Abstract
Fibronectin (FN) circulating in the blood and produced by cells provides the basis of the extracellular matrix (ECM) formed in healing acute wounds. The time-dependent deposition of FN by macrophages, its synthesis by fibroblasts and myofibroblasts, and later degradation in the remodeled granulation tissue are a prerequisite for successful healing of wounds. However, the pattern of FN expression and deposition in skin lesions is disturbed. The degradation of the ECM components including FN in varicose veins prevails over ECM synthesis and deposition. FN is inconspicuous in the fibrotic lesions in lipodermatosclerosis, while tenascin-C containing FN-like peptide sequences are prominent. FN is produced in large amounts by fibroblasts at the edge of venous ulcers but FN deposition at the wound bed is impaired. Both the proteolytic environment in the wounds and the changed function of the ulcer fibroblasts may be responsible for the poor healing of venous ulcers. The aim of this review is to describe the current knowledge of FN pathophysiology in chronic venous diseases. In view of the fact that FN plays a crucial role in organizing the ECM, further research focused on FN metabolism in venous diseases may bring results applicable to the treatment of the diseases.
Keywords
Introduction
Chronic venous diseases are very common in the European population; various forms of them affect more than 80% of men and women. Risk factors for chronic venous disease include heredity, age, female sex, obesity, pregnancy, prolonged standing, and greater height. In spite of the diversity of symptoms associated with chronic venous diseases, all are related to venous hypertension, which may be caused by venous valvular incompetence, venous obstruction or the failure of calf muscle pump. Leukocytes accumulate in legs under conditions of high venous pressure. They adhere to the endothelium of small vessels and migrate through it. The chronic inflammatory state provokes skin changes.1,2 Venous disorders are classified based on clinical manifestations (C), etiologic factors (E), anatomic distribution of disease (A), and underlying pathological findings (P) (CEAP classification). 3
Varicose veins
Varicose veins (C2) that develop mostly in the great saphenous vein are elongated, dilated, and tortuous. They contain atrophic and hypertrophic segments. Both cellular and matrix components are decreased in the atrophic segments. The hypertrophic segments are characterized by increased wall thickness and the accumulation of the extracellular matrix (ECM) and smooth muscle cells.4,5 Collagen synthesis is increased but collagen fibers are disorganized, dispersed, and often fragmented. Elastin content is reduced and elastic fibers are fragmented, which correlates with enhanced distensibility of the veins.5–7 Smooth muscle cells in varicose veins exhibit marked alterations that suggest modulation from the contractile to the proliferative and synthetic phenotype.4,5,8 While the abundance of α-smooth muscle actin (α-SMA) in varicose veins decreases, smooth muscle cells proliferate. 