Abstract
Purpose:
Gingerol inhibits growth of cancerous cells; however, its role in vascular smooth muscle cell (VSMC) proliferation is not known. The present study investigated the effect of gingerol on VSMC proliferation in cell culture and during neointima formation after balloon injury.
Method and Results:
Rat VSMCs or carotid arteries were harvested at 15 minutes, 30 minutes, 1, 6, 12, and 24 hours of fetal bovine serum (FBS; 10%) stimulation or balloon injury, respectively. Gingerol prevented FBS (10%)-induced proliferation of VSMCs in a dose-dependent manner (50 μmol/L-400 μmol/L). The FBS-induced proliferating cell nuclear antigen (PCNA) upregulation and p27Kip1 downregulation were also attenuated in gingerol (200 μmol/L) pretreated cells. Fetal bovine serum-induced p38 mitogen-activated protein kinase (MAPK) activation, PCNA upregulation, and p27Kip1 downregulation were abrogated in gingerol (200 μmol/L) and p38 MAPK inhibitor (SB203580, 10 μmol/L) pretreated cells. Balloon injury induced time-dependent p38 MAPK activation in the carotid artery. Pretreatment with gingerol (200 μmol/L) significantly attenuated injury-induced p38 MAPK activation, PCNA upregulation, and p27Kip1 downregulation. After 14 days of balloon injury, intimal thickening, neointimal proliferation, and endothelial dysfunction were significantly prevented in gingerol pretreated arteries. In isolated organ bath studies, gingerol (30 nmol/L-300 μmol/L) inhibited phenylephrine-induced contractions and induced dose-dependent relaxation of rat thoracic aortic rings in a partially endothelium-dependent manner.
Conclusion:
Gingerol prevented FBS-induced VSMC proliferation and balloon injury-induced neointima formation by regulating p38 MAPK. Vasodilator effect of gingerol observed in the thoracic aorta was partially endothelium dependent. Gingerol is thus proposed as an attractive agent for modulating VSMC proliferation, vascular reactivity, and progression of vascular proliferative diseases.
Keywords
Introduction
Gingerol, a major pharmacologically active component of ginger, has been reported to exhibit anti-inflammatory, antihypertensive, and antioxidant activity. 1 Mounting evidence suggests that gingerol is effective in suppressing the transformation, hyperproliferation, and inflammatory processes that initiate and promote carcinogenesis. 2 Atherosclerosis is a chronic inflammatory disorder that also involves hyperproliferation and inflammation in the vessel wall. 3 Proliferation and migration of vascular smooth muscle cell (VSMC) play a pivotal role in atherosclerosis and restenosis. 4 The VSMCs in the neointima synthesize and excrete extracellular matrix and collagen, which facilitate neointimal lesion formation and degradation of the extracellular matrix associated with vascular remodeling. 5 Thus, therapeutic interventions that inhibit the proliferation of VSMC may also be beneficial in the prevention of intimal hyperplasia and restenosis. The p38 mitogen-activated protein kinase (MAPK) signaling cascade appears to be a unifying pathway for key cellular mechanisms of diverse cardiovascular pathologies. 6
Arterial injury following percutaneous coronary intervention releases various factors that promote VSMC proliferation and migration. 7 It has previously been shown that p38 MAP kinase is significantly activated in balloon-injured artery, and inhibition of p38 MAPK prevents balloon injury-induced neointimal hyperplasia. 8 Gingerol has been shown to prevent activation of p38 MAPK in phorbol ester-stimulated mouse skin. 9 Therefore, in the present study, we hypothesized that gingerol may prevent VSMC proliferation by modulating p38 MAPK and thus prevent progression of neointimal hyperplasia, which involves endothelial dysfunction and robust VSMC proliferation
Materials and Methods
Collagenase type I, methylene tetrazolium (MTT), propidium iodide (PI), Pluronic F127, Mayer’s hematoxylin, eosin, phenylephrine hydrochloride (PE), acetylcholine chloride (ACh), and antibodies including alpha smooth muscle actin, β actin, and anti-mouse horseradish peroxidase (HRP) conjugated secondary antibody were obtained from Sigma (St Louis). ERK pathway inhibitor (U0126), p38 MAPK inhibitor (SB 203580), proliferating cell nuclear antigen (PCNA) antibody, and 6-gingerol were procured from Calbiochem (San Diego, California). Anti-p27Kip1 antibody was purchased from BD biosciences (San Diego, California). p38, p-p38, p-ERK, and p-AKT antibodies were purchased from Cell Signaling Technology (Danvers, Massachusetts). Dulbecco modified eagle medium (DMEM) and fetal bovine serum (FBS) were purchased from Invitrogen (Carlsbad). 2F Fogarty balloon embolectomy catheter was purchased from Edwards Life Sciences (Milan, Italy). Avidin–biotin peroxidase complex (ABC) kit was purchased from Vector Laboratories (Burlingame).
Cell Culture Experiments
The VSMCs were isolated from the thoracic aorta of Sprague Dawley rats (150 to 180 g) by enzymatic digestion technique as described previously.
