Abstract
Venous hypertension due to anatomical venous insufficiency (superficial or deep venous valvular reflux and venous obstruction) and/or functional venous insufficiency (calf dysfunction, dependent edema, lymphatic dysfunction, elevated central venous pressures, etc.) leads to chronic venous insufficiency (CVI) and its sequelae. The most severe clinical manifestation of CVI is venous leg ulcer (VLU), which leads to substantial morbidity and disability. VLUs can present in both typical and atypical forms. However, several other types of leg ulcers can mimic VLUs. Therefore, vascular and wound care specialists must recognize the variability in VLU presentation and understand the differential diagnoses to deliver an accurate diagnosis and optimal care of atypical VLUs and conditions that resemble VLUs. Herein, we review and discuss the broad differential diagnosis of VLUs, supported by illustrative examples.
Introduction
Venous leg ulcers (VLUs) account for approximately 70% of all chronic lower-extremity ulcers. With recurrence rates reported as high as 78%, they result in significant healthcare expenditure and socioeconomic burden.1,2 Wound/vascular specialists often encounter a subset of leg ulcers diagnosed as VLUs that do not respond to the traditional therapies directed at venous hypertension management. These ulcers result from alternate etiologies. 3 This review details the diverse morphologic appearances and the differential diagnosis of VLUs through a pictorial overview. Clinical presentation, key differentiating features, illustrative images, and essentials in managing these differentials will be elaborated with an emphasis on identifying their etiologies to facilitate an appropriate treatment algorithm. This review serves as an atlas for varying presentations of VLUs and their differential diagnosis and general principles of treatment (Table 1).
Venous leg ulcer differential diagnosis.
Venous ulcer presentations
Venous leg ulcers (VLUs)
The standard definition of a VLU is ‘an open skin lesion of the leg or foot that occurs in an area affected by venous hypertension’. 4 Venous hypertension can result from an anatomical venous disease in the superficial or deep veins (venous reflux and obstruction) or functional venous hypertension (calf dysfunction, dependent edema, lymphatic dysfunction, elevated central venous pressures due to heart failure, obstructive sleep apnea, and obesity). The prevalence of VLUs in populations older than 60 years is estimated to be 1–3%, 5 with an annual cost of at least $10.73 billion in the US. 2 VLUs result in impaired quality of life and significant social isolation. 6
Risk factors
Risk factors for VLUs include a history of chronic venous insufficiency (CVI), a history of deep vein thrombosis (DVT), immobility, obesity, advanced age, 7 pregnancy, trauma, and occupations requiring prolonged standing. 8
Pathogenesis and ulcer characteristics
Prolonged venous hypertension transmits pressure into the microcirculation, leading to venous microangiopathy marked by reduced shear stress, a key regulator to maintaining an active endothelium. Changes in vascular tone lead to disruption in the mechanotransduction, which may affect vascular permeability and upregulation and expression of proinflammatory cytokines, enzymes, and adhesion molecules. Inflammatory reactions within the blood vessel wall lead to structural degradation as well as the disruption of redox balance by red blood cell infiltration. 9
This process also triggers the release of inflammatory mediators and proteolytic enzymes, which damage venules and micro-lymphatics. The activation and trapping of leukocytes and platelets further release inflammatory mediators such as Tumor necrosis factor (TNF)-alpha and transforming growth factor (TGF)-beta, leading to stasis dermatitis, hyperpigmentation, lipodermatosclerosis, and eventual ulceration. 10
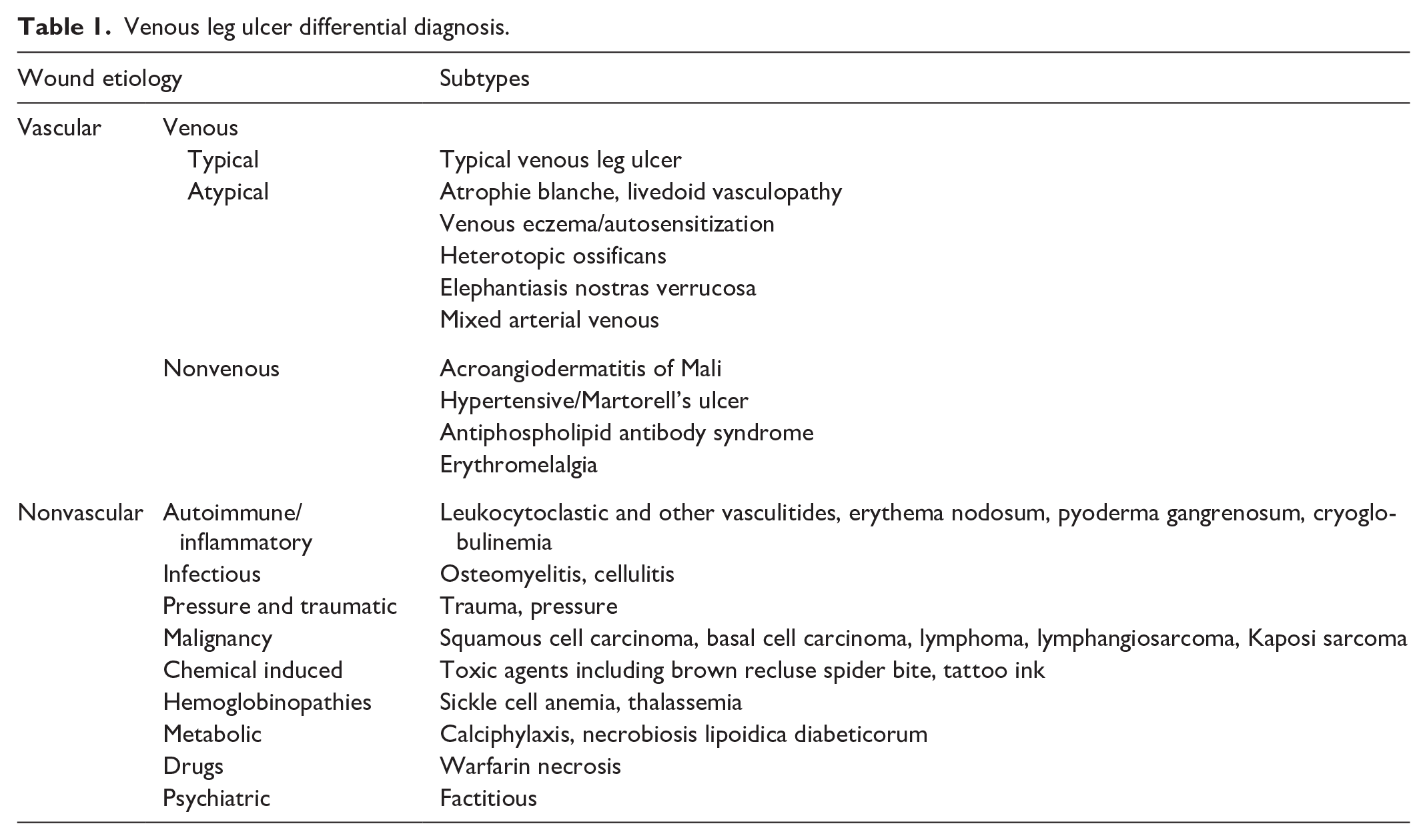
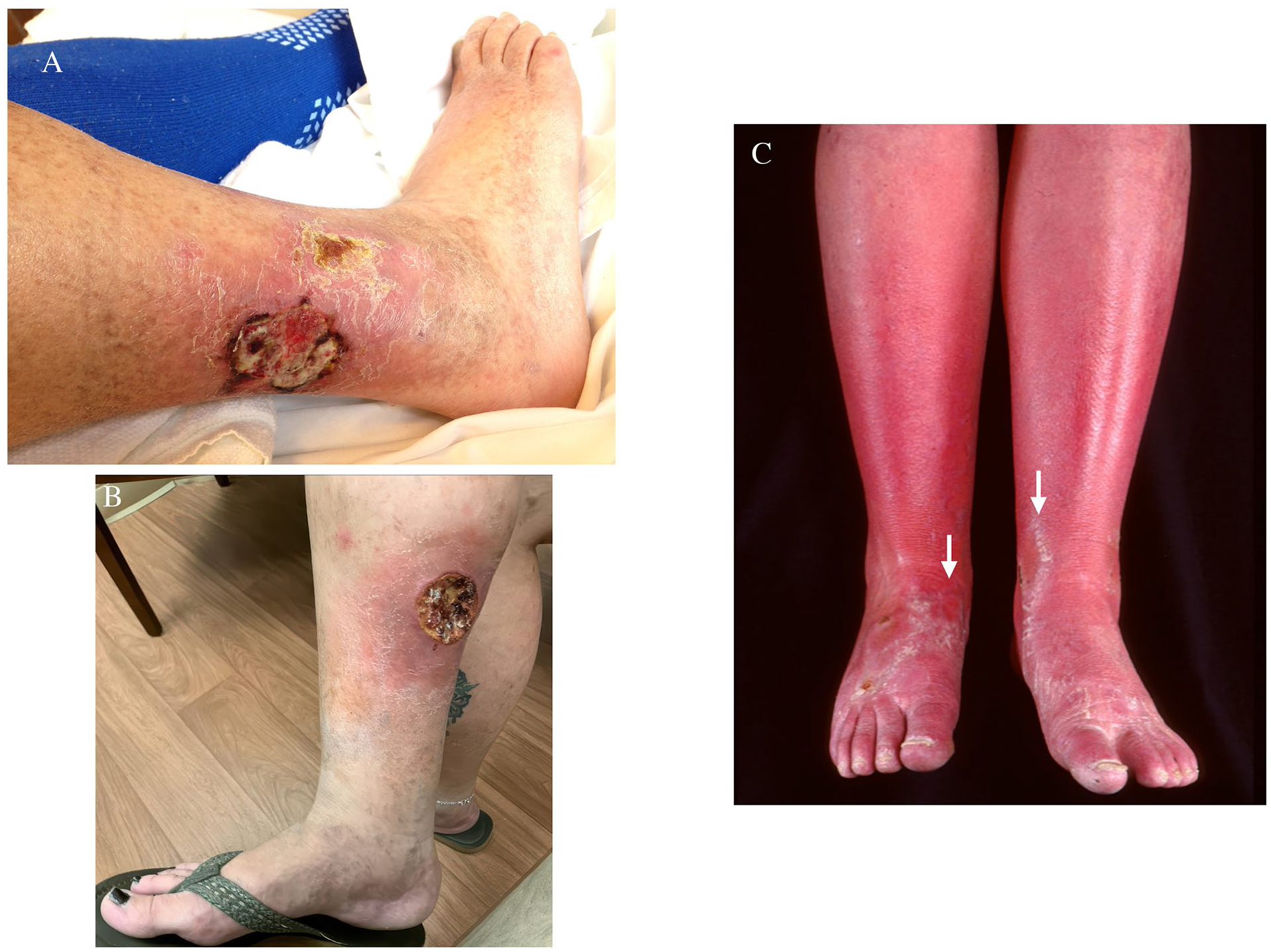
VLUs have been classically described as painless. However, there can be significant pain in VLUs due to the underlying inflammation, constant venous hypertension, and swelling. 10 On exam, ulcer edges are well demarcated, with surrounding skin changes, with or without edema. Cutaneous manifestations commonly associated with VLUs often reflect underlying CVI. These include varicose veins, hemosiderin deposits, lipodermatosclerosis, erythema due to chronic infection, atrophie blanche, corona phlebectatica, and extensive scarring. VLUs are generally located near the peri-malleolar ‘gaiter area’ of the ankle, with the majority occurring medially (Figure 1A). Infrainguinal venous pathologies such as reflux, postthrombotic changes, and obstruction can be detected on duplex ultrasound. This must be done in every patient who has suspected VLU. However, axial imaging may be necessary to assess more proximal ilio-caval pathologies. Physiologic examinations such as air plethysmography can assist in the diagnosis if available. 11 It is also important to evaluate for functional venous hypertension, which may not always be picked up on vascular imaging.
