Abstract
One in 10 independently living adults aged 65 years old and older is considered frail, and frailty is associated with poor postoperative outcomes. This systematic review aimed to examine the association between frailty assessments and postoperative outcomes in patients with vascular disease. Electronic databases – MEDLINE, Embase, and the Cochrane Library – were searched from inception until January 2022, resulting in 648 articles reviewed for potential inclusion and 16 studies selected. Demographic data, surgery type, frailty measure, and postoperative outcomes predicted by frailty were extracted from the selected studies. The risk of bias was assessed using the Newcastle–Ottawa Scale. The selected studies (mean age: 56.1–76.3 years) had low-to-moderate risk of bias and included 16 vascular (elective and nonelective) surgeries and eight frailty measures. Significant associations (p < 0.05) were established between mortality (30-day, 90-day, 1-year, 5-year), 30-day morbidity, nonhome discharge, adverse events, failure to rescue, patient requiring care after discharge, and amputation following critical limb ischaemia. The strongest evidence was found between 30-day mortality and frailty. Composite 30-day morbidity and mortality, functional status at discharge, length of stay, spinal cord deficit, and access site complications were found to be nonsignificantly associated with frailty. With frailty being significantly associated with several adverse postoperative outcomes, preoperative frailty assessments can potentially be clinically useful in helping practitioners predict and guide the pre-, peri-, and postoperative management of frail with vascular disease.
Introduction
As the worldwide population ages, vascular diseases of both arterial and venous systems are expected to increase. The prevalence of any vascular disease is significantly associated with increased age: 2% in those aged 40–50 years old, 3.5% in 51–60 years old, 7.1% in 61–70 years old, 13.0% in 71–80 years old, 22.3% in 81–90 years old, and 32.5% in 91–100 years old (p < 0.0001). 1 Coupled with continuous improvements in techniques and minimally invasive technology, the demand for vascular surgical interventions among the elderly will rise, 2 with a projected 10% increase in vascular surgery patients aged 65–84 by 2040. 3
The elderly population, however, is associated with increased frailty, as approximately one in 10 independently living adults aged 65 years and older are classified as ‘frail’. 4 Frailty is commonly defined as a decrease in physiological reserves with consequent increased susceptibility to adverse health events when exposed to environmental stressors.5 –7 Sarcopenia is the precursor syndrome of frailty, resulting in progressive quantity and quality loss of muscle mass. 8 Important common pathophysiological processes such as chronic inflammation,9,10 sarcopenia, 11 anaemia, 12 malnutrition,13,14 accelerated atherosclerosis, 15 and insulin resistance16,17 underpin frailty and chronic vasculopathy. Frailty is associated with increased mortality, disability, and high resource utilization. 18 Frailty was also found to be the leading cause of death (27.9%) among a 10-year prospective cohort study of 754 community-dwelling elderly individuals aged 65 years old and over, compared to organ failure (21.4%), cancer (19.3%), dementia (13.8%), and other causes (14.9%) 19 in general surgical patients. In surgical patients, frailty is also associated with increased mortality, complications, and longer lengths of stay.20,21 Conceptually and operationally, frailty appears to be related to, but distinct from, disability, comorbidity, and chronological age. 22
As frailty becomes an increasingly pervasive characteristic of elderly patients receiving vascular and endovascular surgery, it is important that practitioners consider adopting the concept of frailty measures both preoperatively and postoperatively to optimize patient outcomes. Therefore, this systematic review aims to examine the association between frailty and adverse postoperative outcomes in vascular and endovascular surgery.
Methods
Search strategy
MEDLINE, Embase, and Cochrane online databases were searched from their date of establishment to January 2022. The search strategy included the following keywords and process: ‘vascular and endovascular surgical procedures’ or ‘vascular and endovascular surgery’ in combination with ‘frailty’. The reference lists from any relevant review articles were cross-matched with the search results to ensure search terms were broad and inclusive of all relevant studies, thus minimizing relevant studies being missed. Conference abstracts were also considered for inclusion if sufficient data were available for meta-analysis.