10
Cells were cultured in DMEM, supplemented with 10% FBS and antibiotics at 37°C in a humidified atmosphere of 95% air and 5% C
Cell Proliferation Assay
Rat aortic smooth muscle cells were seeded into 96-well plates at density of 2 × 103 cells/mL and then cultured in DMEM containing 10% FBS at 37°C for 24 hours. Cells were then incubated with serum-free medium for 24 hours and stimulated by 10% FBS. 11 The proliferation of VSMC was analyzed 24 hours after FBS stimulation. To evaluate the effect of gingerol, quiescent VSMCs were pretreated with gingerol (50-400 µmol/L, for 24 hours), 2 U0126 (10 µmol/L, for 1 hour), or SB 203580 (10 µmol/L, for 1 hour) in serum-free medium and then stimulated by 10% FBS for 24 hours. 12 In other set of experiments, quiescent VSMCs were pretreated simultaneously with gingerol (200 µmol/L, 24 hours) and SB 203580 (10 µmol/L, 1 hour) followed by stimulation with 10% FBS for 24 hours. Cells were washed once and incubated with 0.5 mg/mL MTT at 37°C for 1 hour. Tetrazolium salts are reduced and form an insoluble blue formazan product. After incubation, formazan products were dissolved with dimethyl sulfoxide (DMSO), and absorbance was read at 560 nm using ELISA plate reader (Powerwave XS; Biotek), and the results were expressed as optical density values. 13
Cell Cycle Progression Analysis
Synchronized VSMCs were subcultured into 6-well tissue culture plates at 1 × 106 cells/well. The assay condition was same as described in the section of cell proliferation assay. After being stimulated by FBS, cells were trypsinized at different time points (15 minutes to 24 hours) and centrifuged at 150 g for 5 minutes, and the cell pellets were resuspended in hypotonic PI solution (50 μg/mL with 0.03% NP40 in 0.1% sodium citrate). Samples were examined after 10 to 20 minutes of staining at 4°C. The DNA content in these cells was measured using a flow cytometer (FACSCalibur; Becton Dickinson, San Jose, California) and the CellQuest program and then the cell-phase distribution as well as apoptosis was analyzed by Modfit software (Maine). 14
Immunoblotting Assay
The assay condition was the same as described in the section of cell cycle progression analysis. Cells were harvested after desired treatments and lysed in lysis buffer. Cell extracts were clarified at 15 000g for 5 minutes, and protein contents were measured. Equal amounts of total protein samples of VSMC or rat carotid artery lysate were run on 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis, transferred on a polyvinylidene fluoride membrane and blocked with Tris-buffered saline with Tween 20 (TBST) containing 5% bovine serum albumin (BSA) for 1 hour. 15 After blocking, the membranes were then probed with primary antibodies for PCNA (1:200), p27Kip1 (1:200), p38 MAPK (1:1000), p-p38 MAPK (1:1000), Phospho-AKT (Phospho-AKT, AKT is also known as Protein kinase B), Phospho-ERK1/2 (Extracellular-signal Regulated kinases), and anti-β-actin (1:2000). This was followed by incubation with specific HRP-conjugated secondary antibody. The specific bands were detected by enhanced chemiluminescence. The detected proteins were normalized by β-actin or respective total proteins. The intensities of bands were quantified using Image Quant, LAS 4000 (Amersham, Massachusetts). The Western blot data are representative of 1 of the 3 similar blots from 3 different experiments.
Animal Diet and Treatment Protocol
Male Sprague Dawley rats (250-300 g) used in the present study were obtained from the National Laboratory Animal Centre. The animals received humane care in compliance with the Guidelines for the Care and Use of Laboratory Animals. Rats were kept in polypropylene cages at 24°C ± 0.5°C, 12-hour day–night cycle and were provided ad libitum water and food. Experimental protocols were approved by the Institutional Animal Ethics Committee that follows guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals, which conforms to international norms of Indian National Science Academy. Pluronic F127 was dissolved in sterile water, 30% (w/v), under continuous stirring at 4°C for 24 hours to prepare Pluronic gel. 16 The Pluronic gel remains in a liquid form at 4°C and rapidly forms a gel on exposure to body temperature. On the day of surgery, animals were anesthetized with an intraperitoneal injection of ketamine (50 mg/kg) and xylazine (6.7 mg/kg). Gingerol (200 µmol/L) was mixed in Pluronic gel. The right carotid artery was surgically exposed, and Pluronic gel (200 µL of 30% F127 Pluronic gel) with or without gingerol (200 µmol/L in 30% pluronic gel) was applied to 1 cm segment of common carotid artery at its bifurcation to external and internal carotid arteries. 17 For balloon injured control, vehicle gels were administered which contained DMSO only. After 24 hours of gingerol treatment, a 2F Fogarty balloon embolectomy catheter (Edwards Life Sciences) was advanced along the length of the common carotid artery and retracted 3 times under mild balloon inflation pressure (0.02 mL saline) to ensure endothelial damage. 18,19 Left carotid arteries served as uninjured control. The artery was visualized for patency and pulsation with luminal blood flow as described previously. 18 Ampicillin (50 mg/kg) was given immediately after the surgical procedure. Acetaminophen 10 mg/kg body weight was given orally for postoperative pain relief for 3 days. At the end of study period, the animals were anaesthetized, and carotid arteries were exposed. Pulsation in the artery was again visualized, and we did not observe any vasospasm or blood clot in the artery. Animals were euthanized, and the treated portion of arteries was isolated and fixed with formalin for 24 hours and processed for histology or immunohistochemistry. Carotid artery from different rats treated with or without inhibitors was used for immunoblotting or vasoreactivity studies.
Morphometric Analysis
Carotid artery segments of rats were dissected and embedded in paraffin according to standard procedures. 18 Serial paraffin sections of 5 μm thick were cut at 300 μm intervals throughout the arterial segment. To characterize the general architecture of the arteries, serial cross-sections underwent hematoxylin and eosin (HE) staining. Both balloon injured and uninjured portion of artery underwent similar fixing and dehydration and wax infiltration procedures to nullify the morphological changes induced by solvent treatments in all groups. Intimal area, medial area, neointima/media area ratio, lumen area, and percentage cross-sectional narrowing (CSN) were calculated by blinded investigators employing computer-assisted image analysis software (Leica Qwin version 3.5.1, Switzerland). Morphometric analysis was done as described previously. 20 Briefly, lumen area was calculated as the area of the lumen without plaque. Internal elastic lamina (IEL) area and external elastic lamina (EEL) area were calculated as the area circumscribed by the IEL or EEL traced on stained sections. Intimal area (I) was determined by subtracting lumen area from the area defined by the IEL. The medial area (M) was calculated by subtracting the area defined by the IEL from the area defined by the EEL. Intima by media (I/M) area ratio was calculated by dividing the intimal area by medial area. The CSN was defined as the extent to which the intimal area occupied the potential lumen area and was calculated as % CSN = (Intimal area/IEL area) × 100. Morphometric analysis was performed using bright field microscopy by an Leica DM5000B microscope (Leica Microsystems, Cambridge, United Kingdom) utilizing the 10× and 40× Leica objectives. Imaging was performed using Leica DFC320 camera (Cambridge, United Kingdom). Leica application suite software package version 3.1.0 was used for processing and saving images.