9 The proliferation of smooth muscle cells and endothelial cells can be induced in isolated mouse facial vein segments exposed to a supraphysiological intraluminal pressure. The pressure also induces the expression of matrix metalloproteinase-2 (MMP-2) and its proteolytic activity in smooth muscle and endothelial cells. 10 Smooth muscle cells isolated from human varicose veins proliferate more quickly than cells from control veins, show higher migration, and synthesize more collagen and MMP-2. 8 The expression of MMP-2 and -9, as well as of their inhibitors (tissue inhibitors of metalloproteinases; TIMPs), at the mRNA and protein level, is higher in varicose veins than in normal great saphenous veins. The balance between metalloproteinases (MMPs) and TIMPs may be important for venous wall remodeling. 11
Lipodermatosclerosis
Lipodermatosclerosis (LDS, C4b) combines venous insufficiency and dermatological changes. It is characterized by the palpable induration of the skin caused by the fibrosis of subcutaneous fat. 12 Blood capillaries in patients with LDS are often convoluted or glomerulus-shaped. 13 Dilated spaces between endothelial cells allow for the extravasation of serum and blood cells, mainly erythrocytes, T lymphocytes, and macrophages.14,15 Edema, erythema, and red-brown hyperpigmentation of the lower leg are often found in patients with LDS.12,16 Fibrin is deposited around blood vessels with extravasated erythrocytes. 16 Inflammation is associated with adipocyte necrosis. The subcutaneous adipose layer is replaced by the fibrotic tissue which extends from the septa into the fat lobules. 17 The number of cells expressing procollagen type I mRNA, mainly fibroblasts, is higher in LDS lesions than in control skin samples. 18 The expression and activity of MMPs as well as collagen degradation in the skin lesion also increases and the more rapid matrix turnover may contribute to the development of venous ulcers. 19
Venous ulcers
Venous ulcers (C6) are shallow but large wounds with irregular margins that develop on the lower leg or ankle. The epidermal edge abuts an ulcer base containing necrotic tissue and poor granulating matrix. The ulcer bed is covered with the exudate rich in cytokines and proteinases. 20 Many capillaries in the underlying dermis are surrounded by fibrin cuffs that also contain other matrix components such as fibronectin (FN), tenascin, laminin, and collagen. 21 The border of the ulcer is infiltrated with T-lymphocytes and macrophages, in some cases with mast cells. 22 MMP-9-positive neutrophils and macrophages are found in the ulcer bed 23 and in the wound fluid. 24 The levels and activity of MMPs are higher in the wound fluid in chronic venous ulcers when compared to the wound fluid of acute wounds. 25 The levels of MMP-2, -9, and -12 are higher in the wound fluid in inflammatory venous leg ulcers when compared to the wound fluid obtained from the ulcers in the state of granulation. In contrast, the levels of MMP-1, -7, and -13 are increased in the wound fluid of granulating wounds. 26 MMP-1 and -7 have an essential role in the re-epithelialization of the wounds. 27 The disturbance of the physiological balance between MMPs and TIMPs may lead to the uncontrolled degradation of ECM components, cytokines, and growth factors and their receptors, and contribute to the poor healing of venous leg ulcers. 26
