Abstract
Systemic vascular involvement in children with cerebral arteriopathies is increasingly recognized and often highly morbid. Fibromuscular dysplasia (FMD) represents a cerebral arteriopathy with systemic involvement, commonly affecting the renal and carotid arteries. In adults, FMD diagnosis and classification typically relies on angiographic features, like the ‘string-of-beads’ appearance, following exclusion of other diseases. Pediatric FMD (pFMD) is considered equivalent to adult FMD although robust evidence for similarities is lacking. We conducted a comprehensive literature review on pFMD and revealed inherent differences between pediatric and adult-onset FMD across various domains including epidemiology, natural history, histopathophysiology, clinical, and radiological features. Although focal arterial lesions are often described in children with FMD, the radiological appearance of ‘string-of-beads’ is highly nonspecific in children. Furthermore, children predominantly exhibit intimal-type fibroplasia, common in other childhood monogenic arteriopathies. Our findings lend support to the notion that pFMD broadly reflects an undefined heterogenous group of monogenic systemic medium-or-large vessel steno-occlusive arteriopathies rather than a single entity. Recognizing the challenges in categorizing complex morphologies of cerebral arteriopathy using current classifications, we propose a novel term for describing children with cerebral and systemic vascular involvement: ‘cerebral and systemic arteriopathy of childhood’ (CSA-c). This term aims to streamline patient categorization and, when coupled with advanced vascular imaging and high-throughput genomics, will enhance our comprehension of etiology, and accelerate mechanism-targeted therapeutic developments. Lastly, in light of the high morbidity in children with cerebral and systemic arteriopathies, we suggest that investigating for systemic vascular involvement is important in children with cerebral arteriopathies.
Introduction
Stroke is a leading cause of neurological morbidity and mortality among children. Cerebral arteriopathies, defined as ‘any in-situ pathological abnormality of the arterial circulation not attributable to an exogenous thrombus and not considered a normal developmental variant’,1,2 constitute the primary cause of acute arterial ischemic stroke (AIS) in healthy children, responsible for AIS in 29–53% of affected cases. In addition, arteriopathies serve as the most significant predictor of stroke recurrence and outcome.3,4 The current classification of arteriopathies developed by the International Pediatric Stroke Study (IPSS) group, the CASCADE (Childhood AIS Standardized Classification and Diagnostic Evaluation), mainly relies on clinical and radiological features such as location of the involved artery, laterality (unilateral vs bilateral), disease course (stable vs progressive), presence of collateral vessels and multisystemic involvement rather than pathophysiological mechanisms.1,2,5,6 However, steno-occlusive cerebral arteriopathies which involve the proximal internal carotid artery (ICA) or posterior circulation, rather than the typical intracranial ‘T-junction’ (distal ICA, proximal middle cerebral artery, and anterior cerebral artery) as well as those that involve the systemic vasculature bed, lack clear and consistent terminology in the current classification. In a prospective study of 355 children with AIS (The Vascular Effects of Infection in Pediatric Stroke study, VIPS), 2 30% of the children remained unclassified, thus demonstrating the relatively low reliability of the current diagnostic approach. The lack of unifying terminology for children with multisystemic arteriopathies makes it difficult to accurately group patients and have a consistent approach to classification. This is one of the obstacles to determining the underlying pathobiological mechanisms of these disorders and therefore poses a barrier to the development of mechanism-targeted therapies.
In adults, fibromuscular dysplasia (FMD) is a well-described disease consisting of a heterogeneous group of nonatherosclerotic, noninflammatory arterial lesions which often involves the renal arteries and the cerebrovasculature. In children, however, data on the extent of the vascular bed involvement and the natural history and outcome of FMD are predominantly derived from case reports and small cohorts and confounded by widely varied inclusion criteria.
Here, we review the current literature on pediatric fibromuscular dysplasia (pFMD) and the growing list of monogenic steno-occlusive cerebral and multisystemic vasculopathies. We used PubMed and searched the terms: ‘pediatric/childhood fibromuscular dysplasia’, ‘pediatric genetic/monogenic arteriopathies’, and ‘childhood multi-system vasculopathy’. Based upon current evidence from this literature review (1971–2021), as well as our clinical experience, we propose that pFMD is not the same as adult FMD, and that it shares common clinical, radiological, and histopathological features with other childhood arteriopathies. We also highlight the need for systemic investigation to assess vascular bed involvement in patients with chronic cerebrovascular arteriopathies.
