Abstract

Keywords
It is well-established that coronary artery disease (CAD) is associated with increased perioperative risk in patients undergoing open surgical revascularization for peripheral artery disease (PAD).1,2 Subclinical coronary atherosclerosis (SCA) or incidental coronary artery calcification (CAC) is frequently detected on cross-sectional chest imaging, and incidentally discovered on up to 80% of chest computed tomography (CT) images, although reported in only 25% of imaging reports. 3 Though CAC is an established predictor for long-term cardiovascular events and associated mortality,4–7 its implications for perioperative cardiovascular risk are not well understood. However, small studies suggest that SCA may be as potent as extant CAD for perioperative morbidity and mortality.8,9 We sought to describe baseline characteristics and perioperative outcomes in patients with CAC undergoing surgical revascularization for the indication of PAD.
The Dartmouth-Hitchcock institutional review board approved the study. A retrospective chart review was completed at a single academic institution from 2009 to 2020 using the Vascular Quality Initiative (VQI) database. Patients undergoing supra- and infra-inguinal bypass surgery were included. Retrospective review identified patients with chest CT within 1 year prior to revascularization to determine the presence of CAC. Patients were then categorized: no coronary atherosclerosis, SCA (defined by the presence of CAC by two-physician review of imaging [SS and SH]), and clinical CAD (defined by history of myocardial infarction or coronary revascularization). Categorization occurred based on physician imaging review and was independent of imaging reports. Comorbidities and perioperative outcomes (troponin elevation [myocardial injury; type 1 or 2 myocardial infarction], arrhythmia, heart failure, all-cause mortality) during hospitalization were then compared among groups using either chi-squared or ANOVA tests, as appropriate.
Of 1018 patients undergoing peripheral artery bypass surgery, 199 (20%) had a prior CT available for review and were included in the analysis (Table 1). Of these, SCA was noted in 96 (48%) patients and 72 (36%) patients had clinical CAD. When compared, patients had similar preexisting comorbidities except for hypertension and heart failure, which occurred more frequently in patients with clinical CAD. Individuals with no coronary atherosclerosis and SCA were less likely to be prescribed a beta-blocker, P2Y12 inhibitor, or statin therapy preoperatively compared to patients with clinical CAD. In this retrospective study of patients undergoing open peripheral vascular surgery, we found that postoperative outcomes were similar in patients with SCA compared to clinical CAD. These included troponin elevation (13% in the SCA group and 10% in the clinical CAD group) and heart failure (9% in the SCA group and 6% in the clinical CAD group). These findings were different when compared to those with no coronary atherosclerosis (Table 1). Patients with SCA did have numerically higher postoperative arrhythmia events and in-hospital mortality when compared to patients both with no coronary atherosclerosis and with clinical CAD, though these comparisons did not reach statistical significance.
Baseline characteristics and outcomes based on coronary atherosclerosis (N = 199).
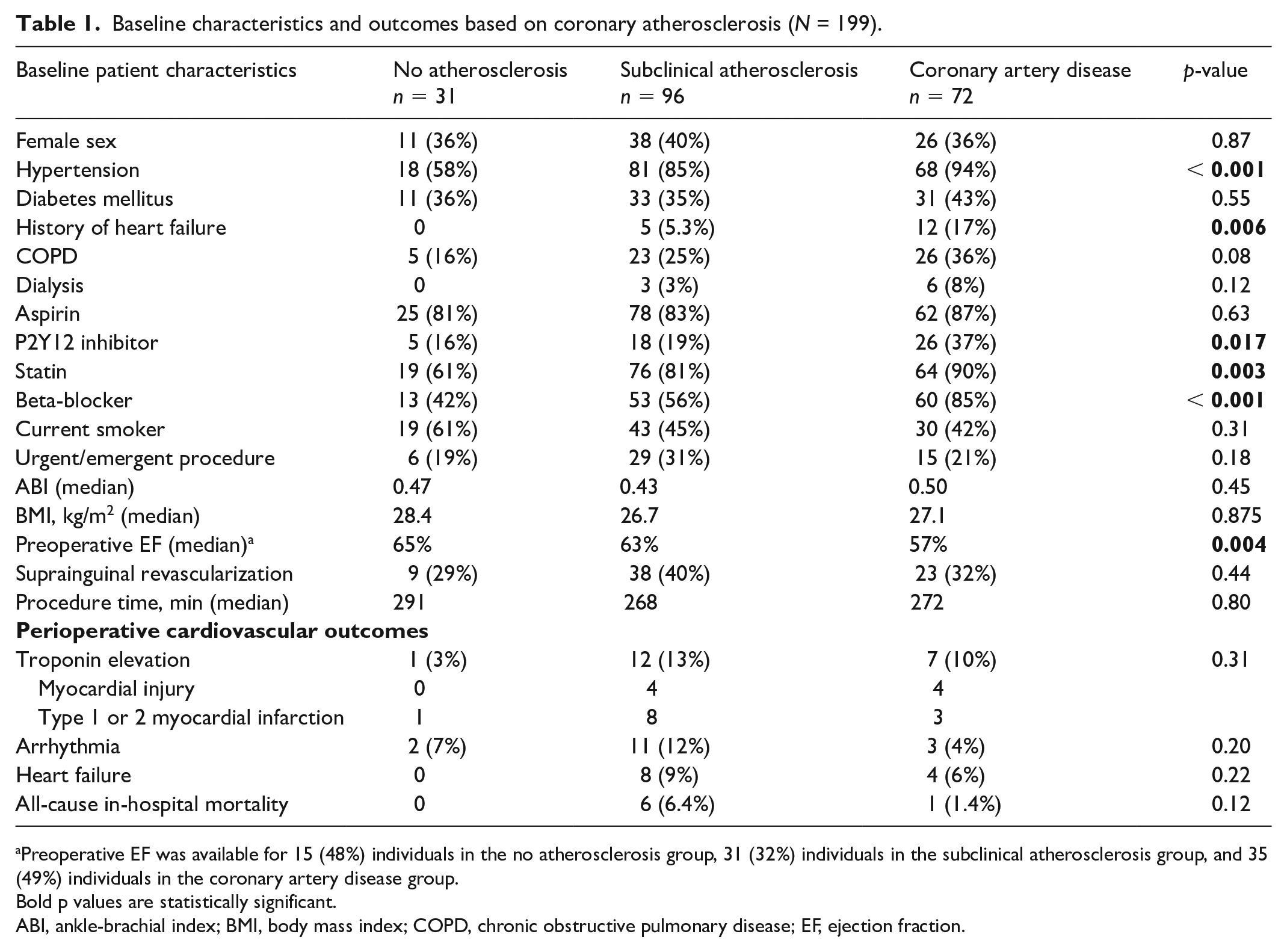
Preoperative EF was available for 15 (48%) individuals in the no atherosclerosis group, 31 (32%) individuals in the subclinical atherosclerosis group, and 35 (49%) individuals in the coronary artery disease group.
Bold p values are statistically significant.
ABI, ankle-brachial index; BMI, body mass index; COPD, chronic obstructive pulmonary disease; EF, ejection fraction.