Aim of the review
FN is a protein that plays a key role in the ECM formation and wound healing. Plasma FN (pFN) interacts with fibrin and forms the provisional matrix that is later replaced with the mature ECM containing an increasing proportion of cellular FN (cFN).28–30 FN interacts with collagen, tenascin, transglutaminase, and other proteins and facilitates their deposition in the ECM. FN regulates matrix organization and stability.31–33 ECM deposition in venous diseases is disturbed when compared to that in normally healing acute wounds.7,19,21 Impaired function of FN, regardless of its reason, hinders the formation of the ECM and wound healing. This review summarizes the results of the studies of FN performed in poorly healing wounds and collected in the databases Web of Science and PubMed. The emphasis was put on the results published in the past decade but as no similar review is available the most important older data were included. The aim of the review is to encourage further research of FN metabolism in venous disorders because its knowledge may be used in designing effective therapy for the diseases.
Fibronectin
Fibronectin structure, synthesis, and degradation
FN is a product of a single gene. Its encoding mRNA undergoes alternative splicing to generate two major forms of FN: pFN and cFN. FN comprises two subunits with the molecular weight 230–270 kDa, depending on the result of splicing. The subunits are joined by two disulfide bonds at the carboxyl end. FN molecules are composed of domains that perform different functions. The domains consist of repeating units (repeats, modules) I, II, and III. cFN contains extra domains EDA and EDB, also termed EIIIA and EIIIB, that are not present in pFN. EDA and EDB are products of single exons. 34 The presence of these regions induces conformational changes in the FN molecule and exposes their cryptic binding sites.35,36 The V domain (variable in length), also called IIICS (type III connecting segment), required for the secretion of FN from the cell, is included in the majority of cFN subunits but is present only in one subunit of the pFN dimer. 37 The scheme of a FN molecule is shown in Figure 1A. pFN is synthesized in the liver by hepatocytes and secreted into the blood. cFN is synthesized by many cell types, including fibroblasts, macrophages, and endothelial cells and is a mixture of isoforms. The alternative splicing of the EDA, EDB, and V domains allows for the expression of different FN isoforms in a tissue-dependent, cell type-specific, and temporally regulated manner. Increased expression of EDA and EDB isoforms is associated with tissue remodeling, including wound healing. 34 Extracellular FN fibrils are cleaved by membrane-type matrix metalloproteinase 1 (MT1-MMP, MMP-14), 38 MMP-2, 39 MMP-9, 40 or by elastase. 41 FN may then be endocytosed by cells and degraded in the lysosomes.38,42
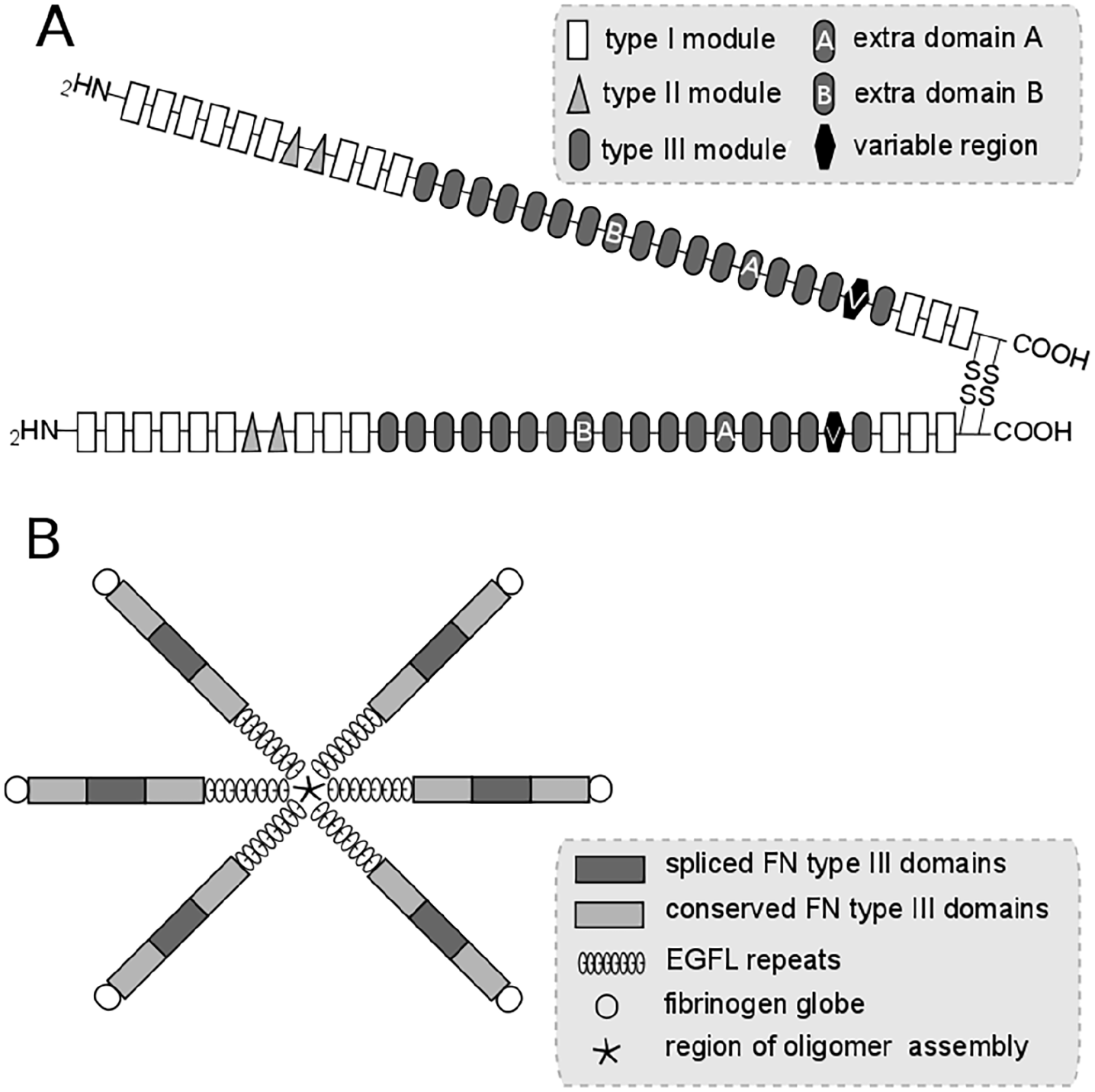
Structure of FN (A) and tenascin-C (B). Each of two subunits of FN consists of modules I, II, and III. Cellular FN contains extra domains A and B that are spliced in plasma FN. The chains of tenascin-C contain FN type III domains. Two of them are constitutively expressed, one can be alternatively spliced.
