Abstract

Chronic limb-threatening ischemia (CLTI) is the most severe stage of peripheral artery disease (PAD) with a high risk of amputation. 1 Among prostacyclin analogues, iloprost has been proposed as an alternative to amputation when revascularization is impossible (i.e., patients with ‘no option’). Nonetheless, there is limited evidence of iloprost interest.2,3 We aimed to assess iloprost benefit and whether it would justify the risks associated to its use in a real-life setting.
The COPART (COhorte des Patients ARTériopathes) prospective multicenter registry recruited consecutively patients hospitalized for atherosclerotic PAD (patients with thromboangiitis obliterans were not included) from three French teaching hospitals’ vascular medicine departments (Bordeaux, Limoges, Toulouse). 4 All patients included in the COPART registry gave their informed oral consent for the collection of their information. Patients enrolled in COPART from October 2006 to January 2021 were eligible for this study. They were included if they had rest pain or trophic disorders associated with a hemodynamic assessment (ankle pressure ⩽ 50 mmHg, toe pressure or transcutaneous pressure of oxygen ⩽ 30 mmHg). Patients with CLTI were considered ‘no-option’ patients if they did not receive revascularization procedures during the time of hospitalization, or if no revascularization was planned after hospital discharge according to the COPART data. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethic Committee of Toulouse University Hospital. Patients exposed to iloprost were matched with up to three unexposed patients according to age, sex, and a propensity score (PS) for iloprost exposure. The effectiveness outcome of interest combined all-cause death and major amputations. One-year survival analysis used Kaplan–Meier estimates and Cox models. Major adverse cardiovascular events (MACE) was the safety outcome of interest; its association with iloprost was estimated using logistic regression. All analyses were performed using SAS Enterprise Guide 7.1 software (SAS Institute Inc., Cary, NC, USA).
Among the 1850 patients with CLTI included in the COPART registry, 201 were treated with iloprost (71.6% men, median age: 72 years; unexposed: 72.1% men, median age: 75 years) and 79.6% (160 patients) of them were from Bordeaux Hospital. Almost half of patients were not treated with statins (Table 1). A total of 146 patients exposed to iloprost could be matched to 397 unexposed patients. Among the matched population, 307 (56.5%) patients were considered ‘no-option’ patients. Fourteen major amputations and 24 deaths occurred in patients treated with iloprost (26%) versus 33 and 46 in unexposed (20%) (Supplemental Figure S1, available online). Treatment with iloprost was associated with an increased risk of these events (hazard ratio (HR): 1.49; 95% CI: 1.01–2.20). The association remained in the subgroup of ‘no-option’ patients (HR: 1.74; 95% CI: 1.01–2.20). MACE occurred in 21 (10.7%) patients treated with iloprost versus 146 (9.41%) unexposed. The association with iloprost was nonsignificant (adjusted odds ratio: 1.23; 95% CI: 0.72–2.11). This study does not support a beneficial effect of iloprost in patients with CLTI in a real-life setting; the results even suggest it could be deleterious. A higher risk of all-cause death and major amputations was found in the exposed group, especially in the ‘no-option’ group. The results are consistent with the Cochrane review by Vietto et al., which found no benefit of prostanoids in patients with CLTI, especially regarding total amputations. 5 If iloprost is theoretically assumed to improve circulation, its pharmacodynamic effects in patients with CLTI are uncertain. Perfusion could indeed be more improved by iloprost in well-perfused areas than in poorly perfused ones. 6 Also, iloprost might have paradoxical thrombotic effects in patients with PAD.7–9 This study used PS to control for potential confounding and strengthen the controlling of a potential indication bias. However, it shows some limitations as all treated patients could not be included, and as information on treatment completion was not available.
Baseline characteristics of patients with chronic limb-threatening ischemia at hospital admission.
