Abstract
Background:
Patients with chronic limb-threatening ischemia (CLTI) and infected leg ulcers are at risk of amputation and postinterventional sepsis.
Methods:
This retrospective, single-center study included patients with CLTI and infected leg ulcers who underwent endovascular treatment (EVT) between 2012 and 2021.
Results:
The study included 712 patients, 286 (40.2%) of whom underwent amputation (minor, n = 212; major, n = 74). Gram-negative bacteria (GNB) were significantly more prevalent in amputees (36.4% vs 30.9%, p < 0.05). Patients with gram-positive bacteria (GPB) had a 4-year freedom from any amputation rate of 72% (95% CI 64–81%) compared to 52% (95% CI 42–66%) in patients with GNB identification (p < 0.05). Cox proportional regression analysis showed that GNB, male sex, mean Wound, Ischemia, and foot Infection (WIfI) score, diabetes mellitus, and end-stage renal disease were independently and positively associated with amputation (p < 0.05). The mean WIfI score and end-stage renal disease were independently and positively associated with death from any cause (p < 0.05). Staphylococcus aureus or GNB, end-stage renal disease, and diabetes mellitus were independent risk factors for sepsis after EVT (p < 0.05). Inpatient-administered antibiotic regimes had significantly higher microbiological activity in cases of GPB identification compared to GNB identification (28% vs 9%, p < 0.05).
Conclusion:
Although the isolation of both GNB and S. aureus is a risk factor for sepsis following EVT, the isolation of GNB is independently associated with higher rates of amputation, demonstrating the importance of identifying pathogens to recognize patients at high risk.
Background
Approximately 200 million people worldwide suffer from peripheral artery disease (PAD). 1 Compared to those without PAD, patients with PAD have a threefold increase in mortality. 2 In patients with chronic limb-threatening ischemia (CLTI), infections are the second most common noncardiovascular cause of death at 17.9%. Besides the increased mortality, patients with PAD are at high risk for amputation, which significantly reduces quality of life. To better identify patients with CLTI at risk for amputation, the Society for Vascular Surgery proposed the Wound, Ischemia, and foot Infection (WIfI) classification, 3 using a three-digit code for each category (wound, ischemia, and foot infection), with increasing severity for each subcategory indicated by a number from zero to three. The final classification of patients with PAD is a two-step process: first, patients are classified according to Rutherford categories based on symptom severity, ranging from stage 1 to 6, indicating increasing disease severity. 4 Second, patients with Rutherford categories 5 and 6 are substratified using the WIfI classification. The foot infection category is associated with an increased risk of amputation. 5 Similarly, wound infection is an independent risk factor for nonhealing in patients with PAD with diabetes. 6 However, little is known about foot infections in patients with PAD. A recent study provided the first insights into the microbiological patterns of infected leg ulcers in patients with PAD, 7 revealing that gram-negative bacteria (GNB) are more frequently isolated in ulcers from patients with PAD compared to those with diabetic foot infection (DFI) (32% vs 20%). 7 As GNB pathogens are significantly more often identified in patients with DFI who underwent amputation, 8 further insights into the pathogen and outcome interaction in patients with PAD and ulcers are crucial for understanding the increased risk of amputation in patients with CLTI and infected leg ulcers.
This study aimed to provide insights into the clinical outcomes of patients undergoing endovascular treatment (EVT) for CLTI with infected leg ulcers, considering their microbiological characteristics. Moreover, we identified risk factors for amputation, death, and postinterventional sepsis.
Methods
Study design
A retrospective analysis was performed using data from patients who underwent EVT for PAD Rutherford categories 5 and 6 with infected leg ulcers between 2012 and 2021. Data were selected from a prospectively maintained database that included the microbiological diagnostics of infected wound specimens from all enrolled patients. Microbiological diagnostics and data storage were performed at the medical laboratories of the private laboratory group Medical Care Center (MVZ; Clotten, Freiburg, Germany). The microbiological data sets were extracted using the hygiene management system HyBASE (epiNET, Bochum, Germany) and stored as pseudonymized Excel files.
Study participants
The study included patients with infected arterial leg ulcers and angiographically confirmed peripheral occlusive disease of Rutherford categories 5 and 6. Only patients with de novo stenosis at study entry were included. Photographic documentation was performed for all patients with ulcers. Upon admission, each patient underwent noninvasive diagnostics, such as tibio-brachial-quotient measurements and duplex sonography, followed by EVT. The wounds of each patient were graded by an independent observer (FI) using the WIfI classification. 3 The grading process involved photographic documentation, laboratory parameters, such as C-reactive protein (CRP) and leukocyte counts, and noninvasive measurements. Patients with a WIfI score of zero were excluded from the study. For every study participant, we obtained complete microbiological test results for the corresponding wound specimens. The microbiological results were assessed by an independent expert in clinical microbiology (TSK) and graded into three categories: (i) pathogens, (ii) potential pathogens, and (iii) pathogens in special circumstances/commensals. Any pathogens graded as commensal were excluded. Thus, this study only included wounds that were infected and originated from ischemia, excluding any noninfected wounds or those caused by other leading factors. Because one patient was infected with more than one bacterium, the number of bacterial isolates exceeded the number of patients.
This study was approved by the Ethical Board of the University of Freiburg, Germany (proposal number: 22-1238-S1-retro). The need for informed consent was waived by the ethics committee.