Fibronectin matrix
The assembly of FN fibrils is mediated by cells. FN dimers bind to the cell surface and their binding to integrins induces the reorganization of the actin cytoskeleton. Cell contractility helps to extend the FN molecule and to expose FN binding sites distributed along the length of the molecule. Myofibroblasts that have a high content of α-SMA after treatment with transforming growth factor-β (TGF-β) display a much higher ability to assemble exogenous FN into a fibrillary matrix than undifferentiated fibroblasts.43,44 Further FN molecules are recruited from the cellular surroundings. FN–FN interactions initially give rise to short fibrils around the cell periphery. Longer and thicker fibrils are then formed and give rise to an insoluble FN matrix. A continual source of FN is required for the maintaining of FN matrix stability.45,46 FN matrix stimulates cell proliferation.47,48 Recombinant FN containing the EDA exon (EDA+FN) is more potent in stimulating DNA synthesis in cells than FN lacking the exon (EDA−FN). 49 FN initiates the assembly of other ECM components. 50 Preformed FN matrix is essential for the formation of a collagen network. 51 FN is able to bind fibrin, collagen, tenascin, and heparan sulfate and chondroitin sulfate proteoglycans, and mediates the interaction between cells and these ECM components. 52 Group A Streptococcus has collagen-like proteins 1 and 2 on its surface. Collagen-like protein 1 binds to the EDA exon of cFN and when the wound is infected with Streptococcus, fibrosis may be delayed or prevented. 53
FN serves as a reservoir for growth factors and is capable of increasing their local concentration. FN may form complexes with positively charged platelet-derived growth factor-BB (PDGF-BB), fibroblast growth factor-2 (FGF-2) or hepatocyte growth factor (HGF) in the presence of negatively charged heparin. 54 TGF-β is present in tissues in a complex with latent TGF-β-binding protein-1 (LTBP-1). FN immobilizes LTBP-1 and stores TGF-β in the ECM. EDA containing FN (EDA-FN) binds LTBP-1 more efficiently than does EDB-containing FN (EDB-FN) and FN lacking splice domains. 55 TGF-β1 and EDA-FN in collaboration with TGF-β receptors and integrins on the cell surface are potent activators of myofibroblasts. TGF-β1 stimulates the production of EDA-FN, which in turn facilitates the incorporation of TGF-β1 into the ECM and its activation. TGF-β1 and EDA-FN form a positive feedback loop. 56
Fibronectin in acute wound healing
pFN is deposited in the wound site within seconds after injury. Since it contains a collagen binding site it may interact with the collagen exposed by the denuding vessel walls. pFN interacts with fibrin to form the early provisional matrix. 57 Macrophages invading the matrix begin to deposit cFN a few hours after wounding. 58 FN provides a conduit for the migration of fibroblasts from collagenous matrix to the provisional matrix. 59 Fibroblasts in the granulation tissue secrete large amounts of cFN at later time intervals. 58 FN fibrils connect fibroblasts in the granulation tissue to the matrix and to each other and facilitate wound contraction. 60 When the granulation tissue begins to form at the base of the wound, FN is synthesized by proliferating endothelial cells and deposited in the blood vessels. 61 The presence of the EDA exon in the cFN is essential for proper healing of the wound. The wound re-epithelialization is impaired, and the granulation tissue shows edematous regions in the mice lacking the EDA exon in FN protein. 62 FN provides a matrix scaffold and its deposition precedes collagen deposition. However, when collagen types III and I accumulate in the granulation tissue, FN disappears. 63
Fibronectin in varicose veins
The amount of FN that can be extracted from varicose veins is around one-third of that obtained from normal saphenous veins. The results of FN determination are supported by immunohistochemistry. These changes are not reflected in the mRNA expression. The remodeling process in varicose veins occurs at the protein rather than at the transcript level. 64 Smooth muscle cells isolated from varicose veins deposit much less FN and collagen type III than the cells from normal saphenous veins when they are cultured in vitro. 65 FN content in the intracellular compartment of the smooth muscle cells from varicose veins is similar to that in normal controls but FN content in the pericellular compartment and in the culture medium is reduced. It is possible that FN is degraded outside the cells. 66 In contrast, the expression of the tenascin-C (TN-C) gene is increased twice in varicose veins compared to control veins. 67 TN-C protein surrounds smooth muscle cells and its quantity is decreased in atrophic portions and increased in the hypertrophic segments of varicose veins. 4
Fibronectin in lipodermatosclerosis
