Abstract
Background:
Endarterectomy (open repair) represents the gold-standard for common femoral atherosclerotic disease (CFAD) treatment. However, with developments like atherectomy, endovascular therapy (endovascular revascularization) may offer an alternative option. The aim of the present study was to evaluate the safety and effectiveness of endovascular versus open therapy for the CFAD.
Methods:
The ARISTON (AtheRectomy-assIsted endovaScular Therapy versus OpeN) study was an 11-center research collaborative in which data from consecutive patients with symptomatic CFAD were analyzed. Retrospective matching was performed for patient-specific characteristics, including age, cardiovascular risk factors and comorbidities, and lesion-specific variables, including lesion calcification and complexity. Primary endpoints were all-cause mortality and freedom from clinically driven target lesion revascularization (CD-TLR). Amputation-free survival (AFS) was a secondary endpoint.
Results:
From 2015 to 2022, 826 patients undergoing endovascular (n = 213 [25.8%]) versus open therapy (n = 613 [74.2%]) were analyzed. The total number of procedural complications was higher with open therapy, whereas hospital stay was shorter with endovascular therapy (p < 0.0001 for both). Major adverse cardiac and limb events at 30-day outcomes were, however, not statistically different (p = 0.06). Bail-out stent rates with atherectomy-assisted endovascular therapy were 5.1%. After matching and during 1.72 (0.9–3.3) years of follow up, all-cause mortality, AFS, and CD-TLR were not statistically different in endovascular versus open therapy (HR = 0.68, 95% CI 0.36–1.29; HR = 1.5, 95% CI 0.59–3.77; and HR = 1.46, 95% CI 0.61–3.49, p = NS for all).
Conclusion:
Endovascular and open therapy exhibit comparable outcomes for the treatment of patients with symptomatic CFAD, including similar CD-TLR in patients with claudication and AFS in patients with chronic limb-threatening ischemia, during short-term follow up. Atherectomy-assisted endovascular therapy may therefore provide a useful alternative for patients who are unfit for surgery.
Keywords
Introduction
Atherosclerotic common femoral artery disease (CFAD) represents a traditional domain for open repair, where open therapy has proven long-term durability,1,2 and is deemed as the gold-standard strategy by intersociety consensus. 3 Mechanical forces due to hip mobility pose concerns for possible stent fracture limiting endovascular therapy, whereas stenting the CFA may compromise flow in the deep femoral artery, potentially resulting in chronic limb-threatening ischemia (CLTI) in cases of stent failure. In addition, stent placement in the groin may ‘burn bridges’ for future interventions as this region serves as an anastomotic or/and access site. 4
Despite indisputable long-term patency results, however, open repair may also be associated with wound infections, nerve damage, and redo-operation rates, whereas 30-day mortality rates may be high in older patients.5,6 Endovascular therapy may therefore represent an alternative, especially in patients at high surgical risk. Comparative studies investigating the two treatment options in CFAD are scarce. Previously, the randomized controlled TECCO trial 7 suggested that endovascular therapy may be a valuable alternative option, demonstrting comparable 2-year patency and clinically driven target lesion revascularization (CD-TLR) rates, albeit with lower complication and mortality rates than open therapy. A recent real-world multicenter trial by the CAULIFLOWER investigators pointed to higher 1-year patency rates with open versus endovascular therapy, especially in the presence of nodular calcification, which further attenuated endovascular results. 8 However, within the TECCO and CAULIFLOWER trials, no debulking techniques such as atherectomy were performed, which has demonstrated promising results for the treatment of CFAD if combined with drug-coated balloon (DCB) angioplasty in single-arm studies.9 –11
We therefore sought to evaluate the role of vessel preparation by atherectomy-assisted endovascular versus open therapy within a multicenter setting.
Methods
Study design and ethic approval
The ARISTON (AtheRectomy-assIsted endovaScular Therapy versus OpeN) study was a multicenter, retrospective research collaborative, with support from the ‘Research Collaborative in Peripheral Arterial Disease’ (RCPAD) network (https://www.rcpad.org/) (see the Appendix for a list of collaborators). Data for consecutive patients who presented with symptomatic peripheral artery disease (PAD) due to CFA high-grade stenosis or occlusion between January 2015 and July 2022 were collected. Approval was obtained from the local ethics committee of the University of Heidelberg and by the Ethical Committee of the Medical Council, Frankfurt; no patient-identifiable data were collected or exchanged. Additional ethical and regulatory approvals were obtained locally at each individual institution based on local regulations, which complied with the declaration of Helsinki. The local principal investigators from each institution were responsible for data collection, transfer, and local approvals. A fully anonymized uniform electronic reporting form was maintained across all institutions. The manuscript was prepared according to the STROBE statement for reporting.
Patient population and inclusion criteria
All centers were asked to provide anonymized data of all-comer consecutive symptomatic PAD patients with CFA high-grade stenosis or occlusion who were treated with open versus endovascular therapy during the study period. In patients with bilateral CFA disease, only the limb during the initial presentation was included. Demographic data and cardiovascular risk factors, severe chronic kidney disease (CKD) (defined as glomerular filtration rate (GFR) < 30 mL/min/1.73 m2 or end-stage renal disease, requiring dialysis), history of coronary artery disease (CAD) and heart failure, and cardiac medications, including antiplatelet and anticoagulation therapies, were recorded. Clinical symptoms of PAD were classified according to the Rutherford categories (RC) and subsection analysis was performed in patients with claudication and CLTI.
Selection of open versus endovascular therapy
All patients included in our study were referred for endovascular or open therapy treatment upon assessment of the primary treating physicians and/or vascular and endovascular specialists. Discussion about possible alternative treatment options took place even in centers primarily practicing one of the two revascularization types. In this context, patients were informed about the possible advantages and drawbacks of endovascular and open therapy.
Preprocedural and angiographic data
Based on preprocedural computed tomography angiography or procedural angiographic images, the Trans-Atlantic Inter-Society Consensus (TASC) classification was used for determining lesion complexity. 12 It should be noted that the TASC classification refers not only to the CFA, but to all peripheral lesions of the index limb, thus considering the concomitant involvement of iliac and femoropopliteal arteries and the presence of chronic total occlusions (CTO). CFA lesion complexity was rated semi-quantitatively based on the classification by Rabellino et al. by readers blinded to clinical and outcome data, 13 including: (i) anatomic localization: type I, isolated CFA lesions; type II, lesions in the external iliac artery (EIA) and the CFA but without involving the femoral bifurcation; type III, CFA lesions involving the femoral bifurcation; and type IV, lesions extending from the EIA down to the femoral bifurcation; (ii) CFA lesion calcification using the peripheral artery calcification scoring system (PACSS) score, as previously reported; 14 and (iii) the presence or absence of CTO. In addition, the length (mm) of the CFA lesion and the number of tibial run-off vessels were analyzed.
Endovascular versus open therapy
Procedures were performed by experienced interventional cardiologists, angiologists, or vascular and endovascular surgeons, dictated by the local institution, and each investigator based on physician and patient preference. Treatment strategies encompassed the following two arms: (A) endovascular therapy, where CFA lesions were generally treated with directional, rotational, or hybrid atherectomy followed by DCB angioplasty and/or bail-out nitinol stent placement.9,11,15 In case of disease extension into the femoral bifurcation, atherectomy-assisted endovascular therapy was performed both in the superficial femoral artery (SFA) and in the deep femoral artery lesions. Other lesion preparation techniques such as lithotripsy or specialty balloons, either as the primary treatment or an addition after atherectomy, were allowed and were used at the discretion of the endovascular specialists. Notably, planned stenting was not considered as a treatment option for the CFA among the endovascular operators from all the participating centers. (B) Open therapy was performed using Dacron, bovine, vein patch, or primary closures and if required including the placement of an interposition graft. In addition, hybrid procedures, including the external iliac artery and treatment of inflow iliac lesions were allowed and performed at the discretion of the vascular specialists. The treatment of outflow lesions beyond the SFA origin was also allowed in both groups.