Fibromuscular dysplasia (FMD)
Vascular abnormalities in FMD may include stenosis, aneurysms, tortuosity, and dissection.7,8 In adults, the disease predominantly affects women of middle age, although a wider age distribution has also been described. The most common sites of involvement are the renal arteries and the internal carotid arteries, but virtually any arterial bed can be involved. Depending on the topological distribution of the lesions and the arterial bed, FMD can lead to a range of symptoms, including renovascular hypertension, arterial aneurysms and dissections, ischemic or hemorrhagic stroke, subarachnoid hemorrhage, or mesenteric ischemia.9,10 Notably, many patients are asymptomatic and are found to have FMD incidentally. The observed natural history of FMD in those patients is reported as being relatively benign. 10
Classification of FMD
FMD was historically diagnosed by the histopathological findings fibromuscular alteration of the vessel wall and classified according to the predominant layer involved.11–13 It may involve the intima, media, or adventitia of the affected artery, resulting in segmental arterial stenosis in multiple arterial beds.9,10 Medial fibroplasia/dysplasia, which is associated with the classic ‘string-of-beads’ radiographic appearance, accounts for ~85% of cases, whereas intimal fibroplasia, which is frequently observed in patients with focal stenosis, is much less common, appearing in fewer than 5% of the patients. In clinical practice, this nonspecific diagnosis is commonly a diagnosis of exclusion in patients with at least one stenotic lesion,8,11,14 but multiple areas of focal disease can be involved and result in vessel stenosis, aneurysms, dissection, and tortuosity. Additional less common histopathological subtypes of adult FMD include perimedial fibroplasia, medial hyperplasia, and adventitial hyperplasia.11–13,15,16 These categories are not mutually exclusive, since involvement of more than one layer in the same diseased artery is not uncommon.
In essence, FMD represents a widely varied spectrum of arterial diseases. Disparity and complexity in the classifications of FMD have recently led to consensus statements based on the phenotypic expression of FMD. Owing to the limited availability of histopathological specimens, an international consensus on the diagnosis and management of FMD supported the diagnosis of FMD solely upon radiographic features of a ‘string-of-beads’ pattern (multifocal type) or focal stenosis (focal type). 17 This diagnosis can be provided after excluding other etiologies, among them atherosclerosis, inflammation, and known genetic disorders, although they may coexist with FMD. 11 The lack of histological proof in most cases as well as the variable and overlapping radiological and histological findings raise the question of whether they are all part of the same disease spectrum or whether focal and multifocal FMD represent two different vascular diseases. 18 An understanding of the unique clinical and imaging findings in patients with FMD is essential to distinguish FMD from other arterial diseases. Notably, there is currently no disease biomarker, and even pathological confirmation may only reflect the downstream disease outcome.
Pathophysiology of FMD
The pathophysiology of FMD remains largely unknown. Fibroblastic transformation of the smooth muscle cells (SMCs), which causes focal disruption, increased deposition of synthesized collagen in the media and degradation of the elastic laminae, results in alternating thickening and thinning of the tunica media or neointimal lesions of cells, ultimately leading to destruction of the arterial wall. 8 Environmental factors such as smoking, mechanical and hormonal influences, and vasculotoxic medications, along with emerging genetic susceptibilities, have been linked to FMD. A complex genetic framework involving common genetic modifiers and genetic-environmental interactions has been suggested. 19 However, studies on the genetic underpinnings of FMD are constrained due to the high prevalence of asymptomatic FMD patients. Only around 5% of adult cases are familial, and a clear genetic association is currently lacking despite extensive research on both familial cases as well as relatively large patient cohorts. 20 A genetic intronic common polymorphism in the phosphatase and actin regulator 1 gene (PHACTR1) associated with FMD was recently identified in a genome-wide association study, which suggested that PHACTR1 may influence the transcription activity of the nearby endothelin-1 gene. 20 Additionally, other monogenic disorders including Grange syndrome (OMIM 602531), caused by a biallelic mutation in the YY1AP1 gene,21,22 were associated with a disorder that encompasses features of FMD. Nevertheless, the results of an exome sequencing analysis from 282 individuals with FMD were conflicting and showed no increased burden of YY1AP1 variants in the FMD-affected subjects. Furthermore, though FMD shares some phenotypic features with monogenic connective tissue diseases, such as Marfan and Ehlers–Danlos syndromes, the yield of genetic testing for known vascular connective tissue disorders was low in patients with FMD.23,24 A potential influence of environmental modifiers was suggested to serve as susceptibility factors (e.g., female hormones, lifetime mechanical stress, and tobacco use).8,25,26 Data from the Defining the Basis of FMD (DEFINE) study (ClinicalTrials.gov Identifier: NCT01967511), a systems biology study in patients with FMD, recently showed a plasma proteogenomic and lipid signature that includes potential causative disease drivers, among them triglycerides and fatty acids, in adults with FMD. That study, however, did not include pediatric patients.
Pediatric FMD
Data on the extent of vascular bed involvement, natural history and outcome of children with pFMD are confounded by widely varied inclusion criteria whereby the diagnosis of pFMD relies upon clinical association with systemic vascular involvement or classic adult radiographic ‘string-of-beads’ appearance rather than on histopathological diagnosis.27,28 Although focal stenosis is considered the dominant form of FMD in children, data on the extent of the cerebral phenotypes of pFMD are scarce and predominantly derived from case reports and small cohorts. In view of the multisystemic involvement and rarity of these disorders, the terminological classification of these vasculopathies is often inconsistent (e.g., multisystemic vasculopathy, complex vasculopathy, and pFMD), thereby making it difficult to accurately classify and group them.