Manifestations of typical and atypical venous leg ulcers.
A typical VLU diagnosis is based on a physical exam and vascular imaging. A biopsy is not routinely necessary, but when done, it displays thickened basement membranes with widened tight junctions and dense fibrosis. Pericapillary spaces are filled with collagen and its precursors, macrophages, and lymphocytes. 10
Treatment
Layered compression therapy, especially multilayered compression, has long been the cornerstone for the management of CVI and VLU.12 –14 The Early Venous Reflux Ablation (EVRA) trial, a randomized control trial, proved that early saphenous reflux treatment can lead to improved ulcer healing, decreased recurrence, and more ulcer-free days. 15 The important takeaway from EVRA is that a large number of patients with ulcer healing also had sclerotherapy, which may indicate that treatment of the periulcer venous network could have significant importance in overall healing. Addressing proximal vein obstruction is also an important component of VLU management in patients who may have external vein compression or postthrombotic syndrome.4,16 However, there is no direct level-1 scientific evidence of improved VLU healing or decreased recurrence with relief of venous obstruction. Comprehensive wound care, encompassing debridement, the application of skin substitutes, and infection management, plays a critical role in the treatment of VLUs4,17 and other wound types described below. However, these topics fall outside the focus of this manuscript.
Atypical presentations of VLUs
Atypical presentations of VLUs are relatively common in clinical practice 3 and may include conditions such as atrophie blanche, venous eczema, heterotopic ossification, elephantiasis nostras verrucosa (ENV), and mixed arterial-venous ulcers. Clinicians should maintain a heightened awareness of these atypical manifestations to facilitate accurate diagnosis. Risk factors for CVI and diagnostic imaging, including venous reflux or obstruction assessments, can play a critical role in confirming these atypical presentations.
Atrophie blanche ulceration (ABU)
ABU is a complex entity associated with venous insufficiency and other underlying conditions that cause capillary occlusions. 18 Extreme pain is a hallmark of these shallow ulcers, often challenging to cure, affecting activities of daily living and sleep, and causing significant morbidity.
Risk factors/associations
ABUs are more common in women than men, with a male-to-female ratio of 1:4. Although they can develop at any age, they most often occur in individuals aged 30–60 years and are primarily associated with CVI. 19 However, they can be associated with livedoid vasculopathy, thrombophilia, and other vasculitides. Atrophie blanche skin changes can precede ulceration. 20
Pathogenesis and ulcer characteristics
Though not definitively established, the accumulation of fibrin, the proliferation of endothelial cells, hyaline degeneration, the aggregation of white blood cells, and the formation of microthrombi within the subpapillary blood vessels that occur in vasculitis and other microcirculatory disorders lead to the obstruction of blood flow within the vessels. This causes localized ischemia with inflammatory infiltrate, causing skin necrosis and breakdown. When this process occurs repetitively, it leads to severe scarring of the skin along with frequent skin breakdown. 20
Atrophie blanche skin changes are characterized by ivory-colored, star- or polyangular-shaped, smooth scars located in the gaiter area of the leg and the feet (Figures 1B and 1C). Surrounding these scars, areas of telangiectasias and hyperpigmentation may be noted. The skin changes can both precede and persist after the healing of an ABU. ABUs are shallow ulcers with pain that is out of proportion to the visual appearance. They often present as fern-shaped ulcers that may be difficult to visualize due to the overlying scab. The pain associated with ABUs significantly improves with compression. Healing can be challenging to determine visually but is suggested by the resolution of tenderness.
Biopsy should be avoided as this may delay healing and further increase pain due to the resultant deeper ABU. However, a histological analysis may rarely be required to distinguish ABU from cutaneous small-vessel vasculitis.
Treatment
Key elements of management include addressing underlying conditions, implementing appropriate compression therapy, pain control, and tailoring treatment to the patient’s individual needs. Additional important components of care involve smoking cessation, considering pentoxifylline, and maintaining meticulous wound care practices. 21 Rivaroxaban and enoxaparin treatment could be considered in patients with ABU and livedoid vasculopathy. 22 When livedoid vasculopathy is suspected (Figure 1D), a workup must be undertaken to assess for underlying autoimmune or connective tissue disorders, thrombophilia, or malignancy. Depending on the underlying cause, immunosuppressive therapies such as cyclosporine, sulfasalazine, and dapsone can be used. Hyperbaric oxygen therapy and prostaglandin use have been reported for treating recalcitrant ABUs associated with livedoid vasculopathy. 23