Immunohistochemistry
Paraffin sections (5 μm) were deparaffinized, dehydrated, and warmed for 20 minutes in citrate buffer (10 mmol/L, pH 6.0) for antigen retrieval, followed by blocking of endogenous peroxidase (3% H2
Assessment of Endothelial Function
Thoracic aorta of normal Sprague-Dawley rats or carotid artery of rats from different groups was excised and immediately placed in ice cold Krebs bicarbonate medium. Aorta and arteries were dissected free of connective tissue and fat and cut into rings (4 mm in length). The rings were mounted vertically between 2 stirrups in organ chambers filled with 10 mL Krebs solution maintained at 37°C±0.5°C constantly bubbled with 95% O2 to 5% CO2. One stirrup was connected to an anchor and the other to a force transducer for recording of isometric tension (SPEL Advanced Isosys organ bath, Experimetria, Hungary). Then rings were allowed for equilibration followed by 10 to 12 washing. To assess the maximum tissue contractility and viability, the rings were exposed to 80 mmol/L KCl Krebs solution. The response was measured before and after the experimental protocol in each aortic ring. After the assessment of maximum tissue contractility, contraction/relaxation response was taken with PE/ACh in control as well as treated groups. 23 To assess the per se effect of gingerol, rat aortic rings were incubated with a wide range of concentrations of gingerol (30-300 µmol/L), which was based on the previous published reports. 24,25 Gingerol was incubated for 30 minutes and dose–response of PE (1-100 µmol/L) was monitored. To assess the per se effect of gingerol (3-300 µmol/L) for their vasorelaxant efficacy, responses were obtained in PE (1 µmol/L) precontracted endothelium intact rings.
Statistical Analysis
Experimental values in the results are expressed as mean ± standard error of the mean. Significance of difference between groups was analyzed by performing one-way analysis of variance followed by Dunett post hoc test. The significance level for Dunett multiple comparison test was set to .05 for 3 or more groups, and the P value less than .05 was considered as statistically significant.
Results
Gingerol Prevents FBS-Induced VSMC Proliferation, Cell Cycle Progression, and Upregulates p27Kip1
For ascertaining the effect of gingerol on VSMC proliferation, MTT assay, cell cycle analysis, PCNA, and p27Kip1 expression were monitored in gingerol pre-treated VSMCs stimulated with FBS. Treatment with FBS-induced a significant increase in VSMC proliferation (~2.2-fold) as compared with unstimulated quiescent VSMCs (Figure 1A). A tendency of decrease in VSMC proliferation was observed at 50 µmol/L (~1.2-fold) and 100 µmol/L (~1.2-fold) gingerol concentration as determined by MTT assay. A significant decrease in VSMC proliferation was observed at 200 μmol/L (~1.65-fold) and 400 μmol/L (~1.7-fold) concentrations of gingerol when compared to FBS-stimulated VSMCs (Figure 1a). Serum deprivation of rat aortic VSMCs for 24 hours resulted in an approximately 90% synchronization of the cell cycle in the G0 phase. A significant increase (~6-fold) in the percentage of cells in the S phase was observed after 24 hours of FBS stimulation (Figure 1B) as compared to quiescent VSMCs. Pretreatment with gingerol at 50 µmol/L (~1.5-fold) and 100 µmol/L (~1.6-fold) dose showed a tendency of decrease in S phase, albeit not significant. Gingerol at concentration of 200 µmol/L (~2.4-fold) and 400 µmol/L (~2.6-fold) significantly prevented FBS-induced increase in S phase (Figure 1B). However, gingerol even at highest concentration of 200 and 400 µmol/L showed negligible cell death (0.58% ± 0.1% and 0.51% ± 0.05% apoptosis, respectively). Results from MTT and cell cycle studies revealed that gingerol exhibited almost similar activity at 200 and 400 µmol/L concentrations, therefore 200 µmol/L concentration was chosen for further studies. Pretreatment of gingerol prevented (1.5-fold) FBS-induced increase (~2.12-fold) in the PCNA protein expression (Figure 1C). To monitor the effect of gingerol at biochemical level, p27Kip1 expression was monitored in VSMCs. Pretreatment with gingerol in VSMCs significantly prevented (~1.64-fold) the FBS-induced decrease in p27Kip1 expression (~1.89-fold; Figure 1D).

Gingerol prevents FBS-induced VSMC proliferation. VSMCs were pretreated with gingerol (50-200 µmol/L) for 24 hours and then stimulated with FBS (10%). Cells were processed for MTT assay, cell cycle analysis, PCNA, p27Kip1, and β-actin blotting. Bar diagram representing (A) MTT assay and (B) cell cycle analysis. Western blot analysis of (C) PCNA and (D) p27Kip1 expression. The results are presented as mean ± SEM of 3 independent experiments, and each analysis was run in triplicates. Graphs in C and D show densitometric analysis of blots (mean ± SEM) from 3 separate experiments. Blots represent one of the 3 similar experiments, which were carried out at least 3 times. ***P < .001 versus unstimulated quiescent VSMCs. ##P < .05, ###P < .001 versus FBS stimulated VSMCs. FBS indicates fetal bovine serum; VSMC, vascular smooth muscle cell; MTT, methylene tetrazolium; PCNA, proliferating cell nuclear antigen; SEM, standard error of the mean.
