Abstract
We aimed to evaluate whether carotid intima-media thickness (CIMT) or the presence of plaque can confer additional predictive value of future cardiovascular (CV) ischemic events in patients with pre-existing atherosclerotic vascular disease. We identified 2317 patients enrolled in the REduction of Atherothrombosis for Continued Health (REACH) registry who had atherosclerotic vascular disease and baseline CIMT measurements. The entire range of CIMT was divided into quartiles and the fourth quartile (≥ 1.5 mm) was defined as carotid plaque. Mean ± standard deviation baseline CIMT was 1.31 ± 0.65 mm. Associated CV ischemic events and vascular-related hospitalizations were evaluated over a 2-year follow-up. There was a positive increase in adjusted hazard ratios (HRs) for all-cause mortality (p = 0.04 for trend) and the quadruple endpoint (CV death, myocardial infarction (MI), stroke, hospitalization for CV events) with increasing quartiles of CIMT (p = 0.0008 for trend), which was mainly driven by the fourth quartile (carotid plaque). HRs for all-cause mortality, CV death, CV death/MI/stroke and the quadruple endpoint comparing the highest (carotid plaque) with the lowest CIMT quartile were 2.09 (95% CI, 1.07–4.10; p = 0.03); 2.49 (1.10–5.67; p = 0.03); 1.71 (1.10–2.67; p = 0.02); and 1.73 (1.31–2.27; p = 0.0001). In conclusion, our analyses suggest that the presence of carotid plaque, rather than the thickness of intima-media, appears to be associated with increased risk of CV morbidity and mortality, but confirmation of these findings in other population and prospective studies is required.
Introduction
Noninvasive high-resolution carotid ultrasound imaging has increasingly been used to measure carotid intima-media thickness (CIMT) and carotid plaque in observational and intervention studies. Previous population studies have shown associations between increased CIMT and cardiovascular (CV) risk factors,1,2 the prevalence of CV disease, 3 the presence of atherosclerosis in other arterial beds, 4 and the prevalence of abdominal aortic aneurysm. 5 Several studies have also shown an association between increased CIMT and incident myocardial infarction (MI) and stroke that is independent of classic atherosclerosis risk factors in patients without pre-existing CV disease.6,7 Measurement of progression or regression of CIMT has also been used effectively as a surrogate endpoint for determining the treatment effect of cholesterol-lowering drugs.8,9 The presence of carotid plaque has also been reported to increase the risk of a cardiovascular event in longitudinal and population studies.10–13 Few studies have evaluated the association between increased CIMT and MI or stroke among patients with clinically diagnosed atherosclerotic disease.14,15 However, most previous studies of CIMT and carotid plaque have been performed in the general population focusing on primary prevention. Thus, there is little information about the predictive value of CIMT and the presence of carotid plaque in assessing the risk for subsequent ischemic events in patients with overt atherosclerotic vascular disease.
In this prospective study, we sought to investigate the association between baseline CIMT and the presence of carotid plaque and incidence of ischemic vascular events in a large international multicenter cohort of patients with established atherosclerotic vascular disease. We hypothesize that increased CIMT and the presence of carotid plaque in patients with overt atherosclerotic disease is associated with increased vascular ischemic events.
Methods
Study population
Study patients were participants in the prospective, multicenter REduction of Atherothrombosis for Continued Health (REACH) registry. The protocol was submitted to the institutional review boards in each country according to local requirements, and signed informed consent was required for all patients. The study design, 16 baseline descriptions, 17 and 1-year CV event rates have been published. 18 Briefly, consecutive outpatients aged ≥ 45 years with coronary artery disease (CAD), cerebrovascular disease (CVD), or peripheral artery disease (PAD), or patients with three or more atherothrombotic risk factors were enrolled by their physicians between December 2003 and June 2004.