Venous eczema
Frequently, CVI can manifest as pronounced inflammation and persistent pruritic eczematous skin lesions. This condition is commonly referred to as venous eczema, gravitational eczema, stasis eczema, or venous dermatitis. Owing to the intense itching and subsequent scratching, it is not uncommon for affected individuals to require hospitalization, either due to secondary cellulitis or misdiagnosed pseudocellulitis. The exact prevalence of venous eczema remains uncertain due to the overlapping occurrence of irritant contact dermatitis and allergic contact dermatitis with VLUs. 24
Risk factors/associations
Most patients have a history of concomitant systemic allergies or contact dermatitis. 25
Pathogenesis and ulcer characteristics
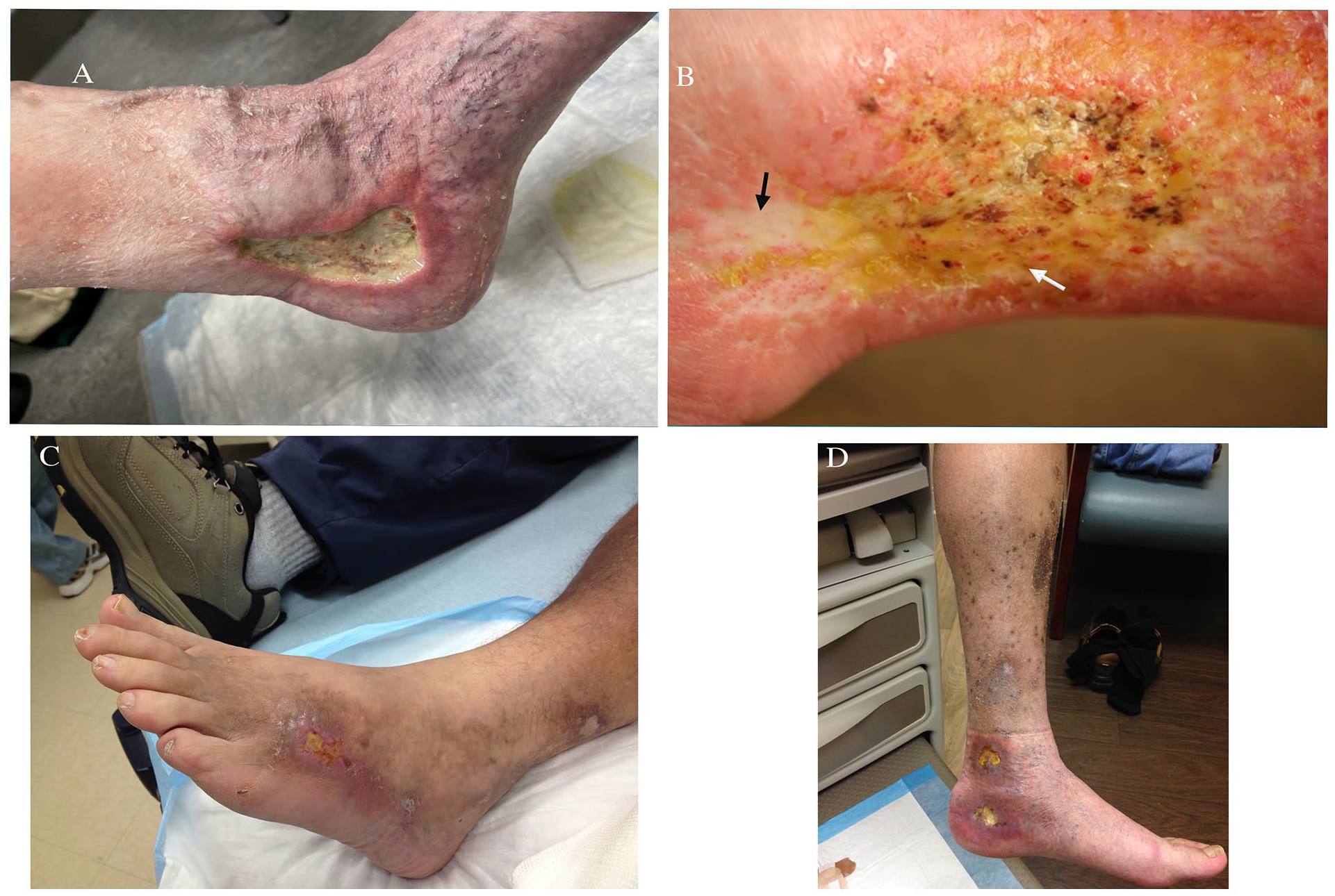
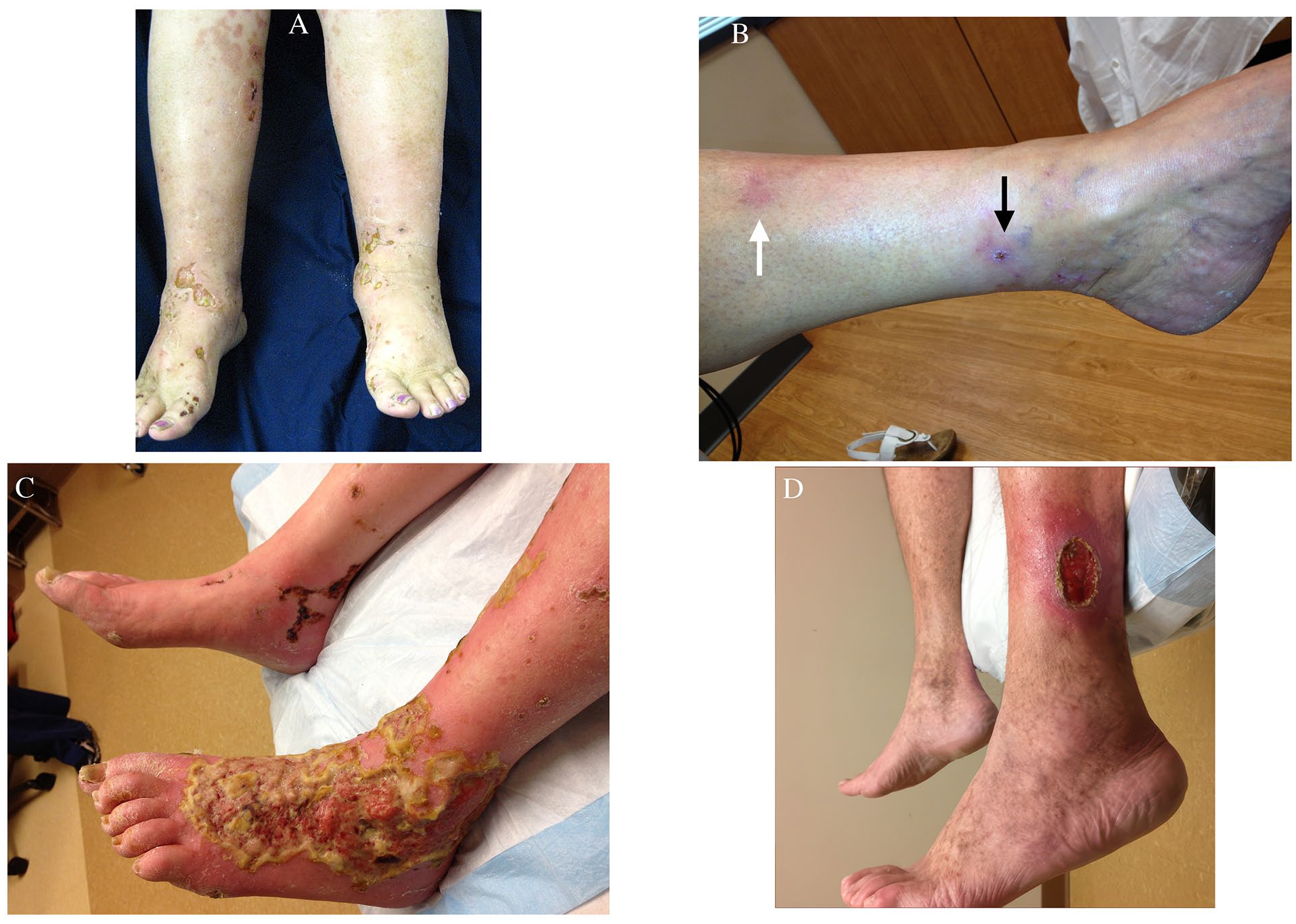
The exact pathophysiology of venous eczema remains unclear. Some theories suggest that it falls into the category of endogenous eczemas, which encompasses atopic dermatitis, discoid eczema, seborrheic dermatitis, eczema affecting the hands and feet, and asteatotic eczema. 26 Ulcers linked to venous eczema typically present as shallow wounds with partial-thickness involvement, characterized by serous exudation (weeping or oozing) and crusting. Ulcers start as small, delicate papules and vesicles that eventually merge, forming superficial exudative ulcers. As the exudate dries, it results in crusting (Figure 2A). Severe venous eczema can lead to autosensitization or Id reaction characterized by a diffuse spread of pruritic papulovesicular eruptions of the upper extremities and trunk (Figure 2B). Prolonged itching and scratching can lead to neurodermatitis, and any resulting scars have the potential to evolve into keloids (Figure 2C).
Additional manifestations of atypical venous leg ulcers.
A biopsy is unnecessary to make a clinical diagnosis of venous eczema, but histopathology shows leukocyte trapping, capillary tufting, and fibrin cuffs around the capillaries, along with hyperkeratosis of the epidermal layers. 24
Treatment
The primary therapeutic objective involves mitigating inflammation 26 via combining potent topical corticosteroids and emollients, transitioning to milder formulations as manifestations improve. Urea cream may be beneficial for hyperkeratotic areas. Compression therapy should be tailored to allow daily application of topicals, with medium-stretch or Velcro wraps often preferred. Caution is advised with spandex-containing garments due to potential contact dermatitis. Once ulcers heal and the eczema resolves, hypoallergenic compression materials like cotton or silver are recommended for ongoing management.
Heterotopic ossificans
Heterotopic ossificans is characterized by the progressive formation of lamellar bone in locations outside the skeletal system. 27 It has been observed in individuals with CVI, presenting as subcutaneous calcification and phleboliths within the vein’s lumen. 28 It has also been referred to as panniculitis with dystrophic calcification, panniculitis ossificans, or Heinz Lippmann disease, named after the authors who described it. 29
Risk factors/associations
Risk factors for heterotopic ossificans in CVI include a long-standing history of severe venous hypertension, diabetes, older age, and obesity. 30
Pathogenesis and ulcer characteristics
The hypotheses for heterotopic ossification pathogenesis include fat necrosis leading to calcification, fat metaplasia, trauma, hematoma formation, hormonal influences, and chronic inflammation30,31; however, this is still unclear.
These ulcers are often characterized by significant pain and are typically full thickness (Figure 2D). Patients may describe the sensation of palpating and extruding calcified or stone-like material from the wound bed. These ulcers may be covered with an eschar, with the surrounding skin displaying areas of stasis dermatitis or atrophie blanche. Calciphylaxis wounds display dusky-colored skin around the edges, making it hard to determine its demarcation. Wounds are full thickness and often develop fat necrosis. Radiographic imaging typically reveals extensive calcification (Figure 2E).
Histopathology findings include the presence of osseous metaplasia, inflammatory cells, and dispersed hemosiderin-laden macrophages. 29 As with most types of VLUs, diagnosis is clinical, and a biopsy is unnecessary.
Treatment
Treating involves removing the calcified tissue through surgical resection. This approach differs significantly from the typical local wound care and compression therapy employed for VLU. Despite diligent management, these ulcers may require an extended healing period of several months. 28 Effective resolution requires a concerted effort from a team of vascular specialists and plastic surgeons.
Elephantiasis nostras verrucosa (ENV)
ENV is a disfiguring form of progressive phlebolymphedema (characterized by a combination of chronic CVI and lymphatic dysfunction) due to severe edema and fibrosis with a high degree of morbidity.
