Abstract
Introduction:
As the role of social determinants in carotid artery stenting (CAS) outcomes remains unclear, we investigated the association between the Distressed Community Index (DCI) (zip-code based) and post-CAS mortality/stroke outcomes.
Methods:
We analyzed patients undergoing CAS from 2015 to 2019 using the Medicare claims-linked Vascular Quality Initiative database. Patients were grouped based on high (DCI ⩾ 60) and low (DCI < 60) community distress. We analyzed 36-month mortality using Kaplan–Meier survival curves and hierarchical Cox regression, and 36-month stroke using cumulative incidence function curves and Fine–Gray models.
Results:
The final cohort included 8717 patients (3032 DCI ⩾ 60), with a mean DCI score of 46.2 (± 28.5) and mean age of 74.7 (± 7.8) years. Most participants were men (64.3%), White (92.7%), and non-Hispanic/Latino (97.7%). There was no significant difference in the 36-month mortality incidence between high and low community distress groups (25.6% vs 23.5%, p = 0.22), and no significant association between high community distress and mortality (unadjusted HR: 1.04; 95% CI 0.90–1.21; adjusted HR: 1.02; 95% CI 0.89–1.17). The high community distress group experienced an elevated 36-month stroke incidence (26.8% vs 22.4%, p = 0.048), but no significant association with stroke was observed (unadjusted sub-HR: 1.12; 95% CI 1.00–1.24; adjusted sub-HR: 1.03; 95% CI 0.92–1.16).
Conclusion:
Our cohort showed underrepresentation in terms of sex, race, and ethnicity, with a skewed DCI distribution towards lower community distress. Contrary to what we know about community distress, no independent association between higher community distress and post-CAS stroke/mortality risk was found. Future work must examine whether accessibility barriers and selective CAS allocation explain our results.
Introduction
Approximately 2.7% or 7.6 million Americans over 20 years old have experienced a stroke, with carotid artery stenosis accounting for up to 15% of these strokes.1,2 For selected patients with carotid artery stenosis, carotid artery stenting (CAS) is an alternative to carotid endarterectomy (CEA). 1 Over 10,000 CAS procedures are performed annually in the United States (US), yet there is limited evidence regarding health disparities in patients undergoing CAS. 2
Overall, individuals with carotid artery stenosis face many healthcare disparities both in terms of treatment accessibility and intervention outcomes. Certain groups of patients with carotid artery stenosis, including women, racial/ethnic minority patients, and low-socioeconomic status patients, are particularly at risk of experiencing disparities such as lower carotid revascularization rates, higher likelihood of receiving care from lower-volume hospitals and surgeons, and higher adverse postrevascularization outcomes such as stroke and mortality.3–5 Although the mechanisms behind these disparities are not well understood, they are likely related to social determinants of health, or the environmental conditions in which people are born, grow, live, work, and age. 6 These factors are greatly impacted by community socioeconomic distress, which has been correlated with adverse outcomes in cardiovascular disease.7–9 One community distress metric is the Distressed Community Index (DCI), first established in 2015 by the Economic Innovation Group. 10 DCI has been associated with increased adverse events and worsened mortality after coronary artery bypass grafting (CABG), and major adverse limb events in patients with peripheral artery disease (PAD).8,11,12 However, at present there are no studies assessing the relationship between DCI and adverse outcomes in patients undergoing CAS.
Thus, in this study, we aimed to examine the Vascular Quality Initiative (VQI) Medicare claims-linked database for carotid artery stenosis patients undergoing CAS and (1) describe the patient population who receives CAS intervention, (2) investigate DCI status as a predictor for long-term all-cause mortality, and (3) the overall stroke risk. We hypothesized that higher community distress would be associated with increased risk of all-cause mortality and stroke following CAS.
Methods
Study procedures and patients
Our study included CAS procedures from the Society of Vascular Surgery and American College of Cardiology VQI database, which contains demographic, clinical, procedural, and up to 1-year outcomes data for patients undergoing vascular disease procedures from over 900 centers across North America, Puerto Rico, and Singapore. Our study incorporated demographic factors, insurance status, comorbidities, prior events (such as previous myocardial infarction [MI] and CEA/CAS procedures), procedural data, and imaging variables available within the database (Supplementary Appendix A, available online). The VQI database was linked with national Medicare claims data, generating the Vascular Implant Surveillance and Interventional Outcomes Network (VISION) database housed at Weill Cornell Medicine, Cornell University, NY. This novel registry claims-linked database was designed to improve the quality of the long-term clinical outcomes data. A Center for Medicare and Medicaid Services (CMS) contractor matched VQI records either directly through social security number verification, or indirectly through facility identification, state, patients’ date of birth, sex, procedural dates, and/or zip codes. Identifying and eliminating inaccurate matches was performed through matching procedural codes as well as admission and discharge dates, which has a > 90% sensitivity and a > 99% specificity. 13
Inclusion criteria were: (1) transcarotid artery revascularization (TCAR) and transfemoral CAS index procedures for asymptomatic/symptomatic carotid extracranial artery stenosis between January 1, 2015 and December 31, 2019; and (2) age ⩾ 18 years old. Exclusion criteria were: (1) patients without Medicare fee-for-service coverage; (2) patients who underwent procedures at centers with high missing data rates (defined as average missing data rate > 3rd quartile + 1.5*interquartile); (3) missing physician or hospital site ID; (4) patients who experienced stenting failure or had missing stenting technical outcome; and (5) DCI missing due to missing zip code or unmatched zip code. The Yale University Institutional Review Board deemed this study exempt due to the deidentified nature of the VQI data.
Exposure: Distressed Communities Index
The DCI is a composite metric of socioeconomic well-being at a US zip-code level, and accounts for 99% of the US population through zip codes containing ⩾ 500 residents. For this study, clinical data were linked with DCI status using patients’ five-digit zip code of their mailing address.
