Abstract
Coronary artery disease (CAD) patients with concomitant peripheral artery disease (PAD) experience more extensive and calcified atherosclerosis, greater lesion progression and more common coronary events compared to patients with CAD only. To characterize the distinct features of this aggressive atherosclerotic disease, we studied novel cytokines that code different stages of atherogenesis. One hundred and eighty consecutive subjects (60 patients into each group of CAD+PAD, CAD and controls) were recruited among patients with stable angina pectoris scheduled for coronary angiography. An ankle–brachial index (ABI) ≤0.9 was determined as occlusive PAD. Fasting serum tumor necrosis factor (TNF)-like antigen 1A (TL1A) and its receptor death receptor 3 (DR3), NOGO-B (reticulon 4B) and its receptor NUS1, high-sensitivity C-reactive protein (hsCRP), A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) 1, 4, 5 and interleukin (IL) 6 levels were determined. Serum hsCRP and DR3/TL1A concentrations were similar and higher than controls in the CAD and CAD+PAD groups. Levels of NOGO-B and its receptor NUS1 were increased and ADAMTS-5 was decreased in patients with CAD+PAD. Independent predictors of ABI in multivariate analysis were smoking (B = −0.13, p = 0.04), NUS1 (B = −0.88, p < 0.001), ADAMTS-5 (B = 0.63, p < 0.001) and SYNTAX score (B = −0.26, p < 0.001). Similarly, smoking (OR = 5.5, p = 0.019), SYNTAX score (OR = 1.2, p < 0.001), NUS1 (OR = 14.4, p < 0.001), ADAMTS-5 (OR = 1.1, p < 0.001) and age (OR = 1.1, p = 0.042) independently predicted the involvement of peripheral vasculature in logistic regression. The diagnostic performance of these cytokines to discriminate CAD+PAD were AUC 0.79 (p < 0.001) for NUS1 and 0.37 (p = 0.013) for ADAMTS-5. We report herein that circulating cytokines can give clues to the ongoing atherosclerotic process and the extent of vascular involvement in which distinct features of ADAMTS-5 and NUS1 make them promising cytokines for future research.
Introduction
Atherosclerosis is a chronic inflammatory disease with lesions ranging from intimal thickening to fibroatheroma. Although the stages of atherosclerosis are well defined, the cause of preferential involvement of certain sites of the arterial tree is still elusive.1,2 In some patients, the disease is confined to a single site, like coronary artery disease (CAD), peripheral artery disease (PAD) or carotid atherosclerotic disease; nevertheless, some patients experience multiple manifestations.
Inflammation, the cornerstone of the atherosclerosis, is orchestrated by endothelial and white blood cells through numerous cytokines. CAD patients with concomitant PAD experience more aggressive disease with extensive and calcified atherosclerosis, greater lesion progression and more common coronary events when compared to patients with CAD only. 3 We hypothesize that this arises from the involvement of different inflammatory pathways. To characterize the distinct features of atherosclerosis in CAD with concomitant PAD, we studied novel cytokines that code different stages of atherogenesis, plaque progression and instability.
Inflammation is triggered by adhesion and transmigration of macrophages into the intima. 1 NOGO-B, a recently discovered membrane protein of endoplasmic reticulum (ER), and its receptor NUS1 regulate leukocyte adhesion and transendothelial migration by intracellular adhesion molecule-1 (ICAM-1) dependent signaling. 4 Once the macrophages settle in the intima, various pathways of inflammatory process are triggered. One activating signal of the major inflammatory nuclear factor κB pathway is through death receptor 3 (DR3) and its ligand tumor necrosis factor (TNF)-like cytokine 1A (TL1A) axis. 5 This ligand-receptor pair is known to promote macrophage foam cell formation, induce apoptosis through metalloproteinase (MMP) expression and produce co-stimulatory signals for T cells which amplify all major pro-inflammatory pathways.6–8 Recent studies have shown that TL1A/DR3 signaling also participates in the regulation of A disintegrin and metalloproteinase with thrombospondin motifs (ADAMTS) proteins 1, 4 and 5. These proteins are secreted enzymes, structurally related to the MMP family, which can cleave vascular proteoglycans, modulating extracellular matrix.9,10 Together, they initiate plaque formation, progression and disruption. NOGO-B/NUS1, TL1A/DR3 and ADAMTS proteins are the cytokines that have not been studied in the PAD patients yet.
The objective of this research was therefore to study the circulating levels of novel cytokines NOGO-B/NUS1, TL1A/DR3 and ADAMTS-1, 4, 5 to characterize plaque progression and stability in atherosclerosis manifested as either CAD or CAD with concomitant PAD, and identify a potential marker for detection of atherosclerotic involvement at multiple vascular territories.
Materials and methods
After obtaining approval from the local Ethics committee, we conducted a prospective study, in which the patient population consisted of patients who were scheduled for coronary angiography (CAG) due to symptoms suggesting stable angina pectoris at our institution. On the day of CAG, the demographic characteristics and cardiovascular risk factors were recorded and the ankle–brachial index (ABI) was obtained to determine the presence of occlusive PAD in each patient. Fasting venous blood samples were collected during the morning hours (between 08:00 and 11:00) prior to CAG for further analysis. Next, all patients underwent the standard CAG procedure. Occlusive CAD was diagnosed when one or more of the epicardial coronary arteries had a diameter stenosis of 50% or more. The SYNergy between PCI with TAXus and cardiac surgery (SYNTAX) score was calculated for all CAD patients to determine the complexity of coronary atherosclerosis by a physician who was blinded to the ABI measurements. 11
The study conformed to the ethical guidelines of the 1975 Declaration of Helsinki, and written informed consent was obtained from every patient prior to enrollment. Patients who experienced an acute coronary syndrome in the past 3 months, patients with left ventricular systolic dysfunction (an ejection fraction <50%), severe valvular heart disease, prior revascularization procedure as either percutaneous coronary intervention or coronary artery bypass grafting, chronic inflammatory disorders, acute infection, known malignancy, chronic liver disease and an estimated glomerular filtration rate <60 mL/min/1.73 m2 were excluded. Patients with uncompressible arteries with an ABI >1.4 were not involved either.
The study was designed to involve 180 patients, categorized into three groups with 60 patients in each group: (1) the ‘CAD+PAD group’ consisted of patients with occlusive CAD and an ABI ≤0.9; (2) the ‘CAD group’ consisted of patients with occlusive CAD and a normal ABI; and (3) the ‘Control group’ consisted of patients with a normal CAG and an ABI within the normal range. Of the consecutive patients who underwent CAG for stable angina pectoris between May and November 2016, and satisfied the inclusion criteria of each group, 180 patients were enrolled into the study. The patient enrollment into each study group was ceased once the objective of 60 patients was reached.