Risk factors/associations
ENV is typically observed in cases of stage III lymphedema. However, it is also commonly associated with CVI and morbid obesity. Most patients with ENV have phlebolymphedema. 32
Pathogenesis and ulcer characteristics
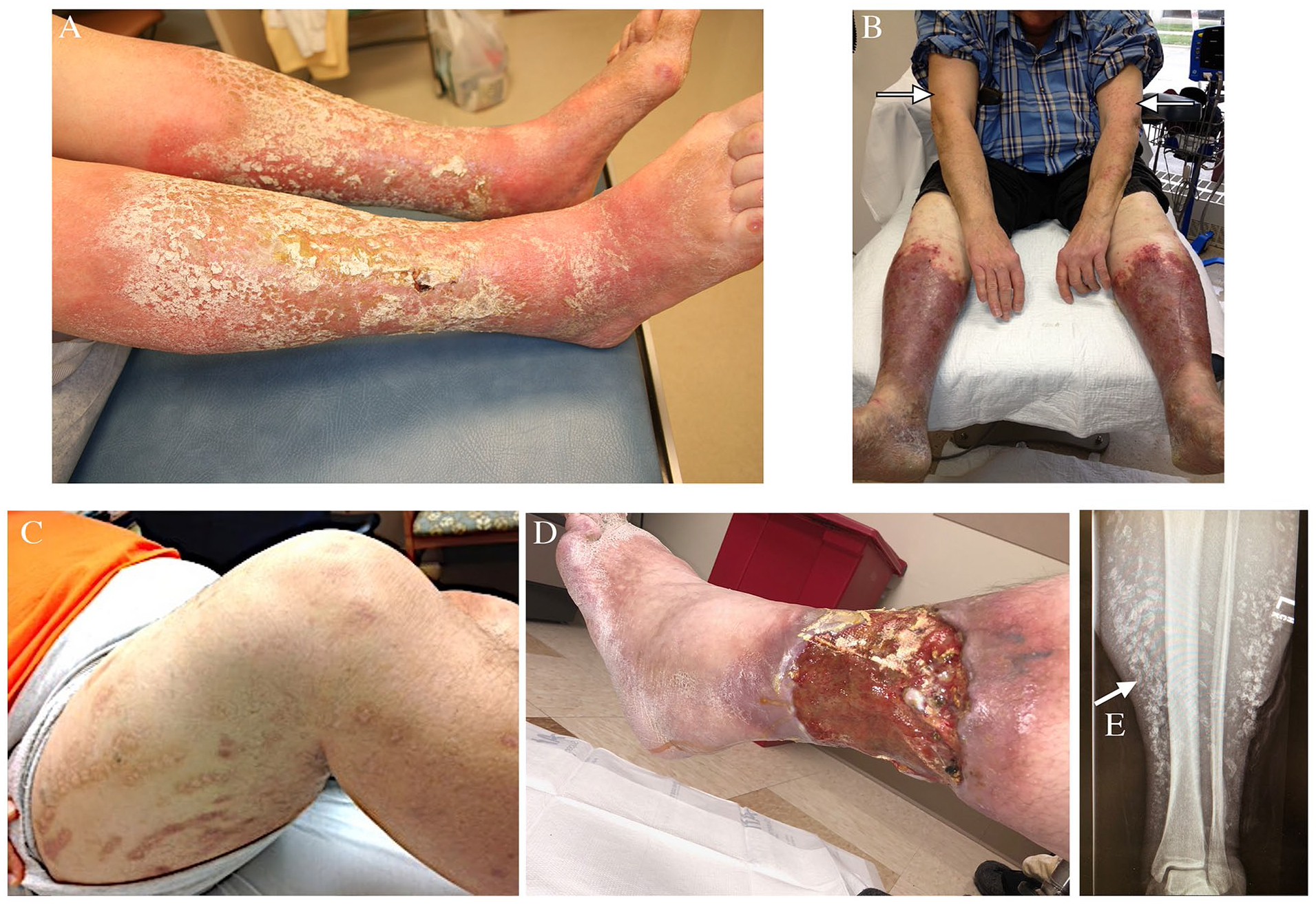
Lymphostasis and trapping of protein-rich fluid in the interstitial space from venolymphatic reflux or obstruction causes chronic inflammation, fibrosis, and eventual ulceration. 33 Profound hyperkeratosis, dermal fibrosis, lichenification (the development of verrucous or papillomatous alterations), and nonpitting edema are hallmark clinical features. These manifest in a distinctive cobblestone-like appearance (Figure 3A). ENV-related ulcers tend to be expansive and lack well-defined borders, often intermingling with verrucous changes. They frequently exhibit significant drainage. 32
Additional manifestations of atypical venous leg ulcers.
A biopsy is unnecessary unless carcinomatous transformation is suspected. Pathologic examination is nonspecific and may demonstrate extremely dilated lymphatic spaces, chronic inflammation, fibroblast proliferation, and pseudoepitheliomatous hyperplasia.
Treatment
General treatment principles include managing venous insufficiency and lymphedema through skin hygiene, limb elevation, manual lymphatic drainage, complex decongestive therapy, and compression therapy. Sequential lymphatic pumping, weight loss, infection control, and treating concomitant elevated central venous pressure are also important. 33
Mixed arterial venous ulcers
The presence of concomitant arterial and venous disease is thought to be nearly 10–20% of all lower-extremity ulcerations. 34 Concomitant arterial ischemia poses a specific challenge with compression therapy. Noninvasive testing is crucial for evaluating arterial disease in patients with peripheral artery disease risk factors or abnormalities on peripheral pulse examination. Clinicians must note that there is variable presentation of ischemic ulcers and mimics of ischemic ulcers as well. 35
Risk factors/associations
Mixed arterial-venous ulcers should be suspected in those with advanced age, diabetes, smokers, and those with risk factors for the development of peripheral artery disease.
Pathogenesis and ulcer characteristics
The pathogenesis is secondary to occlusive atherosclerotic disease, in addition to venous hypertension.
These ulcers may initially present as VLUs but are usually associated with extreme pain and tenderness. Necrosis or eschar formation may occur, leading to a rapid deterioration in both size and depth compared to uncomplicated VLUs (Figure 3B). Pulses are weaker, and distal hair loss can be noted. A biopsy is unnecessary; the diagnosis rests on abnormal noninvasive vascular testing and physical examination. 36
Treatment
Beyond smoking cessation and optimizing medical management for peripheral artery disease, including glycemic control, blood pressure control, and management of dyslipidemia, consideration of arterial revascularization is essential even if the patient does not have lifestyle-limiting claudication for two reasons. The first is to be able to apply effective compression therapy without concern for resting arterial flow, and the second is to increase outflow to promote blood flow to the wound area. Although comprehensive studies are lacking, limited retrospective analyses have indicated enhanced wound healing in patients who undergo arterial revascularization alongside modified compression therapy.37 –40
Other vascular etiologies
Venous ulcers are the most frequent cause of leg ulceration; however, other vascular conditions can also lead to leg ulcers. These include acroangiodermatitis of Mali, livedoid vasculopathy, Martorell’s ulcers, ulcers associated with antiphospholipid syndrome, and erythromelalgia.
Acroangiodermatitis of Mali (AADM)
AADM, often labeled pseudo-Kaposi sarcoma (KS), is a rare angioproliferative disease that presents as purple papules and is mostly seen as case reports. It mimics VLUs in that it presents with ulcers with surrounding hyperpigmentation. However, the hyperpigmentation is violaceous and consists of discrete proximal and distal borders, unlike CVI, in the authors’ experience. AADM presents with skin lesions that are similar to KS but histologically different and usually due to an underlying arterial venous fistula.41,42 This can progress ulcerations in the skin lesions.
Risk factors/associations
AADM is associated with CVI, men, and arteriovenous fistula. There are reports of association with the Prothrombin G20210A mutation, activated protein C resistance, and paralysis of extremities. AADM is also seen in Stewart–Bluefarb syndrome 43 alongside arteriovenous malformations (AVMs) or in Klippel–Trenaunay syndrome with osteohypertrophy.
Pathogenesis and ulcer characteristics
The precise mechanism of AADM is not fully understood, but it is believed to involve chronic venous hypertension, prolonged hypoxia, and subsequent uncontrolled vascular and fibroblast proliferation along with neovascularization. 44 Lesions of AADM are numerous, well-demarcated nodules, papules, or plaques with a dark purplish discoloration (Figure 3C). They are primarily found on the dorsal aspect of the foot and the gaiter surface of the legs. These lesions are disfiguring but painless. If related to CVI, the lesions are likely bilateral. However, if due to underlying unilateral AVMs, they can be unilateral.45,46
Diagnosis is mostly clinical, but a biopsy demonstrates endothelial proliferation and de novo vasculature encasing dermal pericytes along with fibroblastic changes and extravasated RBCs that can resemble KS. However, immunohistochemical staining for CD34 is negative, as would a polymerase chain reaction (PCR) for human herpes virus (HHV)-8. 44
Treatment
Treatment is aimed at the underlying condition. In addition, topical corticosteroids produce a favorable result. Systemic therapy with erythromycin or dapsone has been known to be effective. 43
Martorell’s ulcer
Martorell’s ulcer is a rare type of ischemic ulcer that often mimics venous ulcers. It is characterized by extreme ischemic pain resulting from severe hypertension. 47 The condition is named after Fernando Martorell, who provided a thorough description and characterization of these ulcers. 48
Risk factors/associations
Risk factors include middle age and above, women, and poorly controlled hypertension.