Endpoints
Follow up, including clinical examination as well as Duplex sonography, if indicated, was performed at regular intervals in outpatient centers or by the referring physicians, wherever possible. As surveillance was affected by the pandemic after March 2020 in Europe, missing clinical data were obtained via a telephone call directly to the patient or the referring physician. During telephone contact, a standardized questionnaire was used to assess the patient’s clinical status, limiting symptoms including walking distance and wound healing disorders. These data constituted the base for the calculation of the RC during follow up.
Safety endpoints were perforation, embolization requiring reintervention or surgery, or access site complications requiring further interventions or open repair with endovascular therapy and reoperations, lymphocele, wounds requiring vacuum-assisted closure (VAC) therapy, and persistent nerve irritation with open therapy. Notably, VAC was not used as a primary dressing at the time of wound closure in the form of a preventing treatment.
During the first 30 days of follow up, major adverse cardiovascular events (MACE) and major adverse limb events (MALE) were assessed. MACE included cardiac death and non-fatal myocardial infarction. MALE included acute limb ischemia and above-the-ankle major amputation. Primary endpoints of the study were all-cause mortality and freedom from CD-TLR, the latter mainly driven by limiting clinical symptoms such as a decrease of the pain-free walking distance or ischemic wound progression. Secondary endpoints were major (above-the-ankle) amputation-free survival (AFS) and clinical improvement of at least one RC during follow up. In case of death, a thorough review of the medical records was performed to identify clinically relevant events and possible causes of death.
Statistical analysis
Analysis was performed using MedCalc 20.009 (MedCalc Software Ltd, Mariakerke, Belgium). Following the Shapiro–Wilk normality test analysis, continuous variables were found to have skewed distribution (p < 0.05) and were compared using the nonparametric Mann–Whitney U-test. All corresponding data in the text and tables are presented as median and IQR. Categorical variables were expressed as proportions and were compared using chi-squared tests. Kaplan–Meier analyses were used to evaluate all-cause mortality, AFS, and CD-TLR over time for patients with intermittent claudication (IC) versus CLTI, and endovascular versus open therapy. For comparison of endovascular versus open therapy, adjustment for patient and lesion complexity was performed by case–control matching, considering: (i) age (maximal allowable difference of 2.0 years); (ii) the total number of risk factors, including diabetes mellitus, CAD, heart failure, and CKD (exact match required for the score); (iii) the CFA lesion calcification by PACSS, 14 (exact match required); and (iv) the total anatomical lesion complexity patterns, including the presence of CTO, involvement of CFA bifurcation, the presence of additional inflow lesions or occlusions, and the presence of complex TASC C/D femoropopliteal lesions or occlusions (exact match required for the score). Subsection analysis was performed in patients with claudication versus CLTI and in patients > 70 years. Differences were considered statistically significant at p < 0.05.
Results
Patient demographics and baseline clinical data
After the exclusion of patients who underwent re-do procedures, 826 patients were analyzed in total, including 213 (25.8%) who underwent endovascular therapy and 613 (74.2%) who underwent open repair (Figure 1). The mean age was 72.0 (IQR = 66.0–79.0) years and 234 (28.3%) patients were women. A total of 570 (69.4%) patients had claudication (RC 2 or 3), 120 (14.6%) had ischemic rest pain (RC 4), and 92 (11.2%) and 39 (4.8%) patients, respectively, exhibited ischemic ulcerations (RC 5 and RC 6). In addition, 362 (43.8%) patients had CAD, 181 (21.9%) had heart failure, and 53 (6.4%) had severe CKD. Patients who underwent endovascular therapy exhibited a more complex patient history, including higher age, hyperlipidemia, diabetes mellitus, history of CAD, heart failure, and presence of CLTI (RC 5/6 in 23.7% vs 13.2%, p < 0.001) (Table 1). Diuretics, statins, and oral anticoagulants were more commonly prescribed at baseline in patients undergoing endovascular therapy.
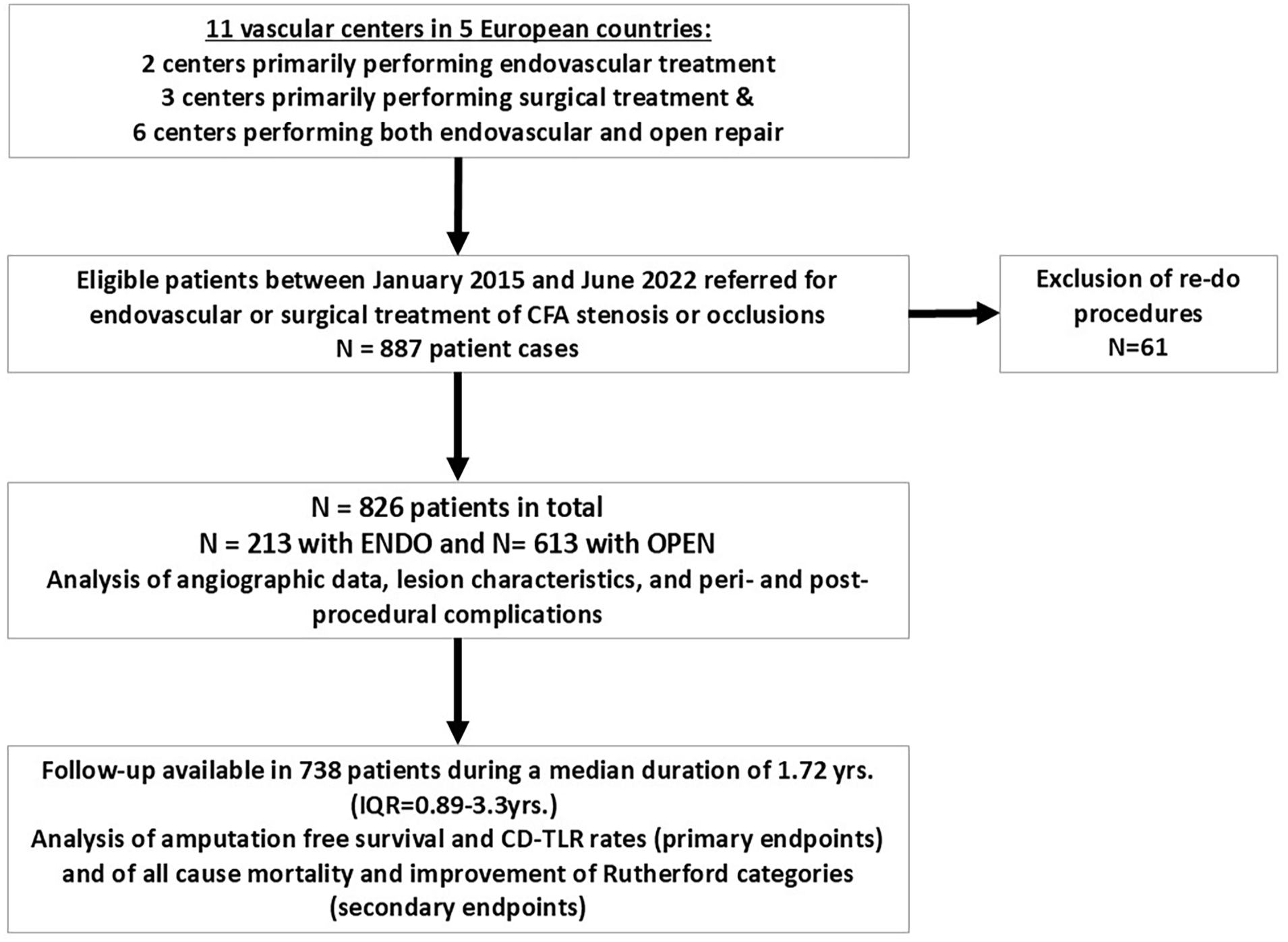
Study flowchart.
