Abstract
The rupture of an abdominal aortic aneurysm (AAA) poses a significant threat, with a high mortality rate, and the mechanical stability of the arterial wall determines both its growth and potential for rupture. Owing to extracellular matrix (ECM) degradation, wall-resident cells are subjected to an aberrant mechanical stress environment. In response to stress, the cellular mechanical signaling pathway is activated, initiating the remodeling of the arterial wall to restore stability. A decline in mechanical signal responsiveness, coupled with inadequate remodeling, significantly contributes to the AAA’s progressive expansion and eventual rupture. In this review, we summarize the main stresses experienced by the arterial wall, emphasizing the critical role of the ECM in withstanding stress and the importance of stress-exposed cells in maintaining mechanical stability. Furthermore, we will discuss the application of biomechanical analyses as a predictive tool for assessing AAA stability.
Keywords
Introduction
The abdominal aortic aneurysm (AAA) is defined as a significant localized dilation, exceeding 50% (or 3 cm) of the normal size, of the abdominal aorta. In contrast to healthy arteries, the AAA wall loses its structural integrity, becoming stiff and fragile. When hemodynamic forces surpass the tissue’s strength limits, AAA ruptures, leading to uncontrollable and life-threatening intra-abdominal hemorrhage. In 2019, global estimates indicated a total of 35.12 million AAA cases, 1 and in 2017, AAA accounted for 167,200 deaths worldwide. 2
Currently, AAA diameter serves as the primary criterion for determining surgical intervention, with thresholds of over 55 mm for men and 50 mm for women. However, autopsy findings reveal that approximately 13% of AAAs with diameters of 50 mm or less rupture, and 60% of AAAs with diameters exceeding 50 mm never rupture. 3 Hence, more accurate indicators are needed to assess the risk of AAA rupture and to guide whether to operate. As for small AAAs (3.0–5.4 cm), periodic diameter surveillance is recommended. 4 Unfortunately, no method can accurately predict the growth rate of small AAAs. Moreover, randomized controlled trials have failed to convincingly demonstrate pharmacological therapy can effectively limit AAA growth.5,6
Factors such as aging, smoking, hypertension, infection, inflammation, and reactive oxygen species (ROS) can adversely affect the arterial wall, resulting in the rupture of elastic fibers, death of smooth muscle cells (SMCs), and degradation of the extracellular matrix (ECM). Under hemodynamic forces, the damaged arterial wall undergoes expansion. Concurrently, due to the degradation of the ECM, the cellular stress shielding effect is disrupted, which activates wall-resident cells, prompting them to produce ECM to restore vascular wall stability – a process referred to as vascular wall remodeling. Detecting and intervening in the remodeling of the arterial wall presents potential avenues for the diagnosis and treatment of AAA. In this review, we will delve into the distribution of stress within the vascular wall, the crucial role played by the ECM in stress-bearing, the cellular response to stress in arterial wall remodeling, and potential clinical implications.
The main stresses experienced by the abdominal artery wall
In nature, the generation and resistance of force are ubiquitous phenomena. When an external force is applied to an object, it induces internal deformations and stresses that counteract the applied force. 7 There are three sources of force exerted on the AAA wall, including pulsatile blood flow, uneven growth during AAA progression, and external compression from surrounding tissues. Correspondingly, several stresses exist within the AAA wall to resist these forces (Figure 1).