Ankle–brachial index (ABI) measurements
The ABI was measured by Doppler technique and an ABI ≤0.9 was considered as occlusive PAD. 12 The ABI of all patients undergoing CAG was measured by an experienced physician prior to CAG using a 8–10 MHz Doppler ultrasound device (Huntleigh Healthcare Ltd, Cardiff, UK). Patients were advised not to smoke on the day of the CAG. Following a 15 minute period of rest in a supine position, brachial and ankle systolic pressures were determined using appropriately sized cuffs placed on the brachial artery and on the ankle above the malleoli. The averaged ankle pressure measured on the dorsalis pedis and tibialis posterior for each limb was used as the numerator and the highest brachial pressure measured from both arms was used as the denominator to calculate the ABI of each leg. A patient was designated to have PAD if either ABI measured for each leg was ≤0.9.
Patients with an abnormally high ABI (ABI >1.4) were considered to have uncompressible arteries due to vascular calcification and were not recruited into the study.
Biochemical analyses
Fasting blood samples were collected from a vein in the antecubital fossa without venous occlusion and were immediately centrifuged. The blood samples were centrifuged at 3000 rpm for 10 minutes. Samples of sera were stored at −86°C until biochemical analysis. TL1A, DR3, ADAMTS 1-4-5, NOGO-B and NUS1 concentrations were determined by the enzyme-linked immunosorbent assay (ELISA) method based on the competition principle and microtiter plate separation. The TL1A kit (human TL1A soluble ELISA kit; SunRed Biotechnology Co. Ltd, Shanghai, China; PCR catalog no. 201-12-7437) had inter-assay and intra-assay CV% as <10%; the minimum detectable dose of human TL1A was 4.857 pg/mL. The death receptor kit (human DR3 soluble ELISA kit; SunRed Biotechnology Co. Ltd; PCR catalog no. 201-12-0618) had inter-assay CV% as 12% and intra-assay CV% as <10%; the minimum detectable dose of human DR3 was 0.238 ng/mL. The ADAMTS-1 kit (human ADAMTS-1 ELISA kit; SunRed Biotechnology Co. Ltd; PCR catalog no. 201-12-1156) had inter-assay CV% as 12% and intra-assay CV% as <10%; the minimum detectable dose of human ADAMTS-1 was 0.377 ng/mL. The ADAMTS-4 kit (human ADAMTS-4 ELISA kit; SunRed Biotechnology Co. Ltd; PCR catalog no. 201-12-3062) had inter-assay CV% as 12% and intra-assay CV% as <10%; the minimum detectable dose of human ADAMTS-4 was 0.345 ng/mL. The ADAMTS-5 kit (human ADAMTS-5 ELISA kit; SunRed Biotechnology Co. Ltd; PCR catalog no. 201-12-3063) had inter-assay CV% as 12% and intra-assay CV% as <10%; the minimum detectable dose of human ADAMTS-5 was 2.205 ng/mL. The NOGO-B receptor kit (human NUS1 ELISA kit; SunRed Biotechnology Co. Ltd; PCR catalog no. 201-12-7317) had inter-assay CV% as 12% and intra-assay CV% as <10%; the minimum detectable dose of human NUS1 was 0.068 ng/mL. The NOGO-B kit (LegendMax™ human soluble NOGO-B ELISA kit; BioLegend, Inc., San Diego, CA, USA; catalog no. 432807) had inter-assay CV% as 5% and intra-assay CV% as <6%; the minimum detectable dose of human NOGO-B was 26.0 ± 8.7 ng/mL. The IL-6 kit (Affymetrix eBioscience human hsCRP ELISA kit; eBioscience, Inc., Vienna, Austria) had inter-assay CV% as 5.2% and intra-assay CV% as 3.4%; the minimum detectable dose of human IL-6 was 0.92 pg/mL.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Macintosh, Version 20 (IBM Corp., Armonk, NY, USA). The continuous variables are presented either as mean ± SD or median (min–max); the categorical variables are presented as number (percentage). The variables were tested for normality of distribution by the Kolmogorov–Smirnov test. The three patient groups were compared by analysis of variance (ANOVA) in normally distributed variables and the Kruskal–Wallis test in abnormally distributed variables. Post hoc analysis in case of significant deviations showed by ANOVA was performed using Tukey’s or Tamhane’s test depending on the homogeneity of variances. Similarly, Dunn’s test was used in the non-parametric pairwise multiple comparisons procedure following the Kruskal–Wallis test. The categorical variables were compared by the chi-squared test. A p-value of 0.017 adjusted by the Bonferroni method was used in pairwise comparisons of categorical variables. The Pearson and Spearman correlation coefficients were calculated to evaluate continuous and non-continuous relationships among biomarkers and other variables. With the ABI set as the dependent variable, multivariate linear regression analysis was conducted, and the variables with a p-value <0.1 were included into the model by backward method to determine the predictors of the ABI. Similarly, the predictors of PAD among patients with CAD were identified by multivariate logistic regression analysis. The receiver operating characteristics (ROC) curves were constructed for biochemical markers to describe their diagnostic properties. A p-value of <0.05 was considered statistically significant.
Results
The demographic data and characteristics of the study population are presented in Table 1, which shows the cardiovascular risk profile of study groups in terms of age, sex, current smoking, diabetes mellitus, hypertension and hyperlipidemia. The circulating levels of cytokines with respect to patient groups are presented in Table 2 and Figure 1. Since the baseline characteristics of the study groups were not homogenous, we first analyzed the possible effects of cardiovascular risk factors on cytokine levels, where we demonstrate that the cytokine levels did not differ with regard to the presence of risk factors (table presented as supplemental file).
Comparison of demographic characteristics in the study population.
