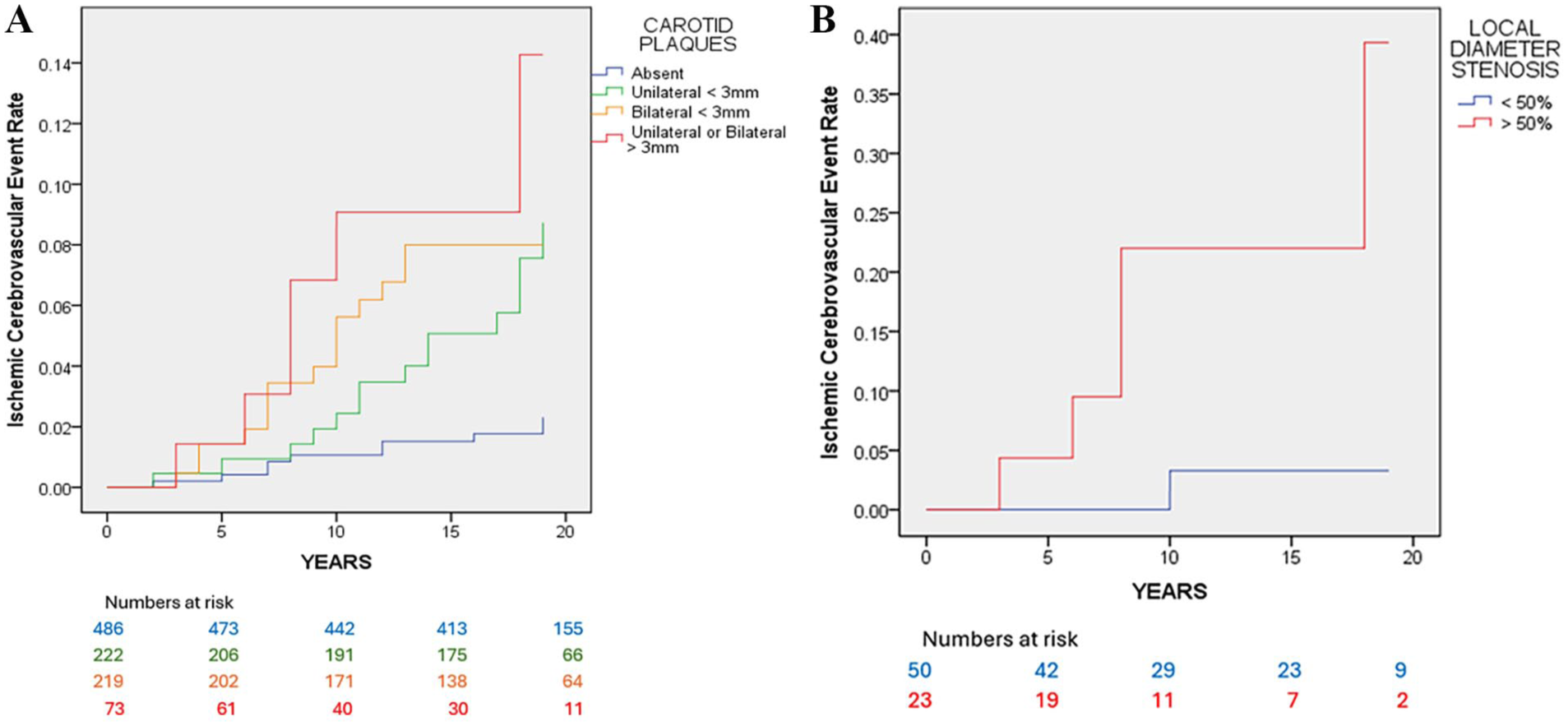
Cumulative ischemic cerebrovascular event (ICE) rate in relation to plaque thickness, sides involved, and stenosis. (
Discussion
The results of this study indicate that compared with the absence of carotid plaques the risk of ASCVD increases progressively with the presence of unilateral < 3-mm plaques, bilateral < 3-mm plaques, and plaques ⩾ 3 mm. The risk was maximum when ⩾ 3-mm plaques were bilateral (Table 2). The HRs also increased for the whole duration of follow up and remained statistically significant after adjusting for conventional risk factors as well as antihypertensive, lipid-lowering, and antiplatelet therapies. This provided further evidence supporting previous studies which demonstrated that the presence of plaques, size, and bilateral involvement are independent predictors of risk.3,4,8 –14
Carotid and common femoral bifurcation plaques are the preferred sites for arterial screening using ultrasound in epidemiological studies because they are superficial and allow the use of high-frequency probes which provide a high resolution; also, because they occur earlier and more often than plaques in long arteries. For example, compared with the carotid bifurcation, plaques rarely occur in the common carotid artery.
Several publications in the last 10 years have demonstrated that carotid plaque thickness, area, and volume are independent predictors of ASCVD, including stroke and significant coronary artery disease.9 –15 We and others demonstrated that CFBP are stronger predictors of risk than CBP.8 –11,14 In the PESA study, the addition of femoral plaque could improve the prediction of ASCVD risk, especially in the low-risk populations. 16
Risk estimation tools using conventional risk factors classify individuals into low, moderate, or high risk, but guidelines acknowledge that all such tools have inherent limitations and that it is reasonable to use additional risk-enhancing factors in cases of borderline or moderate risk: coronary artery calcium (CAC) score 17 and presence of either CAC or carotid plaque. 18 In the Bioimage Study, the predictive ability of carotid plaque burden was shown to be equal to that of coronary calcium scoring. 19 Guidelines recommend risk factor modification for subjects in the moderate-risk group and aggressive risk factor modification for subjects in the high-risk group. 18 The ability to reclassify subjects into a higher risk group by just the presence of two common femoral bifurcations with plaque irrespective of size indicating ‘silent polyvascular’ atherosclerosis and making them candidates for a more aggressive risk factor modification is a new message.