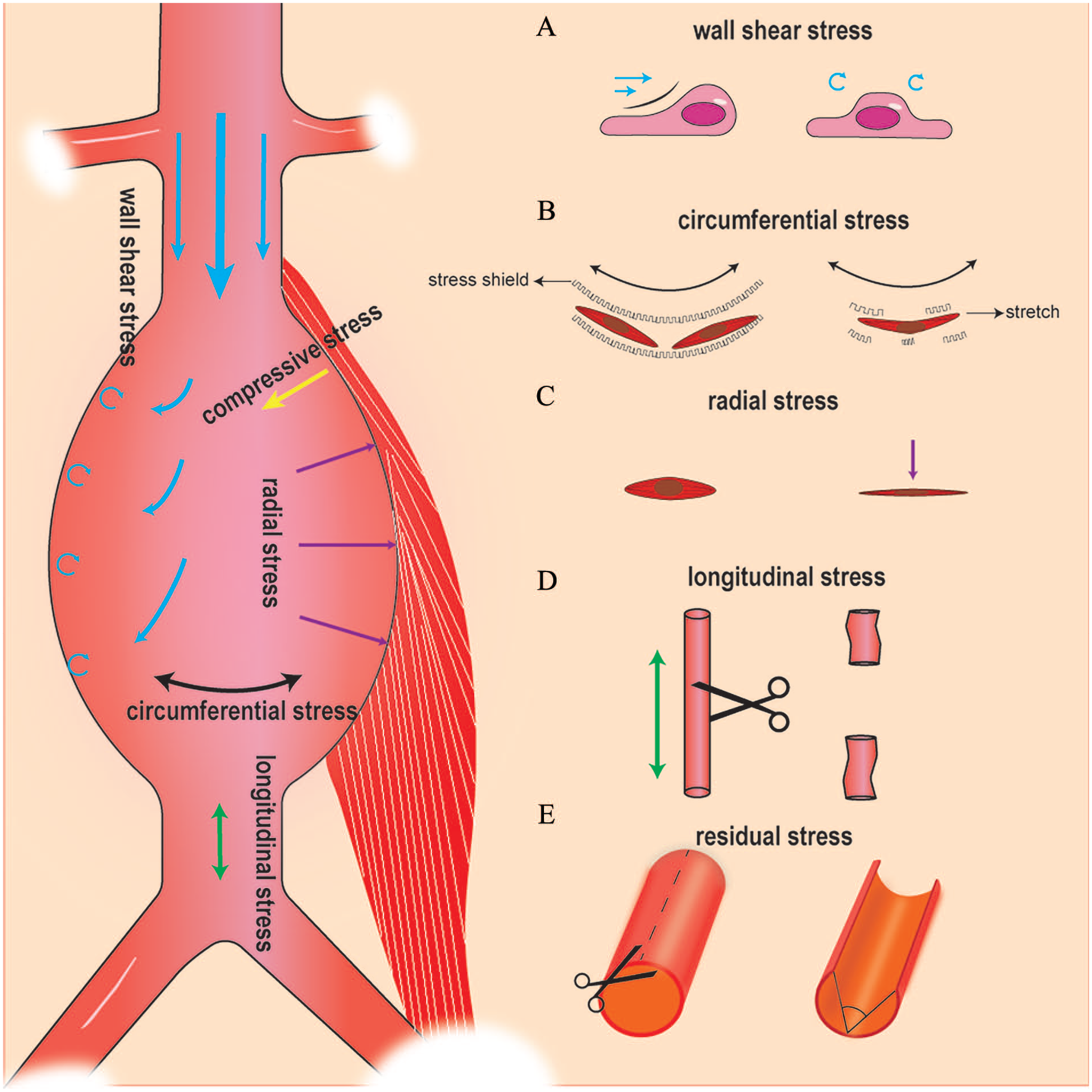
The major stresses abdominal aortic aneurysms withstand.
Wall shear stress (WSS) arises from the frictional drag of flowing blood against the vessel wall and is mainly perceived by the endothelial cells in direct contact with the blood flow. 8 The WSS depends on local vascular geometry, the velocity gradient, and blood viscosity. In the AAA sac, blood enters a larger space, often leading to flow separation phenomena, resulting in disturbed flow patterns, such as vortices or helical flows. These patterns create recirculation zones with a low and oscillating WSS, whereas regions of high-velocity jet flow impingement experience elevated WSS. Moreover, most aneurysms have irregular shapes, creating more complex blood flow patterns. To characterize the variability of WSS, WSS-related parameters, such as the oscillatory shear index (OSI), endothelial cell activation potential (ECAP), relative residence time (RRT), and time-averaged WSS (TAWSS) are introduced.
Under the impact of blood flow, the arterial wall is stretched and expanded in both circumferential and longitudinal directions, while experiencing compressive thinning in the direction perpendicular to the wall. Correspondingly, the wall experiences circumferential, longitudinal, and radial stresses. Circumferential stress is the most significant stress, as described by Laplace’s law, which illustrates the relationship between wall tension (T), radius (r), pressure (P), and wall thickness (d): T = (P * r) / d. As the artery diameter enlarges, circumferential stress increases accordingly.
The circumferential stress exhibits an evident gradient, with the inner wall stress closely matching blood pressure, while the outer wall aligns with abdominal pressure, which is disadvantageous to the wall stability. It is essential to emphasize the existence of residual stress, as evidenced by the artery springing open when the arterial wall is cut radially. This residual stress results from asymmetrical growth and remodeling of the aorta and promotes a more uniform distribution of circumferential stress by transforming it into compressive stress on the inner side and tensile stress on the outer side. 9
In addition to hemodynamic forces, axial stress arises from the axial prestretch of the artery during growth and development. 10 This portion of axial stress manifests as arterial shortening when the artery is severed from the body. It ensures the maintenance of arterial length and blood supply to tissues.
The compressive stress is majorly exerted by muscle compression during hip flexion, which may account for the tendency of AAAs to rupture more frequently on the posterior and lateral sides. 11
In AAAs, the local stress characteristics are more intricate due to the irregular arterial shape, intraluminal thrombus, and calcification. Furthermore, the destruction of the arterial wall structure and its remodeling led to significant alterations in the stress-bearing components, which will be explored in subsequent sections.
The extracellular matrix (ECM) primarily bears the majority of stresses
Circumferential stress is collaboratively born by the ECM, cellular materials, and cellular contractile forces. According to th e constrained mixture theory, the total stress (σ) can be expressed as the sum of the stresses contributed by the cell materials (σcell), elastin fibers (σeln), collagen fibers (σcoll), and cellular contractile forces (σact): σ = σcell + σeln + σcol + σact. Murtada et al. revealed that arterial wall cells maintained a homeostatic state when subjected to 15 kPa. Under this condition, the total stress (σ) is 150 kPa, whereas σact is 0 kPa and σcell is 15 kPa. 12 Obviously, collagen and elastic fibers play a major role in resisting stress within the arterial wall.