Baseline characteristics and clinical data.
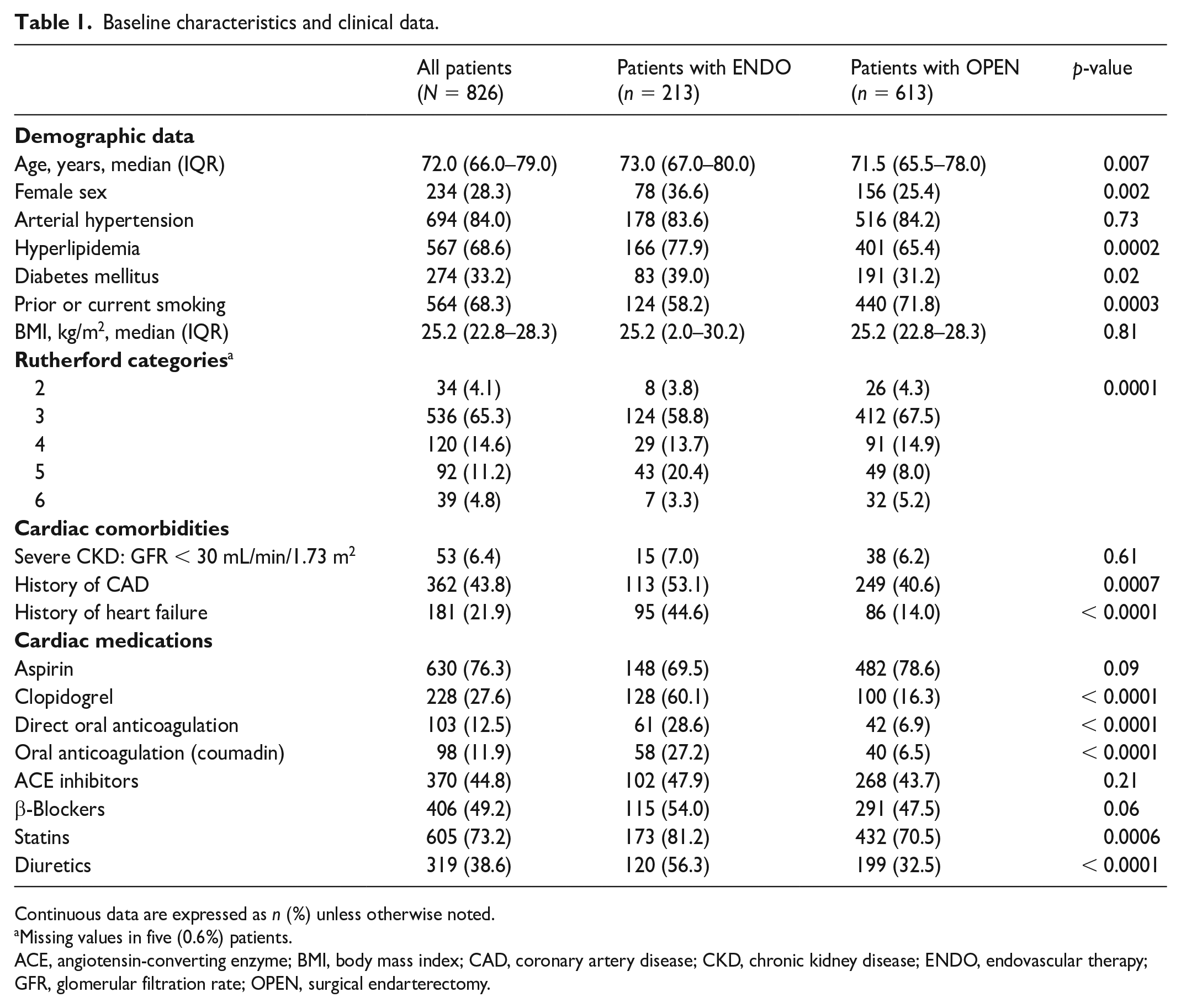
Continuous data are expressed as n (%) unless otherwise noted.
Missing values in five (0.6%) patients.
ACE, angiotensin-converting enzyme; BMI, body mass index; CAD, coronary artery disease; CKD, chronic kidney disease; ENDO, endovascular therapy; GFR, glomerular filtration rate; OPEN, surgical endarterectomy.
Lesion characteristics
Overall, the CFA lesion length was 32.0 (IQR = 22.0–42.0 mm) and 113 (13.7%) lesions were CTOs. CFA lesion types I, II, III, and IV were present in 8.7%, 21.3%, 55.1%, and 14.9% of cases, respectively, whereas lesion calcification was moderate to severe in most cases (2.0 [IQR = 2.0–3.0]). Additional ipsilateral iliac and femoropopliteal (TASC C/D) lesions were present in 231 (28.0%) and 300 (36.3%) cases, respectively. Lesion complexity by CFA lesion length, presence of CTO, lesion calcification, and additional iliac lesions or occlusions was higher in patients undergoing open versus endovascular therapy. The concomitant presence of complex TASC C/D ipsilateral femoropopliteal lesions, however, was more frequent with endovascular therapy (Table 2).
Lesion characteristics.
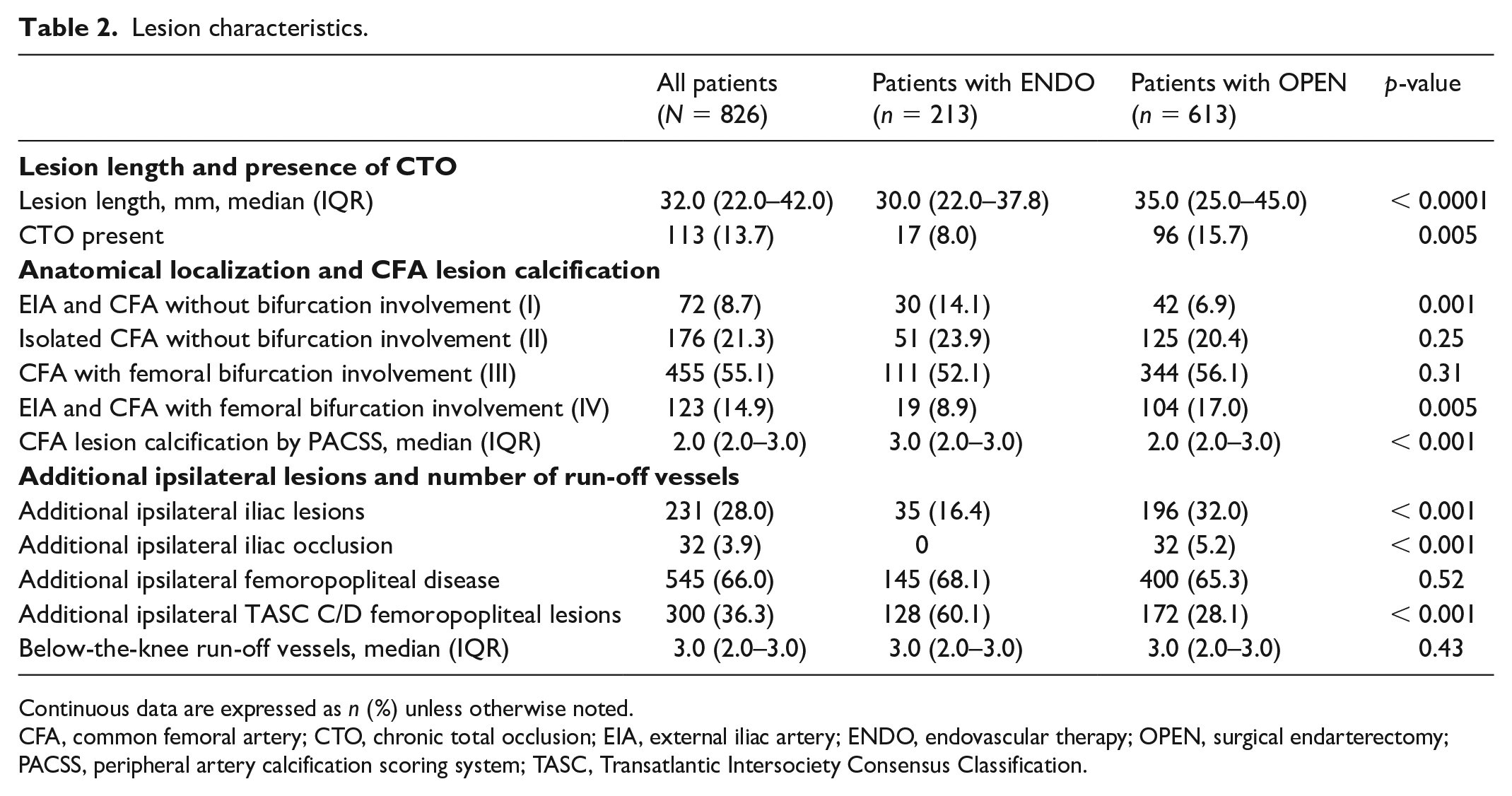
Continuous data are expressed as n (%) unless otherwise noted.