DCI uses the following seven metrics to assess a zip code’s socioeconomic status: percentage of adults aged ⩾ 25 years without a high school diploma, percentage of population living under the federal poverty line, percentage of adults aged 25–54 years not currently employed, housing vacancy rate, median household income ratio, change in employment rate, and change in number of business establishments (Table 1). 8 These seven metrics are derived from the US Census Bureau’s Business Patterns and American Community Survey 5-Year Estimates for the 2015–2019 period.
Components of the Distressed Communities Index. 34
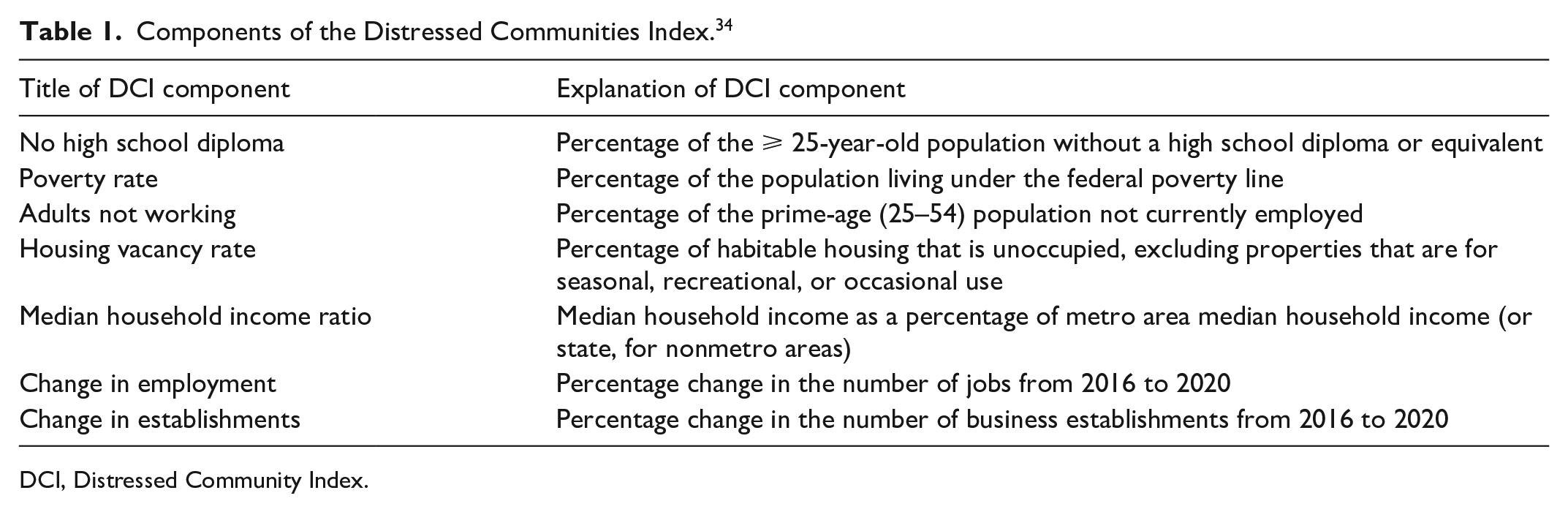
DCI, Distressed Community Index.
As DCI is a continuous score that ranges from 0 (not distressed) to 100 (highly distressed), previous studies have utilized DCI groups, based on national quintiles, or have determined a data-driven DCI value cut-off to create different subgroups.8,9,11,14 The US national-level quintiles have been categorized as: prosperous (quintile 1, [0–20]), comfortable (quintile 2, [20.1–40]), mid-tier (quintile 3, [40.1–60]), at-risk (quintile 4, [60.1–80]), and distressed (quintile 5, [80.1–100]). 15
Outcomes
The 36-month all-cause mortality was derived from Medicare vital status files; the 36-month stroke outcomes were derived from Medicare claims. 16 Stroke outcomes were defined as any peri/postprocedural ipsilateral or contralateral cortical, retinal, or vertebrobasilar ischemic or hemorrhagic stroke based on diagnosis codes and a head computerized tomography (CT) during the same hospitalization. Patient follow-up was 36 months after the index procedure or up to December 31, 2019, or death, whichever occurred first.
Statistical analysis
Patients’ characteristics were described in the overall CAS cohort and by national quintiles of DCI groups. Continuous variables were presented as means with SDs and medians with IQRs, and were compared using Student’s t-test (mean) or Mann–Whitney (median). Categorical variables were presented as frequencies and percentages and were compared using the chi-squared test or Fisher’s exact test where applicable. Patients’ characteristics were also compared by national quintiles of DCI using ANOVA (mean) or Kruskal–Wallis test (median) for continuous variables and using similar tests as described above for categorical variables.
As there is no standardized approach to establishing DCI groups, we categorized patients into two DCI groups: high (DCI ⩾ 60) and low (DCI < 60). We used a time-dependent receiver operating characteristic (ROC) curve for the 36-month risks of both all-cause mortality and stroke. The optimal cut-off DCI point (between 0 and 100 incremented by 5) was then determined using the Youden criterion, which selects the point with the greatest sum of sensitivity and specificity – 1. 17
The 36-month cumulative incidence of mortality by DCI groups was calculated using the Kaplan–Meier method and compared using the Log-rank test. In a preliminary analysis, the association between 36-month mortality and DCI was assessed for linearity using a traditional linear model and a restricted cubic spline with 1 to 4 knots. A linear relationship was observed, as indicated by the lowest Akaike information criterion. Two Cox proportional hazard regression models stratified by site were used to study the association between mortality and DCI. A linear model was used to estimate the hazard ratio (HR) for each 10-point increase in DCI. The second model was used to investigate the association between mortality and DCI group (high or low DCI). The proportional hazards assumptions were met using Schoenfeld residuals. Cox proportional hazard regression models were then adjusted for baseline characteristics listed in Table 2.
Baseline characteristics of the overall complete-case cohort and by national DCI quintiles.