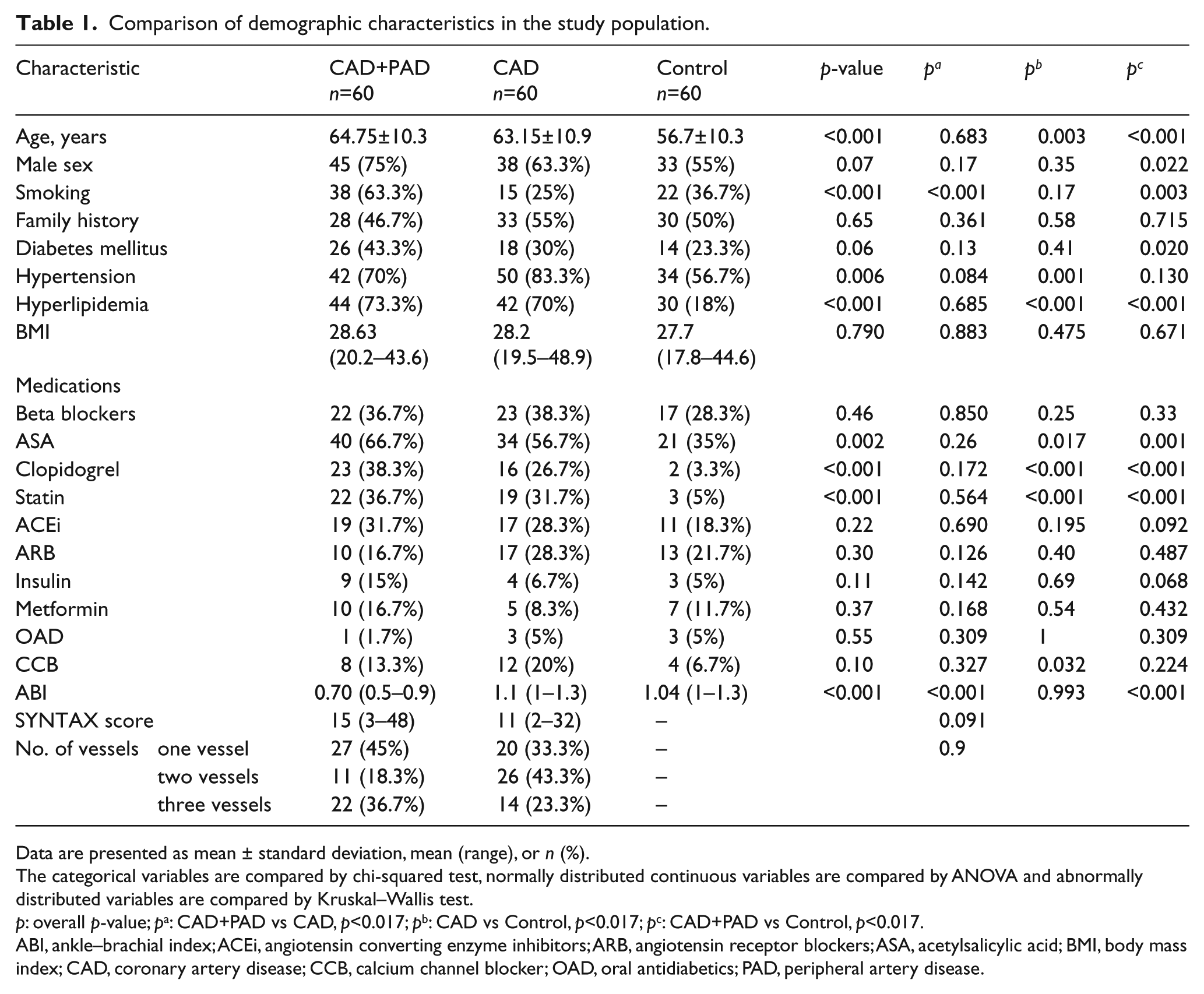
Data are presented as mean ± standard deviation, mean (range), or n (%).
The categorical variables are compared by chi-squared test, normally distributed continuous variables are compared by ANOVA and abnormally distributed variables are compared by Kruskal–Wallis test.
p: overall p-value; pa: CAD+PAD vs CAD, p<0.017; pb: CAD vs Control, p<0.017; pc: CAD+PAD vs Control, p<0.017.
ABI, ankle–brachial index; ACEi, angiotensin converting enzyme inhibitors; ARB, angiotensin receptor blockers; ASA, acetylsalicylic acid; BMI, body mass index; CAD, coronary artery disease; CCB, calcium channel blocker; OAD, oral antidiabetics; PAD, peripheral artery disease.
Circulating levels of cytokines with respect to patient groups.
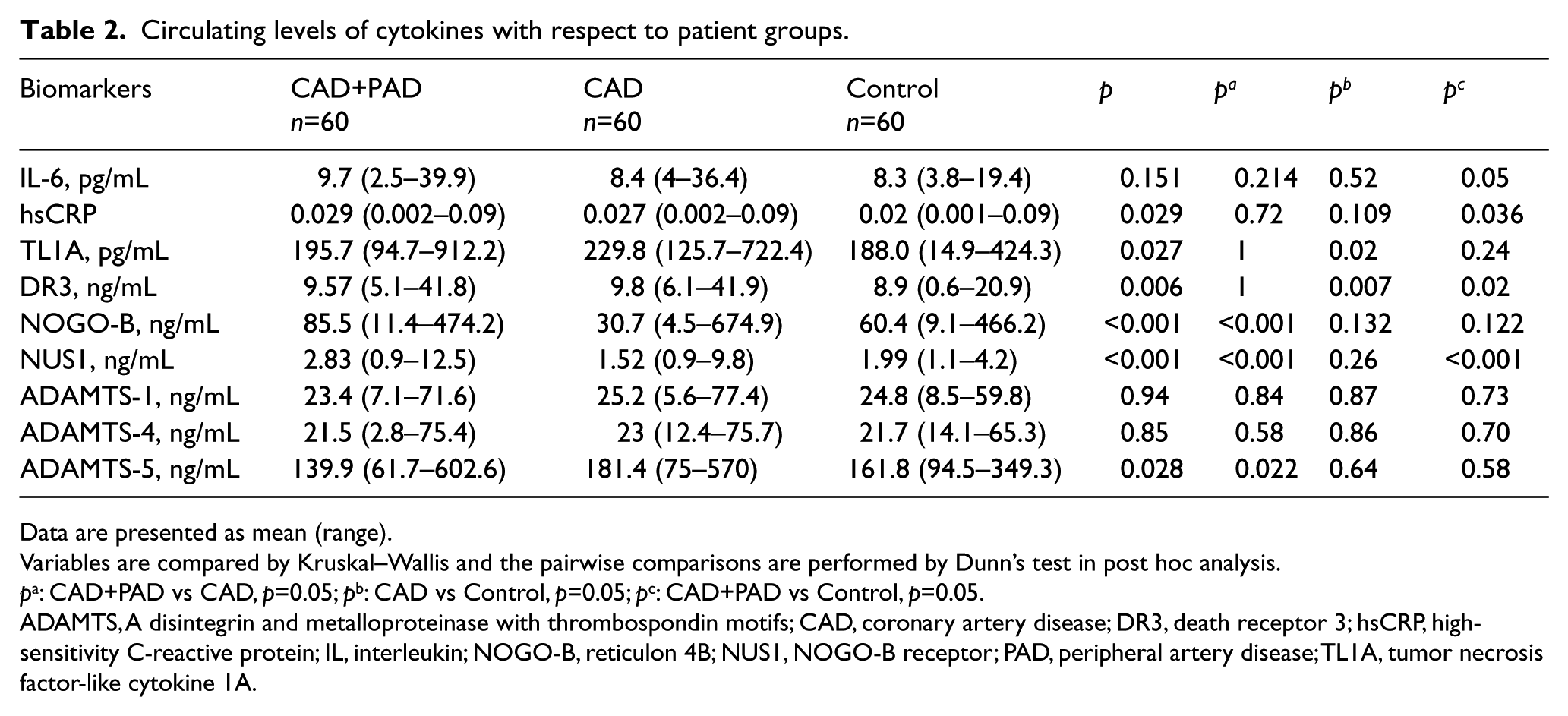
Data are presented as mean (range).