Specimens and microbiology
Wound specimens and swabs were collected by a trained wound manager following a standardized protocol as described previously. 7 If a tissue scrape from the ulcer base was impossible, a deep wound specimen was collected according to the Levine method. 9 Pathogen identification and antimicrobial susceptibility testing (AST) were conducted using automated systems, including MALDI-TOF, VITEK 2, disc diffusion, and microbroth dilution. Only the first isolates per phenotype were included to minimize overestimation due to multiple identifications within the same individual, in accordance with the methodology proposed by Hindler et al. and implemented in the R package AMR (Antimicrobial Resistance).10,11
Statistical analysis
Descriptive analyses of patient characteristics were presented as means with SDs for continuous variables and as counts with percentages for categorical variables. Descriptive analyses of antibiotic regimens were reported as counts and percentages. Time-to-event analyses were performed using Kaplan–Meier curves. The log-rank test was used to estimate statistical differences between the curves. Cox proportional hazards regression analysis was performed to identify the risk of limb loss after revascularization. Owing to the potential for multiple interventions and hospital stays, patients may have multiple wound specimens collected over time. Therefore, only microbiological test results from the last wound swab per leg before the event (amputation, death from any cause, or censoring) were considered for the time-to-event analysis. Gram-negative pathogen identification indicated the isolation of at least one gram-negative pathogen per specimen. Given the strict chronological sequence and temporal proximity to the procedure, risk factors for postinterventional sepsis were identified using multivariable logistic regression analysis. The predictor variables were selected a priori using subject matter knowledge. Hazard ratios (HRs) with 95% CIs and odds ratios (ORs) with 95% CIs were calculated for the Cox proportional regression model and the logistic regression model, respectively. The mean WIfI score, as proposed by Darling et al., was used as a variable for Cox proportional hazard and logistic regressions. 5 Statistical analyses were performed using free statistical computing and graphical software R (R 4.0.3; R Foundation for Statistical Computing, Vienna, Austria). Statistical significance was set at α = 0.05.
Results
Study population
This study included 712 patients (Table 1). The mean age of the study population was 76.4 (SD ± 10.6) years, with a lower prevalence of women (35.8%). Of the total, 286 (40.2%) patients underwent amputation, consisting of 212 (29.8%) minor and 74 (10.4%) major amputations. The median WIfI score for each independent variable—wound, ischemia, and foot infection—was 2. The mean peak leukocyte count per patient during the hospital stay was 14,518.0/µL (Table 1).
Characteristics of the study population.
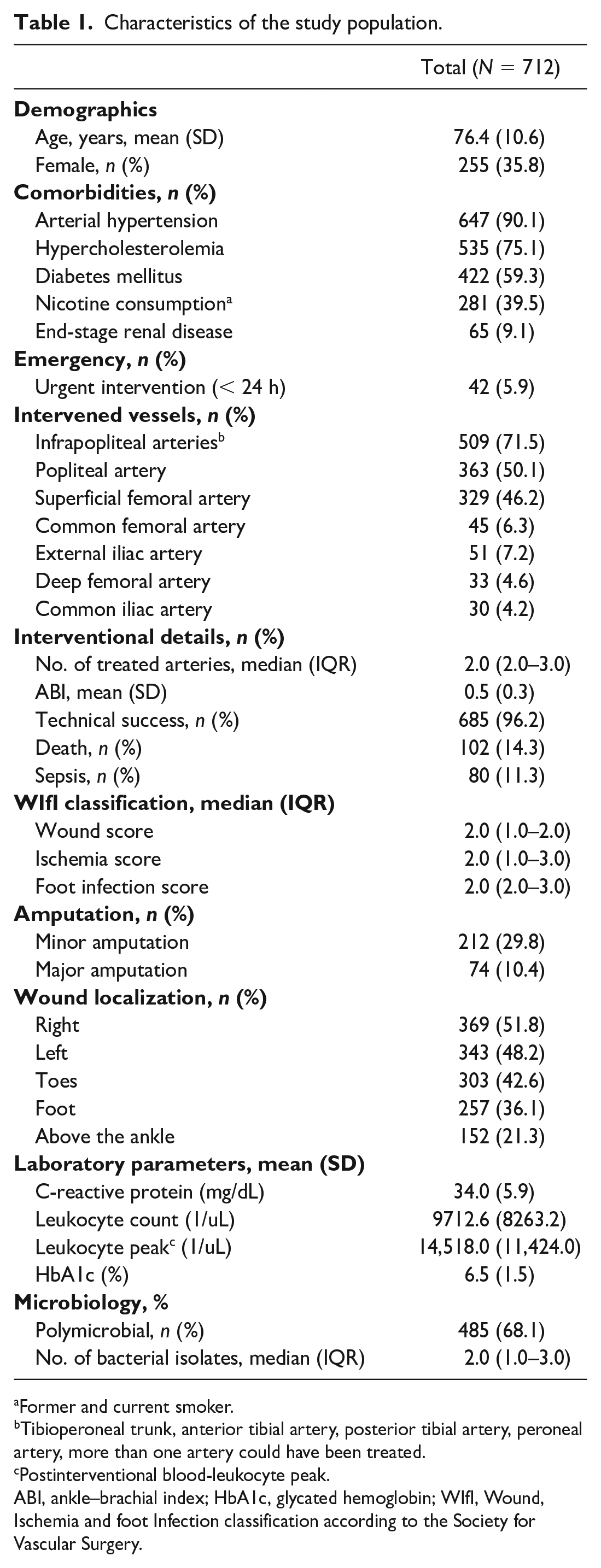
Former and current smoker.
Tibioperoneal trunk, anterior tibial artery, posterior tibial artery, peroneal artery, more than one artery could have been treated.