There are numerous reported differences between pediatric and adult-type FMD in the distribution of the affected vessels, the radiographic appearance of vasculopathy, as well as the histopathological findings (Table 1). The largest study to date investigated various cerebrovascular aspects of pFMD in 81 patients with AIS from the Canadian Pediatric Ischemic Stroke Registry together with cases from published studies. 29 Importantly, that study applied a priori systematic criteria for the terms FMD, fibromuscular hyperplasia, renal artery stenosis (RAS), and hypertension. Systemic vessel involvement was reported in < 1% of children with childhood arterial ischemic stroke, with an incidence of 1:3,000,000 children per year. Of the 81 children included in that study, only 27 had pathologically confirmed FMD, and an intimal fibroplasia subtype predominated in 89% of them. Of note, none had the typical adult-type medial fibroplasia. The other 54 patients were classified as having FMD based on the co-occurrence of renal arteriopathy and stroke. In contrast to adults with FMD, the characteristic radiographic appearance of ‘string-of-beads’ was seen in only six of the 72 children (i.e., > 85% in adults vs < 10% in children) who had undergone cerebrovascular imaging. In addition, of the 27 pathologically confirmed cases, 33% presented in the first year of life and 63% were associated with poor neurological outcome. The patients who lacked a pathological diagnosis of FMD were older and had lower rates of hypertension, higher rates of a ‘string-of-beads’ angiographic appearance and a better outcome. These differences may reflect a selection bias towards performing histopathology in severely ill patients or another arteriopathy subtype but may also represent a changing arteriopathy pattern across the developmental spectrum. Importantly, the ‘string-of-beads’ pattern disappeared on follow-up magnetic resonance angiography (MRA) in a group of school-age children. These clinical characteristics may be suggestive of focal cerebral arteriopathy-inflammatory type (FCA-i), a disease that does not involve systemic arteries – unlike true FMD, is monophasic and usually nonprogressive with favorable prognosis. In addition, this may also explain the ‘purely intracranial FMD’ type that is typically described in children.30–33 In conclusion, these data suggest that the ‘string-of beads’ radiographic appearance described in adults is less commonly reported in children with FMD and highlights its nonspecificity as a radiological biomarker for pFMD.
Adult versus pediatric clinical and radiological characteristics of cervico-cerebral fibromuscular dysplasia (FMD).
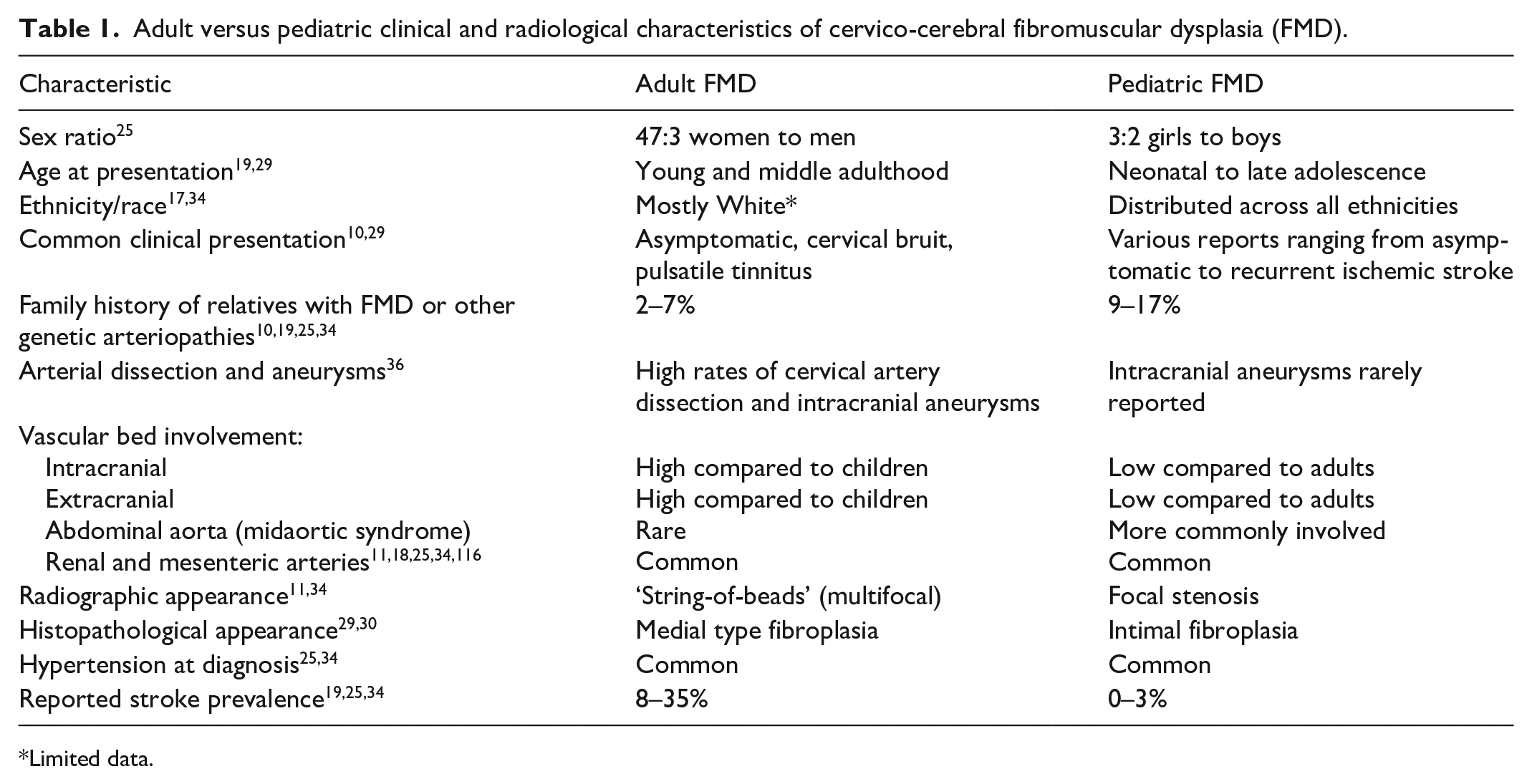
Limited data.
Since the publication in 2013 of the seminal paper by Kirton et al. 29 few data on pFMD have been added, particularly with regard to the cerebrovascular phenotype.25,34,35 Green et al.25,38 investigated 33 pediatric patients enrolled in the United States Registry for FMD. Most of them were diagnosed based upon clinical suspicion in conjunction with assessment of aortic, renal, intra- and extracranial, mesenteric, and coronary vasculature involvement as demonstrated on invasive or noninvasive angiographic imaging studies. The pediatric patients comprised only 3% of the total number of patients in the US registry. Importantly, the type of FMD could not be determined or was unknown in over one-half of the children. Almost all the children (97%) had renal vasculature involvement, 55% had headache, and the extracranial carotid and vertebral arteries were involved in 23% and 25%, respectively, whereas intracranial vasculopathy was reported in only 7%. Only one patient (3%) had sustained a stroke, and none had evidence of dissection or subarachnoid hemorrhage. This is in contrast to cerebrovascular involvement of adults with FMD, in whom FMD is associated with a higher prevalence of intracranial aneurysms, and stroke is most often associated with cervical artery dissection. 