Publication selection
To be included in the systematic review, the following inclusion criteria had to be met: (1) patients underwent a vascular and endovascular surgical procedure; (2) frailty measure was clearly defined; (3) clear postoperative outcomes were measured; and (4) the relationship between frailty and postoperative outcomes was evaluated. Non-English articles were excluded.
Preoperative (i.e., comorbidities) and postoperative factors (i.e., mortality, morbidity, and length of stay) were examined while defining patients. Only patients undergoing vascular surgical procedures were included in the meta-analysis, which was comprised of vascular patients with or without existing medical comorbidities (i.e., chronic renal failure, diabetes mellitus, dyslipidaemia, and hypertension). During the analysis, patients undergoing high-risk open vascular procedures were distinguished from those undergoing low-risk endovascular procedures. Subsequently, the risks from preexisting medical conditions (i.e., cardiac, cognitive dysfunction, difficult pain management, pulmonary and renal) were analyzed separately, as they may confer risk of pre-, peri-, and postoperative complications.23,24
Data extraction
Two reviewers independently screened the studies for inclusion first by abstract and title, and the included articles were reviewed in their entirety in the second stage of screening. Data extraction followed, and the information collected were verified by a third and fourth reviewer. Any conflicts which arose were resolved by consensus, or by appeal to a fifth independent reviewer.
The following data were extracted from the eligible studies: first author, year of publication, sample size, country of origin of the study population, mean/median age, sex of study population, study design, type of surgery performed, frailty measure, postoperative outcomes measured, and association between frailty and postoperative outcomes.
Evaluation of risk of bias
The risk of bias among all studies used was assessed using the Newcastle–Ottawa Scale (NOS). 25 NOS is used to assess the quality of nonrandomized studies to be included in systematic reviews and meta-analyses and has been split into three criteria: selection, comparability, and outcome. Each study is awarded a number of stars for each section, based on predetermined queries. A maximum of four stars can be given to ‘selection’ and ‘outcome’ categories, while a maximum of two stars can be given for ‘comparability’.
Analysis plan
Owing to significant clinical heterogeneity in patient populations, types of vascular surgery, frailty definitions and study methodologies including outcomes, a meta-analysis was not performed. Instead, a narrative synthesis was conducted.