Postinterventional blood-leukocyte peak.
ABI, ankle–brachial index; HbA1c, glycated hemoglobin; WIfI, Wound, Ischemia and foot Infection classification according to the Society for Vascular Surgery.
Microbiological pattern
In total, 1355 isolates were gram-positive, and 673 were GNB (data not shown). Among patients who underwent any amputation, the detection of gram-positive bacteria (GPB) decreased significantly, whereas the identification of GNB increased significantly from 30.9% to 36.4% (p < 0.05). The pathogen patterns did not significantly differ between patients with minor and major amputations, with GNB identified in 36.5% of minor and 36.1% of major amputations (Figure 1 and Supplementary Table 1).
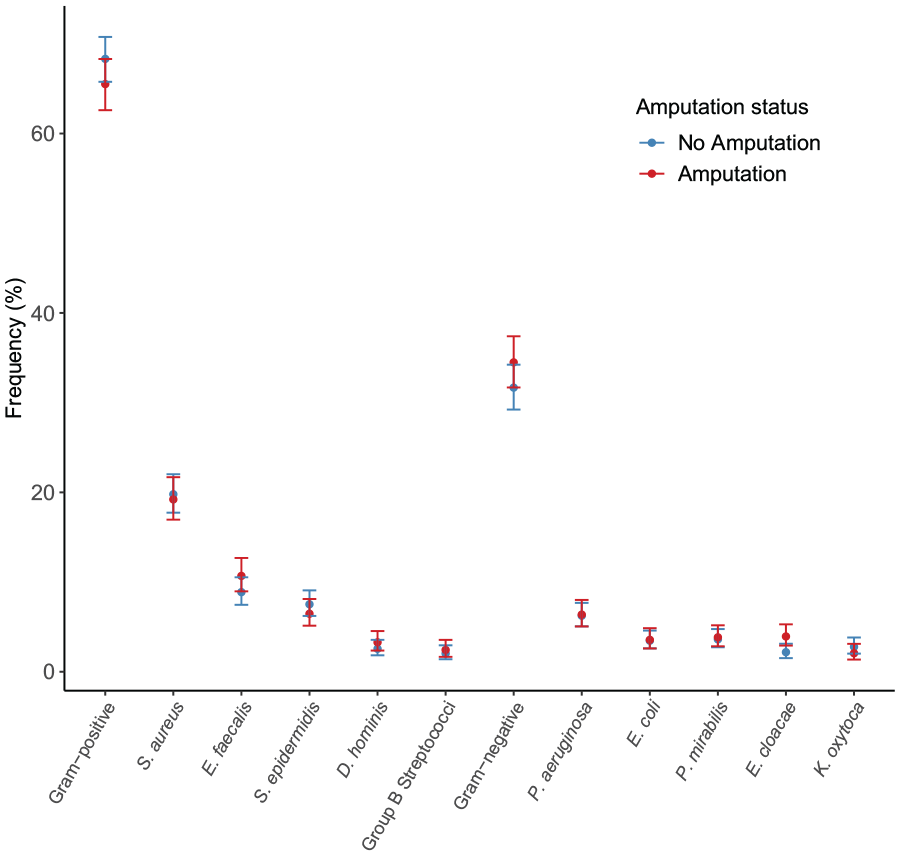
Pathogen distribution stratified by patients’ amputation status.
Limb and survival outcomes
Figure 2 illustrates the time-to-amputation and survival analyses for patients with CLTI, stratified by gram stain and risk factors, using Kaplan–Meier curves. The data indicated that amputation occurred significantly more frequently in patients with PAD with GNB identification (Figure 2A), regardless of amputation extent (Figures 2B and 2C). After 4 years, freedom from any amputation was 72% (95% CI 64–81%) in patients without GNB identification compared to 52% (95% CI 42–66%) in those with at least one isolated GNB (Figure 3A). When stratified by a set of three risk factors (end-stage renal disease, diabetes mellitus, and identification of gram-negative pathogens), freedom from any amputation decreased significantly with an increasing number of risk factors (Figure 2E). Freedom from any amputation at 4 years was 85%, 65%, and 41% for no identified risk factors, one identified risk factor (regardless of which), and two or more identified risk factors, respectively (Figure 3C). For additional information, refer to Figures 2 and 3.