The information about FN expression in LDS is scarce; FN fibers are fragmented. 68 In contrast, TN-C exhibits a widespread deposition in the dermis of patients with LDS.68–70 TN-C is an ECM protein that is upregulated in healing excisional wounds. 71 It has a symmetrical structure called hexabrachion. Each of its six arms contains an epidermal growth factor-like (EGFL) domain, two conserved FN type III (FN-III) domains and one spliced FN-III domain and a globular fibrinogen-like domain (Figure 1B). 72 The EGFL domains have antiadhesive properties and encourage cell migration. 72 The FN-III domains bind TGF-β, PDGF and other growth factors. 73 TN-C supports inflammation 74 and stimulates the migration of human dermal fibroblasts. 75 The response of fibroblasts from TN-C-deficient mice to TGF-β is decreased. Myofibroblast differentiation and collagen synthesis is low in TN-C-null cells compared to wild-type cells. 76 A prolonged expression of TN-C leads to chronic inflammation and tissue fibrosis.77–79
Fibronectin in venous ulcers
Western blot analysis shows that the level of cFN is enhanced in fibroblasts isolated from the skin of patients with venous reflux but no ulcers compared to fibroblasts from patients without reflux. 80 The incorporation of radioactive amino acids indicates that the fibroblasts isolated from the edge of chronic venous ulcers synthesize less collagen than normal dermal fibroblasts but produce equal amounts of total FN. 81 The level of FN mRNA is high at the edges of the ulcers but the expression of the α5 integrin subunit specific to FN in the epidermis is low compared to that in acute wounds. The lack of cell surface receptors may prevent the migration of epidermal keratinocytes and contribute to healing defects in chronic venous leg ulcers. 82 The increased staining for FN is found in the reticular dermis of the margin skin of the ulcer. 68 The expression of FN in the ulcer area of venous ulcers often persists after 1 year. 83 FN can be detected histochemically in the ulcer margins but not at the base of the ulcer. When the ulcer heals and is re-epithelialized, FN is deposited by fibroblasts and the former ulcer base becomes indistinguishable from the adjacent tissue. 21 Neutrophils and macrophages, the sources of MMP-9, are detected in the bed of venous ulcers, in contrast to healing acute wounds where MMP-9 is localized to keratinocytes. 23 FN fragments are found in the wound fluid from venous foot ulcers. 84 FN is extensively degraded in the wound fluid obtained from venous ulcers but not in the acute wound fluid. FN degradation may be the result of the increased level of serine proteinases, mainly neutrophil elastase and proteinase 3. Their inhibitors α1-antitrypsin and α2-macroglobulin are also degraded.85–87
Prospects for the use of fibronectin in the treatment of chronic wounds
The results obtained in animal models suggest that topical application of FN on venous ulcers may be beneficial in their treatment. The healing of excision wounds in the skin of mice exposed to local X-ray irradiation is improved by the application of human plasma FN. 88 The treatment with FN also helps to normalize wound healing in mice partially deficient in FN. 89 However, FN binds a number of various substances 43 and it would be difficult to guarantee the complete absence of contamination (e.g. with viruses). 90 The stimulation of in situ synthesis of FN may help to overcome this problem. The amount of FN and collagen in the excision wounds in the skin of mice is increased after carboxytherapy (subcutaneous application of carbon dioxide around the wound). The numbers of neutrophils and lymphocytes in the wounds decrease after this treatment. 91
Some FN fragments obtained by elastase digestion, synthesis from amino acids or as recombinant peptides have a biological activity. They mimic the ECM when they are used to coat culture substrates and support the adhesion and growth of fibroblasts and other cells.92–94 Peptides containing the cell-binding arg-gly-asp- (RGD) motif and the synergistic pro-his-ser-arg-asn (PHSRN) sequence support the adhesion and migration of normal human fibroblasts and keratinocytes and accelerate the closure of the wounds made in the skin of rabbits. 95 Human umbilical vein endothelial cells proliferate and produce FN when they are cultured on hydrogels containing FN-mimetic peptide sequences gly-arg-gly-asp-ser-pro (GRGDSP) and PHSRN. 96 Certain FN peptides incorporated in hyaluronan hydrogel stimulate the formation of granulation tissue in excisional wounds in the skin of pigs.97,98
FN is a glycoprotein that contains oligosaccharide chains on asparagin residues (N-glycosylation sites) and on threonine residues (O-glycosylation sites). Glycosylation modifies the function of FN domains and is different in pFN and cFN. 99 FN glycosylation modulates fibroblast adhesion and spreading 100 and is associated with various pathological processes.101,102 FN glycosylation has not yet been studied in venous disorders.