Variables are compared by Kruskal–Wallis and the pairwise comparisons are performed by Dunn’s test in post hoc analysis.
pa: CAD+PAD vs CAD, p=0.05; pb: CAD vs Control, p=0.05; pc: CAD+PAD vs Control, p=0.05.
ADAMTS, A disintegrin and metalloproteinase with thrombospondin motifs; CAD, coronary artery disease; DR3, death receptor 3; hsCRP, high-sensitivity C-reactive protein; IL, interleukin; NOGO-B, reticulon 4B; NUS1, NOGO-B receptor; PAD, peripheral artery disease; TL1A, tumor necrosis factor-like cytokine 1A.
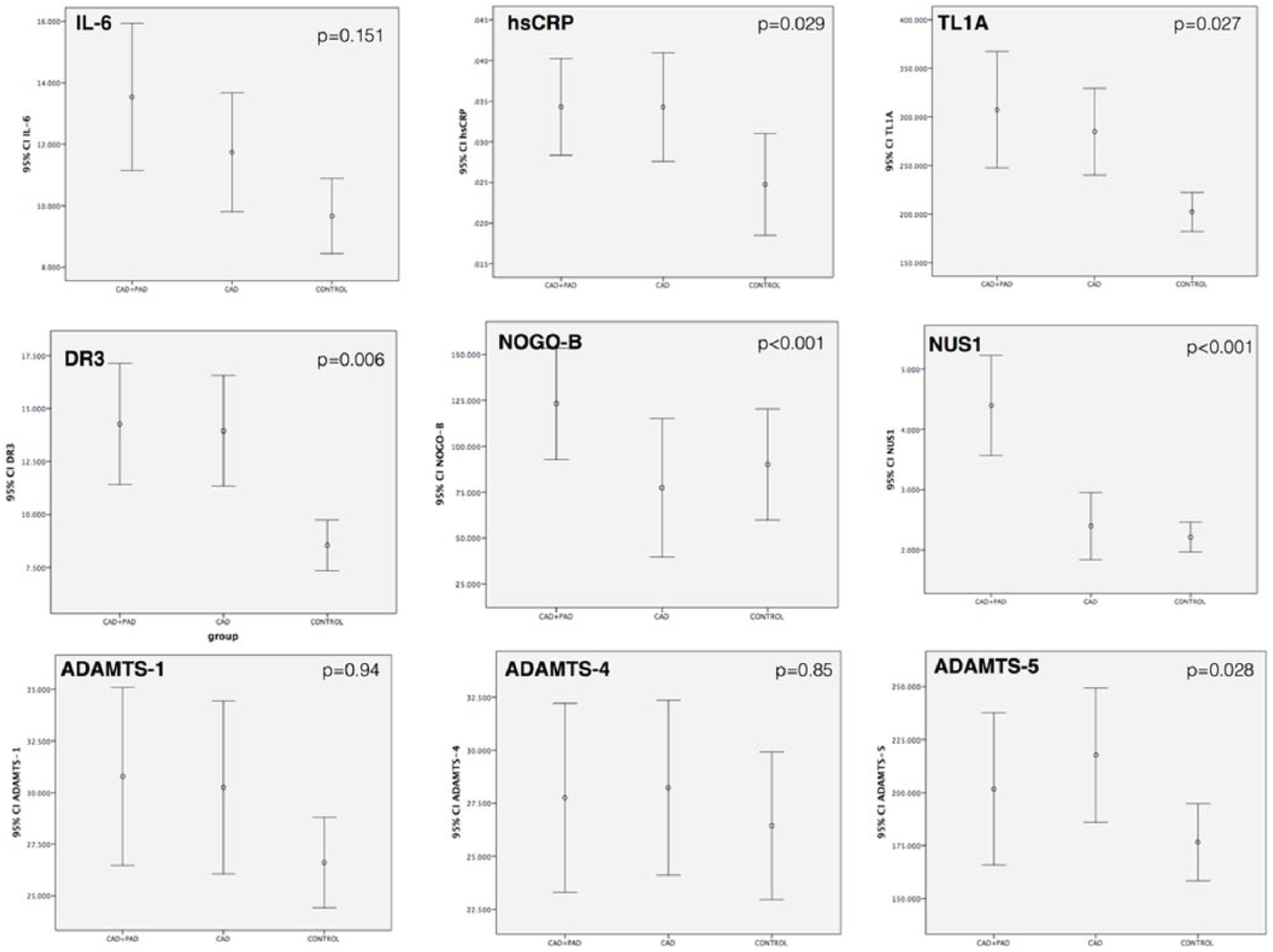
Cytokine concentrations with respect to study groups.
Serum hsCRP and IL-6 concentrations were the lowest in control subjects and increased as the involved territories increased, with the highest concentrations in the CAD+PAD group. The difference, however, was not statistically significant for IL-6, and it was only significant between CAD+PAD and control groups for hsCRP (0.029 [0.002–0.09] vs 0.020 [0.001–0.09], p=0.036).
The TL1A and DR3 levels were higher in the CAD group (229.8 [125.7–722.4] pg/mL and 9.8 [6.1–41.9] ng/mL) than those in the control group (188.0 [14.9–424.3] pg/mL and 8.9 [0.6–20.9] ng/mL). The DR3 levels were also higher in the CAD+PAD patients when compared to control patients (9.57 [5.1–41.8] vs 8.9 [0.6–20.9] ng/mL, p=0.02). There was no significant difference, however, in CAD patients with or without concomitant PAD. The serum concentrations of another ligand-receptor pair, NOGO-B and NUS1, were highest in the CAD+PAD patients (85.5 [11.4–474.2] and 2.83 [0.9–12.5] ng/mL) followed by control subjects (60.4 [9.1–466.2] and 1.99 [1.1–4.2] ng/mL), and finally lowest in the CAD group (30.7 [4.5–674.9] and 1.52 [0.9–9.8] ng/mL) (pairwise comparisons are presented in Table 2).