Pathogenesis and ulcer characteristics
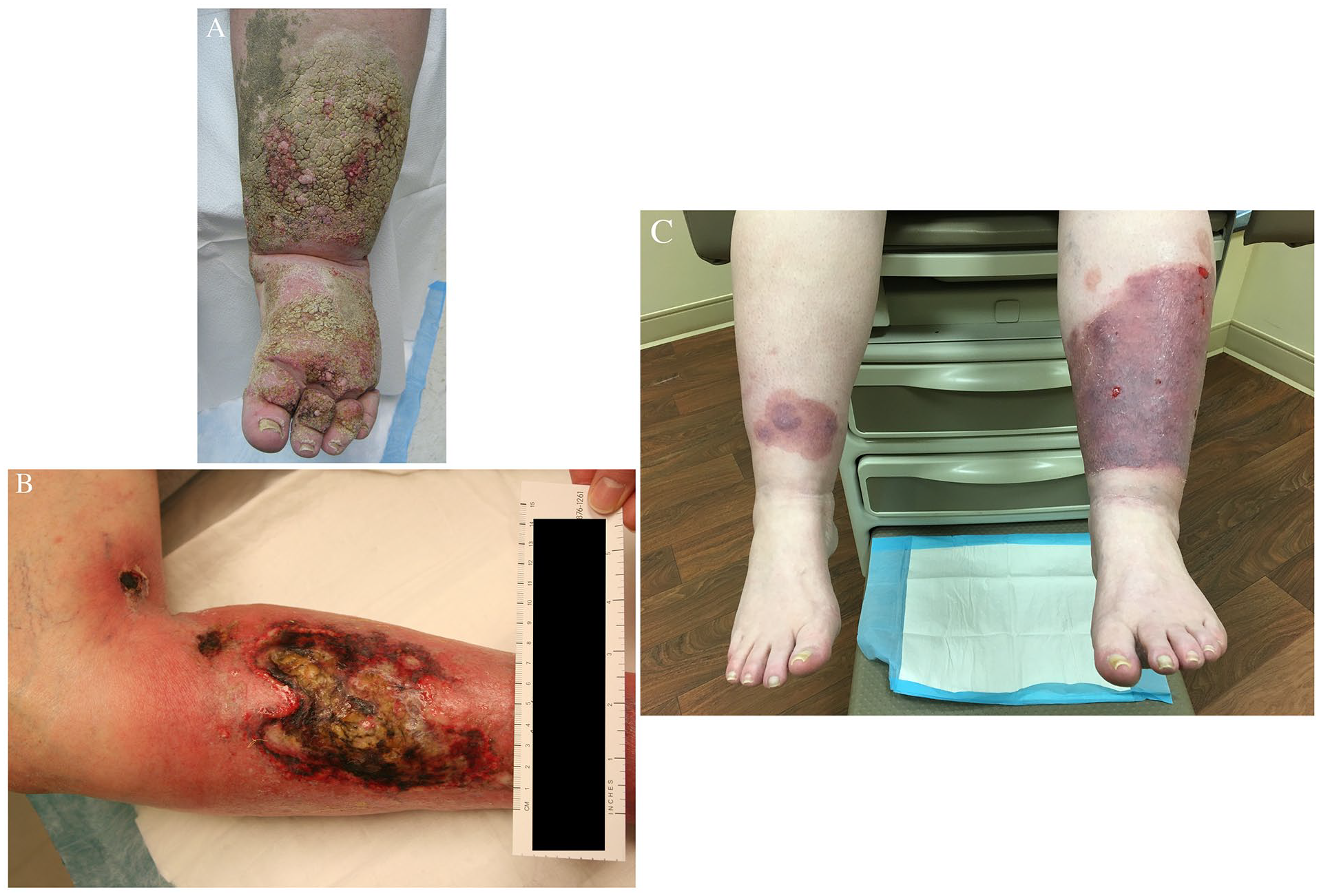
The pathogenesis includes uncontrolled hypertension, leading to arteriolosclerosis, and hyperplasia and thickening of lamina propria. These factors lead to endothelial remodeling and obstruction of blood flow. The severe ischemic pain is out of proportion to the size of the ulcer. Classically, lesions are located on the lateral and distal calf but have been noted on feet or toes (Figure 4A). Martorell’s ulcer generally starts as a painful, erythematous blister after a traumatic event and evolves into a superficial ulceration with a necrotic base and violaceous edge. 49 Histological inspection reveals inflammation with thickened lamina propria with arteriosclerosis, but diagnosis does not require a biopsy. 47
Nonvenous vascular leg ulcers.
Treatment
Treatment requires blood pressure control, often with a vasodilator such as calcium channel blockers. 50 Local wound care and analgesia are crucial; surgical debridement with skin grafting is sometimes necessary.
Antiphospholipid syndrome (APS)
APS is a hypercoagulable disorder that develops both arterial and venous blood clots. Additionally, APS can cause microvascular inflammation, thickening, or vegetation on cardiac valves, and complications during pregnancy. 51 The three primary antiphospholipid antibodies involved are lupus anticoagulant, anticardiolipin, and anti-β2-glycoprotein. 52 With triple positive APS, the risk of blood clots increases. APLA can also result in wounds that resemble venous ulcers.
Risk factors/associations
Primary or secondary APS. The most common secondary cause is systemic lupus erythematosus. Additional secondary causes include other autoimmune diseases, malignancy, infections, and medications.
Pathogenesis and ulcer characteristics
Theories for APS leading to ulcers include molecular dyssynchrony, endothelial disruption, coagulation cascade activation, and upregulation of procoagulants leading to skin ischemia and breakdown. In up to 40% of patients with APS, the initial presentations are dermatological. These can include painful ulcers with necrosis, acrocyanosis, livedo reticularis, superficial thrombophlebitis, or subungual hemorrhage. Notably, these ulcers can vary in size and location but are typically on the legs with irregular, raised violaceous borders and are often misdiagnosed as pyoderma gangrenosum ulcers (Figure 4B). Biopsy is not necessary, but can show diffuse, intravascular occlusion in dermal vessels – though up to a third of biopsies show no sequelae of coagulopathy.53,54
Treatment
Diagnosis of APS in patients who present with ulcerations suggestive of this diagnosis is paramount. Once diagnosed, anticoagulants must be instituted. In patients with APS who are triple positive, anticoagulation with warfarin is preferred, given its larger coverage within the coagulation cascade. Additionally, antiplatelet therapy and statins should be considered. Corticosteroids, plasma exchange, and immunosuppressive therapy have been used in patients with catastrophic APS. 55
Erythromelalgia
Erythromelalgia is characterized by a triad of acral erythema, localized warmth, and a burning sensation. This condition generally affects both lower extremities, although secondary erythromelalgia can present with unilateral symptoms. Though rare, lower-extremity ulcers can occur in patients with chronic erythromelalgia and could be confused with VLUs due to erythro-cyanosis and the location of the ulcers. The pathogenesis is not well understood, but primary erythromelalgia can be associated with a
Risk factors/associations
This disease can be associated with myeloproliferative disorders such as polycythemia vera or essential thrombocytosis, peripheral neuropathy, gout, and connective tissue disorders. 58 Most patients are middle-aged, Caucasian, and women, half active or past smokers. 56 The discoloration usually presents as episodic striking, tender, and hot erythema, often with swelling. When quiescent, the limbs manifest acrocyanosis. Patients frequently describe the pain as intense burning, tingling, or itching. Bathing, exercise, limb dependency, ambient heat, and sleeping under covers can worsen the pain, whereas exposure to cold and leg elevation can provide some relief. Wounds associated with erythromelalgia usually have partial thickness and are seen in the feet or ankles (Figure 4C). These may be associated with maceration from prolonged cold water immersion by the patients. The key to diagnosis is the surrounding skin manifestation and the associated history. 57
Histologic inspection demonstrates diffuse capillary proliferation, perivascular swelling, inflammation, and rare lymphocytic infiltrates. However, due to the variability, a skin biopsy does not aid diagnosis. 59
Treatment
Therapy involves mitigating the underlying disease and avoiding known triggers. High-dose aspirin (325–500 mg) is the first-line treatment with variable results and is most effective in the setting of myeloproliferative syndrome. Nonsteroidal antiinflammatory agents, gabapentin, sympathectomy, and topical lidocaine use have been reported. 57 Amitriptyline and ketamine-based topical compounded creams have also been reported to result in modest improvement in symptoms. 60 Ulcers associated with maceration can eventuate from protracted immersion of the feet in cold water. Patient education is critical to avoid erythromelalgia triggers and limit cool water exposure.
Nonvascular etiologies
Once vascular etiologies are ruled out, based on the negative noninvasive testing and clinical presentation described above, one must consider nonvascular etiologies.
Autoimmune/inflammatory
Given the high prevalence of autoimmune diseases, clinicians need to be aware of the associated complications like leg ulcers. Examples include leukocytoclastic vasculitis (Figure 5A), erythema nodosum (Figure 5B), pyoderma gangrenosum (Figure 5C), and cryoglobulinemia (Figure 5D) (Table 2). Vasculitis can cause skin rashes and ulcers, which often appear in clusters or as large lesions with smaller surrounding ulcers. 57 Accurate diagnosis of vasculitis typically requires a biopsy.58 –62 Erythema nodosum (EN), a common form of panniculitis in patients with underlying conditions, is marked by sudden-onset erythematous nodules and can ulcerate.
Autoimmune manifestitations of venous leg ulcers.
Autoimmune/inflammatory ulcers.

NSAIDs, nonsteroidal antiinflammatory drugs.
Pyoderma gangrenosum is an idiopathic, inflammatory skin lesion with severe morbidity. The hallmark of this disease is a relapsing inflammatory ulcer with a poorly defined, erythematous-violaceous raised border.61,62 Almost two-thirds of cases are associated with a systemic disease. 63 Although the exact mechanism is unknown, its pathogenesis is believed to be a neutrophilic dermatosis without infection. Pyoderma gangrenosum ulcers can be quite challenging to diagnose and require a high degree of suspicion. The PARACELSUS score is a simple, effective, diagnostic tool for identification 64 (Table 3). Treatment is immunosuppression-based and requires multispecialty collaboration. Debridement may worsen pyoderma gangrenosum due to associated pathergy.