36 Louis et al. reported a group of children with nonsyndromic noninflammatory renovascular hypertension who were referred with the diagnosis of FMD. 34 The inclusion criteria in that study included idiopathic or very severe cases of hypertension, or hypertension combined with abdominal bruit. However, none of the children included in the study presented with stroke, though stroke had been reported in higher rates in adult patients. 37 These data, albeit not focused upon the cerebral phenotype of FMD, suggest that the neurological manifestations of pFMD are more subtle and milder than those of adults, and that the extent of the cerebrovascular disease among children seems to be lower compared to that of adults in whom cerebrovascular involvement is reported in 25–80% of the patients. 19 Importantly, as the study by Kirton et al. included only children with evidence of stroke, it most likely reflected the more severe end of the spectrum of pFMD or a different disease entity with systemic vascular involvement. In summary, the inclusion of patients with diverse systemic and cerebrovascular radiologic and/or histopathological profiles across key papers on pFMD, each employing different inclusion criteria, may introduce selection bias and complicate interpretation of results, hindering accurate assessment of the natural history and the extent of involvement of each vascular bed.
Pediatric FMD and other childhood systemic arteriopathies
Overlapping phenotypes and pathological similarities
Vascular abnormalities reported in pFMD include arterial stenosis, tortuosity, aneurysms, and dissections in one or more arterial beds. Many monogenic cerebral and systemic arteriopathies in childhood share these vascular abnormalities as well as other clinical, radiological, and histopathological similarities. For example, intimal hyperplasia or thickening, which is commonly described in cases of pFMD, is also described in Alagille syndrome, neurofibromatosis type 1 (NF1), moyamoya, and Williams syndrome,29,30,38–43 all of which are associated with vaso-occlusive systemic arteriopathy. A loss of structural integrity of the arterial wall including primary or secondary functional transformation of SMCs leading to medial attenuation, thinning or duplication of the internal elastic lamina to create a neointima, degradation of the elastic laminae, and functional changes in collagen deposition and inflammation can be seen in many of the above-cited conditions. The diagnosis of many monogenic syndromes with systemic arteriopathies, such as Grange syndrome and ACTA2-related multisystemic smooth muscle dysfunction syndrome, relies upon unique phenotypic traits (Table 2). However, other patients with systemic arteriopathies often lack definitive features, making the clinical and radiological phenotype indistinguishable from pFMD. Importantly, moyamoya has been described among children with suspected FMD,44,45 and up to 15% of patients with moyamoya are reported to have extracranial/systemic involvement including hypertension.46–51 It is therefore important that children with cerebral occlusive arteriopathies including moyamoya disease are investigated for systemic vascular bed involvement. 52
Genetic causes of cerebral and systemic arteriopathies of childhood (CSA-c).