Serum ADAMTS-1 and 4 concentrations were similar within the study population. Unlike other ADAMTS proteins, ADAMTS-5 was higher in the CAD group (181.4 [75–570] ng/mL) than the CAD+PAD group (139.9 [61.7–602.6] ng/mL, p=0.022).
The correlation analysis (Table 3) revealed significant correlations between ABI and age (r = −0.17, p=0.02), SYNTAX score (r = −0.38, p<0.001), NOGO-B (r = −0.24, p<0.002), NUS1 (r = −0.38, p<0.001) and ADAMTS-5 (r = 0.25, p = 0.006). The SYNTAX score, which reflects the coronary atherosclerotic burden, was significantly correlated with age (r = 0.32, p<0.001), ABI (r = −0.38, p<0.001), TL1A (r = 0.20, p = 0.01) and DR3 (r = 0.16, p = 0.039). The correlations among cytokines revealed significant interactions of the TL1A/DR3 axis with ADAMTS proteins and the TL1A/DR3 axis with the NOGO-B/NUS1 axis.
Correlations of cytokine levels within the study population.
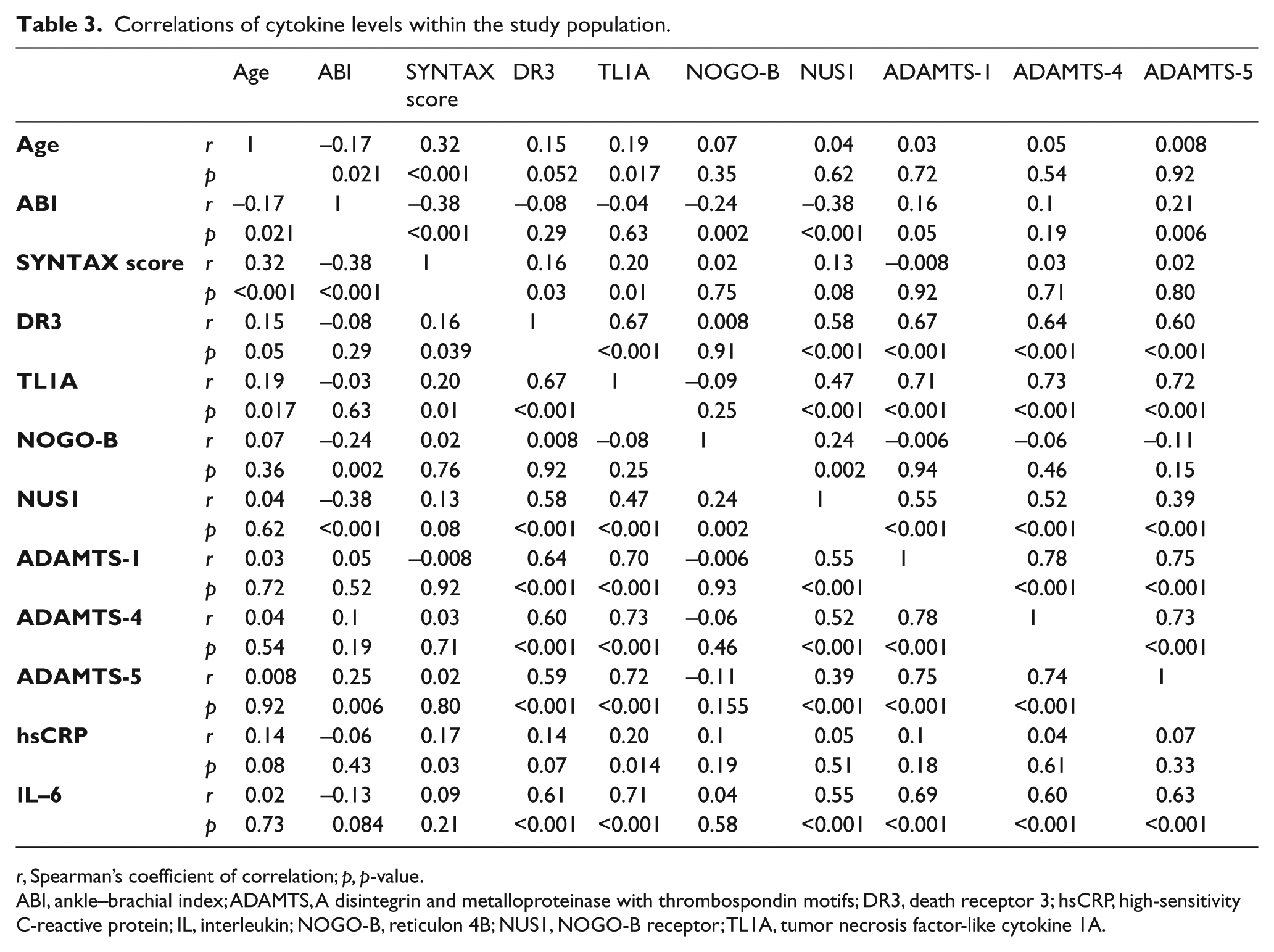
r, Spearman’s coefficient of correlation; p, p-value.
ABI, ankle–brachial index; ADAMTS, A disintegrin and metalloproteinase with thrombospondin motifs; DR3, death receptor 3; hsCRP, high-sensitivity C-reactive protein; IL, interleukin; NOGO-B, reticulon 4B; NUS1, NOGO-B receptor; TL1A, tumor necrosis factor-like cytokine 1A.
Multiple linear regression analysis was conducted to examine the association of variables with ABI, which is summarized in Table 4. The independent predictors of ABI in multivariate analysis were smoking (Β = −0.13, 95% CI −0.11, 0.006; p=0.04), SYNTAX score (Β = −0.26, 95% CI −0.009, −0.003; p<0.001), NUS1 (Β = −0.88, 95% CI −0.1, −0.05; p<0.001) and ADAMTS-5 (Β = 0.63, 95% CI 0.001, 0.002; p<0.001). Logistic regression, which analyzed the predictors of multiple atherosclerotic involvement (manifested as CAD and PAD), confirmed the independent influence of smoking (OR = 5.5, 95% CI 0.04, 0.7; p=0.019), SYNTAX score (OR = 1.2, 95% CI 1.1, 1.3; p<0.001), NUS1 (OR = 14.4, 95% CI 5.0, 41.1; p<0.001) and ADAMTS-5 (OR = 1.1, 95% CI 0.94, 0.98; p<0.001); it also demonstrated that age is another significant predictor (OR = 1.1, 95% CI 1.02, 1.1; p=0.042). Table 5 presents the multivariable logistic model for the predictors of multiple atherosclerotic involvement, manifested as CAD+PAD.