For the purpose of this analysis, 2317 patients with established atherosclerotic vascular disease (defined as documented CAD, CVD or PAD) and available CIMT measurement at baseline were identified and extracted for this subgroup analysis. Documented CAD consisted of one or more of the following criteria: history of stable or unstable angina with documented CAD, history of previous myocardial infarction, percutaneous coronary intervention or coronary artery bypass graft surgery. Documented CVD consisted of a hospital or neurologist report with the diagnosis of transient ischemic attack or ischemic stroke. Documented PAD consisted of one or both criteria: current intermittent claudication with an ankle–brachial index < 0.9 or a history of intermittent claudication together with a previous and related intervention, such as angioplasty, stenting, atherectomy, peripheral arterial bypass graft, or other vascular intervention, including amputation. Data on risk factors, physical examinations, and medications were collected centrally via standardized case report forms. The risk factors consisted of those that were documented in the medical record or for which patients were receiving treatment at the time of enrollment: diabetes, hypertension, hypercholesterolemia, current smoking and obesity.
There were 44 countries participating in REACH and of those, 33 countries provided CIMT measurement. A total of 572 centers/medical offices provided at least one CIMT measurement. CIMT measurements were performed by each site using B-mode ultrasound to calculate the maximum value of intima-media thickness. The measurements were performed in a 10-mm long segment just proximal to the carotid bulb in the common carotid artery. The duplex studies were not standardized across sites. As there is no definition for normal CIMT or pathologic CIMT in patients with established atherosclerotic vascular disease, we analyzed the entire range of the CIMT measurement and divided it into quartiles. The first quartile of CIMT ranged from 0.2 to 0.89 mm and included 501 patients; the second quartile from 0.90 to 1.09 mm (491 patients); the third quartile from 1.10 to 1.49 mm (673 patients); and the fourth quartile from 1.50 to 4.00 mm (652 patients). Mean ± standard deviation (SD) CIMT was 1.31 ± 0.65 mm. The fourth quartile (CIMT ≥ 1.5 mm) is defined as carotid plaque as suggested by the Mannheim CIMT consensus report. 19
Incident CV ischemic events (MI, stroke), mortality (all-cause and CV-related) and vascular-related hospitalizations were determined prospectively at a mean of 24 ± 3 months after enrollment and were provided from participating physicians. Events were not adjudicated; however, reports of ischemic stroke and transient ischemic attack had to be sourced from a neurologist or hospital to ensure a reliable diagnosis. CV death included fatal stroke, fatal MI, and other CV death (other death of cardiac origin; pulmonary embolism; any sudden death unless proven otherwise by autopsy; death following a vascular operation, vascular procedure, or amputation; death attributed to heart failure; death following a visceral or limb infarction; and any other death that could not be definitely attributed to a nonvascular cause or hemorrhage). Any MI or stroke followed by death, whatever the cause, in the subsequent 28 days was considered as a fatal MI or fatal stroke.
The individual endpoints were all-cause mortality, CV death, nonfatal MI, and nonfatal stroke. The composite triple endpoint was defined as CV death, MI, or stroke; and the quadruple endpoint consisted of CV death, MI, stroke, or hospitalization for CV events. Subgroup analyses were performed for patients with CAD alone, CVD alone, and PAD alone.
Statistical methods
Continuous variables are expressed as mean ± SD. Categorical variables are expressed as frequencies and percentages. Event rates are shown as annualized event rates with adjustment for age and sex. Hazard ratios (HRs) were calculated from the 2-year database. CIMT was analyzed as a categorical variable using quartiles. Multivariable-adjusted proportional hazards models were carried out for each outcome to examine the influence of CIMT and the presence of plaque on risk of events. Multivariable models were adjusted for age, sex, diabetes, hypertension, smoking, hypercholesterolemia, and geographic region. For some CIMT quartile categories, HRs could not be calculated because no outcome events had occurred within that subgroup. Statistical significance was considered as a two-tailed probability of < 0.05. Statistical analysis was performed using SAS software version 9.1 (SAS Institute Inc., Cary, NC, USA).