FBS-Induced p38 MAPK Activation is Prevented by Gingerol
To evaluate time course of p38 MAPK activation, VSMCs lysate obtained after 15, 30 minutes and 1, 6, 12, and 24 hours of FBS stimulation was subjected to Western blotting. A significant increase in p38 MAPK phosphorylation was observed at 15 minutes (~3.4-fold), 30 minutes (~2.9-fold), and 1 hour (~2.7-fold) after FBS stimulation, which was subsequently decreased at 6 hours (~1.4-fold), 12 hours (~1.5-fold), and 24 hours (~1.1-fold; Figure 2A). Pretreatment of gingerol prevented (~1.6-fold) FBS-induced increase in the p38 MAPK activation analyzed after 15 minutes of FBS stimulation (Figure 2B). We also monitored AKT and ERK activation in the presence of gingerol, since they are also known to play a role in serum-induced VSMC proliferation. 26 –28 Although serum induced a significant increase in ERK (Figure 2C) and AKT (Figure 2D) phosphorylation, no change was observed in gingerol pretreated and serum stimulated cells. Therefore, serum did induce AKT and ERK activation but gingerol had no significant effect on their activation (Figure 2C and D). To ascertain whether p38MAPK pathway is important for gingerol’s action on serum-induced VSMC proliferation, cells were preincubated with gingerol, SB203580, or both and subsequently MTT assay, cell cycle analysis, PCNA, and p27Kip1 expression was monitored after serum stimulation. Gingerol and SB203580 significantly reduced cell proliferation as evident in MTT assay (Figure 3A) and cell cycle analysis (Figure 3B). Serum-induced PCNA upregulation (Figure 3C) and p27Kip1 (Figure 3D) downregulation were significantly attenuated in gingerol and SB203580 pretreated cells. Although simultaneous addition of gingerol and SB203580 attenuated cell proliferation (Figure 3A and B) and changes in PCNA (Figure 3C) and p27Kip1 (Figure 3d) expression, the changes observed were not significantly different from that observed with gingerol and SB203580 alone.

Gingerol prevents FBS-induced p38 MAPK activation. Quiescent VSMCs were stimulated with FBS (10%) for 15 minutes, 30 minutes, 1 hour, 6 hours, 12 hours, and 24 hours. Cells were processed for p-p38 MAPK, p38 MAPK, and β-actin Western blotting. Western blot analysis of (A) time-dependent p38 MAPK activation. Effect of gingerol (200 µmol/L) pretreatment on (B) p38 MAPK activation, (B) ERK activation, and (C) AKT activation after 15 minutes of FBS stimulation. Gingerol treatment did not show any significant effect on ERK or AKT activation. Graphs show densitometric analysis of blots (mean ± SEM) from 3 separate experiments. Blots represent one of the 3 similar experiments which were carried out at least 3 times. **P < .01, ***P < .001 versus unstimulated quiescent VSMCs. ###P < .001 versus FBS stimulated VSMC. FBS indicates fetal bovine serum; MAPK, mitogen-activated protein kinase; VSMC, vascular smooth muscle cell; SEM, standard error of the mean.

p38 MAPK is key to gingerol’s action. Effect of gingerol in the presence and absence of SB 203580 on cell proliferation as measured by (A) MTT assay, (B) cell cycle analysis, (C) PCNA protein expression, and (D) p27Kip1 protein expression. The results are presented as mean ± SEM of 3 independent experiments and each analysis was run in triplicates. Graphs in C and D show densitometric analysis of blots (mean ± SEM) from 3 separate experiments. Blots represent one of the 3 similar experiments, which were carried out at least 3 times. **P < .01, ***P < .001 versus unstimulated quiescent VSMCs. #P < .05, ##P < .01, ###P < .001 vesus FBS stimulated VSMC. MAPK indicates mitogen-activated protein kinase; MTT, methylene tetrazolium; PCNA, proliferating cell nuclear antigen; VSMC, vascular smooth muscle cell; SEM, standard error of the mean.
Gingerol Prevents Balloon Injury-Induced Proliferation, p27Kip1 Downregulation, and p38 MAPK Activation
To further elucidate the impact of gingerol on proliferative response to vascular injury, the protein expression of PCNA and p27Kip1 was analyzed at 24 hours postballoon injury in uninjured artery and balloon injured artery pretreated with or without gingerol. Injury-induced–enhanced PCNA expression (~3.8-fold) was prevented (~1.5-fold) in gingerol pretreated arteries (Figure 4A). Likewise, gingerol pretreatment also prevented (~1.63-fold) balloon injury-induced decrease in p27Kip1 expression (~1.96-fold; Figure 4B). To further elucidate the impact of gingerol on p38 MAPK activation in response to balloon injury, the expression level of p38 and p-p38 was monitored. A significant increase in phospho-p38 MAPK, normalized to the total p38 MAPK, was observed at 15 minutes (~3.3-fold), 30 minutes (~2.96-fold), and 1 hour (~1.8-fold) after balloon injury (Figure 4C), which was subsequently decreased at 6 hours (~1.26-fold), 12 hours (~1.1-fold), and 24 hours (~1.1-fold; Figure 4C). Gingerol pretreatment prevented (~1.23-fold) injury-induced p38 MAPK activation (Figure 4d) analyzed at 15 minutes after balloon injury (Figure 4D).