Univariable and multivariable models for the predictors of ABI.
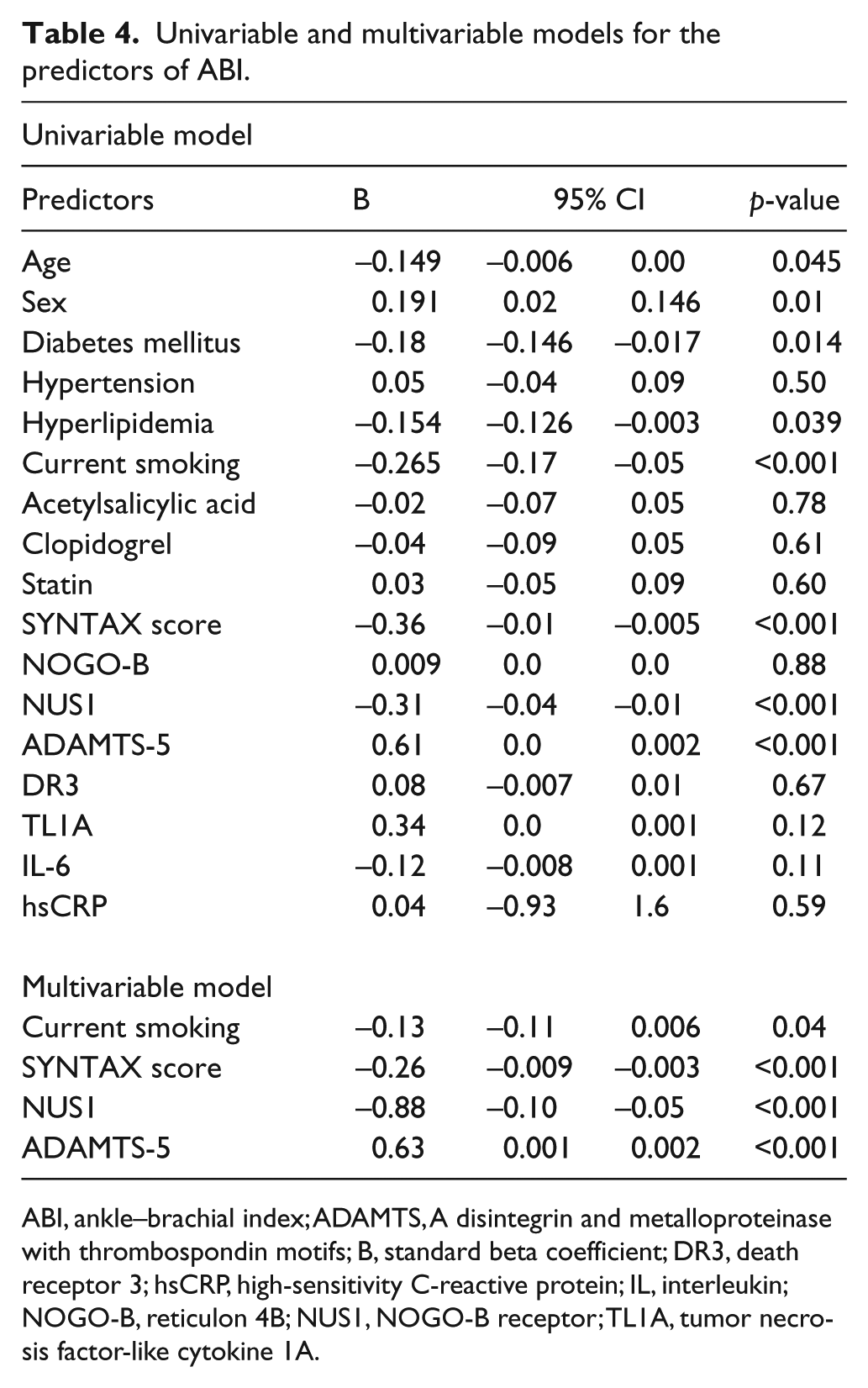
ABI, ankle–brachial index; ADAMTS, A disintegrin and metalloproteinase with thrombospondin motifs; B, standard beta coefficient; DR3, death receptor 3; hsCRP, high-sensitivity C-reactive protein; IL, interleukin; NOGO-B, reticulon 4B; NUS1, NOGO-B receptor; TL1A, tumor necrosis factor-like cytokine 1A.
Logistic regression model for the predictors of multiple atherosclerotic involvement (CAD+PAD).
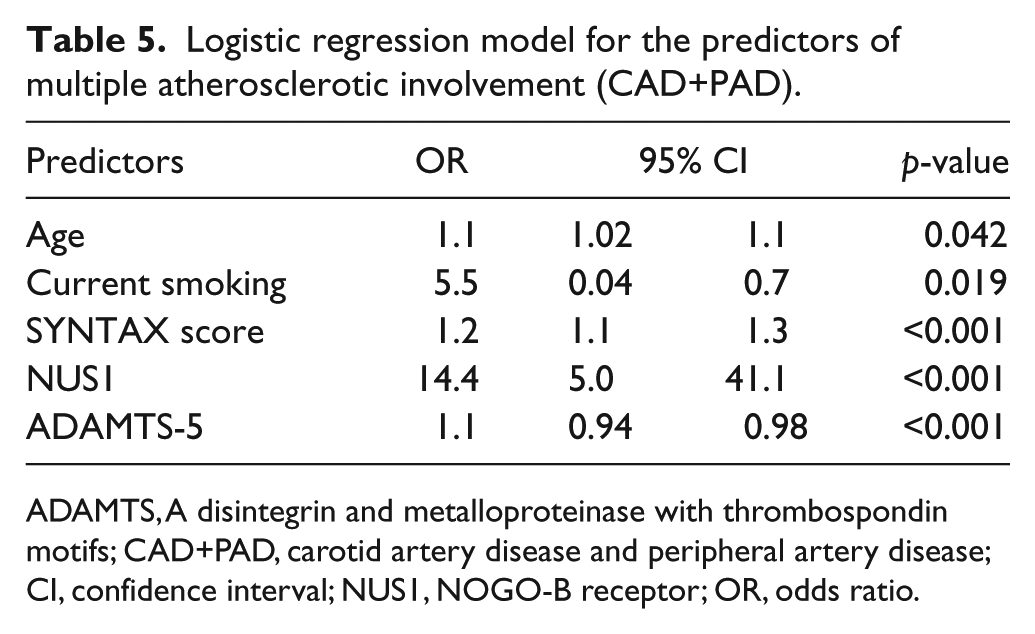
ADAMTS, A disintegrin and metalloproteinase with thrombospondin motifs; CAD+PAD, carotid artery disease and peripheral artery disease; CI, confidence interval; NUS1, NOGO-B receptor; OR, odds ratio.