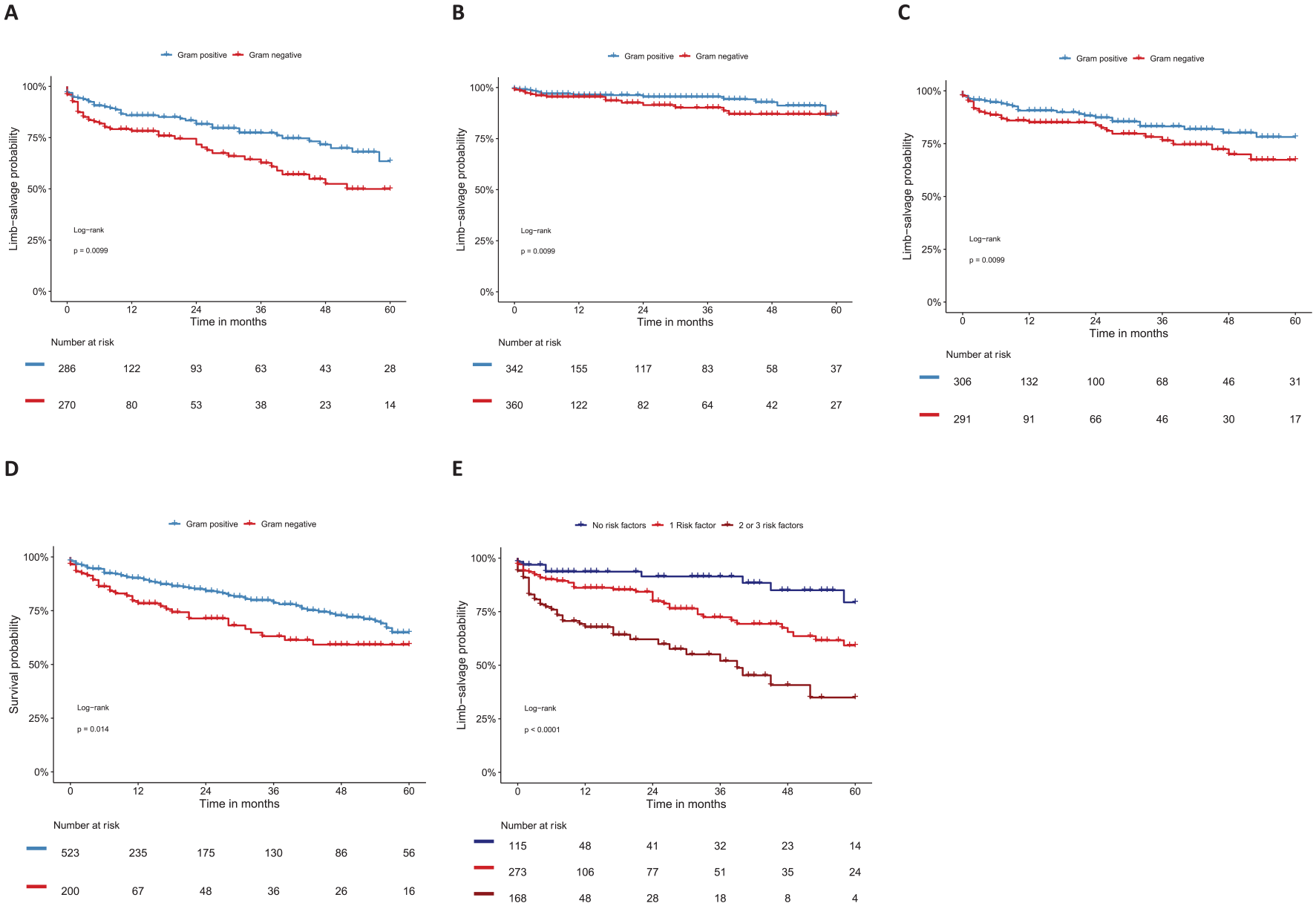
Time-to-event analysis in patients with peripheral artery disease stratified by gram stain and risk factors. (
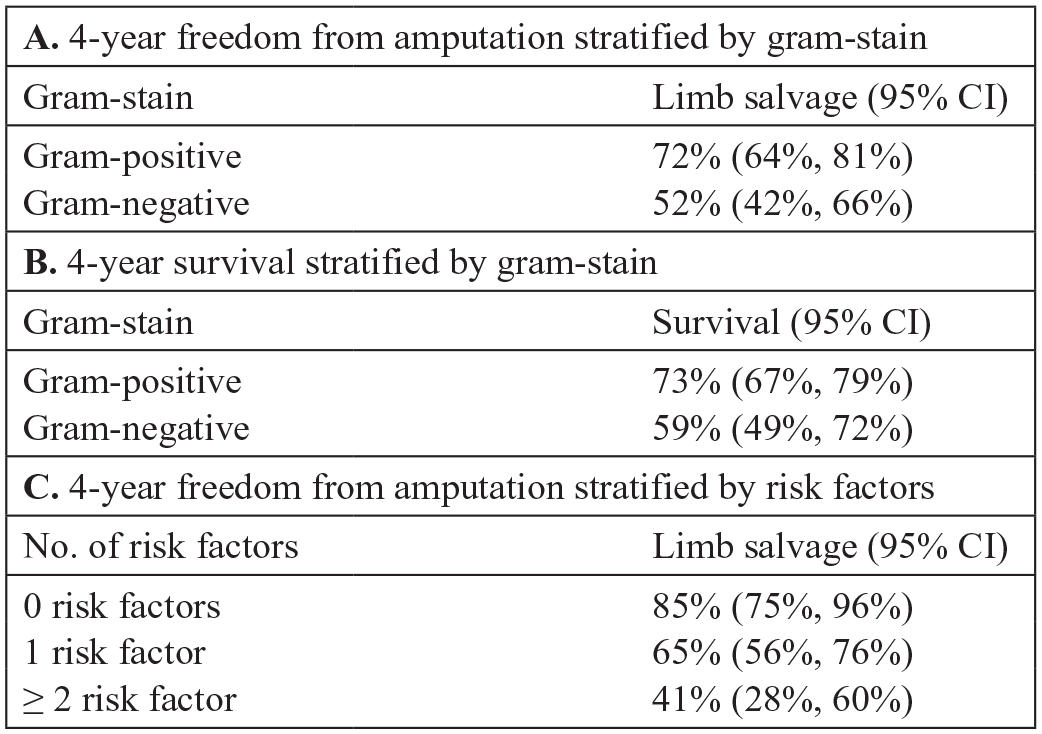
Corresponding numeric values to time-to-event graphs in Figure 2. Figure 3A corresponds with Figure 2B and 2C; 3B corresponds with 2D; and 3C corresponds with 2E.