An instructive stress test result showed that the pure porcine elastic artery and the porcine intact aorta exhibit similar stress responses under lower blood pressures. Whereas under elevated blood pressure, the collagen fibers become active, collaborating with the elastin fibers to resist stress. 13 When elastic fibers are degraded by elastase, collagen fibers participate in stress resistance under lower stress. In normal arterial walls, elastic fibers play a major role under lower stress, with collagen fibers recruited to counter stress under higher stress. In this section, we will delve deeper into the characteristics of elastin and collagen and their alterations in AAA (Table 1).
The difference between elastin fiber and collagen fiber.

Elastin fibers endow the aorta with reversible elasticity
Elastin fibers, a primary stress-bearing component, occupy about 15% of the dry weight of the abdominal aortic artery. 14 The helical molecular organization imparts elastin fibers significant elasticity, whereas the elastin fibers in the media enhance the aorta’s elasticity. Moreover, elastin fibers facilitate uniform stress distribution within the vascular wall. 15
Elastin production primarily occurs at a limited developmental phase, synthesized by SMCs from late pregnancy to birth. 16 Once blood pressure and flow stabilize postnatally, the synthesis of new elastin significantly diminishes. Importantly, elastin fibers possess a remarkable lifespan, with an estimated half-life of approximately 70 years. 17 This longevity ensures the durability and resilience of the arterial wall under continuous mechanical stress imposed by approximately 30 million heartbeats per year in humans. However, once elastin fibers are damaged, they are unable to regenerate fully.
Degraded and fragmented elastin fibers are important pathological manifestations of AAA and are also the basis for its formation. Though elastin production is upregulated in AAA, 18 the newly formed elastin fragments lack the uniformity and efficacy of native elastin fibers in stress resistance. Therefore, protecting elastin from damage is the primary goal of AAA treatment.
Collagen fibers help restrain excessive dilation of the aorta
Collagen fibers are the most abundant ECM in the abdominal aorta, 14 and type I and type IV collagens are mainly concentrated in the intima and medial layers, whereas type III collagen predominates in the adventitia. 19
The elastic moduli of collagen is much greater than that of elastin, which means that collagen fibers experience less deformation under the same stress. 20 Hence, collagen fibers limit excessive dilation during sudden rises in blood pressure, protecting the smooth muscle and elastin. Unlike the elasticity of elastic fibers, collagen fibers exhibit viscoelasticity, especially in their immature state, where they are prone to intermolecular sliding under stress, leading to irreversible creep and the expansion of AAA. 21
The collagen fibers are primarily synthesized by fibroblasts and SMCs, with a half-life of approximately 70 days. 22 Collagen is crucial for maintaining the mechanical integrity of the vascular wall. Mutations of COL3A1 that encode type III procollagen can lead to arterial aneurysm, dissection, and rupture, known as vascular Ehlers–Danlos syndrome (vEDS). 23
Remodeled collagen fibers can provide sufficient strength to the vascular wall
In AAAs, elastic fibers rupture, and the elastin content decreases. To restore stability, the arterial wall undergoes adaptive changes, marked by an augmentation in collagen fibers, even the formation of neointima and neoadventitia. 24 Remarkably, the remodeling AAA wall is not as fragile as one might presume. Gomes et al. conducted an intriguing experiment, applying pressure to the arterial walls of human aortas using an elastic balloon from within. They found that the arterial walls of AAAs exhibited comparable stress-bearing capabilities, far surpassing normal blood pressure levels, reaching up to 590 mmHg. 25 This indicates that the collagen fiber-dominated arterial wall remodeling restores mechanical stability.
The accelerated turnover rate of collagen fibers contributes to AAA instability
Under stress, the collagen fibers will adaptively realign themselves to conform to the stress direction. 26 Biaxial stress tests reveal that collagen fibers transition from their initial axial orientation to a radial or even isotropic pattern in AAA. 27 Moreover, advanced microscopy techniques have observed an increase in abnormal collagen fibers, 28 within the remodeled regions of AAAs. The abnormal collagen fibers and their directional adjustments accelerate their turnover, with a notably shortened half-life of approximately 9 days. 29 This rapid turnover creates a dynamic equilibrium that is easily disrupted, significantly reducing the mechanical stability of the arterial wall.
Cross-linking enhances mechanical and chemical stability of collagen and elastic fibers
The intra- and inter-molecular cross-linking of elastin and collagen fibers significantly enhances their chemical and mechanical stability. Specifically, each collagen unit possesses one to four cross-link sites, whereas elastin boasts 15–20, resulting in elastin’s superior resilience and durability compared to collagen. As the number of mature cross-links accumulates, the newly synthesized fibers gradually transform from viscous to elastic, increasing their mechanical stability. 30 Prolonged exposure of collagen to reducing sugars can strengthen intermolecular cross-linking through the generation of advanced glycation end products (AGEs), thereby bolstering collagen fiber’s stability. This phenomenon explains the protective effect of diabetes on AAA. Although numerous techniques are employed to strengthen cross-linking, 31 their application in treating AAA remains limited.