Gingerol prevents vascular proliferation by modulating p27Kip1 and p38 MAPK activation. Balloon injury was done in the carotid artery of rats pretreated with or without gingerol (200 µmol/L). (A) PCNA and (B) p27Kip1 protein expression was analyzed 24 hours after balloon injury. C, p38 MAPK phosphorylation was determined after 15 minutes, 30 minutes, 1 hour, 6 hours, 12 hours, and 24 hours of balloon injury by Western blotting. D, Effect of gingerol on p38 MAPK phosphorylation was monitored 15 minutes after balloon injury in carotid artery of rats pretreated with gingerol. Blots represent one of the 3 similar experiments, which were carried out at least 3 times. Graphs show densitometric analysis of blots (mean ± SEM) from 3 separate experiments. Each experiment had a minimum of 3 animals in each group. ***P < .001 versus uninjured control artery. ###P < .001 versus balloon injured artery. MAPK indicates mitogen-activated protein kinase; PCNA, proliferating cell nuclear antigen; SEM, standard error of the mean.
Gingerol Prevents Balloon Injury-Induced Neointimal Cell Proliferation and Hyperplasia
The efficacy of gingerol on neointimal hyperplasia was assessed 14 days after balloon injury by determining the area of newly formed intima in carotid artery sections of rats stained with HE. The HE staining in the present study demonstrated blue nuclei and red-pink cytoplasm (Figure 5A-C). There was no neointimal formation in the left carotid artery, which was not subjected to vascular injury (Figure 5A). Balloon injury induced aggressive circumferential neointimal growth and luminal narrowing at 14 days of balloon injury, and no incidence of thrombosis was observed.
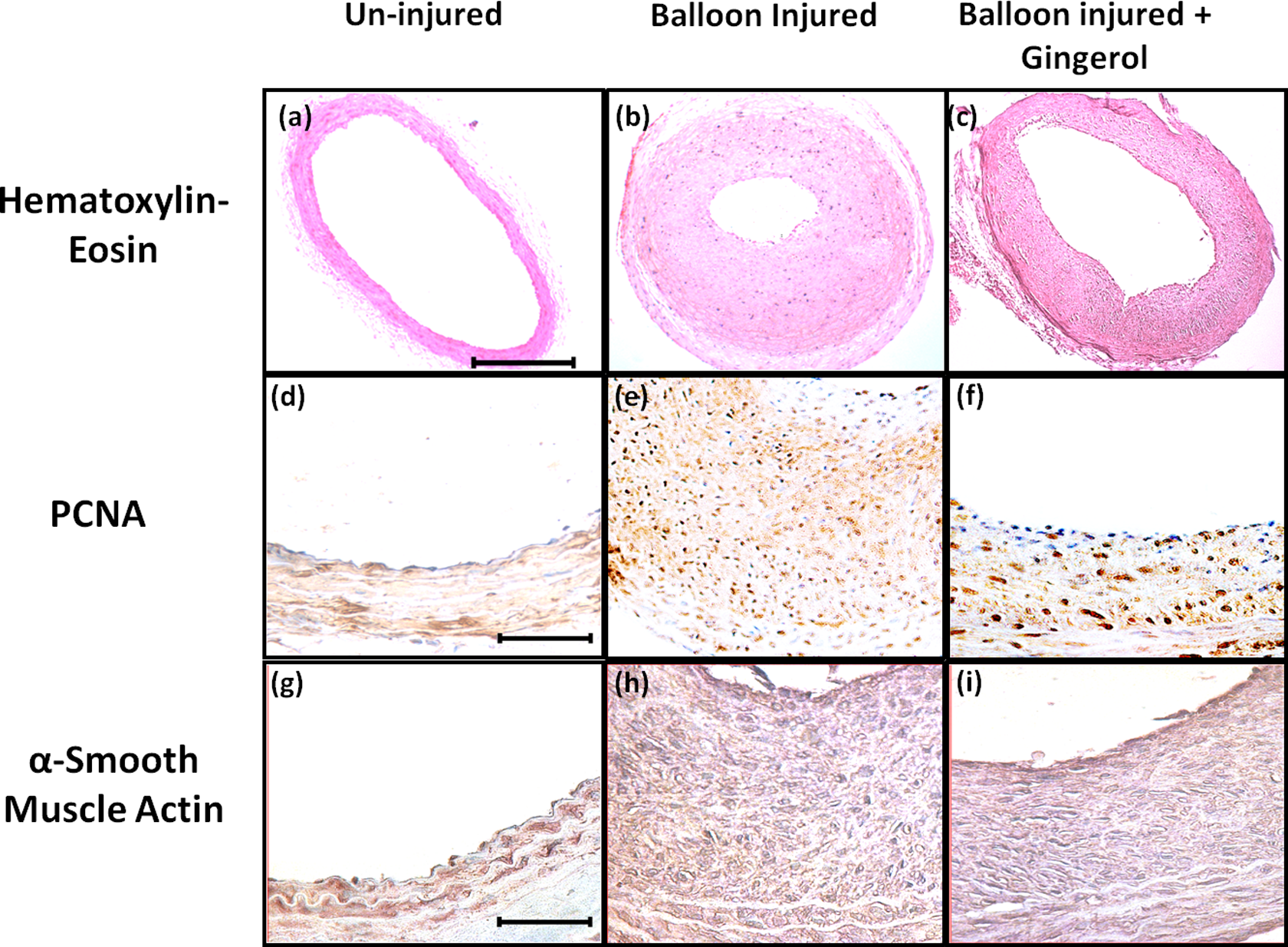
Gingerol prevents balloon injury-induced neointimal cell proliferation and hyperplasia. Carotid arteries pretreated with or without gingerol (200 µmol/L) were analyzed 14 days after balloon injury. Representative images of (A-C) HE staining. D-F, Immunohistochemical staining showing PCNA and (G-I) α-smooth muscle actin. Results are representative arterial sections from 3 independent experiments, and every experiment consisted of 3 rats in each group (n = 9). Magnification of photos is ×100 for HE and ×400 for α smooth muscle actin and PCNA staining. Scale bar in (A) = 100 µm and applies to (B and C). Scale bar in (D) = 20 µm and applies to (E-I). PCNA indicates proliferating cell nuclear antigen; HE, hematoxylin and eosin.