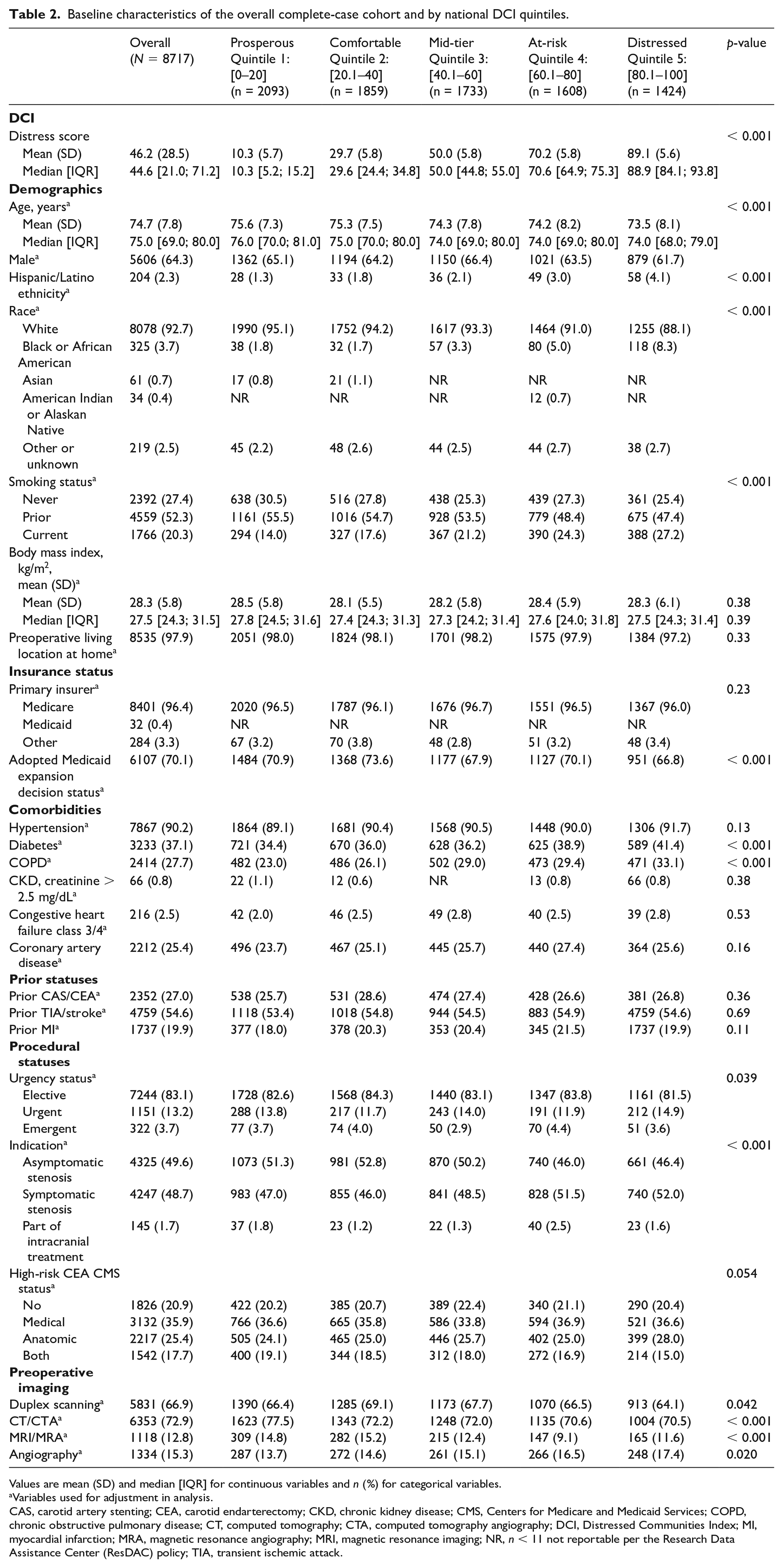
Values are mean (SD) and median [IQR] for continuous variables and n (%) for categorical variables.
Variables used for adjustment in analysis.
CAS, carotid artery stenting; CEA, carotid endarterectomy; CKD, chronic kidney disease; CMS, Centers for Medicare and Medicaid Services; COPD, chronic obstructive pulmonary disease; CT, computed tomography; CTA, computed tomography angiography; DCI, Distressed Communities Index; MI, myocardial infarction; MRA, magnetic resonance angiography; MRI, magnetic resonance imaging; NR, n < 11 not reportable per the Research Data Assistance Center (ResDAC) policy; TIA, transient ischemic attack.
Stroke risk may be overestimated by the Kaplan–Meier method and Cox proportional hazard regressions due to the competing risk of all-cause mortality. 18 To take the competing risk of death into account, competing risk methods were used to assess the association between the 36-month risk of stroke and DCI. The 36-month cumulative incidence function of stroke was calculated using the Aalen–Johansen estimator by DCI groups and were compared using Gray’s test. 19 Preliminary analysis using restricted cubic spline models with the number of knots between 1 and 4 similarly revealed a linear relationship between 36-month stroke and DCI (lowest Akaike information criterion). Sub-distribution hazard ratios (sHR) of stroke at 36 months were then estimated using the Fine–Gray competing risk model for a 10-point increase in DCI and for DCI group. 19 These models were also then adjusted for baseline characteristics listed in Table 2.
A sensitivity analysis accounting for missing data was conducted on an imputed cohort using multiple imputation by the chained equations method generating 10 imputed datasets. 20 Analyses were then replicated in the imputed dataset and results were pooled using Rubin’s rule. 21 Statistical analysis was performed with Stata Statistical Software, Release 17 (StataCorp LLC, College Station, TX, USA). In all analyses, p-values were reported for two-tailed tests with an alpha of 0.05. Sample sizes smaller than 11 were masked to maintain confidentiality per the Research Data Assistance Center (ResDAC) policy.
Results
Patient demographics
The overall complete-case counts included 8717 patients. The cohort derivation flow chart is shown in Figure 1, and patient characteristics by national DCI quintiles are presented in Table 2. For the cohort, the mean age was 74.7 ± 7.8 years, 64.3% were men, and 2.3% were Hispanic/Latino. The racial breakdown was 92.7% White, 3.7% Black or African American, 0.7% Asian, 0.4% American Indian or Alaskan Native, and 2.5% other or unknown. For prior statuses, 27.0% of the cohort had previously undergone either CAS or CEA, 54.6% had previously experienced a stroke/transient ischemic attack (TIA), and 19.9% had previously experienced an MI. For outcomes, a total of 988 (11.3%) patients died and 1396 (16.0%) patients experienced a stroke.