Tables 2 and 3 illustrate the results of Cox proportional regression analysis to identify the hazards of limb loss and death from any cause after EVT, respectively. GNB identification, male sex, mean WIfI score, diabetes mellitus, and end-stage renal disease were independently and positively associated with amputation, with corresponding HRs of 1.9, 2.3, 1.6, 2.8, and 2.3, respectively (p < 0.05) (Table 2). Staphylococcus aureus identification was not associated with limb loss (Table 2). In contrast, mean WIfI score and end-stage renal disease were the only two variables independently and positively associated with death from any cause following EVT, with HRs of 1.8 and 3.0, respectively (p < 0.05) (Table 3).
Cox proportional regression analysis for limb loss following endovascular therapy in patients with peripheral artery disease Rutherford categories 5 and 6 with infected ulcers.
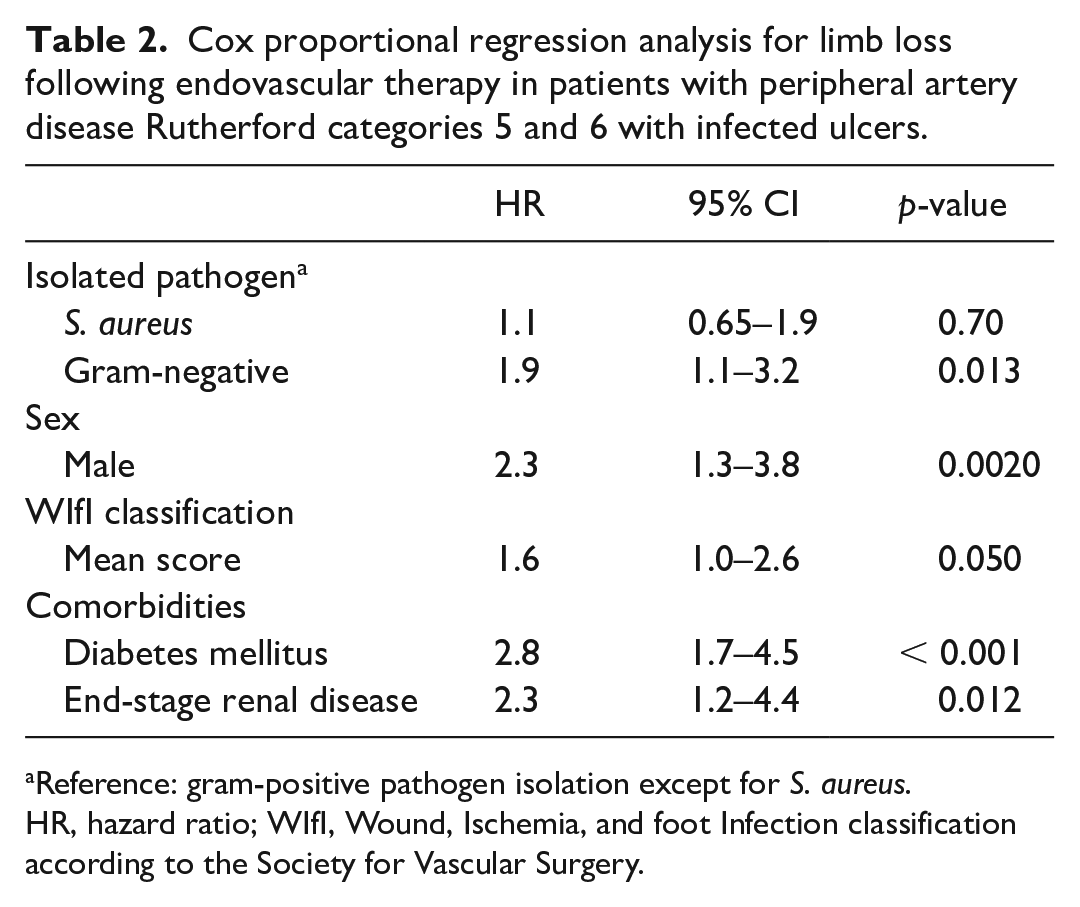
Reference: gram-positive pathogen isolation except for S. aureus.
HR, hazard ratio; WIfI, Wound, Ischemia, and foot Infection classification according to the Society for Vascular Surgery.
Cox proportional regression analysis for death from any cause following endovascular therapy in patients with peripheral artery disease Rutherford categories 5 and 6 with infected ulcers.