PARACELSUS score for pyoderma gangrenosum. 64

Score of ≥ 10: pyoderma gangrenosum is highly likely.
Score of ≤ 10: pyoderma gangrenosum is highly unlikely.
Infectious causes
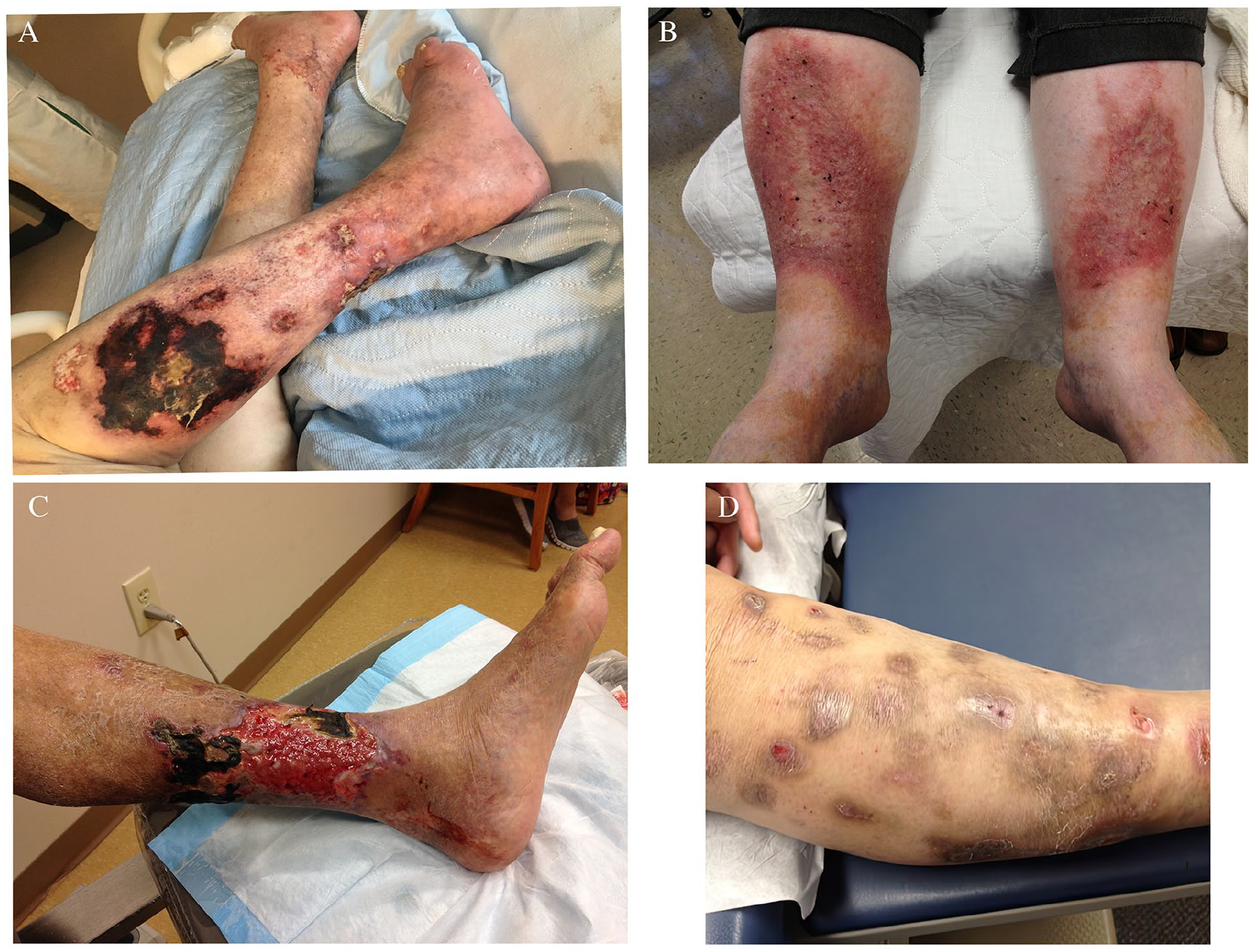
Skin or soft tissue infections and osteomyelitis can sometimes be mistaken for VLUs. Infectious causes of ulcers include animal bites, scratches, trauma, previous surgical scars, and the presence of hardware. Though acute infections such as cellulitis or necrotizing fasciitis typically present with classic inflammatory signs, chronic osteomyelitis may have a more subtle onset (Figures 6A and 6B). In acute infections, the affected area may exhibit drainage, erythema, and swelling (Figure 6C). Osteomyelitis can manifest with systemic symptoms such as fever, chills, localized pain, wound drainage, sepsis, or exposed bone, along with point tenderness over the infected bone.
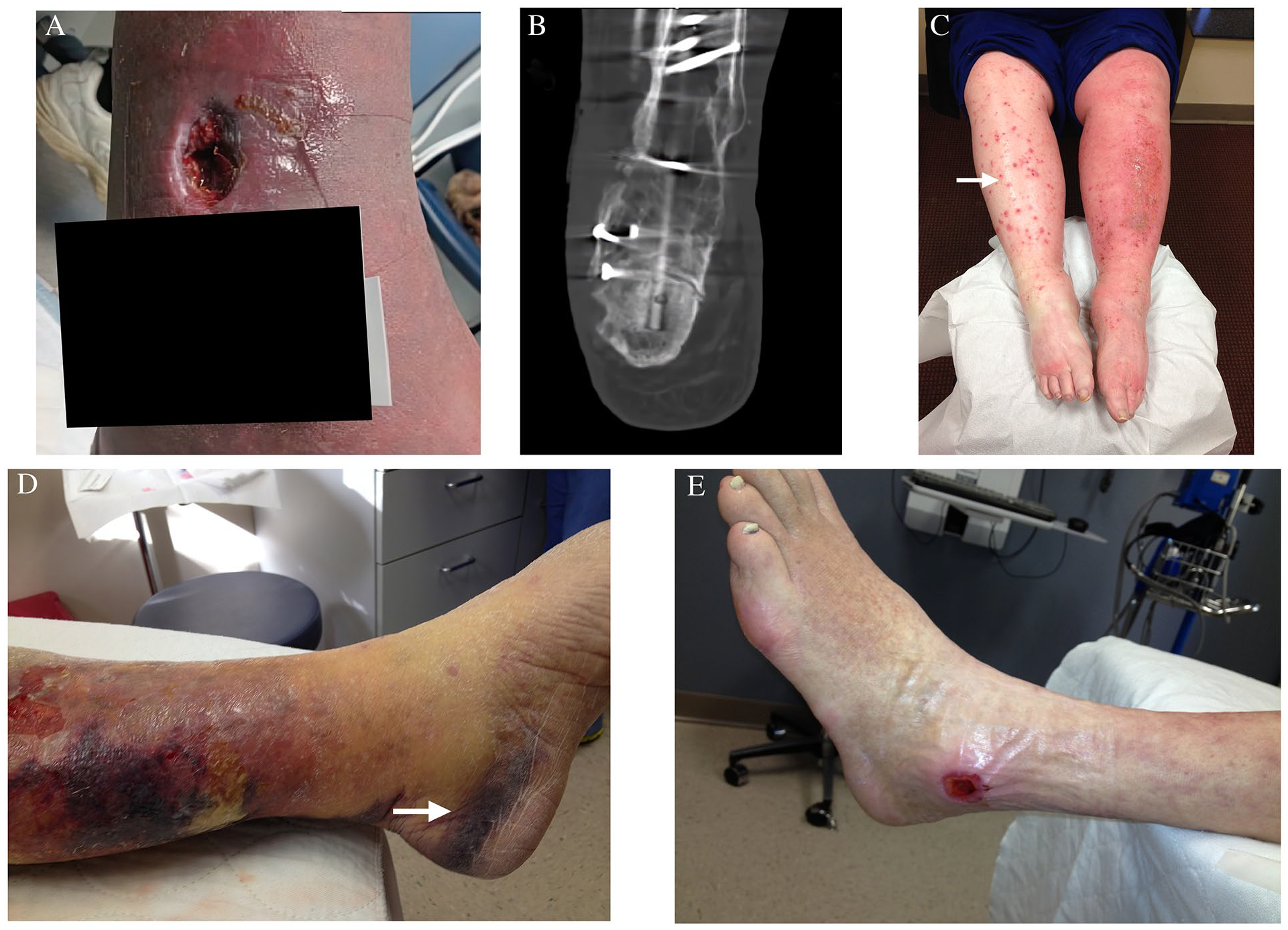
Infectious, traumatic, and pressure ulcers.
Laboratory findings in osteomyelitis often include elevated C-reactive protein, erythrocyte sedimentation rate, leukocytosis, and positive blood cultures. 65 Radiographic imaging may reveal bony destruction, though changes may take up to 3 weeks to become apparent and may not be as sensitive as magnetic resonance imaging (MRI). MRI is the preferred diagnostic modality, as it can detect marrow edema and inflammation. 66
Common causative organisms in wound infections include
Pressure and traumatic ulcers
Traumatic and pressure-related etiologies frequently mimic VLUs. These include trauma-induced skin breakdown (Figure 6D) and pressure ulcers (Figure 6E), particularly common in elderly or bed-bound patients with cognitive impairment. Management typically involves supportive care, removal of trapped blood or coagulum, and protective wound dressings. Biopsy is unnecessary in these types of wounds.