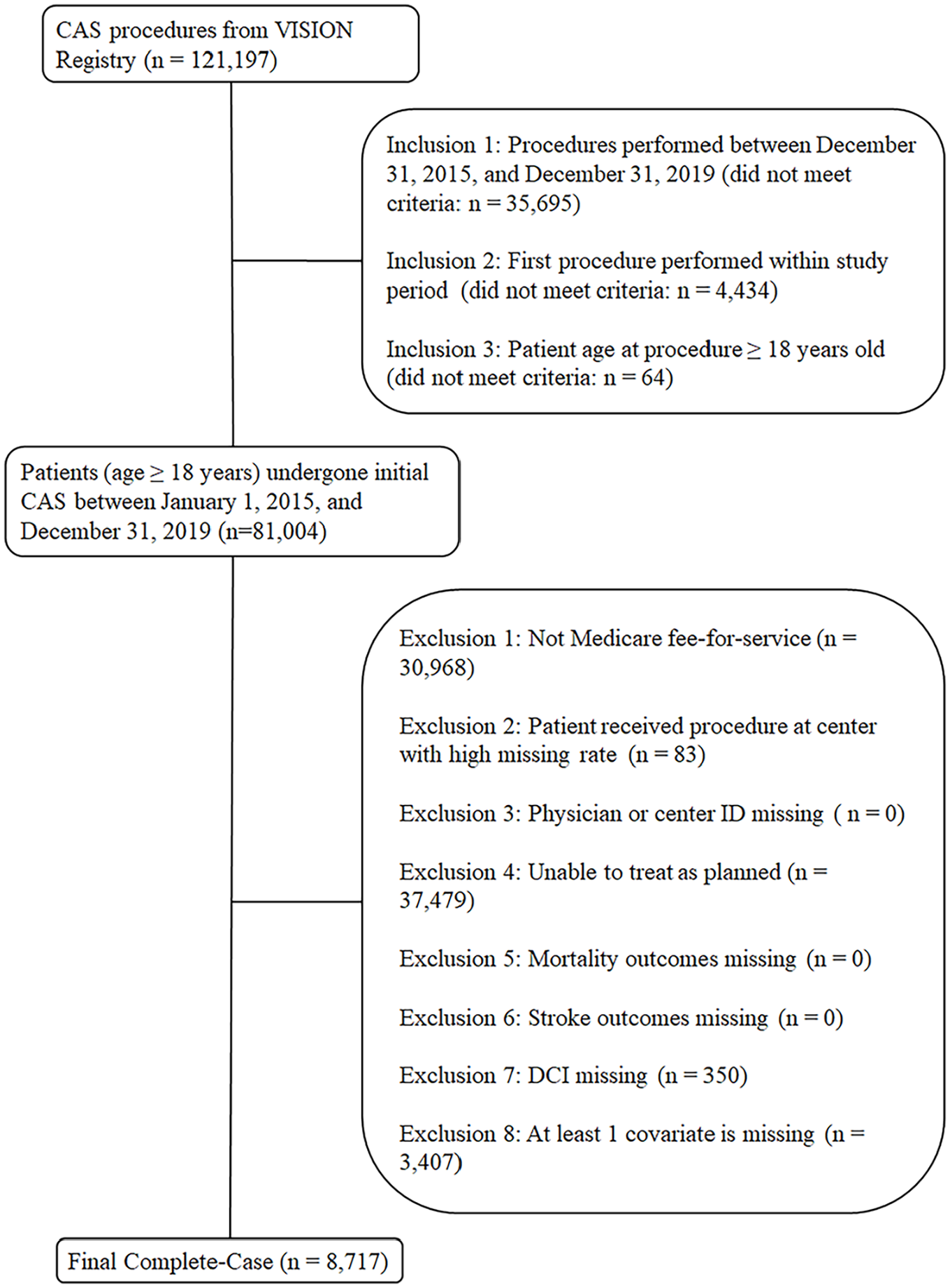
Derivation of study cohort from patients enrolled in the Vascular Quality Initiative linked to Medicare claims data who underwent carotid artery stenting between January 1, 2015 and December 31, 2019.
Patient demographics varied between higher and lower community distress national quintiles. Higher community distress quintiles had higher proportions of Black or African American patients (p < 0.001), as well as Hispanic/Latino patients (p < 0.001). Patients in higher community distress quintiles were more likely to be smokers (p < 0.001), live in a state that did not adopt Medicaid expansion (p < 0.001), and have several pertinent comorbidities such as diabetes, chronic obstructive pulmonary disease (COPD), and prior CABG (p < 0.001, p < 0.001, p = 0.014, respectively). These higher proportions in the prevalence of comorbidities were especially prevalent when comparing the lowest and highest DCI quintiles (p < 0.001, p < 0.001, p = 0.001, respectively).
The median DCI score was 44.6 (IQR: 21.0–71.2), with statistically significant differences between certain demographics. Specifically, female patients had a higher mean DCI score than male patients, Black patients had a higher mean DCI score than White patients, and Hispanic/Latino patients had a higher mean DCI score than non-Hispanic/Latino patients (Figure 2; p = 0.022, p < 0.001, and p < 0.001, respectively). Per national DCI quintiles, 2093 were classified as ‘prosperous’ (DCI quintile 1 (Q1)), 1859 were ‘comfortable’ (DCI Q2), 1733 were ‘mid-tier’ (DCI Q3), 1608 were ‘at-risk’ (DCI Q4), and 1424 were ‘distressed’ (DCI Q5). The median follow-up time was 27.5 months (IQR: 12.3–49.6) and 5.0% of patients were lost to follow-up.