The ROC analysis revealed that the NUS1 and ADAMTS-5 concentrations had good diagnostic performances to discriminate multiple manifestations of atherosclerosis (as both CAD and PAD) within the study population (area under the curve (AUC) = 0.79, SE = 0.045, 95% CI 0.70, 0.88, p<0.001 for NUS1; AUC = 0.37, SE = 0.054, 95% CI 0.26, 0.47, p=0.013 for ADAMTS-5) (Figure 2).
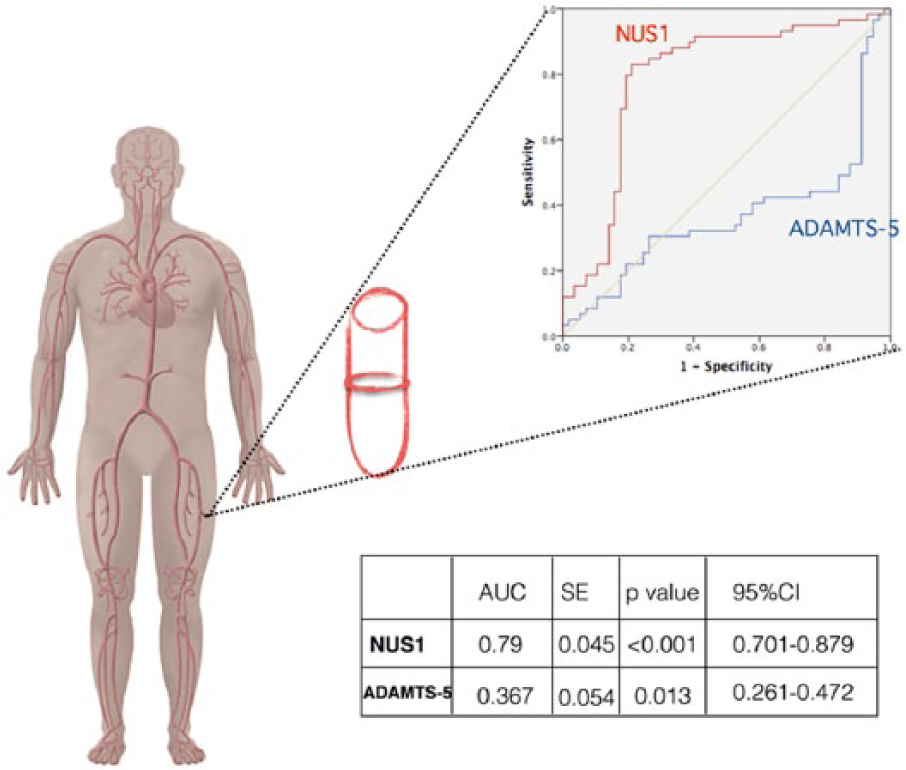
ROC of NUS1 and ADAMTS-5 for the diagnosis of multiple atherosclerotic involvement manifested as CAD+PAD in the patients with CAD.
Discussion
We report herein that circulating cytokines can give clues to the ongoing atherosclerotic process and the extent of vascular involvement, manifested as CAD with or without PAD. Systemic inflammation in CAD expressed by the TL1A/DR3 axis was similar in patients with CAD and concomitant PAD, indicating parallel pathophysiologic mechanisms. NOGO-B and its receptor NUS1, on the contrary, were significantly elevated and ADAMTS-5 was reduced in patients with concomitant PAD. These cytokines may help us explain the pathophysiologic basis of calcified lesions with diffuse vascular involvement. Multivariate analyses provided further evidence for the independent influence of NUS1 and ADAMTS-5 on multiple atherosclerotic manifestations.
Coronary artery disease and PAD are subsets of atherosclerosis that share common cardiovascular risk factors albeit with higher relative risks attributed to age, smoking and diabetes mellitus in PAD than CAD. 13 In our study, the patient groups were heterogeneous in terms of cardiovascular risk profile, with more common occurrence of older age, current smoking, diabetes and hyperlipidemia in patients with CAD and concomitant PAD. Smoking and age were significant predictors of multiple atherosclerotic involvement represented by the CAD+PAD group in multivariate regression analyses. Although diabetes mellitus was associated with ABI on a univariate basis, the association was not consistent in the multivariate model. Prior studies have shown that poorly controlled, longstanding diabetes is more strongly related to PAD. 14 In our study, patients with newly diagnosed diabetes may have diluted its influence on the results. Similarly, we could not demonstrate independent effects of hypertension and hyperlipidemia on PAD, owing to less population risk attributable to these factors. 15
As a unique presentation of atherosclerosis, recent studies have disclosed elevated inflammatory markers in PAD.16,17 Nevertheless, the distinct features of CAD with concomitant PAD imply that the underlying inflammatory pathways are not common in coexistence of PAD. IL-6 and hsCRP levels, which are among the most known inflammatory cytokines, were increased in the CAD+PAD group in the numerical sense, but the difference had statistical significance only for hsCRP when compared to controls.