Results
Study characteristics
In the literature search, 648 articles (180 from MEDLINE, 409 from EMBASE, and 59 from the Cochrane database) were identified. Four additional articles were added upon review of additional journals. Of the 648 articles, 177 duplicate articles were removed. The abstracts, titles, and full texts of the articles were reviewed. Another 412 studies were removed after the abstract and title review; and 43 after full-text review. The references of the selected articles were searched for further eligible studies. Sixteen articles published between 2011 and 2022, comprising a total of 181,416 patients, were included in the final analysis. The study selection process and reasons for exclusion are shown in Figure 1.
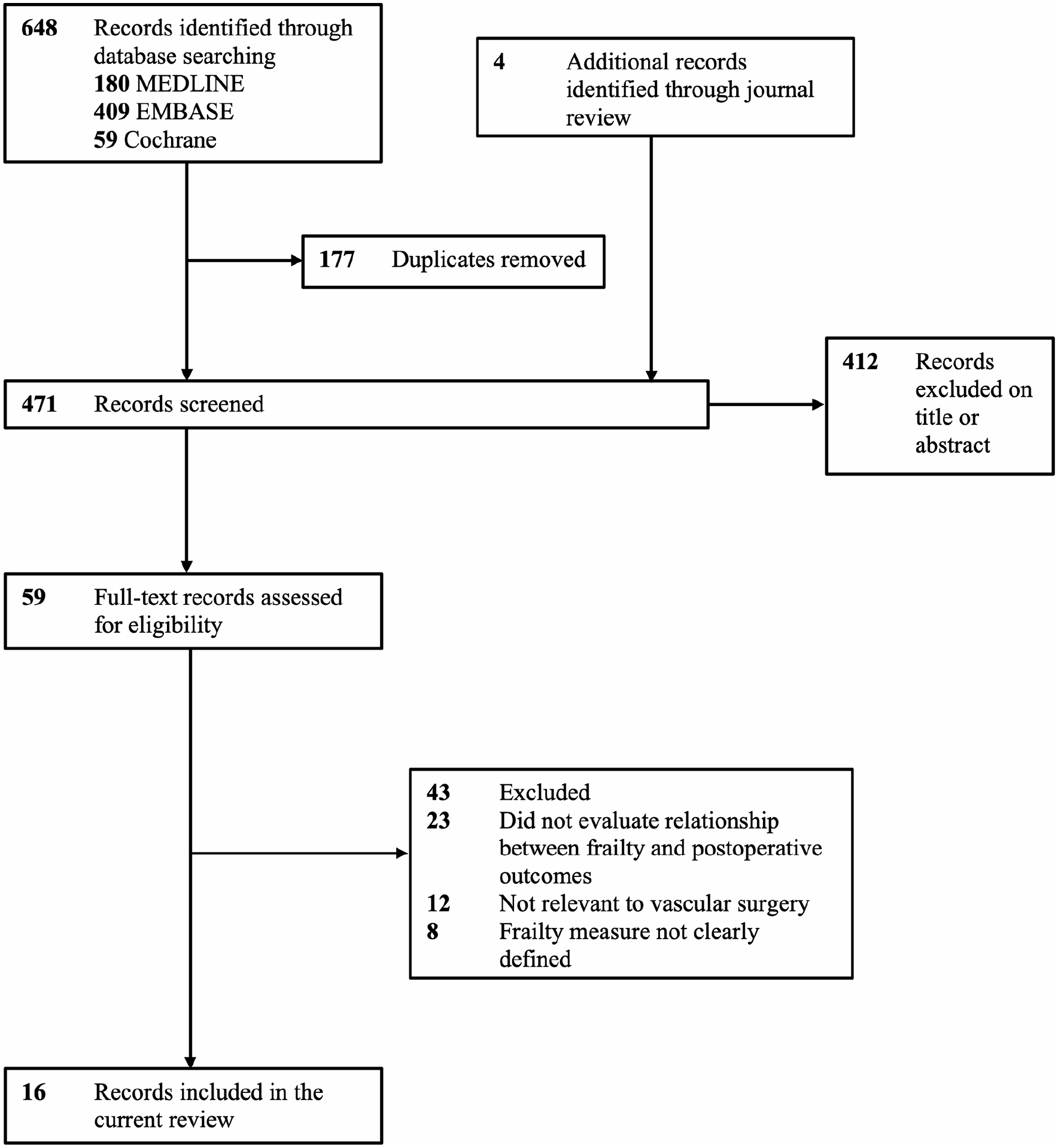
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram for study selection process.
The postoperative outcomes of the study population stratified according to frailty measures are presented in online Supplementary Table 1. The mean and median ages ranged from 56.1 to 76.3 years and from 70 to 77 years, respectively. Of the studies, 11, three, one, and one were conducted in the United States, United Kingdom, Canada, and Japan, respectively. Of the 16 studies, 10 were retrospective, and six were prospective. Although prospective studies have fewer potential sources of bias and allow for the determination of causation and association, 19 they are limited by their high cost and time. Hence, retrospective studies were also included in the systematic review. As retrospective studies are prone to selection bias, the NOS score was calculated for all 16 studies, and all the scores recorded were > 6. 25
Frailty measures
Eight different frailty measures were used across the 16 studies (online Supplementary Table 2) and divided into three groups: the deficit model, frailty phenotype model, and novel frailty markers. The deficit model included Addenbrooke’s Vascular Frailty Score (AVFS) in two studies,31,32 Clinical Frailty Scale (CFS) in two studies,38,39 Edmonton Frail Scale (EFS) in one study, 37 Frailty Index in one study, 36 modified Frailty Index (mFI) in six studies,26 –30,40 and Ruptured Aneurysm Frailty Score (RAFS) in one study. 32 The frailty phenotype model included grip strength measurement in one study. 41 The novel frailty markers included sarcopenia and the total psoas area in three studies.33 –35 The deficit models provided better predictions of postoperative adverse outcomes and the likelihood of recovery than the frailty phenotype model and novel frailty markers.
Frailty associated with adverse postoperative ouctomes following vascular and endovascular surgery are included online Supplementary Table 4.