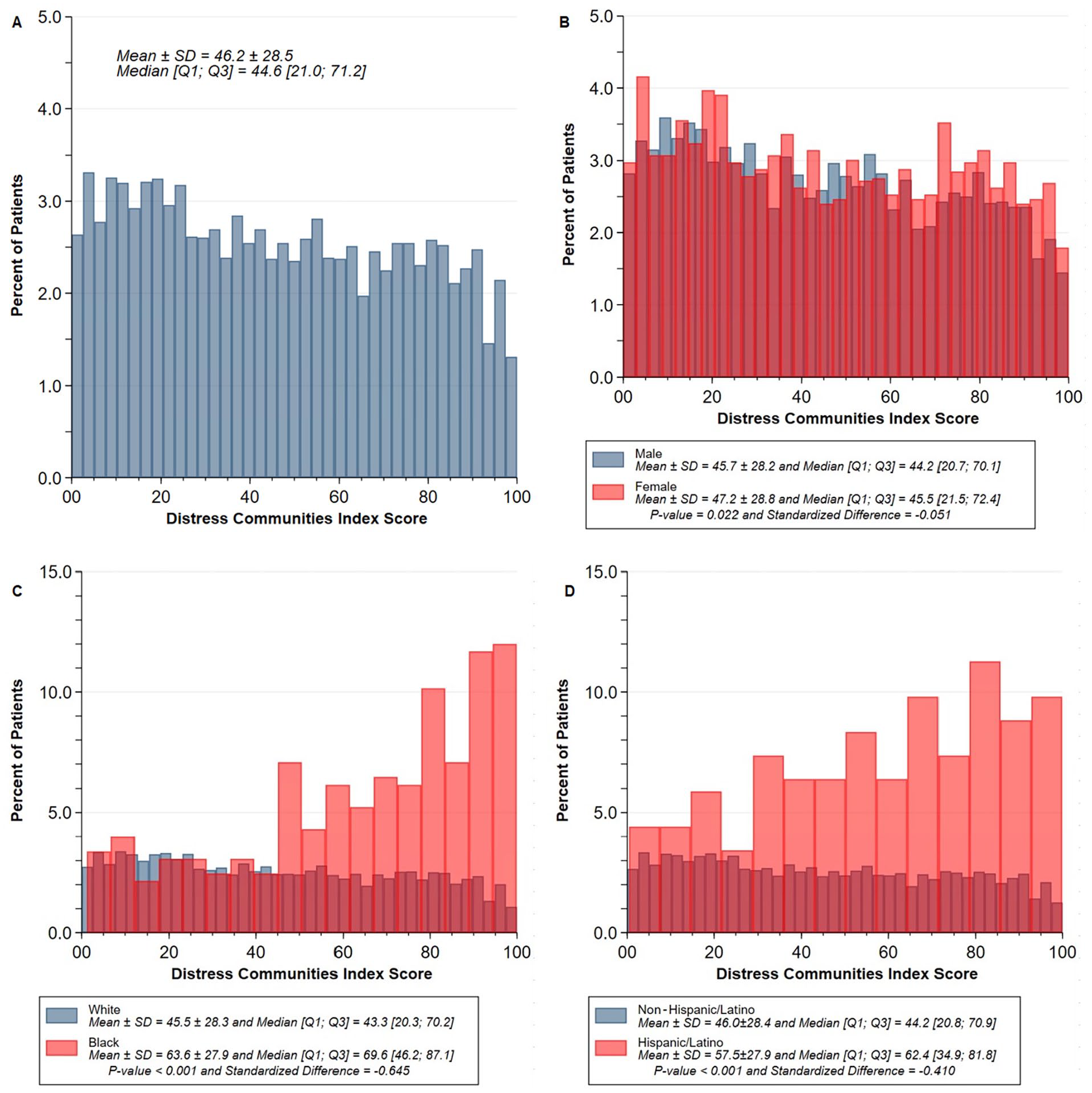
Distribution of DCI for
Patients were categorized into low (n = 16,890) and high (n = 17,019) DCI groups, respectively < 60 and ⩾ 60, based on Youden criterion (36-month mortality sensitivity: 0.33, specificity: 0.71; 36-month stroke sensitivity: 0.36, specificity: 0.72).
DCI and mortality
The 36-month mortality cumulative incidence curves by DCI group are presented in Figure 3. The mortality risk at 36 months, between high (DCI ⩾ 60) and low community distress patients (DCI < 60) was not significantly different (25.6% vs 23.5%; p = 0.22). There was no significant association between high community distress (DCI ⩾ 60) and mortality, even after adjusting for all patient characteristics in Table 2 (unadjusted HR: 1.04; 95% CI 0.90–1.21; adjusted HR: 1.02; 95% CI 0.89–1.17). Similarly, a 10-point increase in DCI was not significantly associated with a 36-month mortality risk (Figure 4; unadjusted HR: 1.00; 95% CI 0.97–1.02; adjusted HR: 0.99; 95% CI 0.97–1.02) even after multiple imputation (adjusted HR: 0.99; 95% CI 0.97–1.01). These results remained consistent after multiple imputation (adjusted HR: 0.99; 95% CI 0.97–1.01) with missing data imputed ranging from 0 to 0.3% for congestive heart failure.