Balloon injury-induced increase in neointimal (I) area (~0.14 mm2) was significantly prevented in gingerol pretreated rat carotid artery (~0.09 mm2; Figure 6A). Medial area (M) was not affected by vascular injury (Figure 6B), which resulted in a significant decrease in I/M ratio in gingerol pretreated arteries (~0.85) as compared to balloon injured control (~1.5; Figure 6C). Cross-sectional narrowing was also prevented in gingerol-treated groups (~48.7%) as compared to balloon injured control (~78.5%; Figure 6D). Balloon injury-induced decrease in lumen area (~0.16 mm2) was significantly prevented in gingerol-treated arteries (~0.30 mm2; Figure 6E).

Histological measurements of carotid artery sections in various experimental groups. Structural changes in each group of animals were assessed 14 days after balloon injury in consecutive sections stained with hematoxylin and eosin using bright field microscopy. Bar diagram representing quantitative analysis of (A) neointimal area (I), (B) medial area (M), (C) I/M area ratio, (D) lumen area (E) percentage of cross-sectional narrowing, (F) percentage of PCNA positive nuclei, and (g) α-smooth muscle actin/neointimal area ratio in uninjured, balloon injured and balloon injured arteries pretreated with gingerol. Results are mean ± SEM of 3 independent experiments and every experiment consists of 3 rats in each group (n = 9). ***P < .001 versus uninjured artery; ##P < .01, ###P < .001 versus balloon injured artery. PCNA indicates proliferating cell nuclear antigen.
To observe whether gingerol has any possible suppressing effect on neointimal smooth muscle proliferation, immunohistochemistry of PCNA and α-SMC actin was done. It was found that the percentage of PCNA-positive nuclei was rare in the uninjured control (~5%; Figure 5D and 6F), but a large percentage of PCNA-positive nuclei could be seen in the neointima of the balloon injured artery (~38.7%; Figure 5E and 6F). A significant reduction in PCNA-positive nuclei was observed in gingerol-treated groups (~32.1%; Figure 5F and 6F). The results also showed prominent α- smooth muscle actin staining in the medial wall among all groups (Figure 5G-I). An increase in the neointimal α-smooth muscle actin area was observed in balloon injured arteries (~0.22; Figure 6G) as compared to uninjured control (Figure 6G). A reduction in α-smooth muscle actin area was observed in gingerol-treated groups (~0.14; Figure 6G). Altogether percentage of PCNA positive nuclei (Figure 6F) and actin/neointima area ratio (Figure 6G) were found to be significantly reduced. These results indicate that protection offered by gingerol is due to reduction in VSMC proliferation.
Gingerol Prevents Balloon Injury-Induced Endothelial Dysfunction
In order to evaluate the effect of gingerol on endothelial dysfunction, carotid arteries from different groups were subjected to PE-mediated contractions and ACh-induced relaxation responses. The contraction of the carotid artery in response to PE (1 to 100 μmol/L) was found to be significantly decreased in balloon injured arteries as compared to normal uninjured control (Figure 7A). Gingerol pretreatment significantly prevented injury-induced decrease in PE-induced contraction in gingerol pretreated balloon injured arteries (Figure 7A). Similarly, Ach- (3 to 3 mmol/L) induced relaxation in PE (1 μmol/L) precontracted rings from balloon injured arteries was significantly reduced as compared to normal uninjured control (Figure 7B). Injury-induced decrease in relaxation response was restored to near control level in gingerol pretreated balloon injured arteries (Figure 7B). Altogether these results indicate that gingerol improves balloon injury-induced endothelial dysfunction.

Gingerol prevents balloon injury-induced endothelial dysfunction. Dose-dependent PE-induced contractions and acetylcholine-mediated relaxations monitored in carotid arteries from different groups were analyzed 14 days after balloon injury to assess the impact of gingerol on endothelial dysfunction. Cumulative concentration-dependent response of (A) PE- (1 nmol/L-100 μmol/L) induced contractions and (b) acetylcholine- (3-300 mmol/L) induced relaxation in all groups. Results are mean ± SEM of 3 independent experiments and every experiment consist of 3 rats in each group (n = 9). *P < .05, **P < .01, ***P < .001 versus uninjured artery. #P < .05, ##P < .01 versus balloon injured artery. PE indicates phenylephrine; SEM, standard error of the mean.
Gingerol Prevents PE-Mediated Contraction and Induces Partially Endothelium-Dependent Vasorelaxant Effect
The direct effect of gingerol on vascular function was also evaluated. Gingerol (3-300 µmol/L) exhibited concentration-dependent relaxation in the PE (1 µmol/L) precontracted endothelium intact rings, similar to that induced by acetylcholine (Figure 8A). To further explore whether the relaxation evoked by gingerol is endothelium dependent, cumulative concentration-dependent responses of gingerol (3-300 µmol/L) were obtained in endothelium denuded rings. Relaxation response of ACh was significantly reduced in endothelium denuded rings (Figure 8A) in comparison to its effect on endothelium intact rings. Relaxation exerted by gingerol was also significantly reduced in endothelium denuded rings in comparison to its effect on endothelium intact rings (Figure 8A). However, effect of gingerol in endothelium denuded rings was still significantly more than that exerted by acetylcholine in endothelium denuded rings. The results showed that relaxation effect of gingerol was partially endothelium dependent. Results obtained also showed that PE- (1-100 µmol/L) induced contraction were reduced in the presence of gingerol at 300 nmol/L concentration albeit not significant (Figure 8B). However, gingerol at higher concentration, that is, 3 µmol/L to 300 µmol/L concentration significantly inhibited PE-induced contraction as compared to vehicle-treated control (Figure 8B).