See online supplementary Table 1 for a complete version of the table (dichotomized based on radiological phenotype).
Accumulating evidence suggests that patients with RNF213 mutations may develop moyamoya disease with systemic involvement, often appearing at a young age and leading to severe outcomes.47,53 This phenotype closely resembles pFMD, raising the possibility that these children were initially diagnosed with pFMD during early life stages and subsequently reported as such in the literature. Notably, there are also similarities between histopathologic findings of moyamoya and pFMD.30,33,54 Intimal thickening, medial attenuation and internal elastic lamina thinning or duplication, which are the histopathological appearance of pFMD and other systemic arteriopathies, likely reflect the end result of heterogenous mechanisms of injury that disrupt vascular homeostasis (i.e., the balance between vessel wall maintenance and repair). The proliferative disease of the intima can have an acquired, genetic, or combined pathological basis. Inherent abnormalities in key mechanisms involved in vascular homeostasis include abnormal circulating endothelial progenitor cell recruitment, regulation of endothelial and vascular SMC (VSMC) proliferation, migration, adhesion, and cell survival, as well as endothelial cell–SMC communication and production of vascular matrix proteins.55–62 The plasticity of the vascular smooth muscle cells (VSMCs) (phenotypic switching) is required for growth and wound healing, but also contributes to vessel wall disease pathology.63,64 These key mechanisms combine to create a heterogenous clinical phenotype. Not uncommonly, causative genes are involved in more than one pathway (e.g., RNF213) resulting in mechanistic overlap. Environmental factors, such as trauma and oxidative stress (e.g., secondary to infection [even trivial]), inflammation, increased shear stress, or even prematurity, likely serve as a second hit, triggering these complex and dynamic homeostatic responses that contribute to vascular pathology. These environmental factors also differ over the lifespan and to those of adult FMD.65–67 The combination of genetic abnormalities that affect multiple pathways including vessel wall structure and environmental factors result in an abnormal response to injury, ultimately leading to the range of vascular abnormalities seen in FMD.
Cerebral and systemic arteriopathies of childhood (CSA-c)
We propose the term CSA-c to describe pediatric patients with systemic vascular involvement which includes both cerebral as well as extracranial vessels (e.g., cervical, renal, and abdominal) (Figure 1). Canonical application of this term would not include FCA-i (also known as transient cerebral arteriopathy [TCA]), or FCA-dissection (FCA-d).68,69 A more refined definition will improve the phenotypic characterization of this cohort of interest and facilitate improved understanding of the genetic basis and pathophysiology of this intriguing group of vascular diseases. Table 3 summarizes the main groups of pediatric cerebral steno-occlusive vasculopathies with their main features and the differential diagnosis of CSA-c, including acquired systemic inflammatory and noninflammatory causes as well as genetic disorders (Table 2, online supplementary Table 1). However, after excluding inflammatory as well as the known genetic entities, most of these patients have unexplained vasculopathy involving cerebral and extracranial vessels. Illustrative images showing various types of cerebral vasculopathies with systemic involvement are shown in Figures 2, 3, and 4.