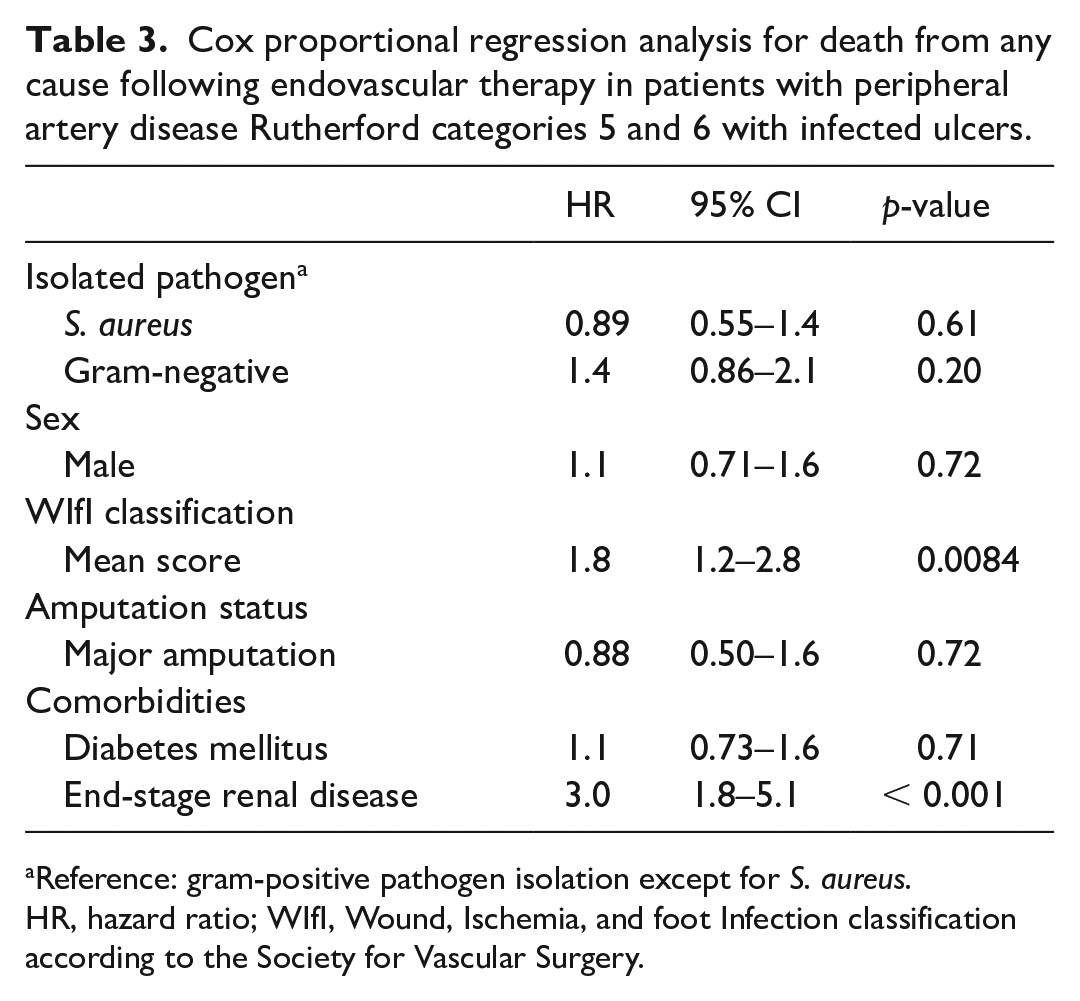
Reference: gram-positive pathogen isolation except for S. aureus.
HR, hazard ratio; WIfI, Wound, Ischemia, and foot Infection classification according to the Society for Vascular Surgery.
Multivariable logistic regression analysis identified the risk factors for postinterventional sepsis in patients with CLTI and infected leg ulcers (Table 4). The development of postinterventional sepsis was independently and positively associated with the identification of either S. aureus or GNB, as well as end-stage renal disease and diabetes mellitus (p < 0.05) (Table 4).
Risk factors for sepsis following endovascular therapy in patients with peripheral occlusive disease Rutherford categories 5 or 6.
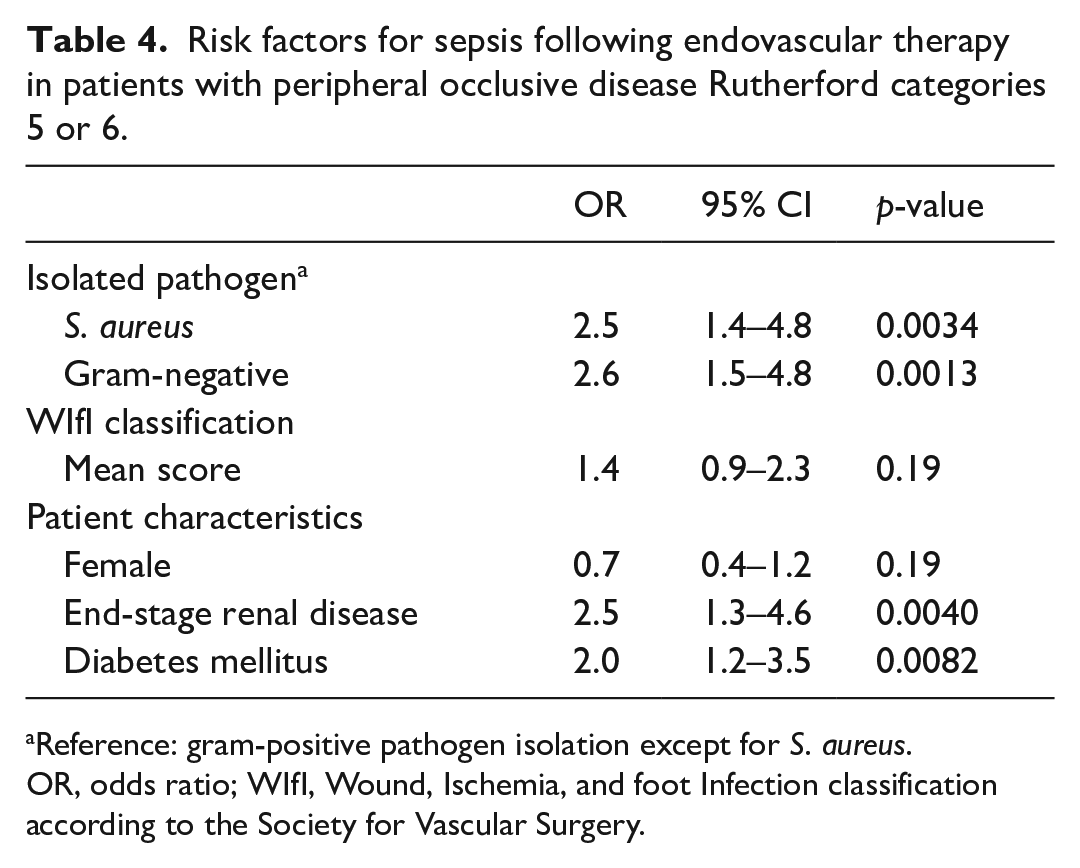
Reference: gram-positive pathogen isolation except for S. aureus.