Effect of gingerol on vascular reactivity. A, Concentration-dependent response of gingerol (3-300 µmol/L) and acetylcholine (3-300 µmol/L) was monitored in endothelium intact and endothelium denuded rat aortic rings precontracted with PE (1 μmol/L). Concentration-dependent response of PE (1-100 µmol/L) was monitored in the presence of gingerol (30-300 µmol/L) in endothelium intact rat aortic rings. Results are mean ± SEM of 6 independent experiments. *P < .001 versus control. ⁁P < .05, ⁁⁁⁁P < .001 acetylcholine endothelium intact vs acetylcholine endothelium denuded. #P < .05, ###P < .001. Gingerol endothelium denuded versus acetylcholine endothelium denuded. @@P < .01, @@@P < .001 gingerol versus gingerol endothelium denuded. PE indicates phenylephrine; SEM, standard error of the mean
Discussion
In our study, we used 10% FBS instead of a single growth factor to induce cell proliferation. Because FBS contains a range of growth factors, including PDGF, FGF, transforming growth factor, serotonin, and thrombin, 29 we used FBS to mimic the multiple factors environment in vivo.
Exposure of smooth muscle cells to growth factors or serum is associated with secretion of inflammatory and growth promoting cytokines 30 such as interleukin-1β and TNF-α, 8 which interact with specific receptors on various cell types and cause direct activation of receptor tyrosine kinase, resulting in activation of Ras/Raf/MEK pathway. 31 Circulating cytokines interact with VSMCs to activate Ca2+, protein kinase C, Rho kinase, and MAPK pathways, which promote cell growth and migration, and VSMC reactivity.
The antiproliferative effect of gingerol was associated with an accumulation of cells in G0/G1 phase of the cell cycle. In an earlier study, 6-gingerol induced intracellular ROS and upregulated p53, p27Kip1, and p21Cip1 levels leading to cell cycle arrest in LoVo cells. 32 To delineate gingerol’s mechanism of action, its effect on cell cycle regulatory proteins was monitored. Cellular proliferation is controlled by multiple holoenzymes comprising a catalytic cyclin-dependent protein kinase (CDK) and a cyclin regulatory subunit. 33 p27Kip1, an inhibitor of cyclin E/CDK2 complex, plays a critical role in cell cycle regulation by binding and inhibiting various CDK/cyclin complex activities. 34 Moreover, p27Kip1 has been reported to play important roles in G2/M checkpoint as tumor suppressor. Overexpression of p27Kip1 results in G1 arrest and inhibition of cell growth. 35 Proliferating cell nuclear antigen is upregulated in proliferating cells and is often used to monitor VSMC proliferation. 36 Therefore to monitor in vitro and in vivo proliferation of the cells, p27Kip1 expression and PCNA index/expression were monitored after 24 hours of FBS stimulation in VSMC and 14 days after balloon injury in rats. 12,37,38 Gingerol pretreatment prevented FBS-induced decrease in p27Kip1 protein expression resulting in cell cycle arrest and inhibition in VSMC proliferation. These results indicate that gingerol modulates p27Kip1 expression, thus preventing the transition from the G1- to the S-phases of the cell cycle. Gingerol also prevented PCNA expression, thus proving its role in the attenuation of DNA duplication and cleavage.
Activation of p38 MAPK mediates the production of a variety of cytokines via transcriptional and posttranscriptional mechanisms. 39 Thus, p38 MAPK appears to play a critical role in the production and transduction of cytokines that are believed to coordinate VSMC proliferation and thus represent a potential target for the modification of the proliferative response. Since FBS-induced p38MAPK phosphorylation was significantly attenuated by gingerol, it can be speculated that inhibitory effect of gingerol on VSMC proliferation can be partly due to its inhibition of p38MAPK. These results are also consistent with previous reports where gingerol has been shown to prevent activation of p38 MAPK in phorbol ester-stimulated mouse skin. 9 To ascertain whether p38MAPK is a key player in mediating suppressive effects of gingerol, experiments were carried out in the presence of both p38MAPK inhibitor and gingerol. Although simultaneous addition of gingerol and SB203580 attenuated cell proliferation and changes in PCNA and p27Kip1 expression, the changes observed were not significantly different from those observed with gingerol and SB203580 alone. From these results, it can be concluded that the effect of gingerol on cell proliferation is indeed due to its inhibition of p38MAPK pathway. Serum also induces AKT and ERK phosphorylation, which are involved in VSMC proliferation. 26 –28 However, since their activation was unaffected in gingerol-treated cells, it can be speculated that majorly p38MAPK plays a role in the suppressive effect of gingerol. To test the in vivo significance of gingerol in regulating VSMC proliferation, experiments were carried out in the rat model of neointimal hyperplasia.
Balloon injury removes the endothelium quickly followed by medial VSMC proliferation. Medial VSMCs then migrate into the intima (4-7 days), after which proliferation of intimal VSMCs results in neointimal hyperplasia. 40 Previous studies from our laboratory as well as others have demonstrated increased PCNA index/expression after 24 hours of FBS stimulation in VSMCs and 14 days after balloon injury in rats. 12,37,38 The authors have therefore evaluated the effect of gingerol on neointimal proliferation at 14 days postballoon injury.
In the present study, balloon injury induced hyperplasic response and morphometric changes were quite similar to earlier reports. 11,41,42 In the balloon-injured rat carotid artery, neointima formation is substantially caused by excessive proliferation of VSMC. 43 In the present study, this was confirmed by immunohistochemical localization of PCNA and α-smooth muscle actin in the carotid artery sections.