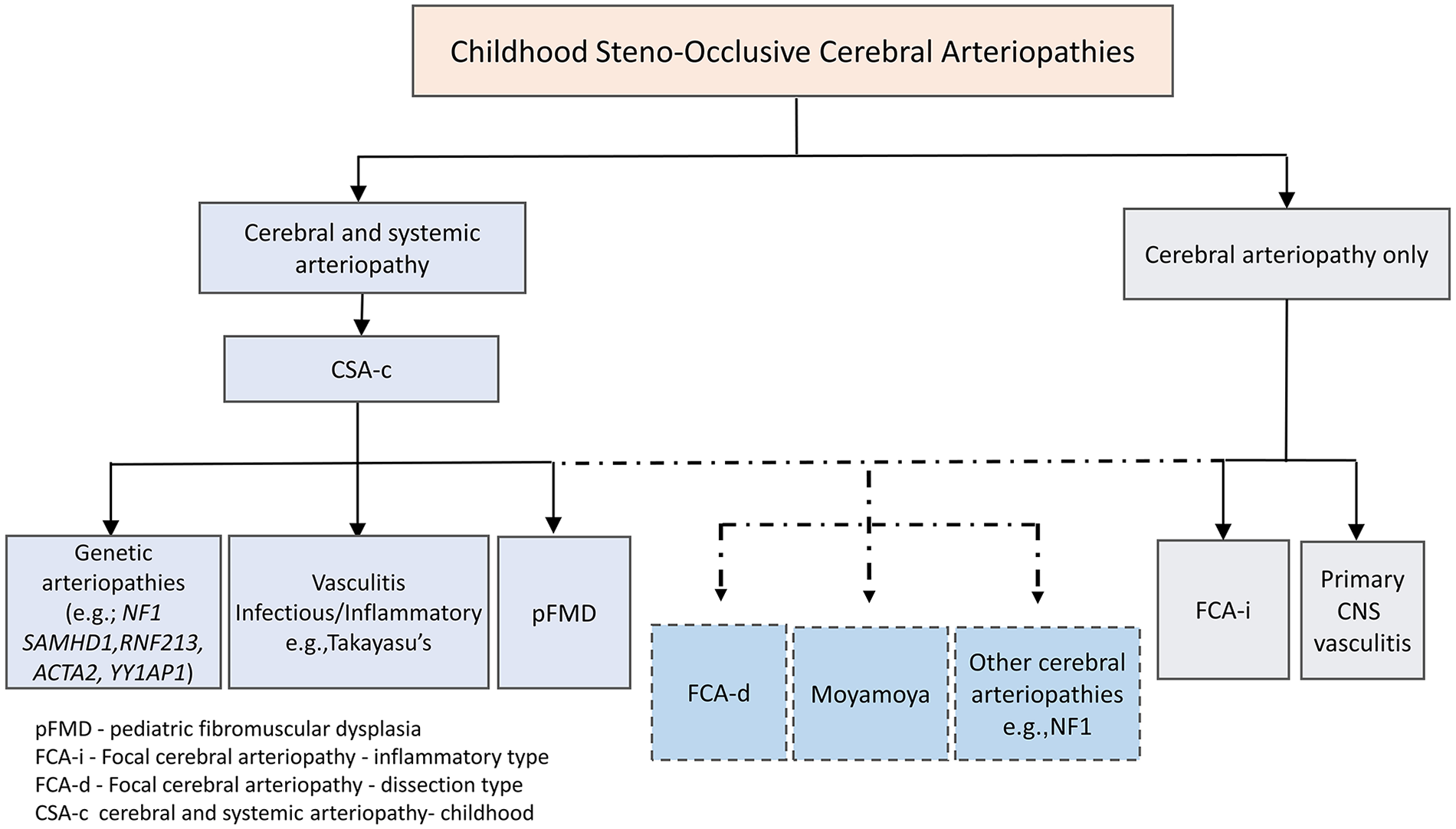
Proposed classification scheme concept of cerebral arteriopathies.
Clinical and radiological presentation of pediatric steno-occlusive vasculopathies with cerebrovascular involvement.
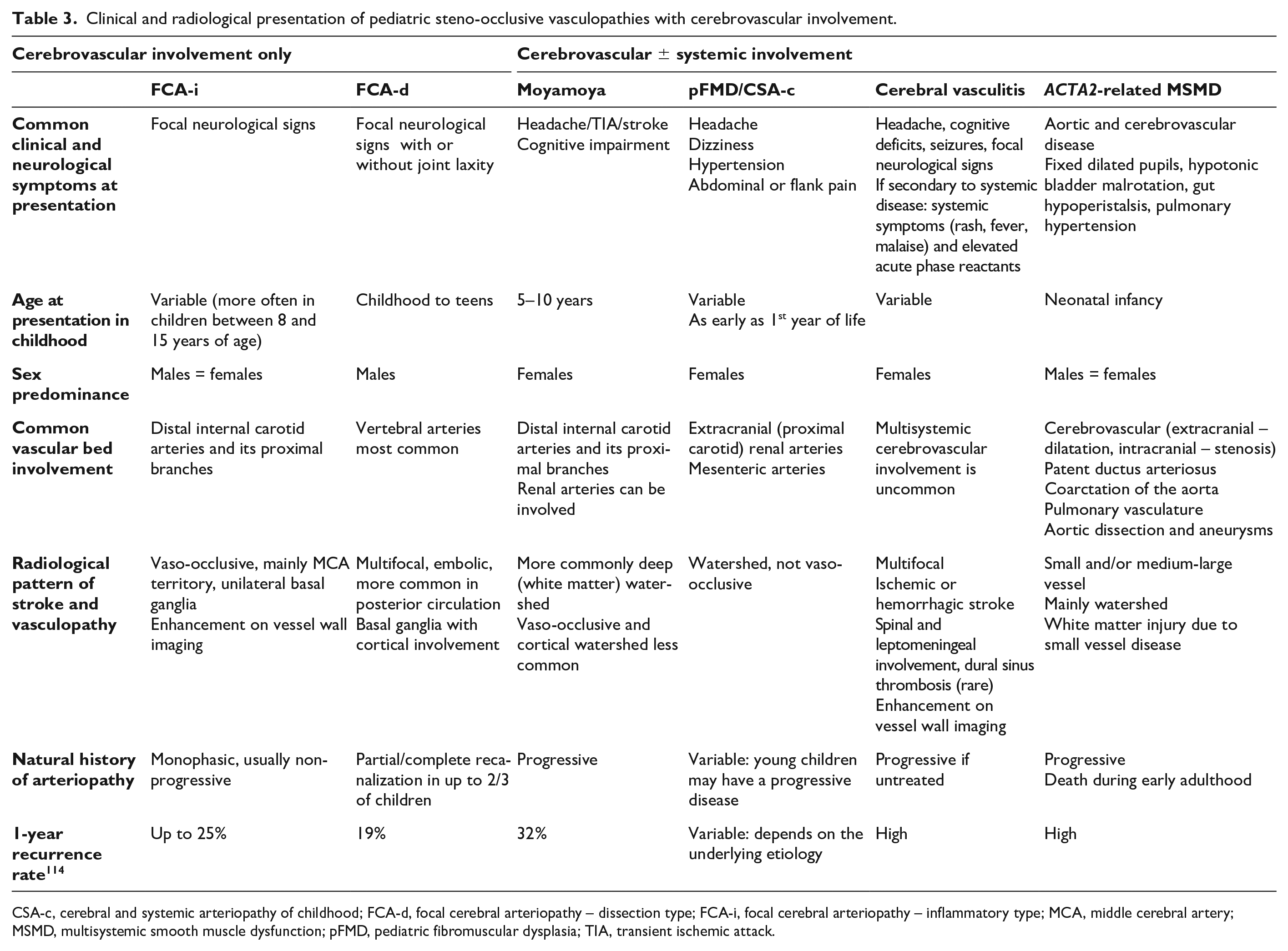
CSA-c, cerebral and systemic arteriopathy of childhood; FCA-d, focal cerebral arteriopathy – dissection type; FCA-i, focal cerebral arteriopathy – inflammatory type; MCA, middle cerebral