OR, odds ratio; WIfI, Wound, Ischemia, and foot Infection classification according to the Society for Vascular Surgery.
Antibiotic regimes
Table 5 illustrates the microbiological activity of in-hospital-administered antibiotic regimes to patients following EVT, stratified by gram stain. Overall, the administered antibiotic regimes were microbiologically active in 17.4% (95% CI 13–23) of patients. When stratified by gram stain, the microbiological activity increased in patients with GPB to 28% (95% CI 20–37) and decreased in patients with GNB identification to 9% (95% CI 5.1–15) (p < 0.05). The most frequently prescribed antibiotics were aminopenicillins with beta (β)-lactamase inhibitors (28.2%) and clindamycin (26.9%). The majority of administered antibiotic regimes were combined (72.8%). Information on antibiotic regimes was missing in 31.9% of cases, including whether antibiotics were prescribed or not.
Microbiological activity of antibiotic regimes administered after endovascular therapy in patients with peripheral artery disease Rutherford categories 5 and 6 with infected leg ulcers by gram stain.
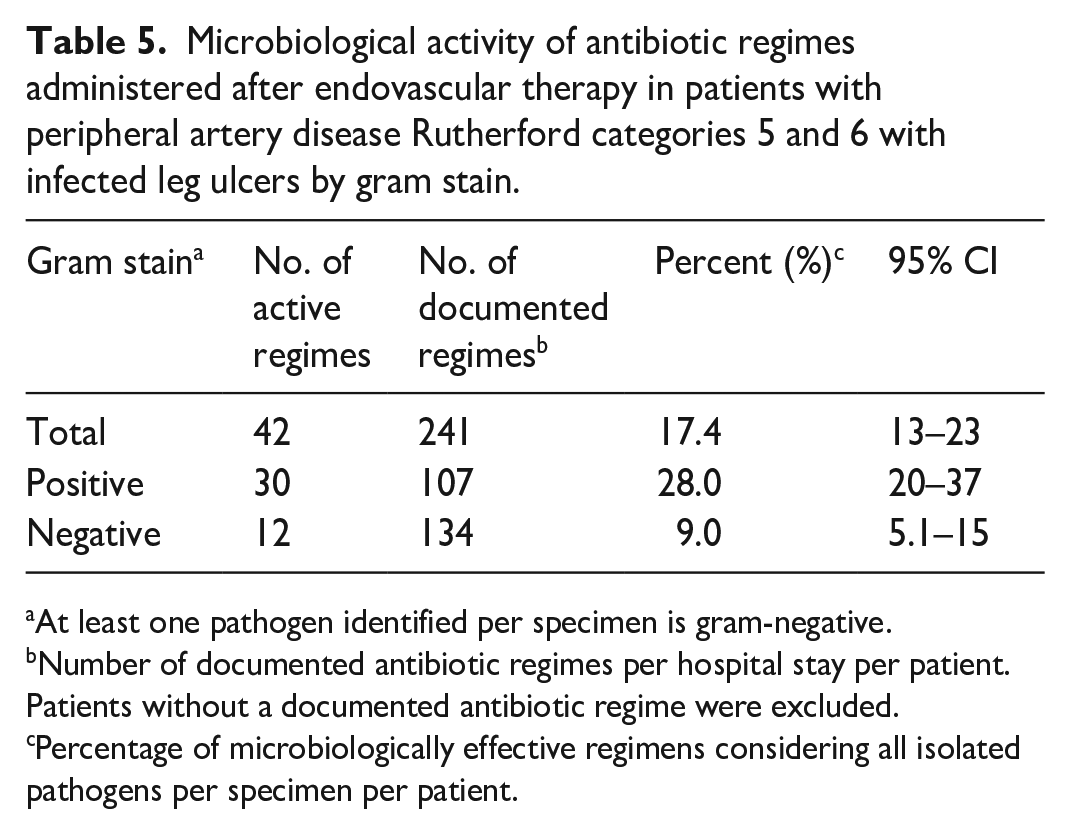
At least one pathogen identified per specimen is gram-negative.
Number of documented antibiotic regimes per hospital stay per patient. Patients without a documented antibiotic regime were excluded.
Percentage of microbiologically effective regimens considering all isolated pathogens per specimen per patient.
Discussion
This study indicated lower 4-year freedom from amputation in patients with CLTI undergoing EVT with GNB isolation compared to those with GPB (52% vs 72%). In this cohort, GNB identification was independently associated with higher rates of amputation than S. aureus identification.
Among patients with DFI, GNB were more prevalent in those who underwent amputation compared to those who did not. 8 This study revealed a similar pathogen–amputation relationship in patients with PAD, regardless of amputation extent. In our study, the isolation of GNB increased from 30.9% to 36.4% in patients with PAD requiring amputation.