CFA, common femoral artery; CTO, chronic total occlusion; EIA, external iliac artery; ENDO, endovascular therapy; OPEN, surgical endarterectomy; PACSS, peripheral artery calcification scoring system; TASC, Transatlantic Intersociety Consensus Classification.
Endovascular versus open procedures and complications
Atherectomy was used in 177 (83.1%) endovascular therapy patients and combined with DCB was used in 173 (97.7%) patients (Table 3). Stent placement in the CFA was performed in 22 (10.3%) patients. The rate of stenting was significantly higher with nonatherectomy cases (13 [36.1%] vs 9 [5.1%], p < 0.0001). In 20 cases, self-expandable nitinol stents were used, whereas interwoven Supera stents were used in two cases. With open repair, Dacron, bovine, vein patch, and primary closure were performed in 220 (35.9%), 278 (45.4%), 35 (5.7%), and 47 (7.7%) cases, respectively.
Lesion preparation strategies and procedural data, including complications.
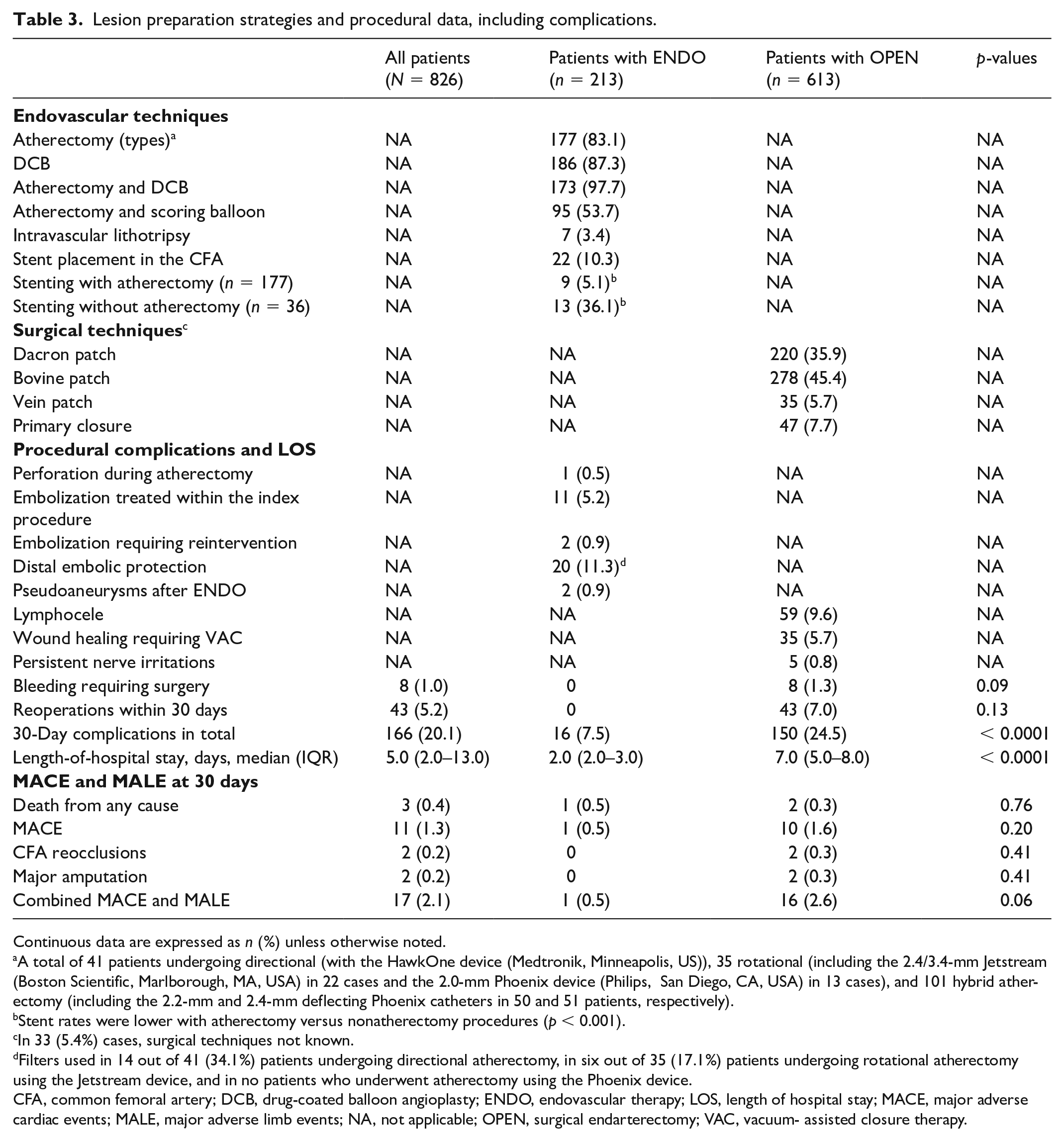
Continuous data are expressed as n (%) unless otherwise noted.
A total of 41 patients undergoing directional (with the HawkOne device (Medtronik, Minneapolis, US)), 35 rotational (including the 2.4/3.4-mm Jetstream (Boston Scientific, Marlborough, MA, USA) in 22 cases and the 2.0-mm Phoenix device (Philips, San Diego, CA, USA) in 13 cases), and 101 hybrid atherectomy (including the 2.2-mm and 2.4-mm deflecting Phoenix catheters in 50 and 51 patients, respectively).
Stent rates were lower with atherectomy versus nonatherectomy procedures (p < 0.001).
In 33 (5.4%) cases, surgical techniques not known.
Filters used in 14 out of 41 (34.1%) patients undergoing directional atherectomy, in six out of 35 (17.1%) patients undergoing rotational atherectomy using the Jetstream device, and in no patients who underwent atherectomy using the Phoenix device.
CFA, common femoral artery; DCB, drug-coated balloon angioplasty; ENDO, endovascular therapy; LOS, length of hospital stay; MACE, major adverse cardiac events; MALE, major adverse limb events; NA, not applicable; OPEN, surgical endarterectomy; VAC, vacuum- assisted closure therapy.
With endovascular therapy, one (0.5%) perforation occurred during atherectomy, 11 (5.2%) embolizations were successfully treated within the index endovascular procedure, two (0.9%) embolizations required reinterventions, and two (0.9%) pseudoaneurysms were successfully treated by manual compression. With atherectomy, distal embolic protection was used in 14 out of 41 (34.1%) patients undergoing directional atherectomy, in six out of 35 (17.1%) patients undergoing rotational atherectomy using the Jetstream device, and in no patients undergoing atherectomy using the Phoenix device (Table 3). In 13 of 177 (7.3%) patients in total suffering from distal embolization, which was either treated within the index procedure or required reintervention, distal filter protection was used in two (15.4%) cases.