The transplantation of cells – keratinocytes, fibroblasts, bone marrow- and adipose tissue-derived stem cells – improves healing of chronic venous leg ulcers. 103 The application of human epidermal basal cells on excision wounds in the skin of rats accelerates healing of the wounds. Precoating of the wound bed with FN leads to further shortening of the healing time. 104 The positive effects of adipose tissue-derived stem cells on wound healing may be caused by soluble factors the cells release but the cells also produce ECM components – collagen types I and III and FN. 105 The expression of the FN gene in adipose tissue-derived stem cells is higher than that in human fibroblasts. 106 The stem cells implanted into the wound need to survive in a harsh microenvironment; they face hypoxia, nutrient deprivation, and the action of death cytokines. Human bone marrow-derived mesenchymal stem cells incorporated into a gel containing hyaluronan and collagen type I and implanted into the wound in mouse skin survive for about a week. This time can be extended when TN-C is added to the gel. 107 Induced pluripotent stem cells are derived from adult cells by in vitro induction of pluripotency. The adult cells are harvested noninvasively and the induced pluripotent stem cells can be transplanted autologously. They are able to differentiate into all cell types in healthy skin and have the potential to accelerate wound healing in non-diabetic and diabetic murine models. 108 A FN-derived peptide containing PHSRN and GRGDSP sequences supports proliferation and attachment of these cells in vitro when it is used to precoat culture plates. This efficient and safe culture may be used to produce induced pluripotent stem cells on a large scale. 109
Discussion
pFN that originates in the plasma and cFN that is synthesized within the tissue play a regulatory role in the formation of the ECM in acute wounds. The expression of FN in healing wounds is increased at both mRNA and protein levels. Most FN is produced by fibroblasts.58,63 FN is also intensively synthesized in the skin surrounding venous ulcers.68,81,83 However, FN is absent at the ulcer base. 21 Neutrophils and macrophages, the sources of serine proteases and MMPs, are located in this area. 23 The activity of neutrophil elastase is higher in the ulcer base and edge than in the surrounding skin. 110 MMP-9, elastase, and proteinase 3 are present in the wound exudate in venous ulcers in contrast to excision wounds. 111 Elastase and proteinase 3 are known to be released from neutrophil granules at the site of inflammation. 112 Neutrophils are also a source of MMP-9 that is released by neutrophil degranulation, while mononuclear cells synthesize MMP-9 after stimulation by cytokines, bacterial products or cellular contacts. 113 The level of MMP-9 is much higher in the wound fluid in chronic venous ulcers than that in the serum or in acute wounds.111,114 The high ratio of MMP-9/TIMP-1 may be a predictor of poor healing of ulcers. 115 When the ulcers heal, protease activity decreases. 116 Besides degrading FN, serine proteinases may support inflammation by activating proinflammatory cytokines TNF-α and IL-1β. 112 Venous ulcer wound fluid has a negative influence on the proliferative activity of dermal fibroblasts. 117 The reduction of MMP levels is associated with ulcer healing; the levels of MMP-1, -2, -3, -8, and -9 decrease after compression therapy. 118 The inhibition of MMPs represents a potential therapeutic strategy in the treatment of venous ulcers. Physiological MMP inhibitors TIMPs, natural antibiotics tetracyclines, a xanthine derivative pentoxyfylline and other agents have been tested in animal models. However, MMPs are needed for proper healing and care must be taken to find a balance between MMP activity and inhibition. 119
Fibroblast dysfunction appears to play a key role in poor healing of venous ulcers. 120 Fibroblasts and other cells become senescent when they are exposed to stress factors such as inflammatory cytokines, oxidative stress, DNA damage, chromatin disruption, chemotherapeutic drugs or the activation of certain oncogenes.121,122 Senescent cells can be detected in tissues by using senescence-associated β-galactosidase or other markers. 122 Fibroblasts isolated from venous ulcers display an irregular shape, larger size, and slow growth in vitro. The lifespan of senescent cells is significantly reduced when compared with that of normal fibroblasts and their response to growth factors PDGF-BB, FGF-2, and EGF is diminished.120,122,123 Cultured fibroblasts from normal human skin respond to TGF-β1 by increased collagen synthesis. Fibroblasts isolated from the wound bed of venous ulcers or from lipodermatosclerotic skin do not respond in this way. 124