Results
Among 2317 patients with CIMT measurements, 1622 (70.0%) were men; mean ± SD age was 66.3 ± 9.5 years. The average age of the entire cohort is similar at 68.6 ± 10.1 (Table 1). A high proportion of patients in the CIMT subgroup as well as in the entire study cohort had hypertension, hypercholesterolemia, diabetes, were overweight or obese, or were former/current smokers (Table 1). Overall, the CIMT subgroup appears to be younger, has a greater percentage of males, was less obese and had less diabetes, and had less lipid abnormality than the entire cohort. Detailed baseline demographics, concomitant atherosclerotic risk factors, geographic distribution, and CIMT among all patients with atherosclerotic disease and among CAD alone, CVD alone, and PAD alone are given in Table 1.
Baseline demographics of the entire study cohort and the CIMT subgroup
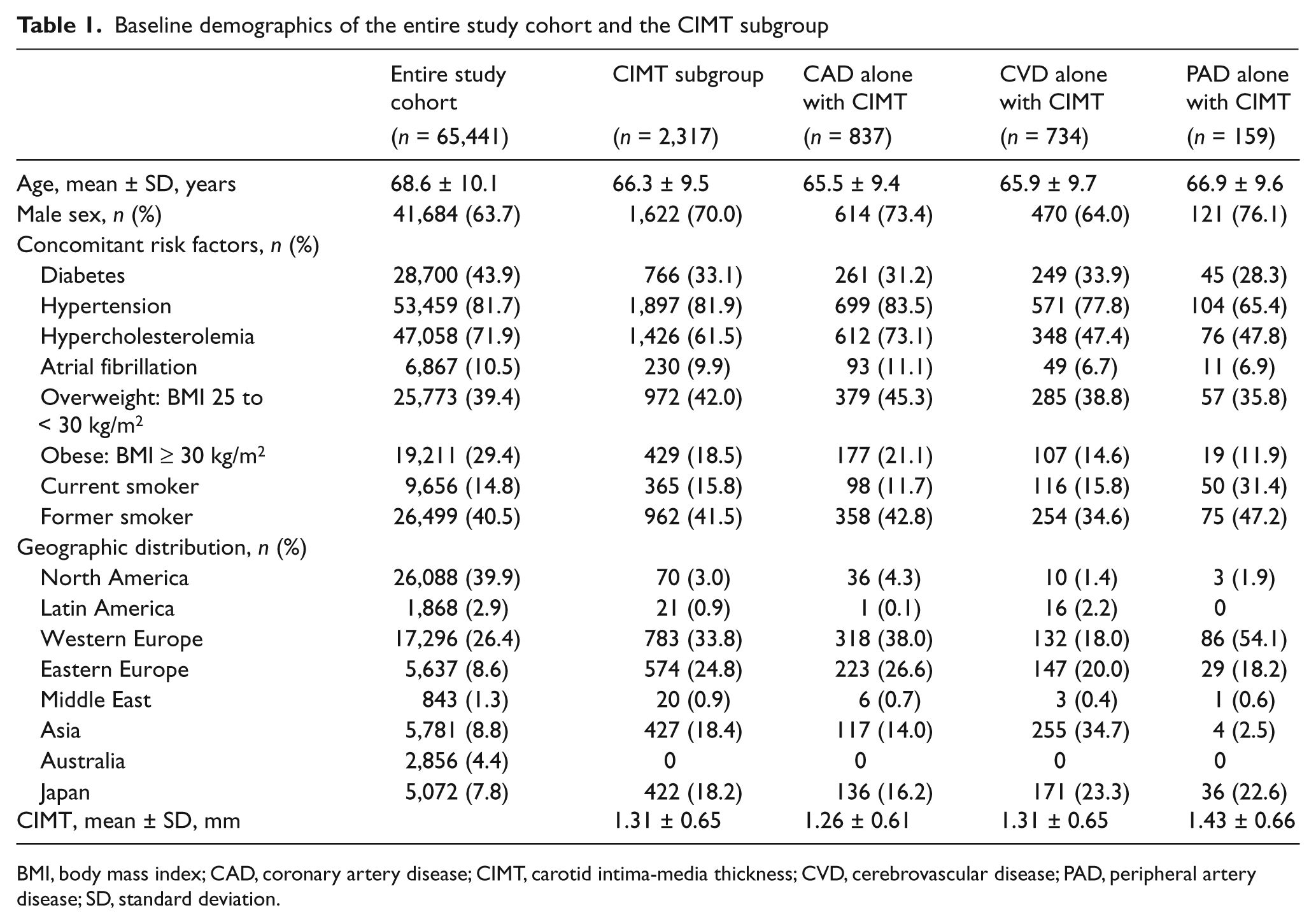
BMI, body mass index; CAD, coronary artery disease; CIMT, carotid intima-media thickness; CVD, cerebrovascular disease; PAD, peripheral artery disease; SD, standard deviation.