The ECM in the AAA model
The application of elastase to adventitia or high-pressure perfusion can induce AAA in animal models. 32 Remarkably, when elastase is applied alone, elastin fiber fragmentation is observed; however, the abdominal aorta only expands within the initial 2 weeks of modeling and shows no further enlargement over a 100-day observation period. 33 Hence, despite the absence of intact elastin fibers, the arterial wall can maintain stability after remodeling. However, once the balance between remodeling and stress is disrupted, aneurysm progression can occur. When mice are orally administered β-aminopropionitrile (BAPN), a lysyl oxidase (LOX) cross-linking enzyme inhibitor, AAAs undergo continuous expansion. 33 Similarly, suppression of transforming growth factor-β (TGF-β) or upregulation of miR29 impairs arterial wall remodeling, exacerbating AAAs in mice. 34 However, ECM alone is insufficient to maintain the stability of the arterial wall. Through transplanting the decellularized aortic xenograft to the normal aorta, with intact elastic fibers and collagen fibers, the arterial wall still experiences dilation within 3–4 weeks. 35 Next, we will discuss the crucial role of cells in maintaining the stability of the arterial wall.
Stress regulation of vascular wall remodeling achieves mechanical equilibrium
Cells continually interpret stress signals from the extracellular environment and strive to establish a preferred stress environment, a phenomenon coined as ‘mechanical homeostasis’. 36 In the arterial wall, the wall-resident cells shield the vascular wall from stress by synthesizing the extracellular matrix, negating the need for continuous cell actin contraction activity, which is energetically advantageous.
Normal arteries rely heavily on adequate mechanical sensitivity and responsiveness for stability. As age progresses and tissue perfusion requirements increase, arteries undergo remodeling accordingly: the arterial diameter gradually enlarges, and the arterial wall thickens to accommodate the rising blood flow and blood pressure during development. Moreover, in pathological conditions, hemodynamics also modulate arterial wall remodeling, exemplified by arterial wall thickening in hypertension and the arterialization of veins following fistula creation. 37 Next, we will discuss how vascular wall cells restructure the arterial wall in response to stress in the context of AAA.
Circumferential stress induces adaptive cellular responses that protect against AAA
In AAA, the circumferential stress borne by the intramural cells increases significantly. SMCs, the most critical cells in the arterial wall, sense the circumferential stress and respond appropriately to maintain the mechanical homeostasis of the artery.38,39 Single-cell sequencing data suggest that in angiotensin-induced aneurysms and dissections, SMCs adaptively overexpress genes involved in wound healing, elastin and collagen production, proliferation, migration, cytoskeleton organization, and cell-matrix adhesion, thereby maintaining the stability of the arterial wall. 40
The cellular experiments also demonstrate that under supraphysiological stretching, SMCs undergo a phenotypic transition from a contractile to a synthetic state, enhancing their proliferative and migratory capabilities. 41 However, cyclic tensile stretching also increases the apoptosis. 42 Recently, Qian et al. employed an ultrasound tweezers-based micromechanical system and discovered that SMCs transition into a mechanically solid-like state under force perturbation, exhibiting decreased sensitivity to mechanical stress. 43 The differences in results may be attributed to varying experimental stress conditions. In the context of complex and individualized AAA wall stress, whether SMCs are to synthesize ECM to stabilize the arterial wall or apoptosis and decreased mechanical sensitivity could be key factors in determining AAA stability.
Given the significant decrease in medial SMCs in AAA, the role of adventitial cells in the remodeling of AAA becomes apparent. In the context of arterial injury, adventitial Sca1+ cells transform into SMCs and migrate to the media, participating in the repair of the arterial wall. 44 In AAAs, due to the attenuation of elastic fibers in the media, the major stress is transmitted to the adventitia and imposed on the fibroblasts. Under mechanical stimulation, the fibroblasts transform into myofibroblasts and increase the ability to migrate, proliferate, and synthesize ECM. 45 With the synthesis and maturation of collagen fibers, the stress is shielded, thus achieving mechanical homeostasis.
Low TAWSS leads to arterial wall instability
Appropriate WSS is crucial for maintaining the function of endothelial cells. However, in the AAA sac, the TAWSS significantly decreases. Low WSS leads to dysfunction of the endothelial barrier, inducing the expression of adhesion molecules and chemokines, while also causing metabolic disorders and increased production of ROS in endothelial cells, all of which contribute to the damage of the arterial wall. 46 Moreover, the disturbed blood flow pattern prolongs the flow residence time, and combined with endothelial cell injury, increases the risk of thrombosis. In turn, intraluminal thrombus with biological activity has a significant impact on the hemodynamics and mechanics of the AAA, as highlighted in the comprehensive reviews conducted by Boyd. 47 In summary, low TAWSS contributes significantly to the instability of AAAs. Consistently and clinically, AAA dilation, rupture, 48 and increased vascular wall permeability 49 often occur in areas with lower TAWSS.