With open therapy, bleeding requiring surgery occurred in eight (1.3%) cases, reoperations within 30 days in 43 (7.0%), lymphocele in 59 (9.6%), wound healing requiring VAC in 35 (5.7%), and persistent nerve irritation in five (0.8%) cases. The total number of complications was higher with open therapy, whereas hospital stay was shorter with endovascular therapy (p < 0.001 for both; Table 3). During 30 days, MACE and MALE were low, without significant differences between the two arms (p = 0.06; Table 3).
All-cause mortality, AFS, and CD-TLR
Clinical follow up was available in 738 (89.3%) patients during a median duration of approximately 2 years (1.72 years, IQR = 0.89–3.3 years). The follow-up duration was similar in patients undergoing open versus endovascular therapy (p = 0.18). During follow up, 84 (11.4%) patients died, 17 (2.3%) underwent major above-the-ankle amputation, and 86 (11.7%) underwent CD-TLR. Death was related to cardiovascular causes, sepsis, and cancer in 23 (27.4%), 22 (26.2%), and 20 (23.8%) patients, and to unknown causes in the remaining 19 (22.6%) cases. Mortality was significantly higher, whereas AFS and freedom from CD-TLR rates were lower in patients with CLTI versus claudication (Figure 2A–C).
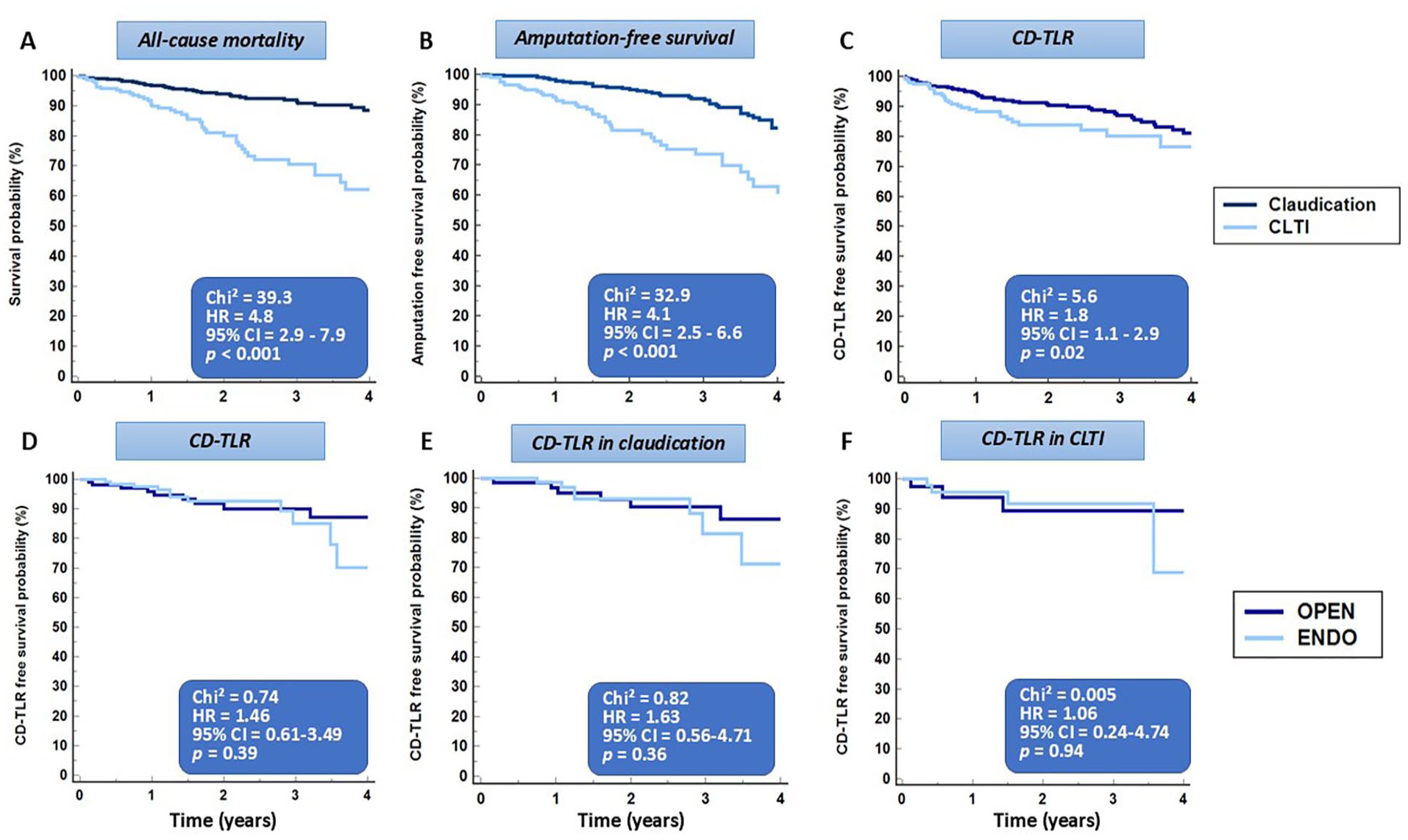
All-cause mortality was higher, whereas amputation-free survival and freedom from CD-TLR rates were significantly lower in patients with CLTI versus claudication
Primary outcomes by revascularization type
After matching patient history and PAD complexity, 270 patients were available for analysis (supplemental Table S1). All-cause mortality was similar with endovascular versus open therapy, (Figure 3A), whereas subsection analysis revealed similar mortality in claudication, but higher mortality rates for open therapy in patients with CLTI and in those > 70 years, in 358, 239, 119, and 224 patients, respectively, matched for clinical characteristics (Figure 3B–D). Notably, after matching patient and lesion characteristics, in 150 patients with claudication and 85 with CLTI, CD-TLR rates were similar for endovascular versus open therapy, in all patients, in patients with claudication, and in those with CLTI (Figure 2D–F).