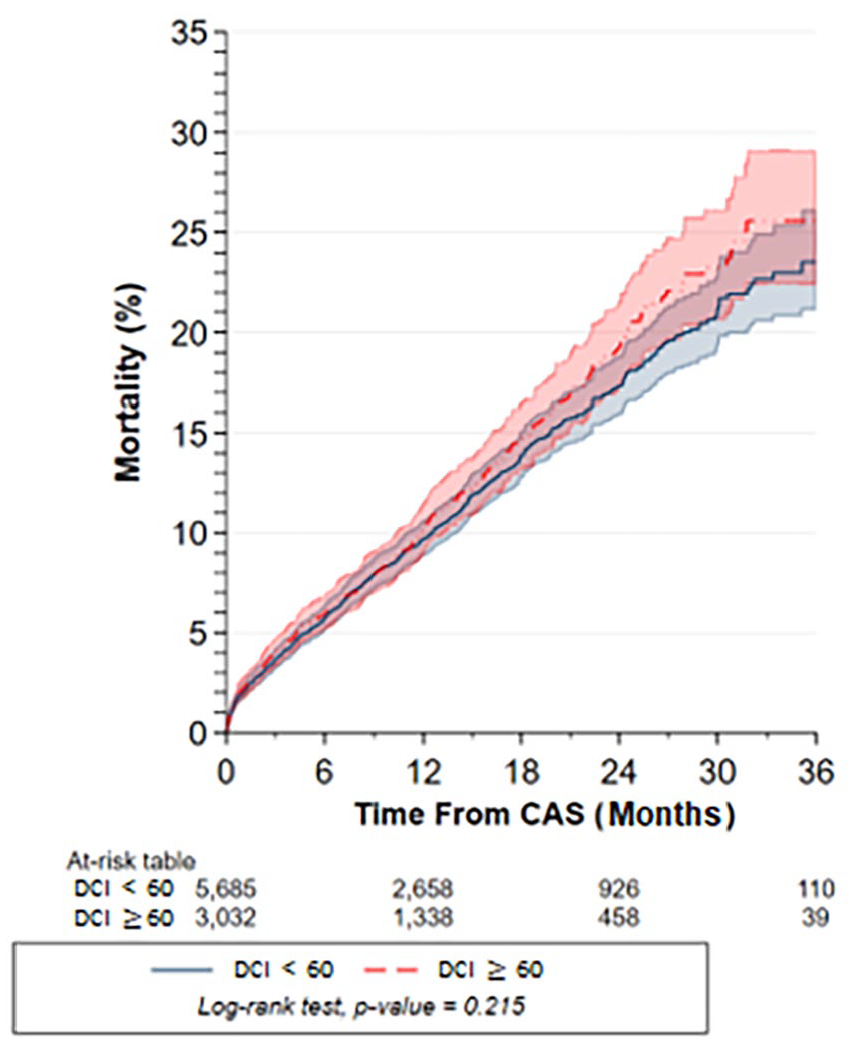
Cumulative risk of all-cause mortality up to 36 months after CAS by DCI groups (DCI < 60 [prosperous to mid-tier] vs DCI ⩾ 60 [at-risk to distressed]) using Kaplan–Meier estimators and compared using the Log-rank test.
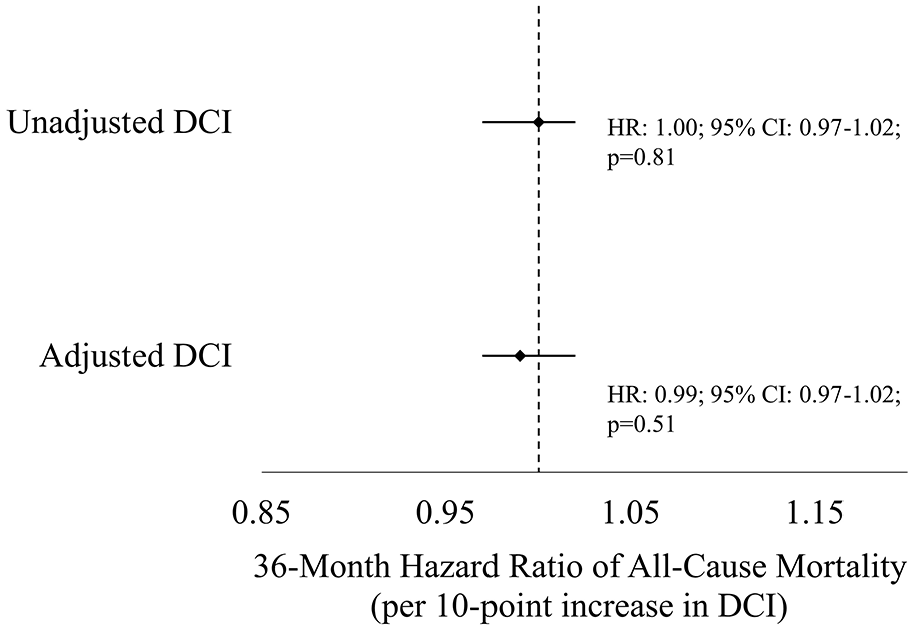
Unadjusted and adjusted association of a 10-point increase in DCI with 36-month mortality using the Cox proportional hazard regression model.
DCI and stroke
The 36-month stroke cumulative incidence curves are presented in Figure 5. At 36 months, high community distress patients (DCI ⩾ 60) experienced higher stroke risk compared with low community distress patients (26.8% vs 22.4%; p = 0.048; unadjusted sHR: 1.12; 95% CI 1.00–1.24). After adjusting for patient characteristics, this association became nonsignificant (adjusted sHR: 1.03; 95% CI 0.92–1.16). A 10-point increase in DCI was also not significantly associated with a 36-month stroke risk (Figure 6; unadjusted sHR: 1.01; 95% CI 0.99–1.03; adjusted sHR: 1.00; 95% CI 0.98–1.02). These results remained consistent after multiple imputation (adjusted sHR: 1.01; 95% CI 0.99–1.02).