artery; MSMD, multisystemic smooth muscle dysfunction; pFMD, pediatric fibromuscular dysplasia; TIA, transient ischemic attack.
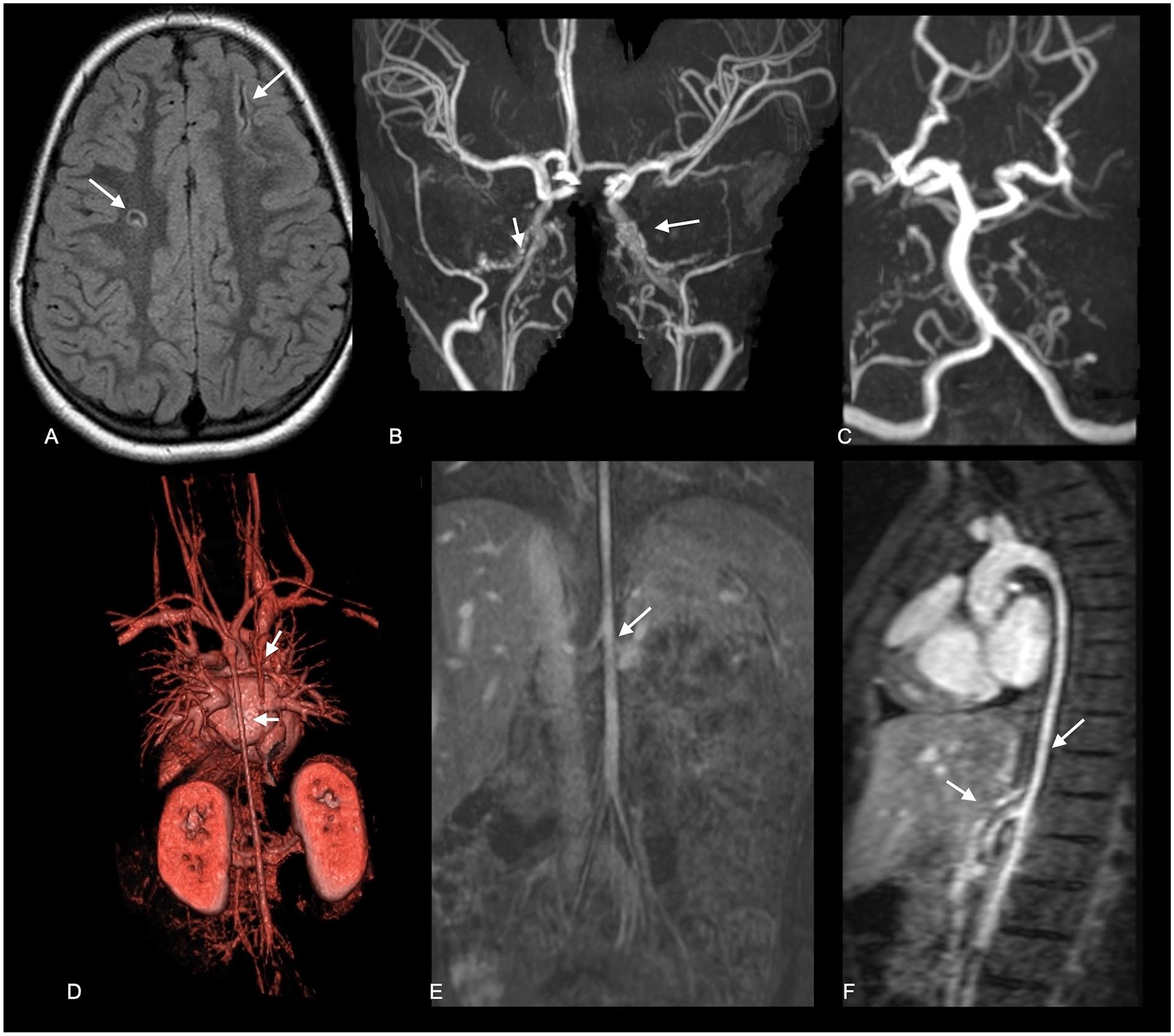
An illustrative case of cerebral vasculopathy with systemic involvement and stroke. Case 1 is a 13-year-old girl with an antenatal diagnosis of coarctation of the aorta and peripheral pulmonary artery branch stenosis. Clinical presentation was with headache and episodes of dizziness at age 5 years.