Novel cytokines in PAD
TL1A/DR3 axis
TL1A and its receptor DR3 have been shown to induce macrophage foam cell formation, T cell activation and MMP mediated apoptosis in atherosclerotic lesions. 5 Our study has determined that the emerging TL1A/DR3 axis was up-regulated in CAD patients but demonstrated no further increase in patients with concomitant PAD. This suggests that the TL1A/DR3 axis does not have an additional role over CAD in PAD. Stamatelopoulos et al. reported elevated TL1A levels in CAD patients, which was correlated with the extent of atherosclerosis, expressed as the Gensini score. 18 In this article, we have shown the positive correlation of TL1A and DR3 with the SYNTAX score and reported no correlation with ABI. The correlation analyses also unveiled significant interactions of the TL1A/DR3 axis with the NOGO-B/NUS1 axis and with ADAMTS proteins. These findings support the literature on the ability of members of the TNF superfamily, like TL1A/DR3 proteins, to trigger a wide variety of signaling pathways including the NOGO-B/NUS1 axis and ADAMTS proteins.9,19,20
NOGO-B/NUS1 axis
NOGO-B (reticulon 4B) is neurite outgrowth inhibitor protein located in the ER of endothelial and smooth muscle cells of the vessel wall and is involved in vascular homeostasis and inflammation. 21 The basic biological role of NOGO-B/NUS1 interaction is to regulate sphingolipid synthesis by inhibiting the rate-limiting enzyme serine palmitoyltransferase. The reduction of sphingolipids plays an important role in the onset of endothelial dysfunction, hypertension and pathological cardiac hypertrophy. 21 NOGO-B and NUS1 propagate early stages of atherosclerosis, wherein they mediate the adhesion and transmigration of leukocytes into the intima. 22 In early fibroatheromas, in which the necrotic core is relatively small, high amounts of diffuse NOGO-B expression were demonstrated and this was inversely related to the plaque area. 23 Rodriguez-Feo et al. have also shown that the NOGO-B levels in carotid plaques were inversely related to the presence of large lipid pools. 24 There are several studies which have analyzed the levels of circulating NOGO-B in diseases like pulmonary arterial hypertension or liver cirrhosis, and only a single study in CAD.25–27 Gao et al. found increased NOGO-B concentrations in patients with acute coronary syndromes and stable angina pectoris with respect to healthy controls. 27 The current study is the first to evaluate NOGO-B levels in PAD. Furthermore, there are no data on the involvement of its receptor NUS1 in atherosclerosis in the literature. In our study, the serum levels of NOGO-B and NUS1 were higher in the PAD+CAD group compared to the CAD group. This can be explained by the abundant number of lesions at the early stages of the atherosclerotic process in patients with PAD. By multivariate analyses, we showed that circulating NUS1 is a good index of the extent of vascular territories involved in atherosclerosis.
Although there is no conclusive data on the association of vascular calcification with the NOGO-B/NUS1 axis, there are several ways the two pathological pathways intersect. Sphingolipids, which are regulated by NOGO-B, have recently been shown to modulate vascular smooth muscle cell matrix mineralization. 28 Moreover, NOGO-B is an important determinant of intracellular calcium homeostasis through store operated calcium entry.29,30 Endoplasmic reticulum stress, which is propagated by NOGO-B, is known to trigger vascular calcification. 31 Given that PAD is closely associated with diffusely calcified lesions, increased NOGO-B/NUS1 in PAD may contribute to the more calcified nature of atherosclerosis in patients with both CAD and PAD. 30
ADAMTS proteases
This is the first study to expose circulating ADAMTS proteases in patients with both CAD and PAD, in which we demonstrated decreased ADAMTS-5 in PAD patients. Although ADAMTS-5 levels were elevated in the CAD group, the coexistence of PAD had a negative influence on circulating ADAMTS levels. The exact role of ADAMTS proteases in atherosclerosis is not straightforward. Experimental studies revealed that ADAMTS proteases are expressed on macrophages during the differentiation of monocytes to macrophages – a process regulated by TL1A. 9 Likewise, our findings proved strong correlations between TL1A/DR3 protein and ADAMTS protease levels (Table 3). These proteases modulate the extracellular matrix through the catabolism of low-density lipoprotein (LDL)-binding proteoglycans, which has several implications: catabolism of LDL-binding proteoglycans may cause release of LDL as well as destabilization of plaque. 10 Didangelos et al. showed reduced levels of ADAMTS-5 in a murine model of atherosclerosis which resulted in accumulation of biglycan and versican. 32 Moreover, they proved the release of LDL from human aortic samples when treated with recombinant ADAMTS-5. 32 Therefore, the role of ADAMTS-5 may vary on different territories depending on the different post-signaling mechanisms. A decrease in the early stages of atherosclerosis may be followed by and increase as the plaque structure changes. A decrease in ADAMTS-5 levels in our study seems to reflect the ongoing extensive atherosclerotic process in patients with concomitant PAD. This also provides evidence that explains why adverse events related to plaque stability is a more common feature of CAD than PAD.
Study limitations
Current findings yield insights on the pathogenesis of PAD in patients with established CAD. There are, however, some limitations of the study. First, there are limited data regarding the influence of different factors on circulating levels of these novel cytokines, such as circadian variation. We have tried to control for this by limiting the collection of blood samples to the morning hours. Additionally, the atherosclerotic spectrum usually presents as multi-vascular involvement. In order to control for the occult overlap of different manifestations, we chose the study population among patients with stable angina pectoris, at the expense of having patients with multiple cardiovascular risk factors and endothelial dysfunction in the control group. The levels of inflammatory cytokines were, consequently, elevated in the control group, blurring the distinction, particularly in hsCRP and IL-6. Intriguingly, the control patients had high concentrations of NOGO-B and NUS1, which were previously demonstrated to participate in endothelial dysfunction and hypertension through decreasing production of sphingolipids. 21 The occurrence of PAD without the involvement of any other vascular territory is a rare event. Therefore, we based our hypothesis on the inflammatory milieu of PAD over CAD, as it is in real life.
Conclusion
In conclusion, we have demonstrated that CAD with concomitant PAD shares the same denominator of inflammation that is reflected by the novel TL1A/DR3 axis. When compared to the CAD patients, CAD and PAD patients had increased NOGO-B/NUS1 concentrations, which suggests ongoing inflammation with a larger vascular surface area affected by atheromas at early stages of atherosclerosis and a more calcified plaque structure in PAD. The ADAMTS-5 concentration, on the other hand, is decreased in patients with extensive involvement of the arterial tree. Further studies are needed to explore the diagnostic or therapeutic roles of these cytokines in atherosclerosis.
Supplemental Material
VMJ763096_Supplementary_Table_1_ – Supplemental material for The role of novel cytokines in inflammation: Defining peripheral artery disease among patients with coronary artery disease
Supplemental material, VMJ763096_Supplementary_Table_1_ for The role of novel cytokines in inflammation: Defining peripheral artery disease among patients with coronary artery disease by Demet Ozkaramanli Gur, Savas Guzel, Aydin Akyuz, Seref Alpsoy and Niyazi Guler in Vascular Medicine
Footnotes
Acknowledgements
We thank Ramazan Bilge for organizing the recruitment of blood samples.
The abstract of this article was presented at the 33th Turkish Cardiology Congress (5–8 October 2017, Antalya).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Scientific Research Projects Council of Namik Kemal University (NKUBAP.02.GA.16.044).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