Table 2 shows 2-year event rates of the entire population, the CIMT subgroup and CIMT subgroup separated by CAD, CVD, and PAD alone. Overall mortality is significantly higher in the entire population than the CIMT subgroup. Nonfatal stroke and the quadruple endpoint occurred more frequently in the CIMT subgroup than the entire population (Table 2).
Two-year vascular ischemic event rates among the entire study cohort and among patients with established arterial disease
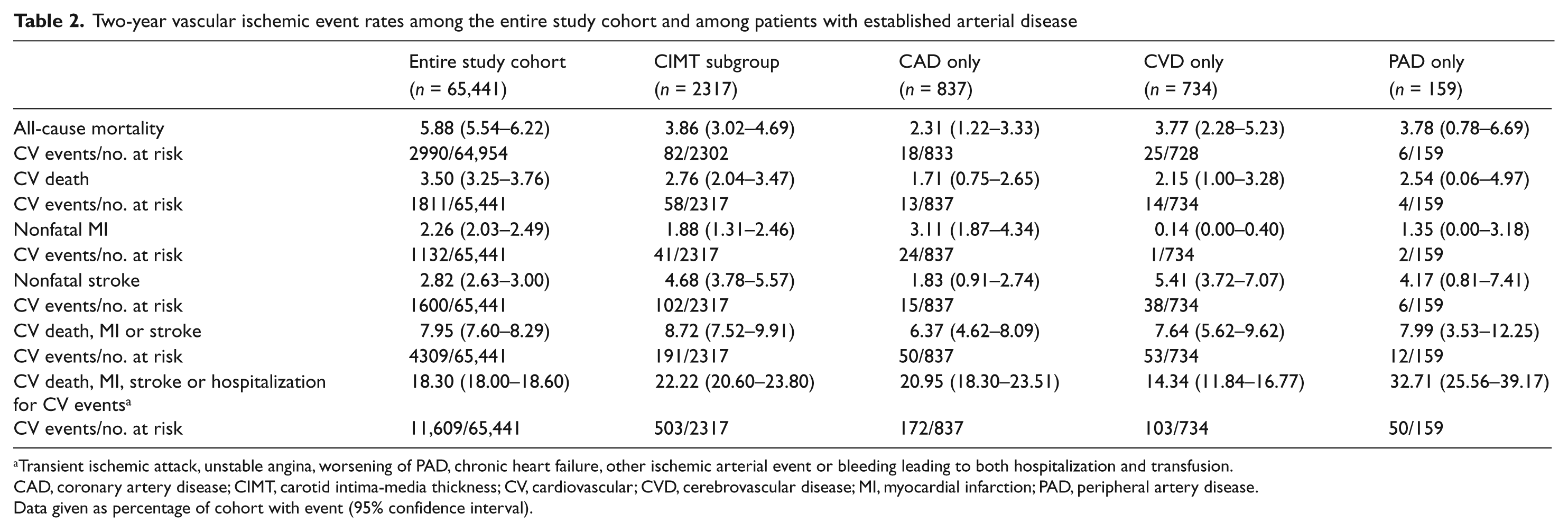
Transient ischemic attack, unstable angina, worsening of PAD, chronic heart failure, other ischemic arterial event or bleeding leading to both hospitalization and transfusion.