Effect on 30-day, 90-day, 1-year, 2-year, and 5-year mortality
Fourteen studies assessed the effect of frailty on postoperative mortality.26,27,29 –36,38 –41 Frailty and 30-day mortality were significant related (p < 0.05).26,27,29,30,36,39 The risk of 30-day mortality increased from 1.4 to 13.7 times in frail patients compared with nonfrail patients (OR, 1.4–13.7), except for those who underwent endovascular abdominal aortic aneurysm repair (EVAR) (p = 0.36) and peripheral vascular surgeries (p = 0.49). 30
Frailty was significantly associated (p = 0.003) with 90-day mortality, showing a one-third decreased risk of 90-day mortality per 1000 mm2 increase in psoas area (OR, 0.33; 0.95 CI, 0.16–0.68) after elective open AAA repair. 33 Frailty was also significantly associated with increased risk of 1-year mortality.31,32,36 Frail patients had a 4.5-fold increased risk of 1-year mortality compared with nonfrail patients. 36
Intermediate and high CFS scores were significantly associated with approximately 1.6- and twofold increased risks of 2-year mortality, respectively. 38 Frail patients were found to have a fourfold increased risk of 5-year mortality compared with nonfrail patients. 34
Effect on 30-day morbidity
Postoperative 30-day morbidity was measured in seven studies.26,27,29,30,35 –37 Frailty and increased risk of postoperative morbidity showed a significant relationship.27,29,30 Frail patients had an approximately threefold increased risk of any of the listed morbidities (postoperative myocardial infarction (MI), stroke, progressive renal failure, and graft/prosthesis/flap failure) and a fourfold increased risk of postoperative MI. 26 Frail patients had an approximately twofold increased risk of gastrointestinal (GI), renal, and respiratory complications; a threefold increased risk of neurological complications; and an eightfold increased risk of cardiac complications. 35
Effect on 2-year amputation-free survival
The two-year amputation-free survival rate was significantly associated with frailty. Patients with intermediate and high CFS scores had 1.72- and 2.34-fold increased risks of amputation, respectively. 38
Effect on failure to rescue
The failure-to-rescue (FTR) rate, defined as the likelihood of in-hospital complications, was measured in two studies.27,39 In patients without complications, the overall FTR rate was 0.37% (EVAR: 0.29%; open abdominal aortic aneurysm repair (OAR): 0.67%). Patients with complications had an almost tenfold increase in FTR rate, with an overall FTR rate of 10.28% (EVAR: 9.62%; OAR: 11.05%). 27
Effect on length of stay
Length of stay (LOS) 42 was assessed in five studies.31,32,34,36,37 Frailty was found to have no significant association with postoperative LOS in four studies. However, the area under curve (AUC) of 0.74 (0.95 CI, 0.74–0.83) 31 indicated that AVFS is a good predictor of postoperative LOS.
Effect on nonhome discharge and postdischarge care
Nonhome discharge was assessed in six studies.28,31,35,36,38,40 Frail patients had approximately twofold,28,35 fourfold, 36 and 12-fold 38 increased risks of nonhome discharge compared with nonfrail patients. Moreover, the AUC of 0.78 (0.95 CI, 0.77–0.90) 31 indicated that AVFS is a good predictor of nonhome discharge.
Increased risk of postdischarge care was significantly associated with frailty (p < 0.05). The AUC of 0.70 and RAFS of 0.60 indicated that both are good predictors of postdischarge care requirements in frail patients. 32
Effect on other postoperative outcomes
Combined composite 30-day morbidity and mortality, 36 functional status at discharge, 37 bleeding, infection, spinal cord deficit, and access site complications 35 showed no significant association with frailty.
A summary of the strengths and weaknesses of frailty measures for assessing mortality, morbidity, amputation-free survival, failure to rescue, length of stay, non-home discharge, post-discharge care and other postoperative outcomes are included in online Supplementary Table 5.
Risk of bias
Two authors independently assessed the potential risk of bias using the NOS. All 16 studies had a similar risk of bias (online Supplementary Table 3) and a low risk of bias in the ‘selection’ domain. Most had a low risk of bias in ‘outcome’ and a moderate risk of bias in ‘comparability’ as they had only one controlled factor (e.g., age).