Senescent cells transiently appear in the proliferative phase of acute wounds and have a beneficial influence on wound healing. They secrete PDGF-AA that induces myofibroblast differentiation. 125 Senescent cells limit fibrosis at the late stage of healing because they cease to proliferate but secrete MMPs that degrade the ECM and promote the remodeling of the granulation tissue. 126 However, the protracted presence of senescence cells impairs healing. 122 The time necessary to heal venous ulcers correlates with the percentage of senescent cells in the fibroblast population. 127 Fibroblasts with reduced growth rates and positive for senescence-associated β-galactosidase can also be found in patients with venous reflux and no ulcers. 80 Therapeutics are sought that would eliminate the negative effects of senescence. 128
A number of proteins are able to bind to FN and modify its functions. TN-C counterbalances certain activities of FN in vitro. FN is an adhesive protein – cells spread on a FN substratum. TN-C is antiadhesive – cells plated on TN-C are rounded. 129 The EGFL domains in TN-C branches are responsible for the antiadhesive properties of TN-C. 130 FN-III domains in TN-C bind a number of other ECM components including FN. Alternative splicing provides diversity in the function of TN-C polypeptides. 72 TN-C expression by fibroblasts is enhanced by profibrotic cytokines. TN-C elicits an increased expression of collagen type I and α-SMA in the cells. TN-C content is elevated in the skin of patients with systemic sclerosis. Contrarily, skin and lung fibrosis are attenuated in TN-C-deficient mice. 78 TN-C expression is increased in the dermis of patients with LDS68–70 and may play an important role in the disease development and persistence. Changes in the metabolism of FN and of TN-C in chronic venous diseases are summarized in Table 1.
Changes in FN and TN-C metabolism in chronic venous diseases.

FN, fibronectin; TN-C, tenascin-C.
Periostin is a protein that binds to both FN and TN-C. 131 It activates dermal fibroblasts and accelerates wound healing. 132 Periostin supports adipose tissue inflammation and fibrosis. 133 On the other hand, its expression is decreased in nonhealing skin wounds. Periostin is present in the dermis proximal to the wound but its immunoreactivity is decreased in the wound bed. 134 Osteopontin is a proinflammatory and profibrotic cytokine closely associated with adipose tissue. 135 However, it has not yet been studied in LDS, whose prominent feature is the fibrosis of the adipose tissue. 12 Other FN-binding ECM components (e.g. thrombospondins) play a role in tissue repair, but only limited information on their metabolism in nonhealing wounds is available. 136 A better knowledge of the metabolism of individual ECM components and their interaction with fibroblasts and other cells in chronic wounds may open up new approaches to the treatment of such wounds.
Conclusion
FN is an extracellular matrix protein that plays an important role in the healing of acute wounds. Its metabolism is disturbed in chronic venous diseases. The degradation of FN prevails over its synthesis in varicose veins. Little is known about the fate of FN in lipodermatosclerosis lesions in which tenascin-C appears to have an important function. The cells in the surroundings of venous ulcers synthesize ample amounts of FN but the protein is not deposited at the ulcer bed. The reason may be the degradation of FN by an excessive level of proteinases in the wound fluid or the dysfunction of the ulcer bed fibroblasts.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Ministry of Education Youth and Sports of the Czech Republic, grants SVV-2017, No. 260392; the project ‘Fighting Infectious Diseases’, No. CZ.02.1.01/0.0/0.0/16_019/0000787; and by Charles University, grant GA UK No. 128417.