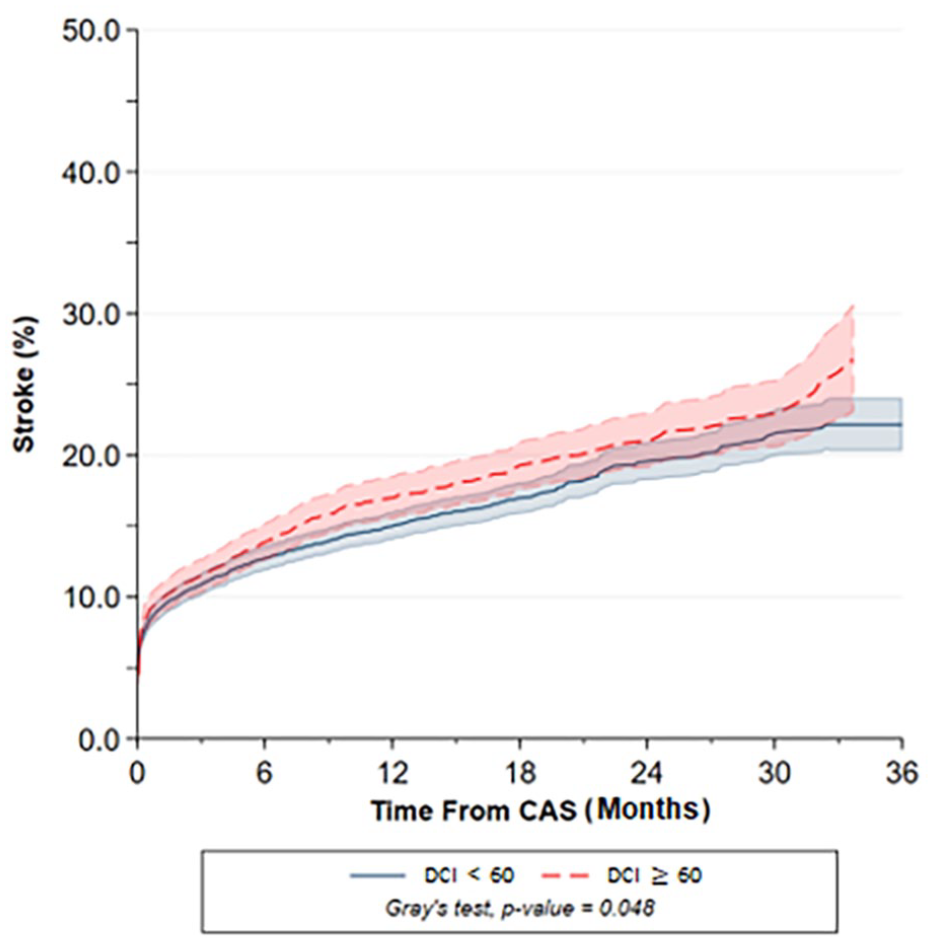
Cumulative risk of stroke up to 36 months after CAS by DCI groups (DCI < 60 [prosperous to mid-tier] vs DCI ⩾ 60 [at-risk to distressed]) using the Aalen–Johansen estimator and compared using Gray’s test.
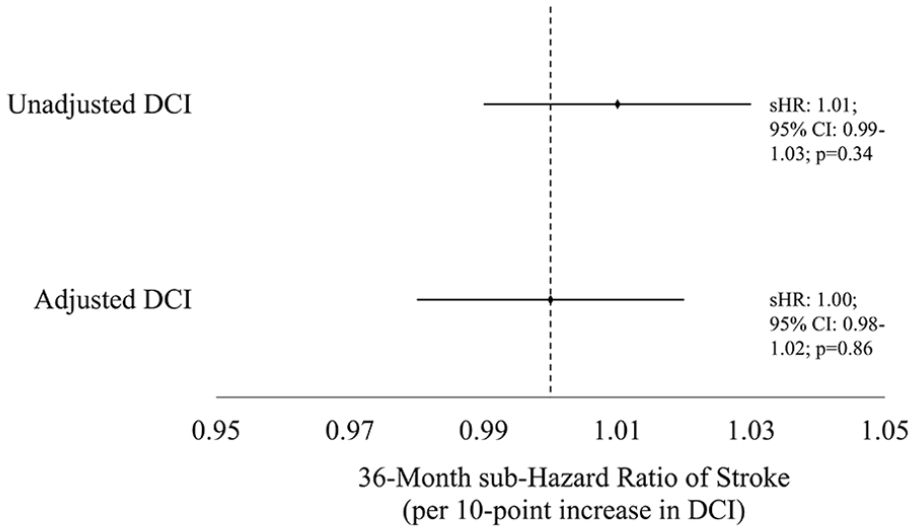
Unadjusted and adjusted association of a 10-point increase in DCI with 36-month stroke using the Fine–Gray competing risk regression model taking all-cause mortality into account.
Discussion
Our study revealed that the predominant demographics in this large, multicenter post-CAS cohort were non-Hispanic/Latino, White men, and skewed towards lower community distress. Though higher DCI stress was associated with a higher risk of 36-month stroke, the relationship was not maintained after adjusting for relevant confounders. No association between high community distress and mortality could be established either. Over nine out of 10 patients were White or non-Hispanic/Latino, and both the mean and median DCI (46.2 and 44.6, respectively) fell within the range of low community distress (DCI < 60), underscoring the select nature of individuals who get access to CAS.
Our study’s findings are limited due to the underrepresentation of certain demographic groups, but our identification of this discrepancy is a first step to addressing and investigating the cause of this gap. Our cohort diverges significantly from national demographics: our cohort was only 35.7% women, 3.7% African American or Black, 2.3% Hispanic, and 0.7% Asian, compared to higher national prevalences. 22 This discrepancy prompts an inquiry into whether the prevalence of carotid stenosis is lower in racial/demographic minorities or if it reflects the known healthcare accessibility issues faced by these groups. One study, where participants self-referred and personally funded the required vascular testing, reported lower rates of carotid artery stenosis in African American and Hispanic patients, but other studies have indicated higher stroke risk and comorbidities in these groups, as well as lower revascularization rates when asymptomatic.23–27 Although the skewed demographic representation limits the generalizability of our study, it provides concrete data on this discrepancy and underscores the need for further investigations into what the root causes of these selection biases consist. Such investigations could involve assessing the true prevalence of carotid artery stenosis in different racial/ethnic groups and exploring potential healthcare barriers minorities face before intervention. Additionally, we can further investigate the relationships between DCI, race/ethnicity, and healthcare accessibility within carotid artery stenting populations.