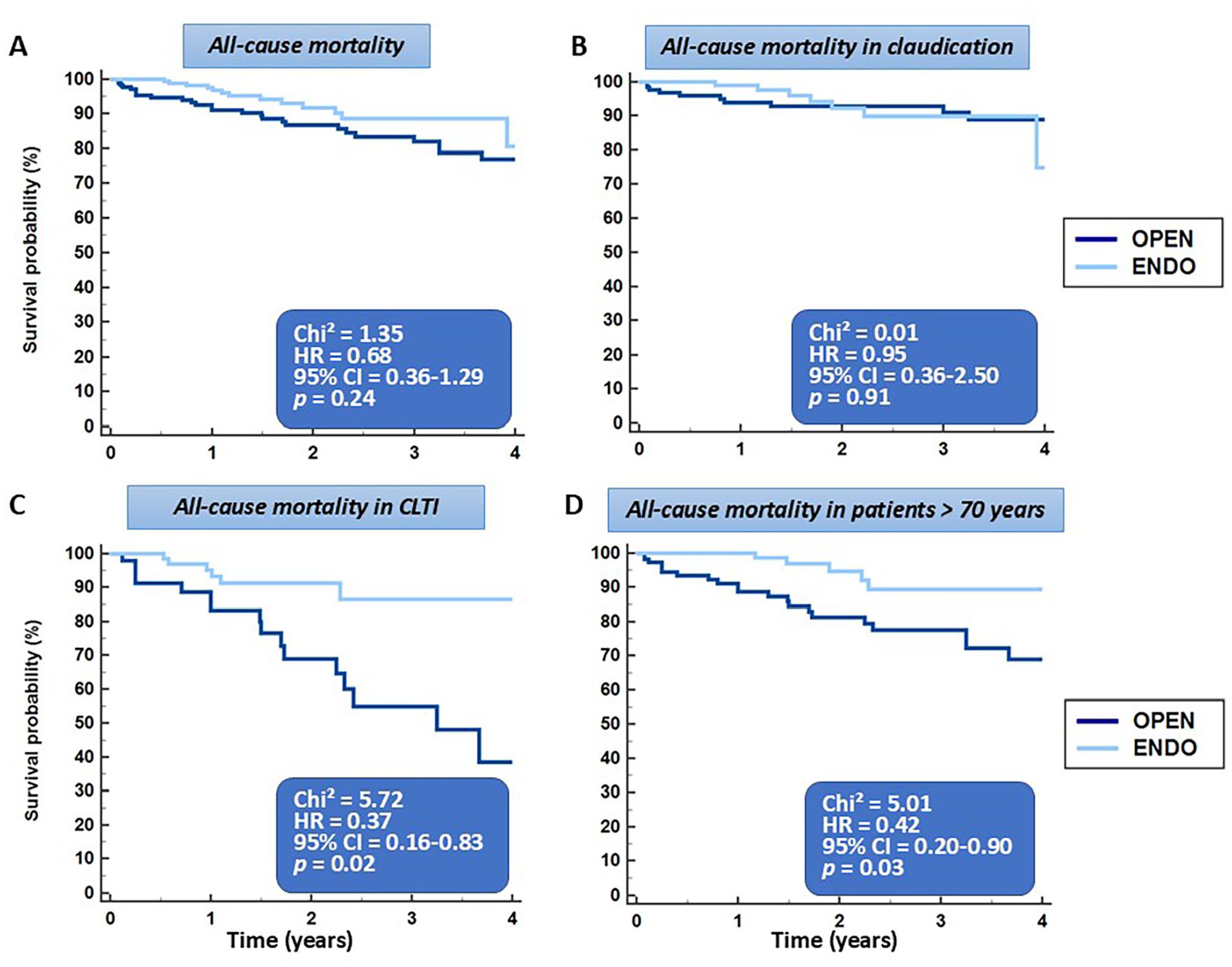
After matching, all-cause mortality was similar with ENDO versus OPEN
Secondary outcomes by revascularization type
Similar results were noted for AFS rates between endovascular and open therapy in the matched cohort, including 160 patients with claudication and 88 with CLTI (Figure 4A–C). During follow up, RC improvement was noted in 173 (90.6%) patients who underwent endovascular versus 372 (85.7%) patients who underwent open therapy (p = 0.09). RC improvement was similar in patients with claudication (p = 0.81) but favored endovascular therapy in patients with CLTI (p = 0.001) (Figure 4D–F).
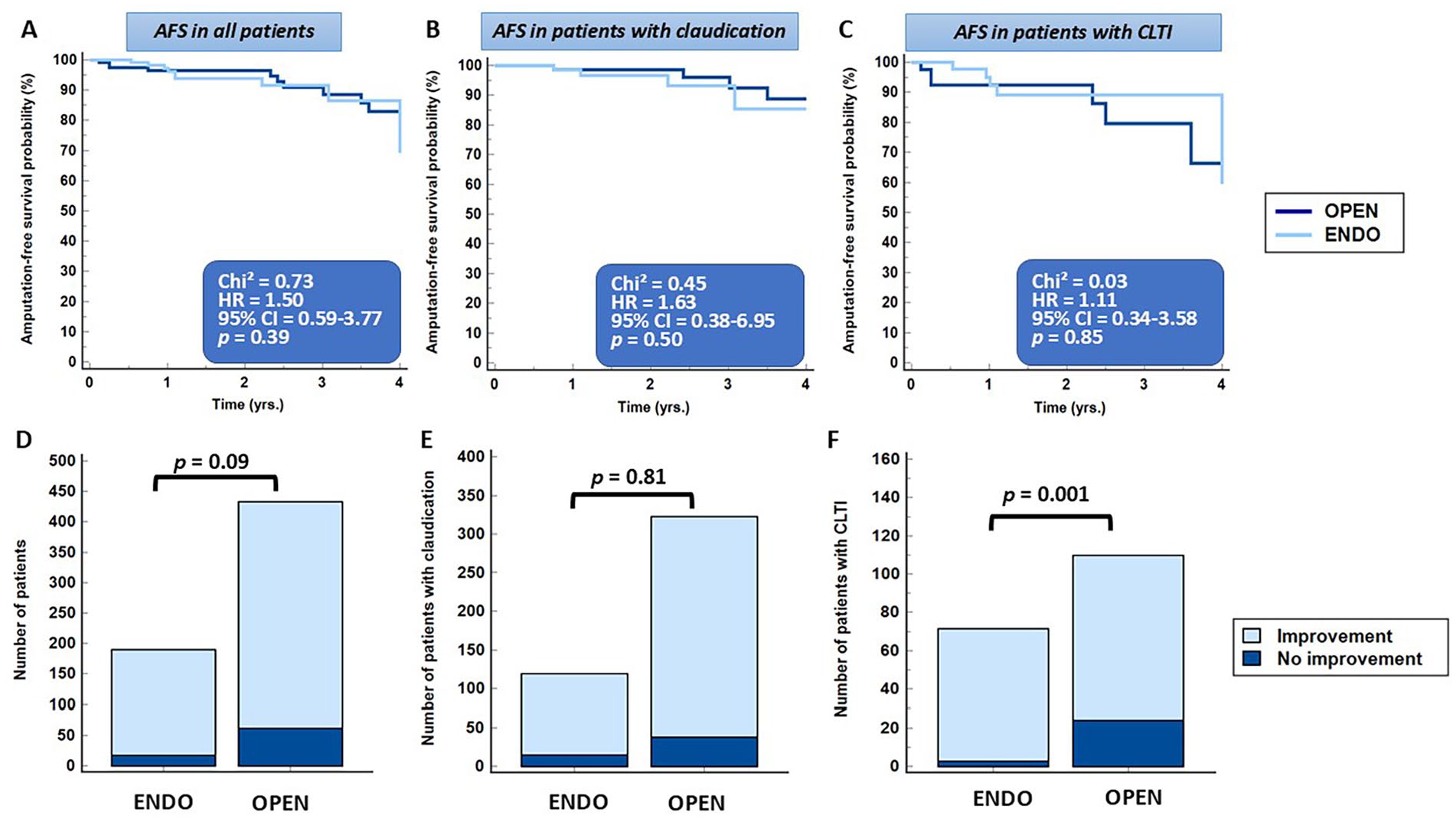
Similar AFS rates were noted between ENDO and OPEN in all patients
Discussion
To our knowledge, this is the largest multicenter study describing contemporary management strategies and their outcomes in patients presenting with symptomatic atherosclerotic CFAD, which for the first time in the current literature included a large proportion of patients treated with atherectomy-assisted endovascular techniques. The ARISTON study demonstrates that both open and endovascular therapy are safe and exhibit comparable results in terms of freedom from CD-TLR and clinical success with sustained RC improvement during follow up (Figure S1). Importantly, most lesions in the endovascular therapy arm were types III or IV, thus involving the femoral bifurcation, so that our trial studied the most complex CFA disease, not just focusing on focal or nonbifurcation lesions. On the other hand, despite higher invasiveness with open procedures, MACE and MALE rates at 30 days were not significantly different, whereas hospital stay was shorter with endovascular therapy. Notably, wound infection was not registered using prespecified criteria in our study, and only wound infections requiring VAC therapy were registered, so the number of wound infections may have been underreported in the open arm.