Induction of high WSS through femoral arteriovenous fistula in AAA mouse models results in a proliferation of endothelial cells and SMCs, 50 a reduction in medial macrophage infiltration, 51 and a decrease in adventitial neovascularization, 52 thereby protecting the AAAs. Conversely, femoral artery ligation mice (low WSS) exhibited AAA progression. Based on these findings, the proposal of enhancing lower-limb exercise to improve AAAs was made. However, it is important to note that Sun et al. discovered that lower-limb exercise is potentially associated with the renal artery stenosis of postendovascular aneurysm repair (EVAR) surgery because it decreased the WSS in the renal artery. 53
Stiffness of the ECM aggravates the AAA
During arterial remodeling, the accumulation of collagen fibers leads to significant stiffness of the aorta. 54 In a harder environment, aortic fibroblasts produce more interleukin-6 (IL-6), and their proliferation is inhibited, 55 impairing the repair function of the arterial wall. Similarly, SMCs exhibit cellular senescence, endoplasmic reticulum, and mitochondrial dysfunction in a stiffer ECM.56,57 Furthermore, segmental stiffening of the abdominal aortic wall increases axial stress, exacerbating arterial wall damage and AAA progression. 58 Though arterial wall remodeling by collagen fibers provides strength, it also introduces a stiffer extracellular microenvironment that promotes inflammation and aging.
Mechanical signal transduction: a double-edged sword
As mentioned above, remodeling is essential for maintaining AAA stability. However, the remodeling process of the ECM is accompanied by the infiltration of inflammatory cells, 59 leading to unexpected inflammation. The balance between strength and inflammation relies on appropriate mechanical signal transduction. Abnormal mechanosensing and mechanical regulation are prone to aneurysm lesions. 60 Although the mechanical stresses in the arterial wall are difficult to change, the process of transducing mechanical signals into biological signals can be regulated and potentially serves as a therapeutic target. In this section, we will discuss the mechanical signal transduction and potential therapy targets in SMCs and fibroblasts (Figure 2).
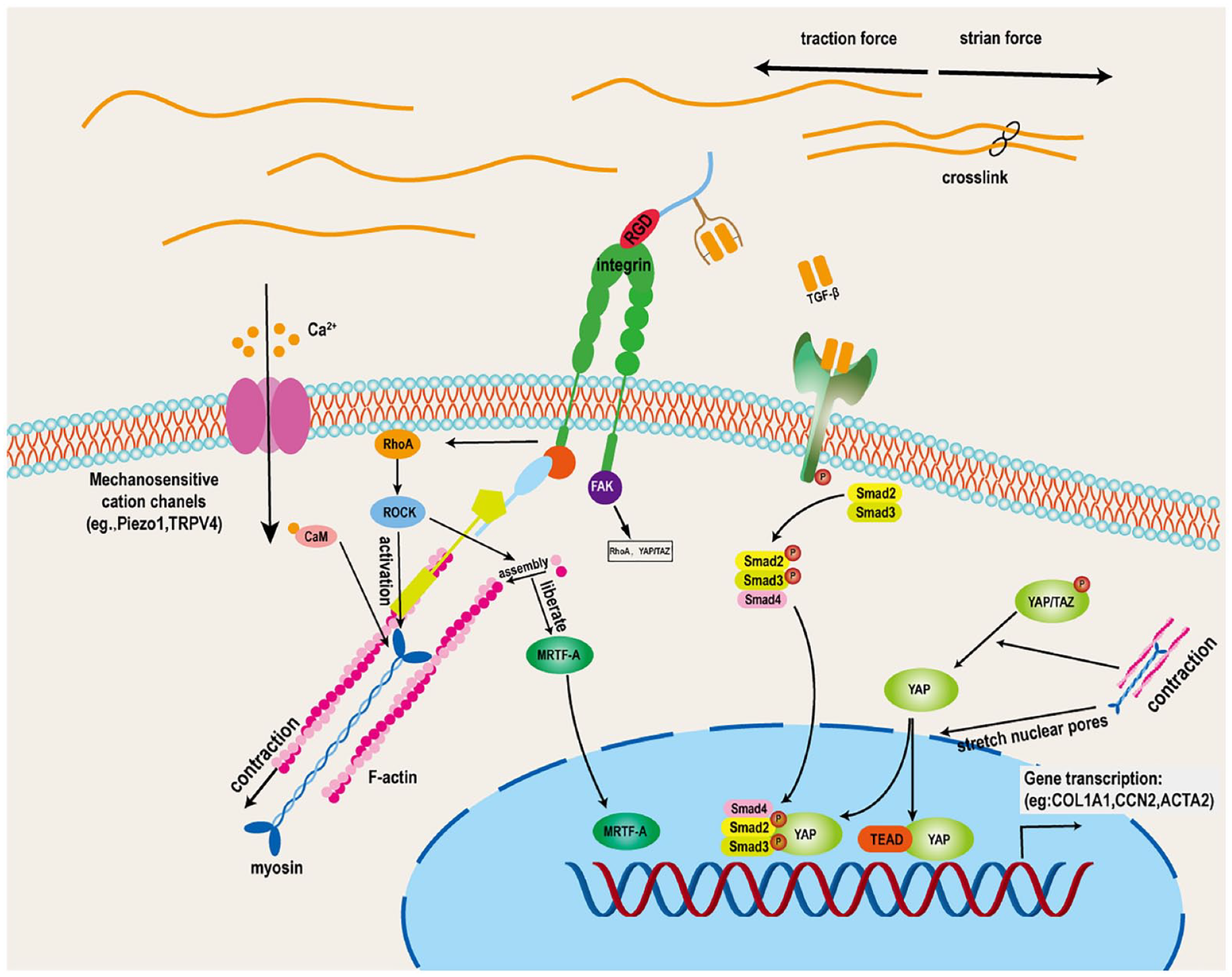
Mechanical signal transduction in fibroblasts and smooth muscle cells. The arginine-glycinea-aspartate (RGD) sequence within the extracellular matrix binds to integrins, linking them to the cytoskeleton through integrin-associated proteins and activating kinases such as focal adhesion kinase (FAK). Following integrin-mediated mechanosignaling transduction, ras homolog family member A (RhoA) RhoA activates Rho-associated kinase (ROCK), facilitating the polymerization of G-actin into F-actin. During this actin assembly process, the transcription factor myocardin-related transcription factor A (MRTF-A) is released, enters the nucleus, and triggers the transcription of remodeling-related genes. Concurrently, RhoA promotes cytoskeletal contraction. The contractile forces and strain on the cytoskeleton stimulate the release of transforming growth factor-β (TGF-β) stored in the extracellular matrix. TGF-β binds to