Pressure ulcers are defined as localized skin and subcutaneous tissue destruction resulting from immobility. Risk factors include extended care facility residence, acute illnesses, significant comorbidities, advanced age, poor nutrition, and impaired mobility. 68 The pathogenesis is multifactorial, primarily involving tissue that is sandwiched between a bony prominence and external pressures that compromise blood flow, leading to oxygen and nutrient deprivation. Poor nutrition and continued pressure contribute to ulcer progression. 69
Pressure ulcers of the legs typically develop over bony prominences such as the heels or lateral malleoli. Ulcers located on the malleolus can be confused with VLUs. These ulcers usually present with clear punched-out edges without any undermining. Unless there is concomitant CVI, these ulcers do not demonstrate skin changes such as stasis, hyperpigmentation, or dermatitis. They are classified into four stages 70 :
Stage I: nonblanching erythema
Stage II: shallow, open ulcerations
Stage III: full-thickness tissue destruction with visible adipose
Stage IV: exposure of underlying bone, fascia, and muscle.
Treatment approaches include regular wound cleansing, nutritional support, and, in rare cases, surgical intervention. Addressing the patient’s immobility and underlying conditions is crucial for effective management.
Malignancy
It has been proposed that 10% of nonhealing leg ulcers are secondary to malignancy. 71 Therefore, it is imperative to include malignancy in the differential diagnosis of all chronic ulcers. Basal cell carcinoma (BCC) is not only the most common cancer but also the most common malignant mimicker of VLUs. 72 Other malignancies that can mimic VLUs include squamous cell carcinoma (SCC) (Figure 7A), basal cell carcinoma (Figure 7B), cutaneous T-cell lymphoma (Figure 7C), 73 lymphangiosarcoma (Figure 7D), and KS. Though the histologic characteristics and treatments are beyond the scope of this article, it is crucial to recognize the features of malignant ulcers and the possibility of malignant transformation in VLUs and obtain biopsies based on clinical suspicion.
Malignancy-related ulcers.
Several risk factors increase the likelihood of BCC and SCC, including advanced age, wound chronicity, immunosuppressant therapies, repeated UV radiation exposure, fair complexion, family history, actinic keratosis, and a history of sunburns. Men, African American race, and rheumatoid arthritis are associated with T-cell lymphoma. 74 The variant of KS, which closely mimics VLUs, is highly associated with HIV/AIDS patients receiving immunosuppressive therapy or organ transplant recipients. 75 KS typically presents as numerous dark violet to brown nodular or plaque-like lesions that may eventually ulcerate (Figure 7E).
Characteristics of malignant ulcers often include rolled edges, hypergranulation tissue, malodor, and brisk bleeding. The hypergranulation tissue can be friable and enlarge easily, leading to a fungating mass. Intense pruritus is particularly associated with BCC.
A high degree of suspicion, timely biopsy, and the involvement of a dermatopathologist in the diagnosis of cutaneous malignancy are essential. Additionally, the care of these patients should include the involvement of a dermatologist or a plastic surgeon.
Chemical-induced ulcers
VLUs may be mimicked by chemical-induced ulcerative lesions, leading to potential misdiagnosis. In certain geographical regions, envenomation from
Brown recluse spider bites
Brown recluse spiders are among the most clinically significant venomous arachnids in the United States, primarily found in central and southeastern regions. These nocturnal creatures typically inhabit undisturbed, warm areas and become dormant during colder months. 76 Diagnosis can be challenging due to the often painless nature of the bite. Symptoms may manifest up to 6 hours postbite, characterized by sharp or burning pain. The venom includes various enzymes that degrade collagen and basement membranes, with sphingomyelinase-D being particularly associated with dermal necrosis. 77
The bite site may initially present with puncture marks, surrounding erythema, and edema (Figure 8A). A characteristic ‘bull’s eye’ lesion may develop, progressing to a necrotic ulcer. Systemic symptoms can include rash, fever, and malaise.
Ulcers related to toxins and hemoglobinopathies.
Management is individualized and primarily supportive, involving wound care, immobilization, and tetanus prophylaxis. Advanced cases may warrant dapsone, systemic glucocorticoids, hyperbaric oxygen therapy, or surgical intervention. Healing typically occurs over several months. 76
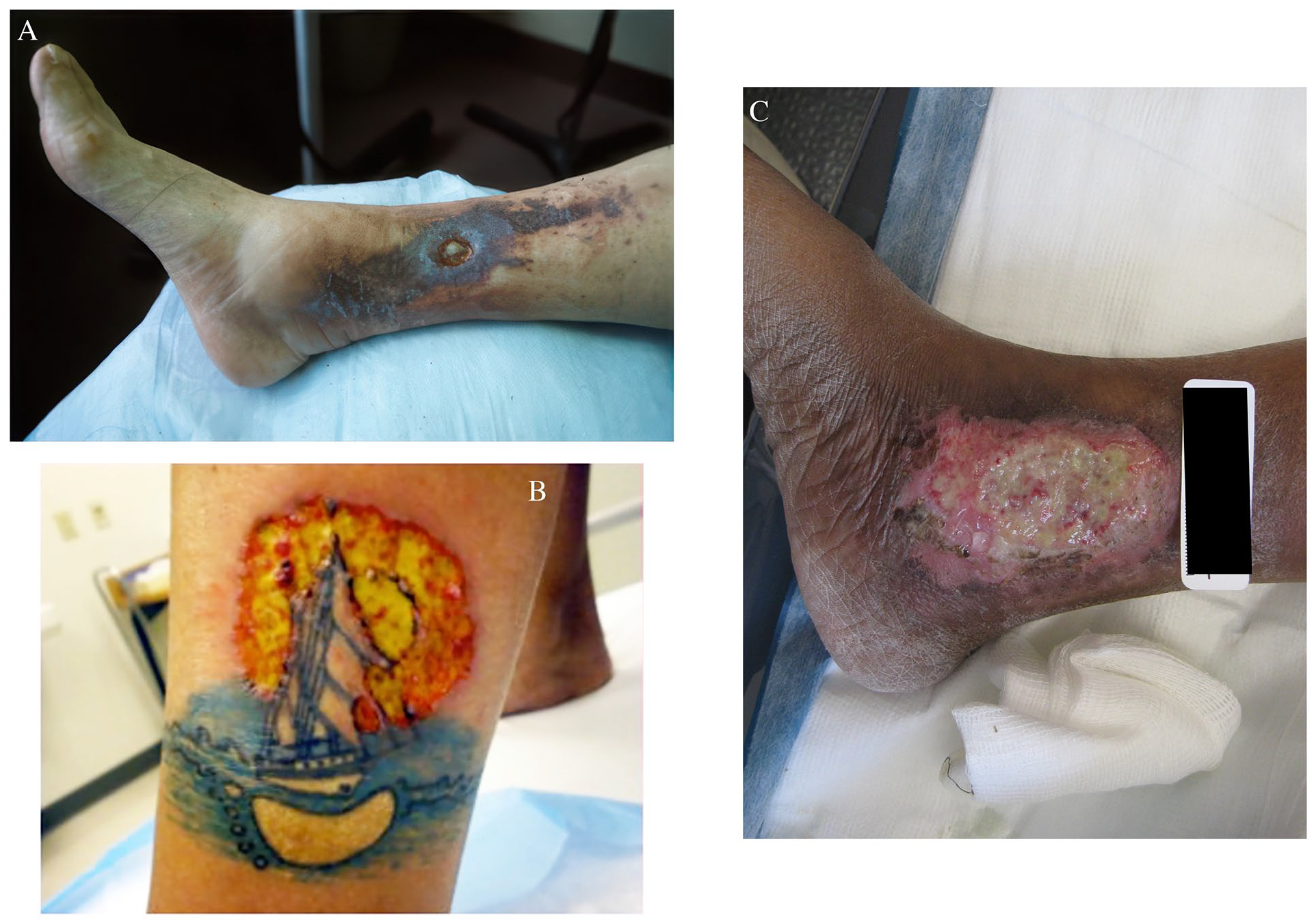
Tattoo ink and subcutaneous injection by intravenous drug users
Ulcers are an uncommon adverse effect of tattooing but can occur due to contamination, infections, allergic reactions, or inflammatory responses to tattoo pigments. 78 With the increasing prevalence of tattoos, vascular and wound care providers may encounter these ulcers, sometimes misidentified as VLUs. These ulcers typically appear in areas of previous tattoos (Figure 8B) or local injections related to intravenous drug use. Their appearance can range from inflammatory to granulomatous, and they may resemble pyoderma gangrenosum. Treatment is tailored to the specific type of rash and may involve topical immunosuppressive therapy and, in some cases, surgical intervention.
Ulcerations from hemoglobinopathies
Hemoglobinopathies, particularly sickle cell disease (SCD) and certain thalassemias (intermedia and major), 79 may produce VLU mimicry. Clinicians should include these hereditary conditions in their differential diagnosis, especially when typical VLU features are absent and/or standard treatments fail.
In SCD, leg ulcers often result from microvascular occlusion and ischemia due to hemoglobin S sickling.80,81 These painful, slow-healing ulcers typically appear near the medial malleoli. They are characteristically round, punched out, and with necrotic slough and surrounding hyperpigmentation similar to VLUs (Figure 8C). Unlike VLUs, SCD ulcers may worsen with compression therapy. Histopathology is nonspecific and unnecessary.
Management of SCD-related ulcers is complex, with reported treatments including pentoxifylline, hydroxyurea, pain management, hyperbaric oxygen, and skin grafting. 82 The role of hydroxyurea in ulcer development remains controversial, as it can both treat SCD and potentially contribute to ulcer formation 83 in some patients.