Our study reflects the systemic healthcare barriers that prevent underrepresented communities from accessing necessary care, making DCI less effective in identifying disparities within skewed demographic populations. Though previous studies have found a positive association between higher community distress and worse revascularization outcomes, our findings, along with two other studies, suggest that DCI is not a reliable predictor for adverse outcomes within a cardiovascular population.8,9,11,12,14,28 Notably, one of the studies that did not find an association between DCI and adverse outcomes examined patients undergoing transcatheter aortic valve replacement (TAVR) in one of the most affluent areas in New York. Here, the authors concluded that access to care might be a key factor contributing to this disparity and subsequently they were unable to study the truly distressed patient population. Similarly, our results may have been influenced by this same limitation given our racially/ethnically/socioeconomically skewed population. In addition to uniquely identifying the demographic skew in patients undergoing CAS within a Medicare database, our study further demonstrates how a racially/ethnically/socioeconomically nonrepresentative cohort impacts DCI as a predictor metric for adverse cardiovascular outcomes. Future research should investigate healthcare barriers faced by racial/ethnic minorities and lower-income populations to improve their access to CAS. By identifying accessibility barriers, we can work towards extending these therapies to diverse minority populations. Our findings thus underscore the importance of diversity in real-world cohorts and investigating potential intervention accessibility issues non-White minorities may encounter.
This study has uniquely furthered our current knowledge of patient representation in those undergoing CAS in terms of their demographics and indicators of social determinants of health. With a larger sample size of over 8000 participants, our study surpasses the scale of most studies examining DCI and revascularization outcomes, improving our study’s statistical power as well as the overall US population’s representation. Utilizing a national database also ensures geographic diversity, offering representation of diverse population structures (i.e., rural vs urban) and state policies (i.e., Medicare expansion). Additionally, by capitalizing on longitudinal clinical outcomes data from Medicare records, we gain valuable insights into the long-term implications of CAS as well as socioeconomic status, frequently a chronic lifetime exposure. This approach underscores the importance of leveraging existing large-scale healthcare databases to explore complex health disparities for lifesaving medical interventions. By utilizing the Medicare outcomes data, a longitudinal and data-rich source, our study has the unique ability to analyze patient outcomes over time, capturing their experiences as they return to their communities and navigate varying levels of community distress.
Considering our findings, it is crucial for clinicians, policymakers, and healthcare administrators to address cohort diversity and CAS accessibility. With CMS proposals advocating for expanded CAS coverage, it is essential to assess the demographics of patients currently accessing this intervention and identify who can benefit. 29 Clinicians should also engage in shared decision-making with racial/ethnic minority patients eligible for carotid revascularization and address individual barriers to treatment. In addition, hospital systems and policymakers can initiate CAS outreach in underserved communities, improve screening for comorbidities, increase public transportation, and eliminate food deserts.30–32 As patients from underrepresented groups undergoing CAS also have higher stroke risk, it is essential to take additional proactive measures. Thus, both clinicians and systems-level experts can improve cardiovascular screening programs for vulnerable communities, increase cardiovascular health education and awareness campaigns to improve understanding of stroke risk factors and symptoms, and strengthen preventative healthcare services in these communities. By addressing these issues at both individual and systemic levels, healthcare providers can improve patient diversity and promote equity in treating carotid artery stenosis.
Limitations
This study has several limitations. First, as the DCI metric and long-term outcomes were derived from Medicare data, our analysis was limited to patients receiving fee-for-service Medicare and thus may not be generalizable to uninsured patients or patients under 65 years old. Investigating these patient populations is essential as they often have elevated risk for peripheral vascular disease. Previous data have shown that younger patients with severe peripheral vascular disease are more likely to be uninsured or have a psychiatric disorder. 33 Additionally, uninsured/Medicaid patients with carotid artery stenosis are less likely to receive carotid revascularization procedures compared to privately insured counterparts. 27 Thus, it is essential that future studies explore these patient populations to provide a more comprehensive understanding of carotid artery stenosis treatment outcomes.
In addition, several other limitations must also be considered. Next, our study only involved patients undergoing CAS; thus, this study does not analyze healthcare accessibility disparities prior to CAS intervention. Third, DCI scores are assigned at a community-level via zip codes, and thus a patient’s assigned DCI score may not correlate to their individual situation. Fourth, the stroke outcome included all forms of stroke (i.e., ipsilateral or contralateral cortical, retinal, or vertebrobasilar ischemic or hemorrhagic stroke), potentially broadening the scope of adverse events beyond those directly associated with CAS intervention. Additionally, the stroke outcome data within the Medicare database is not currently validated, so future efforts should focus on validating these outcomes. Finally, the DCI metric, though expansive, may not capture every socioeconomic disparity a community may face. To address these limitations, future studies can explore additional patient populations as well as investigate the association between individual socioeconomic status and adverse outcomes following CAS.
Conclusion
In this predominately non-Hispanic/Latino, White, male cohort, high community distress was not independently associated with mortality, or stroke after CAS. We observed that only a highly select group of individuals, in terms of their demographics and reduced exposure to community distress, preferentially get CAS, which may explain a lack of association between community distress and adverse outcomes otherwise observed in other cardiovascular populations. Further awareness of barriers in access to care and preventive strategies, as well as newer evidence on the comparative effectiveness of carotid artery stenosis management, that can be shared in a patient-centric way are ways to address the potential healthcare disparities that exist in CAS.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241292545 – Supplemental material for Lack of diversity in patients undergoing carotid artery stenting: Implications for the Distressed Community Index
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241292545 for Lack of diversity in patients undergoing carotid artery stenting: Implications for the Distressed Community Index by Carmen Pajarillo, Gaëlle Romain, Jacob Cleman, Lindsey Scierka, Scott Grubman, Christopher Schenck, Jonathan Kluger, Kim G Smolderen and Carlos Mena-Hurtado in Vascular Medicine
Footnotes
Acknowledgements
We gratefully acknowledge the invaluable contributions of the authors and other individuals involved in this project. We also gratefully extend this deep-hearted appreciation to our study participants, whose contributions made this research possible.
Data availability statement
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Mena-Hurtado reports unrestricted research grants from Philips, Shockwave, Abbott, and Merck; he is a consultant for Abbott Vascular and Cook. Dr Smolderen reports unrestricted research grants from Philips, Shockwave, Abbott, and Merck; she is a consultant for Cook and Happify. The remaining authors have no conflicts of interest.
Funding
This publication was supported by the Richard K Gershon, MD, Endowed Medical Student Research Fellowship at the Yale School of Medicine.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