its receptors, leading to the phosphorylation of SMAD2 and SMAD3, which then translocate from the cytoplasm to the nucleus to exert transcriptional regulatory effects. Furthermore, cytoskeletal contraction also facilitates the dephosphorylation and nuclear translocation of PDZ-binding motif (TAZ) and Yes-associated protein (YAP), allowing it to exert transcriptional effects by binding to the transcriptional enhanced associate domain (TEAD) and SMAD. Under mechanical stress stimulation, calcium ion (Ca2+) entry cells through mechanosensitve cation channels such as Piezeo1 and transient receptor potential vanilloid 4 (TRPV4). Then Ca2+ binds to calmodulin (CaM), which activates cytoskeletal contraction.
Integrins connect cells to the ECM
Integrins connect with the arginine-glycinea-aspartate (RGD) sequence of the ECM on one side, and on the other side, they form integrin adhesion complexes (IACs) with the cytoskeleton through a series of proteins such as talin, kindlin, and vinculin, and to kinases like focal adhesion kinase (FAK) and integrin-linked kinase (ILK), thereby linking the cell to its surrounding environment. Under strain force, IACs rapidly assemble and trigger the activation of corresponding enzymes and transcription factors. 61 Once integrin signaling transduction is blocked, the adaptive reaction of the arterial wall is impaired. For instance, legumain (LGMN) blocks the connection between integrins and the ECM, leading to the progression of thoracic aortic dissection in mice. 62 Similarly, the deletion of the ILK gene in SMCs leads to aorta aneurysms in mice. 63 Meanwhile, the excessive activation of mechanical signaling pathways also leads to AAA instability. For instance, overactivation of FAK signaling leads to increased production of monocyte chemoattractant protein-1 (MCP-1) and matrix metalloproteinase-2 (MMP-2); pharmacological inhibition of FAK can reduce inflammation in AAA mouse models. 64 Similarly, in mouse models of Marfan syndrome (MFS), blocking mechanical signaling by inhibiting the integrin αV subunit extends the lifespan of the mice.65,66
Activation of RhoA enhances cell contraction
RhoA, one of the small GTPases, is closely related to the generation of intracellular force. Upon cellular tension stimulation, RhoA transforms from an inactive guanosine diphosphate (GDP) state to an active guanosine triphosphate (GTP) state. GTP-bound RhoA enhances the assembly of myosin and actin filaments by stimulating Rho-associated kinase (ROCK) and promotes myosin adenosine triphosphatase (ATPase) activity, thereby increasing the contractile force exerted by myosin on actin filaments. 67 As monomeric G-actin polymerizes into F-actin, myocardin-related transcription factor A (MRTF-A) is released into the nucleus and binds to serum response factor (SRF), triggering the expression of α-SMA and collagen. Therefore, RhoA activation is essential for ECM adaptive remodeling. Recently, some targets such as nuclear receptor corepressor 1 (NCOR1), 68 small GTP-binding protein GDP dissociation stimulator (SmgGDS), 69 and Polo-like kinase 1 (PLK1) 70 have proven to affect the development and progression of AAA in mice by regulating RhoA activation.