Recent studies show that perivascular application of MAPK inhibitors, 17,44 small-interfering RNA, 29 or plasmid DNA 45 using Pluronic gel prevented intimal hyperplasia. By using a similar approach in the present study, an inhibitory effect of gingerol on balloon injury induced PCNA upregulation and p27Kip1 downregulation was observed, thus indicating an antiproliferative effect of gingerol in vivo. One of the well-recognized molecular events during neointimal hyperplasia associated with vascular injury is activation of p38 MAPK pathway. 46 Early increase in p38 MAPK phosphorylation following balloon injury was therefore not surprising. Since gingerol application also prevented p38 MAPK phosphorylation, it is quite possible that this pathway is key to the antiproliferative effect of gingerol in vivo.
Inhibition of p38 MAPK in FRTL-5 thyroid cells inhibited cell proliferation and decreased Cdk2-cyclin E activation by regulating the subcellular localization of Cdk2. 47 Recent studies also demonstrate that inhibition of allograft inflammatory factor 1, a positive regulator of nuclear factor-kappaB/cyclin D1 pathway and cell proliferation, 48 reduces development of neointimal hyperplasia and p38MAPK kinase activity. 49 Since activation of Cdk2/cyclin E may also lead to phosphorylation and degradation of p27, 50 it is quite possible that inhibition of Cdk2/cyclin E activity due to inhibition of p38MAPK may lead to an increase in p27 levels, resulting in inhibition of cell cycle. Also studies performed in the rat suggest that inhibitors of p38 MAPK may act to reduce neointimal hyperplasia by inhibiting interleukin-1β production. 8 Further studies are needed to clarify the precise mechanism by which inhibition of p38 MAPK by gingerol results in the prevention of neointimal hyperplasia after balloon angioplasty.
Gene transfer of dominant negative c-junNH2-terminal kinase prevents neointimal formation in balloon injured rat artery, 26 suggesting importance of this kinase in neointima formation and VSMC proliferation. However interestingly, deficiency of mixed lineage kinase 3, an upstream kinase and positive regulator of JNK, promotes neointimal hyperplasia. 51 Therefore, role of JNK needs careful and detailed evaluation during neointima formation.
Since previous studies demonstrate that to prevent neointimal hyperplasia, a combination of re-endothelialization and reduction in VSMC proliferation are required, and the present study clearly demonstrates that beneficial effects of gingerol in vivo are at least due to the inhibition of VSMC proliferation. Such an effect will favor prevention of neointima formation. However, different mechanisms may account for differential regulatory effect on endothelial, as well as VSMC which may involve p38 MAPK.
A recovery in the vessel function due to an inhibition in the intimal hyperplasia was evident since PE-induced contractions and Ach-induced relaxations of the vessel wall were significantly improved in the gingerol-treated animals. However, it is quite possible that gingerol has a direct beneficial effect on the vascular tone. To ascertain any direct vasodilator effect on the vessel wall, experiments were carried in endothelium intact or denuded rat thoracic aortic rings mounted in isolated organ bath. Interestingly, gingerol exerted vasodilatory effect, which was slightly reduced in endothelium denuded preparations indicating that the vasodilator effect of gingerol is partially mediated through endothelium derived factor like nitric oxide. Previously, it has been shown that ginger extract causes vasorelaxation in rabbit aortic preparations by inhibiting the voltage-dependent Ca++ channels. 52 It has also been shown that ginger extract induced relaxation in PE precontracted rings was reduced in the presence of L-NG-Nitroarginine methyl ester (L-NAME). 53 The results regarding inhibition in PE-induced contraction was similar to previous published reports where ginger extract has been shown to inhibit both the receptor-operated channels and the intracellular Ca++ channels, 52 which are involved in PE-induced contractions. 54 6-Gingerol, which is an active constituent of ginger extract, may be responsible for such an effect. The present study thus identifies gingerol as one of the constituents which may mediate vasodilator effect of ginger extract. An antithrombotic effect of gingerol can also facilitate its antiatherosclerotic effect. Gingerol can exert an anti-platelet function by inhibiting thromboxane formation. 55 In a different study undertaken in the laboratory, gingerol (200 µmol/L, by oral route) did show ~48% (data not shown) protection in a mice model of collagen epinephrine-induced pulmonary thromboembolism.
Synthetic or secretory phenotype is observed in VSMCs present in atherosclerotic lesions 56 and in those stimulated by serum in cell culture. 57 The phenotype switch of SMC from contractile to synthetic phase also impairs vessel contraction. In our experiments, gingerol pretreatment significantly prevented balloon injury-induced reduction in PE-mediated vessel contraction. Therefore, it will be quite logical to conclude that an inhibition in proliferation by gingerol is also accompanied by an inhibition in the phenotype of the VSMC. However whether such an inhibition will be solely due to the inhibition of p38MAPK pathway needs experimental validation.
Conclusion
Gingerol attenuates FBS and balloon injury induced VSMC proliferation and neointimal hyperplasia by inhibiting p38MAPK. Gingerol can independently exert a partially endothelium-dependent vasodilator effect on the thoracic aorta. The antiproliferative potential of gingerol can be exploited for preventing the progression of vascular proliferative diseases.
Footnotes
Authors’ Note
Jain contributed to design, contributed to acquisition and analysis; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Singh contributed to design; contributed to acquisition and analysis; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Singh contributed to design; contributed to acquisition; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Maurya contributed to acquisition; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Barthwal contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Acknowledgments
We gratefully acknowledge to Council of Scientific and Industrial Research (CSIR), New Delhi, India, for the award of research fellowships to A. S., V. S., and P. M.; Indian Council of Medical Research, New Delhi, India to M. J. We are grateful to Mr M.P.S. Negi from biometry and statistics division, CSIR- Central Drug Research Institute for helping in the statistical analysis of the data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from BSC0102, BSC0103, and CSIR-Central Drug Research Institute is acknowledged.