These findings represent a twofold problem: the undertreatment of cardiovascular risk factors in a high-risk cohort and, by extension, suboptimal perioperative risk modification in patients prior to surgical revascularization. Despite all patients in our study having a strong indication for statin therapy due to the presence of severe PAD (median ABI of 0.43–0.5), only 61% of patients with no coronary atherosclerosis were prescribed statins (and 81% in those with CAC). This highlights the medical undertreatment of patients with PAD. 10 Secondly, this is also important when considering perioperative risk factor modification. The 2014 ACC/AHA guideline on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery recommend continuation of statins in patients previously prescribed statins (Class I recommendation) and consideration for statin initiation in all patients undergoing vascular surgery (Class IIa).11,12 Our findings suggest that despite all patients with a preexisting indication for statins, there was a substantial number of patients who did not receive statin therapy as part of their cardiovascular risk reduction prior to surgery.
Similarly, the 2014 ACC/AHA perioperative guidelines support continuation of beta-blockers in those with preexisting indications (Class I) and consideration for beta-blocker initiation in patients with three or more Revised Cardiac Risk Index (RCRI) factors (Class IIb). 11 Current RCRI scoring only includes clinical CAD, such as prior myocardial infarction or ischemia. However, given the similar cardiovascular outcomes between patients with SCA and clinical CAD undergoing revascularization for PAD, further investigation is needed to determine if SCA should be considered an equivalent cardiac risk factor in the RCRI preoperative risk assessment that necessitates equally aggressive risk factor modification.
There are some important limitations of this study. First, given its single-center retrospective nature, the study is limited by small sample size and is thus likely underpowered to reach statistically significant conclusions. We evaluated for presence of cross-sectional chest imaging in the 1 year preceding vascular surgery, which also resulted in a smaller cohort. Other studies may consider expanding preoperative imaging to a longer period (i.e., 3–5 years) to enlarge the cohort and further understand the implications of incidental SCA. Certainly, coronary artery calcium scoring may be considered in such scenarios as well, and its use in perioperative risk assessment prior to major vascular surgery. An additional limitation is that our study reviewed prescription history as a surrogate for risk factor modification. This does not account for patient compliance, indicate if risk factor goals were met prior to surgery, or clarify if patients were prescribed alternative medications for risk factor modification due to statin intolerance (i.e., proprotein convertase subtilisin/kexin type 9 inhibitors). Further studies are needed to evaluate the impact of cardiovascular risk factor optimization on outcomes for patients with subclinical coronary disease. Finally, owing to granularity of VQI data, we were unable to differentiate between type 1 and type 2 myocardial infarction. Although characterization of myocardial injury and infarction is important for treatment considerations, both myocardial injury and type 2 myocardial infarction carry a significant risk of mortality. 13
In summary, our findings support that the mere presence of coronary calcification, even if asymptomatic, signals increased risk for perioperative cardiovascular events that parallel clinical CAD. Though future studies are needed to better understand this cohort, our study supports the need for aggressive secondary prevention measures in patients undergoing surgical peripheral artery revascularization with specific attention to cardiovascular risk modifying strategies.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