TGF-β signaling contributes to remodeling of arterial walls
TGF-β is synthesized and latently bound to latent TGF-β binding protein (LTBP) in the ECM. Activated cells contract and pull the ECM, which promotes the release of TGF-β. 71 TGF-β binds to its receptors, leading to the phosphorylation of SMAD2 and SMAD3, and then they enter the nucleus from the cytoplasm to exert transcriptional regulatory effects, including the upregulation of collagen-related genes such as COL1A1 and COL1A2. Numerous mice experiments have shown that inhibiting TGF-β and its downstream Smad proteins will lead to the progression of AAA.34,72 Conversely, overexpression of TGF-β can contribute to the stabilization of AAA. 73 Mechanically, the TGF-β signaling pathway plays a role in antiinflammation and promotion of arterial wall remodeling, 74 thereby protecting AAA. However, abnormal activation of the TGF-β pathway is observed in connective tissue disorders prone to aortic aneurysm formation, typified by Loeys–Dietz syndrome (LDS) and MFS. 75
Yes-associated protein (YAP) activation protects against aneurysm development
YAP is a mechanosensitive transcriptional regulator. It enters the nucleus and binds to the transcriptional enhanced associate domain (TEAD) and SMAD to exert its transcription factor function. Under mechanical stress, F-actin polymerizes and contracts, regulating YAP phosphorylation, which influences the rate of YAP influx into and efflux from the nucleus. Meanwhile, the contraction and stretching enlarge the nuclear pores, resulting in increased nuclear entry of YAP. 76 YAP has been proven to be crucial in mechanosignal-mediated fibroblast activation and remodeling in various diseases, including aneurysms and dissection. 40 The knockout of YAP in SMCs leads to AAA in mice. 77 Conversely, overexpression of YAP in SMCs protects against AAA in mice. 78 Therefore, supplementing YAP or promoting YAP into the nucleus may be a potential therapy target for AAA.
Mechanosensitive calcium ion channels exacerbate arterial wall inflammation and damage
Piezo1 and transient receptor potential vanilloid 4 (TRPV4) are mechanosensitive calcium channels that are overactivated in mice with abdominal aortic AAA. The excessive influx of Ca2+ into the cells exacerbates the destruction of the ECM and inflammation of the arterial wall. By inhibiting TRPV4, Peizo can mitigate abdominal aortic AAA in mice.43,79
Biomechanical analyses enhance accuracy of AAA risk prediction
The wall stress undergoes significant change in AAA, which can be estimated using numerical approaches based on imaging data such as ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI). In this way, several patient-specific indices are available and have shown advantages in predicting AAA rupture risk. However, it is noted that approximations and assumptions are necessary in these methods, which may sometimes lead to deviations from actual conditions.
Finite element analysis (FEA) is a well-established method for estimating wall stress. Numerous studies have shown that AAA rupture and symptomatic occurrences are correlated with higher peak wall stress (PWS). 80 The histological result also showed that high-stress regions exhibit more fragile features than low-stress areas identified through FEA. 81 In addition, Li et al. discovered that higher shoulder stress in AAA was associated with the rapid expansion of AAA. 82 However, some reports suggest that the rupture location does not necessarily coincide with areas of high wall stress. 83 A meta-analysis also revealed that it is not PWS but rather the peak wall rupture index (PWRI), which combines wall strength and wall stress, that enhances the accuracy of rupture risk prediction. 84
Doppler ultrasound and phase-contrast MRI can capture hemodynamics information, allowing for the calculation of WSS. Another approach is simulating and obtaining hemodynamic information through mathematical modeling based on vascular geometric models, such as computational fluid dynamics (CFD). Additionally, fluid-structure interaction (FSI) combines flow hemodynamics and arterial deformation data, enabling a more precise estimation of WSS. WSS and WSS-related parameters enhance the accuracy of AAA rupture risk prediction. Regarding the application of WSS and its derivative parameters in AAA, one can refer to the comprehensive review conducted by Mutlu et al. 85
With the remodeling of AAA and the accumulation of collagen fibers, the stiffness of the AAA increases, and its compliance decreases. Pulse wave velocity (PWV) and magnetic resonance elastography (MRE) can be used to assess aortic stiffness, yet the results of current clinical trials remain controversial (Table 2). Most findings suggest that the abdominal aorta in the AAA group exhibits higher PWV. However, some results are negative or contradictory due to confounding factors such as diameter and blood pressure. There are relatively few studies on MRE in AAA. Preliminary study results indicate that MRE can predict the rupture risk of AAA.
Clinical trials to determine the stability of abdominal aortic aneurysms using pulse wave velocity and magnetic resonance elastography.