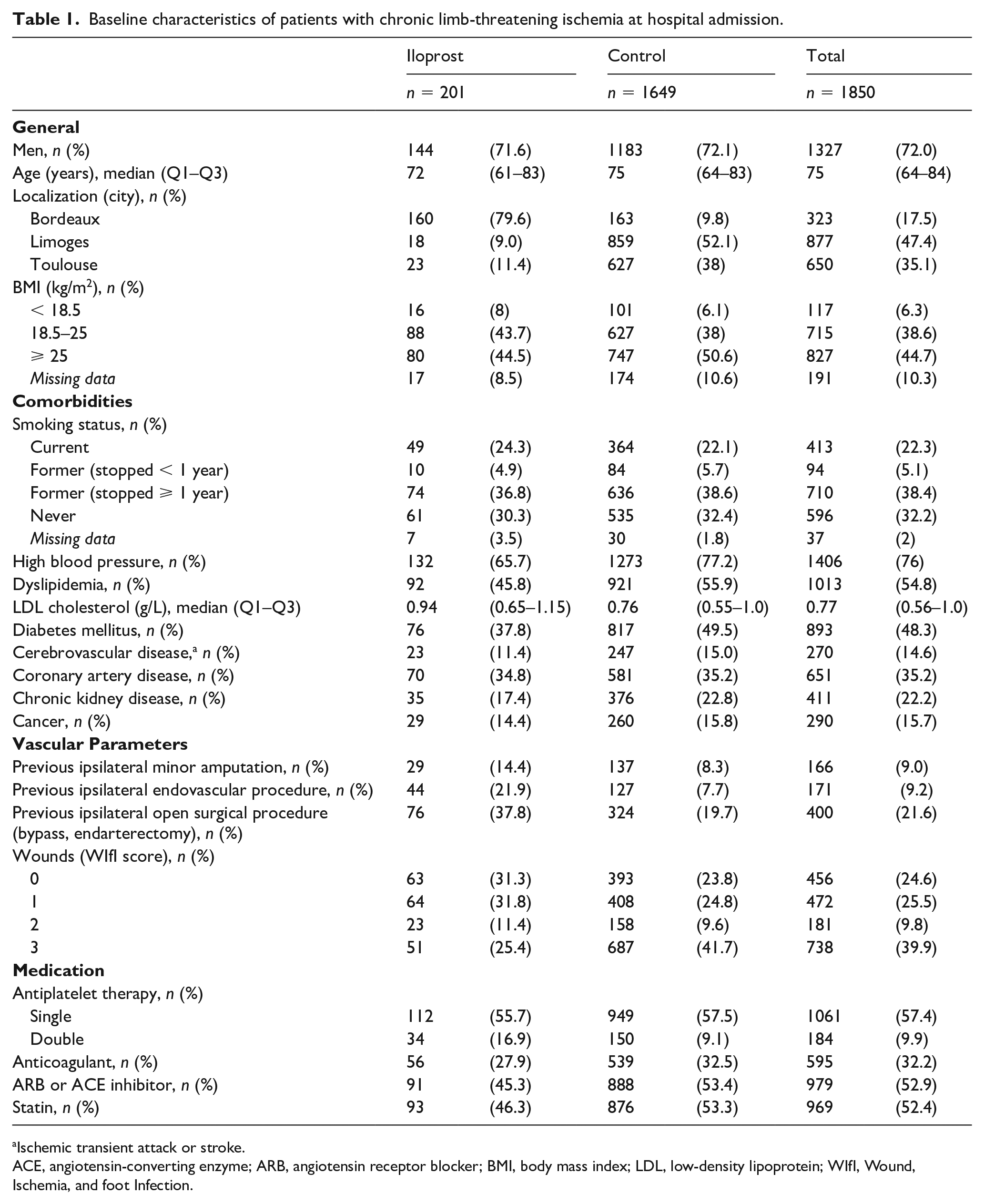
Ischemic transient attack or stroke.
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; LDL, low-density lipoprotein; WIfI, Wound, Ischemia, and foot Infection.
In conclusion, this study did not support the use of iloprost in patients with CLTI, especially in ‘no-option’ patients.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231209263 – Supplemental material for Real-world effect of iloprost in patients with chronic limb-threatening ischemia (CLTI): A cohort study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231209263 for Real-world effect of iloprost in patients with chronic limb-threatening ischemia (CLTI): A cohort study by Dari Loubna, Joël Constans, Carine Boulon, Caroline Caradu, François-Xavier Lapébie, Alessandra Bura-Riviére, Lucie Chastaingt, Philippe Lacroix, Julien Bezin and Antoine Pariente in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr Lapébie and Dr Bura-Rivière reports the receipt of personal fees from Bayer HealthCare SAS outside the submitted work. The other authors have nothing to disclose.
Funding
The COPART registry is supported by the French Society of Vascular Medicine and by grants from AstraZeneca France, Bayer France, Bristol-Myers Squibb France, and Sanofi-Aventis France companies.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