GNB isolation was identified as a risk factor for amputation in a study of patients with DFI. 12 Similar to our results, de Vries et al. did not find an increased risk of amputation in patients with isolation of S. aureus only. However, both S. aureus and GNB isolation were independent risk factors for postinterventional sepsis. These findings align with the high potential of S. aureus for invasive infections. 13 Moreover, these findings indicate that, unlike isolated S. aureus infections, ischemic foot ulcers with GNB isolation pose a dual risk: they have a high potential for local destructive infections, leading to amputation, and an increased risk for invasive infections, resulting in postinterventional sepsis. Multiple recent studies have revealed male sex, increased mean WIfI score, diabetes mellitus, and end-stage renal disease as independent risk factors for amputation in patients with PAD, irrespective of their pathogen identifications.5,14–17 This study confirmed the previous findings and demonstrated a significant decrease in freedom from amputation after EVT with an increasing number of risk factors, including GNB identification, end-stage renal disease, and diabetes mellitus. The freedom from any amputation at 48 months decreased from 85% with no risk factors to 41% with two or more risk factors. These results emphasize the importance of these three risk factors for clinicians.
In a recent study, the risk factors for amputation and all-cause mortality differed. Only mean WIfI score and end-stage renal disease were identified as independent risk factors for death from any cause, whereas the type of identified pathogen was found irrelevant, suggesting that the likelihood of death is determined by the severity of the foot infection rather than the type of bacteria, as it is a component of the WIfI score.
A higher mean WIfI score emerged as a risk factor for both amputation and death from any cause and not for postinterventional sepsis. Nevertheless, recent research indicated that sepsis ranked as the second most common cause of 30-day readmission in patients with PAD after EVT with 8%. 18 This study included patients at all PAD stages, highlighting the significance of infection-associated complications. Although the WIfI score was designed to predict amputation, its reliability as a predictor of postinterventional sepsis remains uncertain. However, all patients in our cohort exhibited a WIfI grade greater than or equal to 1, indicating a preselected group that could introduce bias into the estimates. Eventually, we could not rule out the possible predictive value of the WIfI score for postinterventional sepsis.
Wound healing plays a chronic role in preventing amputation among patients with CLTI. Evidence suggests that managing biofilm is a crucial aspect of wound care, with notably higher healing rates observed in patients receiving biofilm-suppressive wound care compared to standard care alone. Biofilm management is an additional measure of potential antimicrobial therapy; however, it can be performed simply by mechanical debridement using cotton pads.19,20
Our study revealed a microbiological activity rate of only 17.4% for cultured organisms in postinterventional antibiotic regimes. As patients treated for PAD at our clinical center are often discharged the day after the procedure, antimicrobial test results are not available for all cases. Therefore, empirical antibiotic therapy is often used. This high rate of inadequate antibiotic regimes may be partly attributed to the frequent prescription of clindamycin, accounting for 26.9% of all prescribed antibiotics. Notably, clindamycin lacks activity against GNB, and resistance of S. aureus to clindamycin is prevalent among patients with CLTI, occurring in 11% of cases. 7 Therefore, empirical antibiotic therapy with clindamycin is likely to be ineffective. Consistent with this, the microbiological activity increases to 28% when considering patients with GPB isolation; however, it decreases to 9% for those with GNB isolation. Importantly, aminopenicillins with β-lactamase inhibitors were the most frequently prescribed antibiotic regimen, constituting 28.2% of prescriptions. Unlike clindamycin, these broad-spectrum antibiotics are not affected by the limitations mentioned above. However, information regarding the antibiotic regimen was obtained by scanning discharge letters, leading to a lack of information on the administered regimen or whether antibiotics were given at all for 246 hospital stays out of a total of 771. Therefore, acknowledging the potential for bias and interpreting the results regarding microbiological activity should be approached with caution.
This study had several limitations, including its single-center and retrospective design, which introduced bias. Additionally, single-center microbiological studies may have reduced external generalizability due to potential regional differences in pathogen distribution. Moreover, the analysis relied on final microbiological test results, raising the possibility of pathogen preselection, such as the suppression of potential commensals or irrelevant pathogens by clinical microbiologists. Furthermore, retrospective analyses also limited access to comprehensive patient data, potentially resulting in incomplete information, particularly regarding amputations conducted at external clinics without our knowledge.
Conclusion
Although the isolation of both GNB and S. aureus are risk factors for sepsis following EVT, GNB isolation is independently associated with higher rates of amputation. The low microbiological activity of the administered antibiotic regimes raises concern. The use of clindamycin as an empirical therapy is highly questionable due to the risk of amputation and sepsis associated with the identification of GNB. These findings underscore the significance of infection-related outcomes and emphasize the importance of adequate microbiological testing in patients with CLTI.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X241268692 – Supplemental material for Clinical outcomes in patients with chronic limb-threatening ischemia and infected ulcers following endovascular therapy are pathogen dependent: A single-center experience from 2012 to 2021
Supplemental material, sj-docx-1-vmj-10.1177_1358863X241268692 for Clinical outcomes in patients with chronic limb-threatening ischemia and infected ulcers following endovascular therapy are pathogen dependent: A single-center experience from 2012 to 2021 by Jonas Salm, Franziska Ikker, Tanja Böhme, Elias Noory, Ulrich Beschorner, Tobias Siegfried Kramer, Siegbert Rieg, Dirk Westermann and Thomas Zeller in Vascular Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge the following colleagues for their help and support: Dr Peter Christian Flügel (data extraction), Dr Karlheinz Bürgelin (data extraction), Dr Börries Jacques (data extraction), Dr Kolja Sievert (data extraction), Anja Thoma (wound management), Eugen Ritz (data extraction), and all employees from the MVZ Clotten Freiburg (data extraction).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