Atherectomy-assisted endovascular therapy in combination with DCB angioplasty exhibited lower bail-out stent rates compared to nondebulking-assisted endovascular therapy. Thus, this may be the preferred endovascular treatment regimen for the groin as it combines the advantage of ‘leave nothing behind’ in a mobile zone, with simultaneously lower CD-TLR rates. Although stenting may not directly affect CD-TLR and MALE rates, stents may fracture in the long term or limit endovascular access and surgical options in the future, which may have relevant implications for the cardiovascular and limb health of the patients receiving permanent metallic implants in the groin. After matching patient characteristics and lesion complexity, endovascular therapy exhibited similar CD-TLR and AFS compared to open therapy. Notably, all-cause mortality after open therapy was higher in patients with CLTI and in patients older than 70 years.
With continuous technical developments and the introduction of new materials, 16 endovascular revascularization has emerged as the state-of-the-art therapy for a vast majority of patients with PAD. However, there is ongoing debate about the specific requirements of the CFA region, which due to mobility of the hip joint is exposed to strong biomechanical forces.17,18 In the CFA, open therapy is considered as the reference standard because it yields robust long-term results regarding vessel patency and limb outcomes.2,19 However, open therapy was also associated with a significant burden of complications, such as redo-procedures, nerve damage, seromas, and wound infections. Thus, in a retrospective analysis, which evaluated 30-day outcomes in 1843 CFA endarterectomy patients from the National Surgical Quality Improvement Program database between 2005 and 2010, postoperative mortality and wound-related complications were 3.4% and 8.0%, respectively. 5 In an observational two-center study involving experienced operators on the other hand, open therapy showed very high success and low rates of complications. 20 These results are highly favorable compared to previous reports, setting a high standard for the treatment of the CFA by experienced operators. An overview of the surgical complication rates of the TECCO, 7 the CAULIFLOWER, 8 and of the CFA registry by Nguyen et al. 5 is provided in Table 4. In the present study, wound healing disorders after open therapy were comparable to the CFA registry, lower than in the TECCO trial but higher compared to the CAULIFLOWER study. This may be attributed not only to differences regarding the patient cohorts but also to the way wounds were registered. The CAULIFLOWER study reported only on wound infections and in our analysis only wounds requiring VAC were recorded. In addition, mortality rates were low in our study, like all other studies except for the CFA registry, where a high mortality rate of 3.4% was reported 30 days after open therapy. Finally, redo surgery or reintervention rates were similar to the CFA registry but higher compared to the CAULIFLOWER study. The total rate of major complications was comparable to the CFA registry and to the TECCO trial, thus underlining the real-world character of our study.
Overview of open therapy complications at 30 days compared to the ARISTON trial.
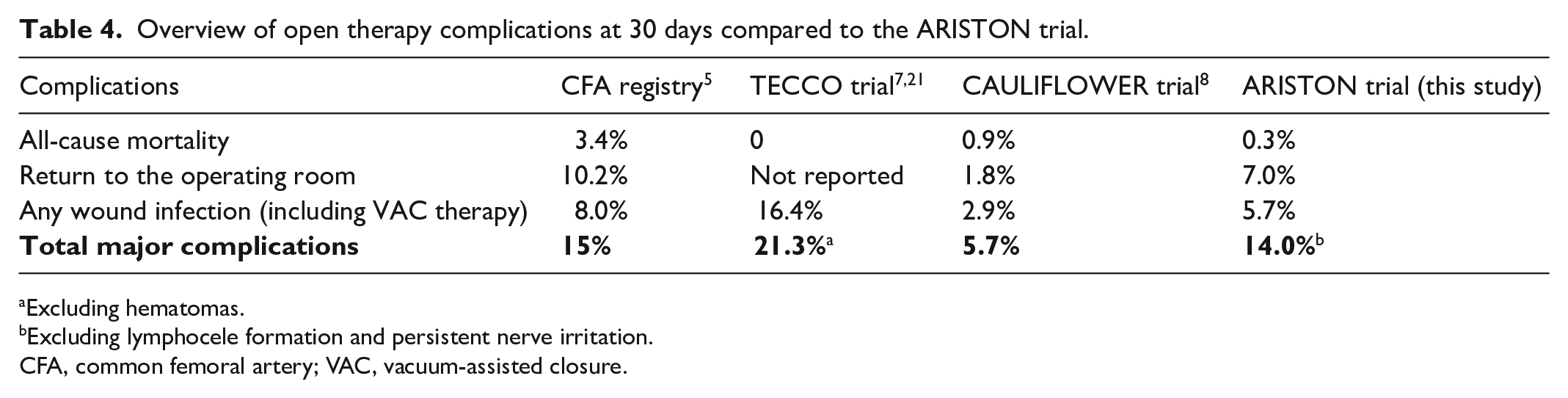
Excluding hematomas.
Excluding lymphocele formation and persistent nerve irritation.
CFA, common femoral artery; VAC, vacuum-assisted closure.
Gouëffic et al. 7 reported on the first randomized controlled TECCO trial of CFA open versus endovascular therapy. In this, multicenter study involving 17 centers in France, the primary outcome of operative morbidity and mortality at 30 days was significantly lower in the stenting group based on both intention-to-treat (12.5% vs 26%; p = 0.05) and per-protocol (6.4% vs 26%; p = 0.005) analyses. During a median follow up of 2 years, there were also similar rates of clinical improvement, patency, and CD-TLR between the two groups. In addition, freedom from CD-TLR was similar with stenting of anatomically complex versus simple CFA lesions. 21 Despite these results for stenting, long-term consequences need to be considered with stenting in this region because the groin is the traditional access site for coronary, vascular, and structural heart interventions and an important landing zone for bypass surgery.
Another recent real-world multicenter comparative trial by the CAULIFLOWER investigators evaluated 1193 consecutive patients in 66 centers, who underwent open (n = 761) versus endovascular therapy (n = 432), the latter mainly consisting of plain old balloon angioplasty (POBA), DCB, and if required stent placement. 8 After matching, periprocedural 30-day complications were higher in the open versus endovascular group, whereas the length of hospital stay was significantly shorter with endovascular therapy. Importantly, residual 25–50% stenosis on the final angiogram was significantly more frequent with endovascular versus open therapy (39.3% vs 1.4%, p < 0.001). As residual stenosis has been reported as a predictor for higher CD-TLR rates with endovascular therapy, 9 it may have triggered significantly lower 1-year primary patency compared to the open group (82.3% vs 96.6%; p < 0.001). Indeed, the presence of nodular calcification, which is probably most difficult to treat by angioplasty or stent placement and without the prior use of lesion preparation strategies, further attenuated the results of endovascular therapy, favoring open therapy. 8 Patients presenting with nonambulatory status, on the other hand, exhibited similar outcomes irrespective of the revascularization type.
In our study, endovascular therapy involved the use of atherectomy techniques in 177 of 213 (83.1%) cases. Notably, most of these patients underwent Phoenix atherectomy (Philips, San Diego, CA, USA) using the 2.2 and 2.4-mm catheters which combine directional and rotational properties due to the ability of deflection.11,15,22 In the present study, the rate of bail-out stenting was indeed significantly lower with atherectomy- compared to nonatherectomy-assisted endovascular procedures (5.0% vs 31.6%, p < 0.001). Notably, bail-out stent placement rates were similarly low to recent studies, which reported stent rates between 3% and 8% for the CFA after usage of rotational/hybrid and directional atherectomy, respectively.9,14,22 Notably, in patients with atherectomy-assisted endovascular therapy, 97.7% and 53.7%, respectively, received adjunct treatment with DCB and scoring balloon angioplasty, which highlights that atherectomy is only part of the endovascular procedure and not a stand-alone approach.