CAD, coronary artery disease; CIMT, carotid intima-media thickness; CV, cardiovascular; CVD, cerebrovascular disease; MI, myocardial infarction; PAD, peripheral artery disease.
Data given as percentage of cohort with event (95% confidence interval).
In the proportional hazards models, CIMT values at the common carotid artery were not significantly associated with any of the individual or composite endpoints when adjusted for age and sex, except for the quadruple endpoint. There was a positive increase in the HRs of the quadruple endpoint with increasing quartiles of CIMT (first quartile defined as reference; second quartile 1.19 [95% CI 0.89–1.60]; third quartile 1.38 [1.06–1.80]; fourth quartile 1.50 [1.15–1.96]; p = 0.02 for trend). The HR of the quadruple endpoint comparing the highest with the lowest quartile of CIMT was 1.50 (95% CI 1.15–1.96; p = 0.003).
In the fully adjusted model there was a positive increase in HRs of all-cause mortality and the quadruple endpoint with increasing quartiles of CIMT (Table 3). However, this positive increase in HRs is mainly driven by the fourth quartile indicating that carotid plaque rather than CIMT is associated with a substantial increased risk. Increasing quartiles of CIMT were not associated with the individual endpoints of nonfatal MI or stroke, or with the triple endpoint. Although not consistently linear, there was a positive increase in HRs of CV death with increasing quartiles of CIMT. The positive increase of these HRs is also mainly driven by the carotid plaque (i.e. fourth quartile of CIMT). The adjusted HRs of all-cause mortality, CV death, and the triple and quadruple endpoints comparing the highest (carotid plaque) with the lowest CIMT quartile were all significant (Figure 1, Table 3).
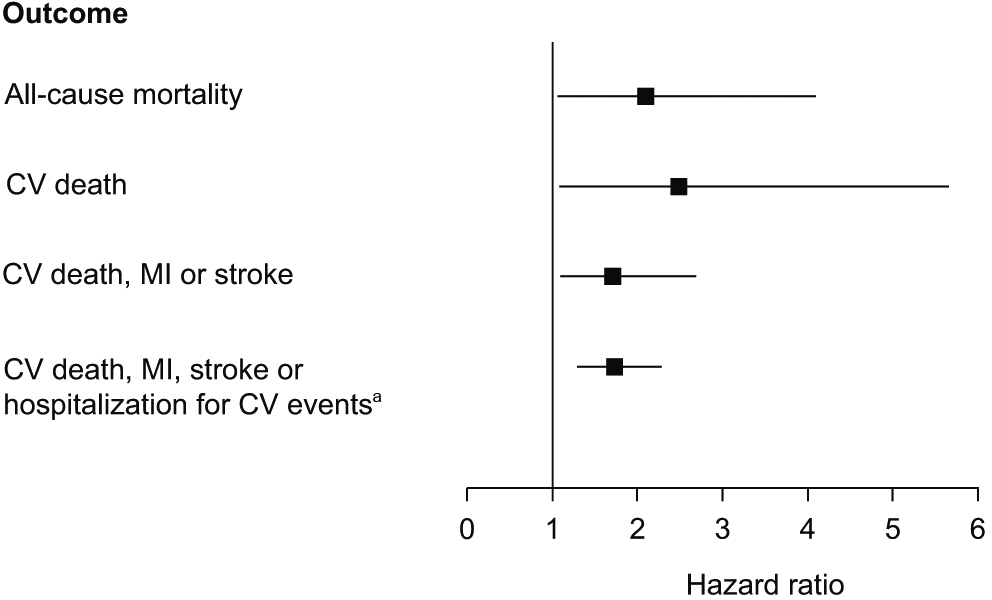
Adjusted HRs and 95% CIs for CV outcomes of the fourth (carotid plaque) (versus first) quartile among patients with atherosclerotic vascular disease. All HRs were significant (p < 0.05). aTransient ischemic attack, unstable angina, worsening of PAD, chronic heart failure, other ischemic arterial event, or bleeding leading to both hospitalization and transfusion. (CI, confidence interval; CV, cardiovascular; HR, hazard ratio; MI, myocardial infarction; PAD, peripheral artery disease.)