Given the intricacies of SCD management and the potential treatment impact on ulcer development, close collaboration with hematology specialists is essential. This ensures appropriate evaluation of hydroxyurea use and consideration of other SCD-specific interventions, such as blood transfusions.
Metabolic disorders
Metabolic disorders such as calciphylaxis and necrobiosis lipoidica diabeticorum (NLD) should be included in the differential diagnosis of VLUs. They should be suspected in patients with associated metabolic conditions.
Calciphylaxis
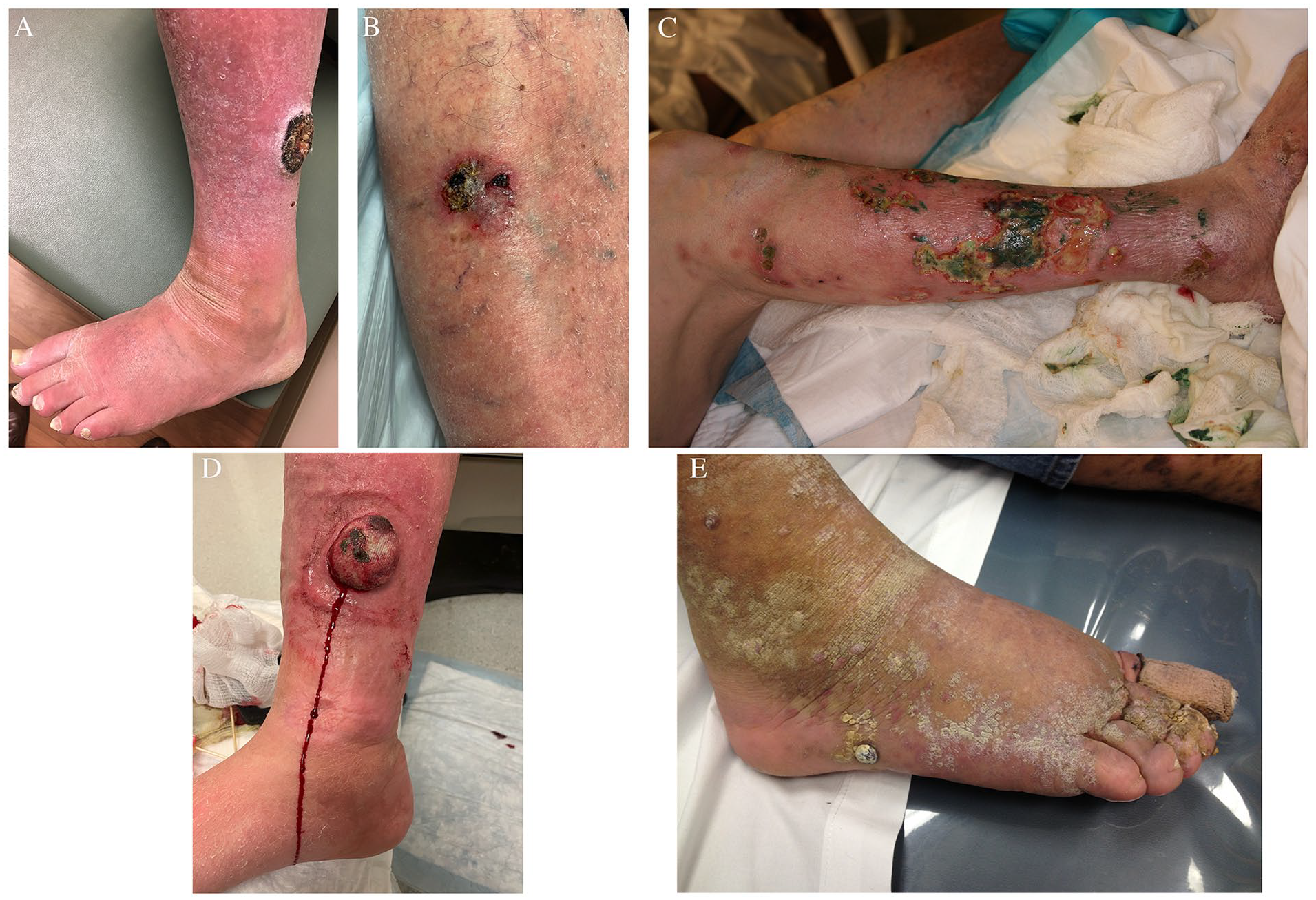
Calciphylaxis, characterized by vascular calcification and skin necrosis, is primarily seen in patients with end-stage renal disease on dialysis. Although less common, nonuremic calciphylaxis is diagnostically challenging in patients with characteristic ulcers but no renal dysfunction. 84 Conditions such as hyperparathyroidism, multiple myeloma, cirrhosis, and rheumatoid arthritis, as well as obesity, diabetes, autoimmune diseases, and certain medications (e.g., warfarin, steroids, iron-dextran, erythropoietin), are associated with calciphylaxis. 85 One-year mortality rates can be as high as 50%, with sepsis being a leading cause of death. 86 The pathogenesis involves severe arterial hypoperfusion due to endoluminal calcification, fibrin deposition, and microthrombi, leading to distal ischemia and painful ischemic and necrotic ulcerations (Figure 9A). Commonly affected sites include the legs, trunk, buttocks, and genitals. Diagnosis is primarily clinical with supportive histology displaying intravascular calcium and fibrin deposition. Treatment focuses on local wound care, pain management, sodium thiosulfate to dissolve loco-regional calcium salts, and bisphosphonates to reduce calcium levels and systemic inflammation. 85
Additional nonvascular manifestations.
Necrobiosis lipoidica diabeticorum (NLD)
NLD is an idiopathic granulomatous skin condition linked to microangiopathy and collagen degeneration. Over 50% of patients with NLD have diabetes, yet its prevalence among those with diabetes is less than 1%. 87 Lesions are well-circumscribed, erythematous plaques with yellow-brown centers and raised violaceous borders, typically found on the distal anteromedial shins or dorsal feet 88 (Figure 9B). Pathergy is common, and about one-third of cases may ulcerate. The clinical course is challenging, with frequent relapses. Pentoxifylline, corticosteroids (topical or intralesional), infliximab, hyperbaric oxygen therapy, and photodynamic therapy have been reported as treatment options. Tight glycemic control and trauma avoidance may improve outcomes. 89
Drugs
Drug-related ulceration is a rare mimicker with devastating morbidity and mortality. Warfarin-induced skin necrosis and heparin-induced thrombocytopenia can result in these ulcerations. Hydroxyurea-related ulcerations have been discussed in the context of SCD treatment.
Warfarin-induced skin necrosis
Warfarin-induced skin necrosis (WISN) is a rare but serious complication affecting approximately one in 10,000 patients on warfarin. Risk factors include women, obesity, liver dysfunction, hypercoagulable states, and large initial doses of warfarin without adequate bridging with enoxaparin. 90 The rapid inhibition of protein C, which has a short half-life (6–10 hours), leads to a prothrombotic state and microthrombi formation. 91
WISN begins as painful, erythematous areas that progress to purple, maroon, blue, and black discolorations due to microvascular hemorrhages and occlusions (Figure 9C). These lesions can evolve into ulcerative necrosis with eschar and bullae formation, commonly affecting fatty areas with high blood supply, such as the breasts, abdomen, thighs, and buttocks, but can also appear on the legs.
The diagnosis is primarily clinical, supported by a biopsy showing fibrin microthrombi and surrounding hemorrhage. Immediate discontinuation of warfarin and administration of vitamin K or fresh frozen plasma (FFP) are crucial, along with initiating enoxaparin. Local wound care is essential, and severe cases may require surgical debridement or even amputation. 92
Psychiatric
Although rare, an overlooked etiology is psychiatric-induced ulceration, which is difficult to diagnose and harder to treat. Such an example is dermatitis artefacta.
Dermatitis artefacta
Dermatitis artefacta is a complex psychocutaneous disorder characterized by self-inflicted skin lesions serving an underlying psychological need. This condition presents diagnostic challenges due to its diverse pathology and manifestations. Dermatitis artefacta predominantly affects women and adolescents, often coexisting with other psychiatric conditions such as borderline personality disorder and anorexia.
Patients frequently deny their involvement in creating these lesions and may provide vague or inconsistent histories. The wounds affect accessible skin and are conspicuously absent in the interscapular region. They often exhibit unusual shapes, geometric or linear patterns, and varying stages of healing and may be accompanied by scars or keloids suggestive of chronic manipulation (Figure 9D).
Dermatitis artefacta is primarily a diagnosis of exclusion. Management of dermatitis artefacta necessitates a collaborative approach involving dermatologists and mental health professionals. To avoid exacerbating the condition, it is crucial to limit unnecessary diagnostic tests and treatments once dermatitis artefacta is suspected. 93
Conclusion
Key points to remember are summarized in Table 4. VLUs are the most common type of lower-extremity ulceration, yet they can present in both typical and atypical forms. Alternative diagnoses should be considered when ulcers do not respond to standard venous treatments or when noninvasive testing fails to confirm a venous origin. Importantly, early recognition of VLU mimics is essential and requires a comprehensive diagnostic approach, including a detailed patient history that accounts for potential autoimmune, inflammatory, or metabolic conditions. Biopsy remains a valuable diagnostic tool for identifying autoimmune disorders, malignancies, inflammatory conditions, and rare vascular diseases such as AADM, though its effectiveness depends on proper integration with the clinical context and interpreter expertise. Additionally, assessing for soft tissue and bone infections is critical to comprehensive VLU management, ensuring accurate diagnosis and optimizing treatment outcomes (Figure 10).
Key points to remember about venous leg ulcers (VLUs).

Algorithm for suspected venous leg ulcer (VLU).
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