Comparing open to endovascular therapy after matching, no differences were noted in terms of CD-TLR and AFS during follow up, whereas mortality, amputation, and CD-TLR rates were higher in patients with CLTI versus those with claudication. Looking at patient subgroups, for CD-TLR and AFS, no differences were observed between endovascular therapy and open therapy for patients with CLTI or claudication. Furthermore, the total number of 30-day complications was higher with open therapy, whereas the length of hospital stay was lower with endovascular therapy, which agrees with previous studies.7,8 Although the level of experience was not controlled by study protocol in our observational trial, it should be noted that atherectomy-assisted endovascular therapy was typically performed by experienced and board-certified endovascular specialists (GK, JK, and MA), whereas open procedures may have been performed by less experienced operators. In addition, the higher mortality rate with open therapy in patients with CLTI and older patients merits further in-depth analysis, including adjunct medical treatment of the patients in future studies. Recently, a subsection analysis of the BASIL-2 trial reported on increased mortality in patients with CLTI undergoing surgery, which was mainly attributed to increased rates of cardiovascular deaths not directly after surgery but during the follow-up period. 23 RC improvement, on the other hand, was higher in patients undergoing endovascular versus open therapy due to CLTI, whereas no differences were observed in this context in patients with claudication. As patients who are older or/and present with CLTI may be more frail and therefore more prone to complications with open therapy,5,6 these patient subgroups may profit more from less invasive strategies.
Limitations
Our study has some limitations. Although centers were asked to provide consecutive patients presenting with symptomatic CFAD, the exact mode of enrollment in each center was not controlled. Thus, patient heterogeneity issues and the lack of randomization, as well as the relatively short follow-up period, all need to be considered, especially when interpreting the comparative value of endovascular versus open therapy. Furthermore, the reasons for the initial treatment choice were influenced by selection and treatment biases. Although matching reduced such biases as much as possible, some unknown confounding variables such as patient ‘frailty’, patient medications, and fibrosis of the atherosclerotic lesions were not assessed and may have influenced patient or limb endpoints. Furthermore, analysis of subgroups within the matched cohort included smaller numbers of patients. Also, different levels of experience for the physicians performing the endovascular versus open therapy may have influenced the results. In addition, angiographic analysis of the lesions has been performed in a nonblinded fashion and without core-lab adjudication. Endovascular results showed a sharp worsening in terms of CD-TLR after 3 years, which requires more in-depth analysis in future longer-term follow-up studies. In addition, patency rates of the CFA and the deep femoral artery were not assessed by Duplex-sonography during the follow-up duration, and the analysis of concomitant SFA disease and the type of revascularization for these lesions was not controlled by the study protocol. Future randomized controlled trials are now therefore warranted to allow for an equivalent evaluation of the two revascularization types over longer time periods.
Conclusions
The ARISTON study implements the latest developments of vessel preparation with atherectomy-assisted endovascular therapy for the treatment of patients with symptomatic CFAD. The present study highlights the durable and reproducible early-to-mid-term clinical outcomes of open therapy, simultaneously providing early-to-mid-term results for atherectomy-assisted endovascular therapy as a potential alternative treatment option. Endovascular therapy may represent a good alternative option, especially in unfit surgical candidates, such as patients with a complex history, advanced age, and CLTI. The results of our study need to be interpreted in the context of the relatively short follow-up duration, whereas prospective randomized trials with a rigorous inclusion of patients should be the next step of evaluation and scientific goal.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251323508 – Supplemental material for Atherectomy-assisted endovascular therapy versus open repair for atherosclerotic common femoral artery disease: The multicenter ARISTON study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251323508 for Atherectomy-assisted endovascular therapy versus open repair for atherosclerotic common femoral artery disease: The multicenter ARISTON study by Grigorios Korosoglou, Jason T Lee, Martin Andrassy, Drosos Kotelis, Marco V Usai, Mario D’Oria, Raphael Coscas, Nicola Troisi, Bahaa Nasr, Athanasios Saratzis, Solon Antoniades and Konstantinos P Donas in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X251323508 – Supplemental material for Atherectomy-assisted endovascular therapy versus open repair for atherosclerotic common femoral artery disease: The multicenter ARISTON study
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X251323508 for Atherectomy-assisted endovascular therapy versus open repair for atherosclerotic common femoral artery disease: The multicenter ARISTON study by Grigorios Korosoglou, Jason T Lee, Martin Andrassy, Drosos Kotelis, Marco V Usai, Mario D’Oria, Raphael Coscas, Nicola Troisi, Bahaa Nasr, Athanasios Saratzis, Solon Antoniades and Konstantinos P Donas in Vascular Medicine
Footnotes
Appendix. The ARISTON Collaborative
Grigorios Korosoglou1, Jason T Lee2, Martin Andrassy3, Drosos Kotelis4, Marco V Usai5, Mario D’Oria6, Raphael Coscas7, Nicola Troisi8, Bahaa Nasr9, Athanasios Saratzis10, Solon Antoniades11, Konstantinos P Donas12,13, Gergana T Taneva12,13, Apostolos Pitoulias12,13, Jan Karcher1, Christoph Schöfthaler1, Konstantinos Avranas12, Christian Zielasek4, Corinne Kohler4, Dimitrios D Papazoglou4, Thomas Schönefeld5, Austermann Martin5, Arne Schwindt5, Beatrice Grando6, Sandro Lepidi6, Raffaella Berchiolli8, and Hany Zayed14
1Department of Cardiology and Vascular Medicine, GRN Hospital Weinheim, Weinheim, Germany
2Division of Vascular Surgery, Stanford University Medical Center, Stanford, CA, USA
3Department of Cardiology and Angiology, Fürst Stirum Clinic Bruchsal, Bruchsal, Germany
4Department of Vascular Surgery, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
5Department of Vascular and Endovascular Surgery, Franziskus Hospital Münster, Münster, Germany
6Department of Clinical Surgical and Health Sciences, University of Trieste, Trieste, Italy
7Department of Vascular Surgery, CHU Ambroise Paré, Assistance Publique – Hôpitaux de Paris (AP-HP), France and University Versailles-Saint Quentin, University Paris-Saclay, Paris, France
8Department of Vascular and Endovascular Surgery, University of Pisa, Pisa, Italy
9Department of Vascular and Endovascular Surgery, CHU Brest Hospital, Brest, France
10Department of Cardiovascular Sciences, University of Leicester, Glenfield Hospital, Leicester, UK
11Department of Vascular and Endovascular Surgery, Isar Klinikum München, Munich, Germany
12Department of Cardiology and Vascular Medicine, Langen, Langen, Germany
13Department of Vascular and Endovascular Surgery, Rhein Main Vascular Center, Asklepios Clinics Langen, Paulinen Wiesbaden, Seligenstadt, Langen, Germany
14Department of Vascular Surgery, Guy’s and St Thomas’ NHS Foundation Trust, London, UK
In addition to the authors’ disclosures, the remaining ARISTON collaborators disclosed the following potential conflicts of interest. Hany Zayed receives honoraria from Abbott Ltd, Gore Medical, Bentley, Boston Scientific, Cordis, and Cook Medical; research grant support from Abbott Ltd; proctors for Gore Medical; and consults for LimFlow SA. The remaining collaborators have nothing to disclose.
Acknowledgements
A commentary by Dubosq-Lebaz and Secemsky accompanies this article. 24
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Grigorios Korosoglou: received modest speaker honoraria from Philips, Boston Scientific, and BARD Peripheral Vascular Inc. Raphael Coscas: consulting/speaking honoraria from Medtronic, Boston Scientific, Biotronik, Abbott, BD, and Shockwave. Nicola Troisi: receives honoraria from Biotronik SE & Co. and B Braun SE & Co. Athanasios Saratzis: funded by the National Institute for Health Research (salary support) and British Heart Foundation (research support); receives research and salary support by Abbott Ltd; receives honoraria from Shockwave Ltd. All other authors have no conflicts of interest to declare.
Funding
This study was supported in part by an institutional research grant from Philips to the GRN Hospital Weinheim (Grigorios Korosoglou).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