Two-year adjusted HRs of CV ischemic events according to increasing quartiles of CIMT and comparing fourth with first quartile of CIMT among any patients with established arterial disease and patients with CAD alone
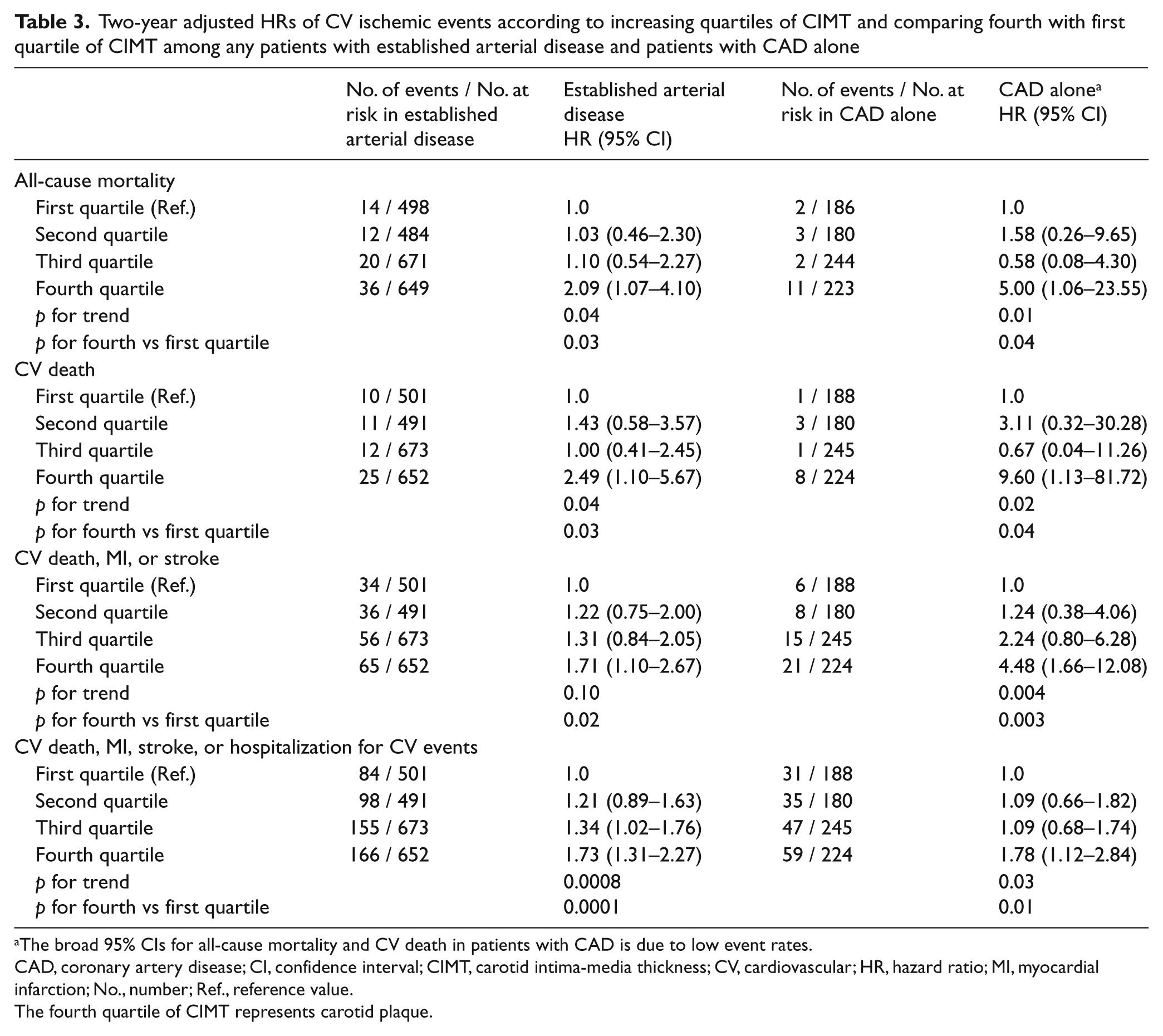
The broad 95% CIs for all-cause mortality and CV death in patients with CAD is due to low event rates.