Polyvascular disease, which is defined as the presence of symptomatic ASCVD in two or more territories, 20 is now recognized as a risk factor for increased rates of cardiovascular death, myocardial infarction, and stroke. In addition, there is a stepwise increased risk with each additional vascular territory involved. 21 It is not surprising that there is an increased risk also in subjects with silent atherosclerotic plaques in two common femoral bifurcations, indicating the presence of silent polyvascular atherosclerosis.
The increased 10-year rate of ICE from 1% in the absence of carotid plaques to 3% in individuals with unilateral < 3-mm plaques, 6% in those with bilateral < 3-mm plaques, and 9% in those with unilateral or bilateral 3–5-mm plaques with associated increases in the adjusted HRs validates the 3-mm cut-off point recently proposed in the novel classification system ‘Carotid Plaque-RADS’. 7 The observed ‘dose–response’ relationship strengthens the likelihood of causality for the observed associations.
The relatively high 10-year risk of ICE of 22% in individuals with 3–5-mm plaques producing ⩾ 50% local stenosis compared with the 10-year risk of only 3% in individuals with stenosis < 50% despite the same plaque size-range emphasizes the importance of measuring not only the MPT but also the percentage diameter stenosis.
A limitation of the study is that power calculations were based on the published mortality rates and annual incidence of cardiovascular disease in the year 2000 as well as the primary endpoint, which was a composite of ASCVD. As a result, the study was not powered to investigate ICE alone. Thus, the results on the relationship between ICE and plaques with an MPT of < 3 mm and 3–5 mm need to be confirmed by larger studies.
Another limitation of the study is that the subjects recruited were confined to the Greek Orthodox population of Cyprus, an island in the Eastern Mediterranean and one of the smallest countries in the European Union. At the time of recruitment, access to the Turkish Cypriot population in the north-eastern part of the island, which was under Turkish administration, was not possible. Although the population studied was uniform in terms of race, we cannot claim generalization for populations in other countries.
At recruitment, primary prevention with lipid-lowering therapy and antihypertensive therapy were suboptimal by today’s standards. They were similar to those found in the Aragon Workers’ Health Study, where although 78% of subjects free from ASCVD had subclinical atherosclerotic plaques, only 31% had any form of prophylaxis and only 14% were on statins – more or less reflecting primary prevention practice in the first decade of the 21st century in some countries in Europe. 22 Although there was a marked improvement in prophylactic medications during follow up, it is unlikely that treatment targets were close to the ones currently recommended and this may also limit the generalizability of our findings.
Although ultrasound is operator-dependent, plaque thickness in superficial vessels is a highly reproducible measurement.13,22 MPT is recommended by the American Society of Echocardiography as the measurement of choice for the assessment of carotid plaque in the evaluation of cardiovascular risk 22 and presence of carotid plaque is recommended by the European Society of Cardiology for reclassification of asymptomatic individuals in low or moderate risk groups. 18
Despite the above limitations, ultrasound which is free from ionizing radiation remains a relatively inexpensive and quick diagnostic imaging tool, readily available in all vascular diagnostic departments and portable. Given all these attributes, it makes it an ideal method for improving on risk prediction by determining the presence of additional CFBP irrespective of whether carotid plaques < 3 mm or ⩾ 3 mm producing a < 50% diameter stenosis are detected on ultrasound, MRI, or CT.
Conclusions
The presence of small atherosclerotic carotid plaques with a maximum thickness of < 3 mm (Plaque-RADS 2) in asymptomatic individuals should not be ignored and should not be reported as ‘small plaques of no hemodynamic significance and therefore of no clinical importance,’ as often done after duplex scanning in the presence of normal blood velocities. Such plaques are markers of an increased ASCVD risk. In addition, the presence or absence of two common femoral bifurcations with plaque indicates a more extensive systemic subclinical atherosclerosis and improves risk prediction. This in turn can provide better guidance on lifestyle modifications and prophylactic risk factor therapy as recommended in recent guidelines.
Footnotes
Acknowledgements
We are grateful to the late Dr Charis Fessas (The Cyprus Institute of Cardiology, Nicosia, Cyprus) for his help in setting up the study, recruitment, and follow-up.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Research reported in this publication was supported in part by a grant (41/50ΠE-2002) from the Cyprus Research Promotion Foundation, Nicosia, Cyprus, a grant from the Ministry of Health, Government of Cyprus, Nicosia, Cyprus, a grant from the Cyprus Cardiovascular Disease Educational and Research Trust, UK, and a grant from the AG Leventis Foundation, London, UK. Funders had no role in the study design, interpretation, and manuscript preparation.
Disclaimer
Richard M Martin is a National Institute for Health and Care Research (NIHR) Senior Investigator. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Data availability
De-identified data supporting the findings of this study are available through the corresponding author or the Trustees of the Cyprus Cardiovascular Disease Educational and Research Trust (CCDERT), a registered charity at 2, Kyriacou Matsi Street, 2368 Nicosia, Cyprus, upon request.