Discussion
Our systematic review confirms that preoperative frailty is associated with adverse postoperative outcomes following vascular and endovascular surgery. Significant associations were established between frailty and increased mortality at 30 days, 90 days, 1 year, and 5 years – with the strongest evidence for 30-day mortality.
Preoperative frailty was also associated with 30-day postoperative morbidity, nonhome discharge, adverse events, FTR, requiring care postdischarge, and amputation following limb ischaemia. Of the eight frailty measures included, the deficit model, mFI, was more efficient at evaluating frailty than novel frailty markers (e.g., sarcopenia and total psoas area). High mFI scores were associated with a greater risk of postoperative morbidity and mortality.30,43
Similar systematic reviews for cardiac 44 and general 20 surgery corroborate our findings; the latter also found the strongest association between frailty and 30-day mortality. 20
Clinical relevance
The significant associations found between frailty and postoperative outcomes provide three major benefits. First, risk-stratifying patients can potentially predict and prevent adverse postoperative outcomes. Clinicians can optimize the outcomes of patients identified as frail with preoperative conditioning, 45 nutrition and pharmacological therapy. 21 Hall et al. found that a facility-wide Frailty Screening Initiative for elective surgical patients was equally effective and feasible in reducing mortality among frail patients; the 30-day mortality rate decreased from 1.6% to 0.7% after implementation. In frail patients, the mortality rate dropped from 12.2% to 3.8%, while mortality decreased from 1.2% to 0.3% in nonfrail patients. In addition, rescue therapies, including ventilator management and immediate dialysis after surgery, were better maintained. 46 Preoperative frailty assessments may therefore benefit patients by optimizing their medical and physical functions pre- and postsurgery, thereby reducing the risk of postoperative outcomes.
Second, our results emphasize the importance of using frailty instead of age, which is more valid and accurate in predicting postoperative outcomes in vascular surgery. 21 As frailty is multifactorial, additional variables (e.g., female sex, low body mass index (BMI), chronic respiratory and cerebrovascular disease) have also been found to be associated with frailty, 47 suggesting that additional variables can be added to the age variable for future risk assessments.
Finally, frailty measurements can help clinicians and patients make informed decisions. Medical treatments are often not generalizable to elderly patients due to age-related physiological decline and potential comorbidity and exacerbation risk, which alter the risk-to-benefit analysis. Our systematic review indicates that frailty assessments can assist physicians with more accurate predictions and risk-to-benefit analysis and assist patients to make more informed decisions about their treatment (e.g., understanding they are at higher risk of mortality if frail). 21
Study limitations
Our study has several limitations. First, most frailty models have been developed in White populations.5,6 The prevalence of frailty differs among racial/ethnic groups (e.g., higher in Southern Europe 48 and older Mexican Americans 49 ), suggesting that different cut-off points may be required to define frailty categories for different ethnicities. Second, no clear consensus on the definition of frailty nor on the ‘gold-standard’ tool for measuring frailty exists. Third, our literature search was limited to English, potentially missing relevant publications in other languages. Finally, a meta-analysis was not conducted due to the heterogeneity of frailty measures, cut-offs, and reported outcomes; hence, the most effective frailty measurement for predicting postoperative outcomes could not be evaluated.
Conclusions
Frailty scores are clinically useful for predicting and guiding the pre-, peri-, and postoperative management of vascular and endovascular surgery patients. Identifying frail patients before surgery facilitates the prediction of operative benefits and risks and helps personalize their peri- and postoperative treatment plans to reduce the risk of postoperative complications and mortality. The optimal goal is to maintain patients’ functional status, mobility, and nutritional needs during transitional care.
Further research should be undertaken to determine which frailty measures are best suited to different surgical settings and the association between individual frailty components and postoperative outcomes. This would enable interventions to be better targeted, with the overall aim of improving clinical outcomes while using surgical resources efficiently.
Although frailty measures can act as effective prediction tools, we must understand that medical care should ultimately be founded upon individual patients’ diseases, goals, expectations, and preferences; any operational decisions should be dependent on the patient’s autonomy and informed decision-making.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
*
These authors contributed equally to this work.