CAD, coronary artery disease; CI, confidence interval; CIMT, carotid intima-media thickness; CV, cardiovascular; HR, hazard ratio; MI, myocardial infarction; No., number; Ref., reference value.
The fourth quartile of CIMT represents carotid plaque.
Patients with CAD alone revealed a positive increase in HRs of the triple and quadruple endpoints with increasing CIMT quartiles. Although not consistently linear, there was a positive increase in HRs of all-cause mortality and CV death with increasing CIMT quartiles (Table 3). No significant association between CIMT and the individual endpoints of nonfatal MI or nonfatal stroke were found among CAD-only patients. The adjusted HRs of all-cause mortality, CV death, and the triple and quadruple endpoints comparing the highest with the lowest CIMT quartile were all significant (Table 3). In this CAD-only subgroup, the increased HRs are also mainly driven by the fourth quartile, which by definition is carotid plaque.
There was no statistically significant association of HRs of any endpoints with increasing CIMT quartiles among patients with CVD or PAD alone. However, when comparing the highest (carotid plaque) with the lowest CIMT quartile, the quadruple endpoint showed a statistically significant association among patients with CVD (2.03; 95% CI 1.05–3.93; p = 0.04) but not among those with PAD.
Discussion
This study assessed whether CIMT and the presence of plaque is associated with vascular ischemic events and all-cause mortality in patients with established atherosclerotic disease during a 2-year follow-up. Our results suggest that carotid plaque rather than the thickness of intima-media is associated with an increased risk of all-cause mortality, CV death, and the composite endpoint of CV death, MI, stroke, or hospitalization in a stable outpatient population with atherosclerotic disease. CIMT values in the fourth quartile (i.e. carotid plaques) were associated with an increased risk of all-cause mortality, CV death, CV death/MI/stroke, and CV death/MI/stroke/hospitalization by 1.7–2.5 times compared with CIMT values in the first quartile. As the strongest association between carotid plaque and risk of adverse events was observed for the quadruple endpoint, carotid plaque seems to be a marker of generalized atherosclerotic burden rather than a more specific indicator of increased stroke or MI risk. The results may underestimate the true association of carotid plaque and vascular ischemic events, as the duration of follow-up in this study was relatively short. Except for the quadruple endpoint, intima-media thickness failed to predict any ischemic events, whereas carotid plaques tended to predict all-cause mortality and CV morbidity and mortality.
Our findings are in contrast to other studies of CAD, coronary bypass surgery, or stroke patients that have found a positive association between increasing CIMT and recurrent vascular ischemic events.14,20–22 However, another study of intima-media thickness divided in tertiles failed to predict CV events in patients with stable angina, thus supporting our findings. 23 Recently, Kablak-Ziembicka et al. 24 demonstrated in an observational study of 652 patients with established atherosclerosis that a mean CIMT value ≥ 1.25 mm is associated with a 2.5-fold increased risk of adverse CV events and CIMT quartiles ranged between ≤ 1.02 and ≥ 1.61 mm, which by definition included patients with carotid plaques. To our knowledge, there is only one study that evaluated the prognostic implications of carotid plaque in patients with known CAD concluding that carotid plaque predicts CV events, thus supporting our results. 23 Recently, carotid atherosclerotic plaque composition has been shown to predict systemic cardiovascular outcome. 25
An important finding was that patients with CAD alone had a significant increase in HR of all-cause mortality, CV death, and the triple and quadruple endpoints with carotid plaque. Thus, the additional measurement of CIMT to define carotid plaque could be a useful tool to identify patients with CAD who would be at increased risk of future vascular ischemic events. Carotid plaque in patients with CVD was associated with the quadruple endpoint. CIMT or the presence of carotid plaque in patients with PAD alone was not associated with any endpoints. These nonsignificant results could be explained by the small sample size in the subgroups and the limited duration of follow-up.