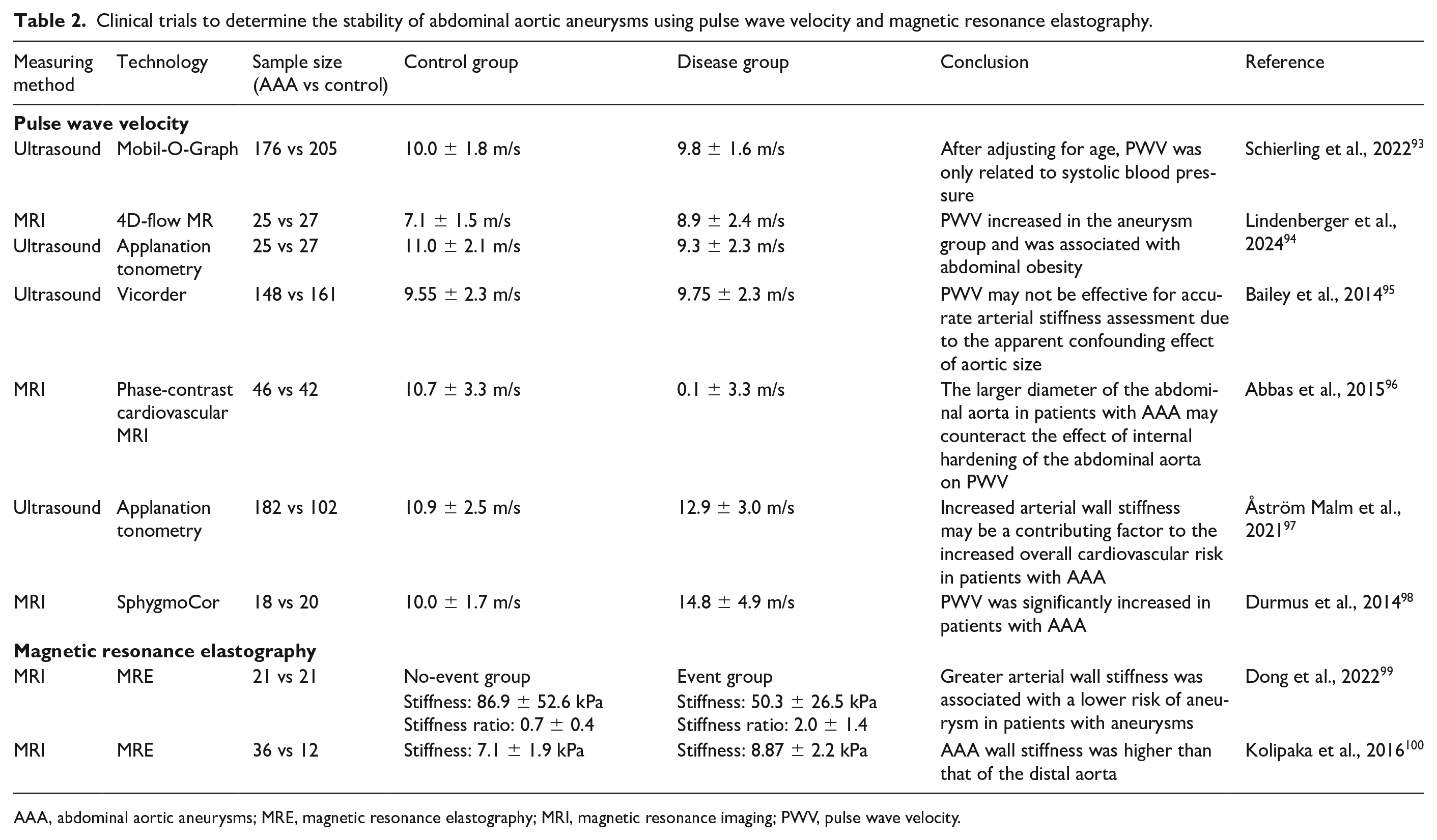
AAA, abdominal aortic aneurysms; MRE, magnetic resonance elastography; MRI, magnetic resonance imaging; PWV, pulse wave velocity.
Arterial compliance can be calculated from ultrasound imaging data, and the linear growth model suggests that compliance is a strong predictor of aneurysm growth. 86 Another study shows that the medium level of pressure-normalized principal wall strain is associated with faster AAA growth. 87 Moreover, the increase in strain heterogeneity may be an indicator of aneurysm instability. 88
Utilizing machine learning models and deep learning to extract features from clinical, morphological, and biomechanical data provides a promising approach to accessing the stability of AAA. 89 Some attempts have yielded encouraging results, and as the volume of data increases, the accuracy of the model’s predictions will improve significantly.90–92
Conclusion
Circumferential stress and WSS are the primary types of stress that the vascular wall experiences, and they exhibit the most significant changes in AAAs, making them the most extensively studied. The collagen fibers bear the majority of the circumferential stress in AAAs. Normal production and cross-linking maturation of collagen fibers provide the basis for resisting stress. Enhancing the cross-linking of collagen fibers in the arterial wall can improve its mechanical and chemical stability, protecting the AAA.
Owing to the destruction of the arterial wall, the wall-resident cells are exposed to abnormal stress environments, activating the mechanical signaling pathway that facilitates the restoration of arterial stability through remodeling of the arterial wall. When the mechanical sensitivity and responsiveness decrease, the repair processes are compromised, leading to the progression of AAA. Regulating the mechanical signal transduction pathways is expected to restore the mechanical reactivity of wall-resident cells and reshape the stability of the arterial wall.
Through biomechanical analyses, the accuracy of AAA rupture risk can be improved, providing better evidence for clinical decision-making. In summary, a better understanding of the role of stress on the AAA wall is expected to address the key issues in the diagnosis and treatment of current AAA.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work received support from the Regional Cooperation Program of Shanxi Province, China (202204041101038); The Leading Talent Team Building Program of Shanxi Province, China (202204051002010); Construction and Demonstration of Molecular Diagnosis and Treatment Platform for Vascular Diseases in Shanxi Province, China (SCP-2023-17); Translational Medicine Engineering Research Center for Vascular Diseases of Shanxi Province, China (2022017); The Shanxi Provincial Science and Technology Department Centralized Guided Local Projects (YDZJSX2021C026); and The National Natural Science Foundation of China (81770695 and 81870354).