CIMT and the presence of carotid plaque can be assessed by B-mode ultrasound using a relatively simple technique, yielding information on wall changes that may not be obtained by conventional contrast angiography, computed tomography (CT), or magnetic resonance imaging (MRI). 26 Furthermore, angiography is invasive and, like CT, requires substantial radiation and contrast agents. MRI is time-consuming, technically challenging, and not available at all facilities. These diagnostic modalities are also associated with high costs. The usefulness of CIMT measurement to refine CV disease risk in patients at a modified intermediate Framingham risk score of 6–20% has already been suggested in patients without pre-existing CV disease.27,28 Therefore, carotid plaque measurement might be a good indicator for those with established atherosclerotic vascular disease to identify patients with incremental risk for vascular ischemic events, thus providing the basis of closer monitoring and aggressive lowering of CV risk factors and motivating the patients to treatment adherence. However, additional large-scale, population-based studies are needed to confirm our results before widespread use in routine practice can be recommended.
Despite the large sample size, there are some limitations to this study. CIMT and carotid plaque measurement was not performed using a common protocol at each site, and might therefore be subject to selection bias as evidenced by the different baseline characteristics. Follow-up was relatively short and thus might reduce the ischemic event rate and attenuate the association with the endpoints. The CIMT subgroup appeared to be younger, was less obese and had less diabetes and lipid abnormality than the entire population, which might reduce the subsequent ischemic event rate. Despite these limitations, our results provide additional information of the association between carotid plaque and vascular ischemic events in patients with established atherosclerotic vascular disease given the limited data currently available. Second, despite the absence of a centralized protocol and quality control for the CIMT and carotid plaque measurement, this study provides valuable information in terms of relevance in testing the practice-based usability of a CIMT and carotid plaque measurement in an international cohort.
In conclusion, our analyses suggest that the presence of carotid plaque, rather than the thickness of intima-media, appears to be associated with an increased risk of all-cause mortality and CV morbidity and mortality, but confirmation of these findings in prospective studies is required.
Footnotes
Acknowledgements
The REACH Registry is endorsed by the World Heart Federation. A complete list of REACH investigators is accessible online at ![]() . The REACH Registry enforces a no ghost-writing policy. We thank the REACH Editorial Support Group for providing editorial help and assistance in preparing this manuscript including editing, checking content and language, formatting, referencing and preparing tables and figures.
. The REACH Registry enforces a no ghost-writing policy. We thank the REACH Editorial Support Group for providing editorial help and assistance in preparing this manuscript including editing, checking content and language, formatting, referencing and preparing tables and figures.
The REACH Registry is sponsored by sanofi-aventis, Bristol-Myers Squibb, and the Waksman Foundation (Tokyo, Japan).
Drs Keo and Duval and Blandine Pasquet: none. Dr Baumgartner reports receiving honoraria as speaker from sanofi-aventis and Bristol-Myers Squibb. Dr Hirsch has received research grants from Bristol-Myers Squibb and sanofi-aventis. Dr Steg reports receiving research support from Servier, and honoraria as consultant or speaker from Astellas, AstraZeneca, Bayer, Boehringer-Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Medtronic, MSD, Otsuka, Roche, sanofi-aventis, Servier, The Medicines Company and being a stockholder in Aterovax. Dr Bhatt discloses the following relationships: research grants – AstraZeneca, Bristol-Myers Squibb, Eisai, sanofi-aventis, The Medicines Company. Dr Roether reports receiving honoraria as consultant or speaker from Boehringer-Ingelheim, Bristol-Myers Squibb, Lundbeck and sanofi-aventis.