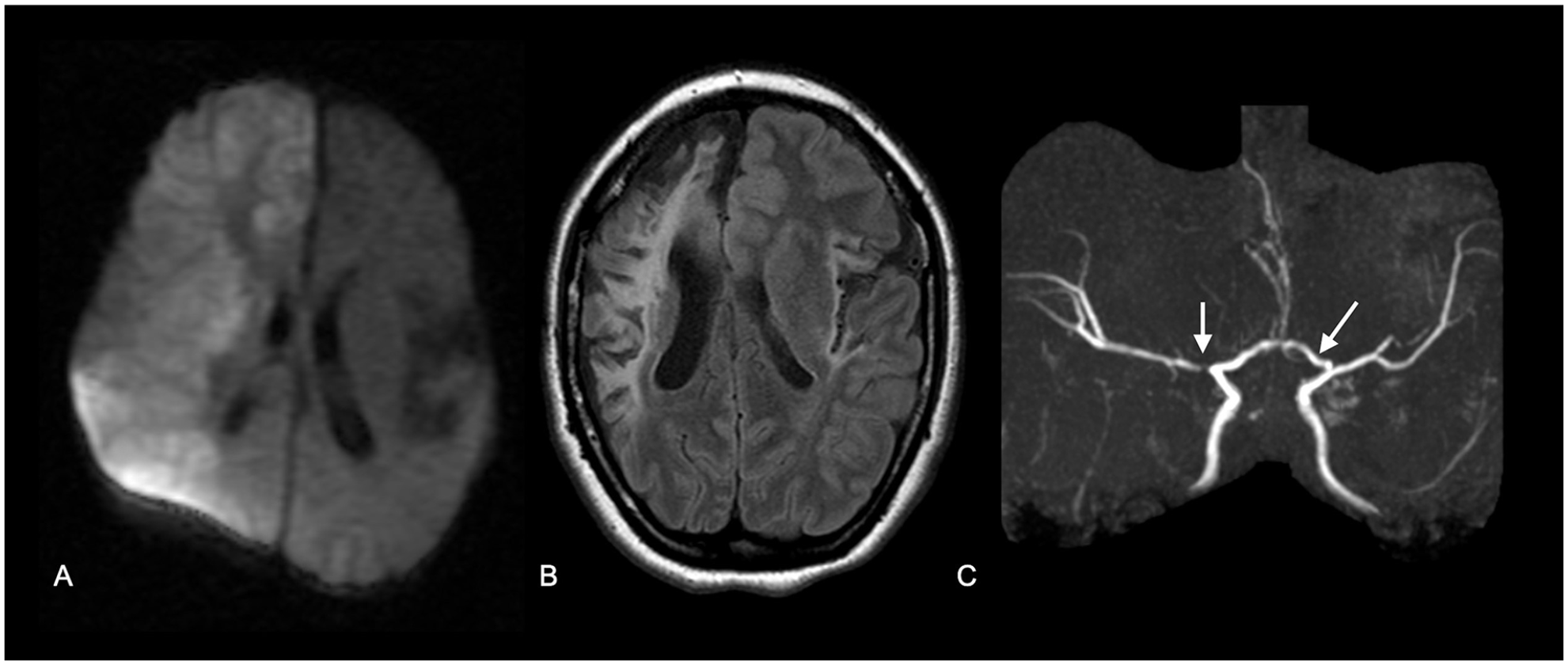
Illustrative images showing cerebral vasculopathy with systemic involvement and stroke. Case 2 is a 15-year-old girl who presented at 2 months of age with acute right MCA territory AIS and a remote left MCA stroke
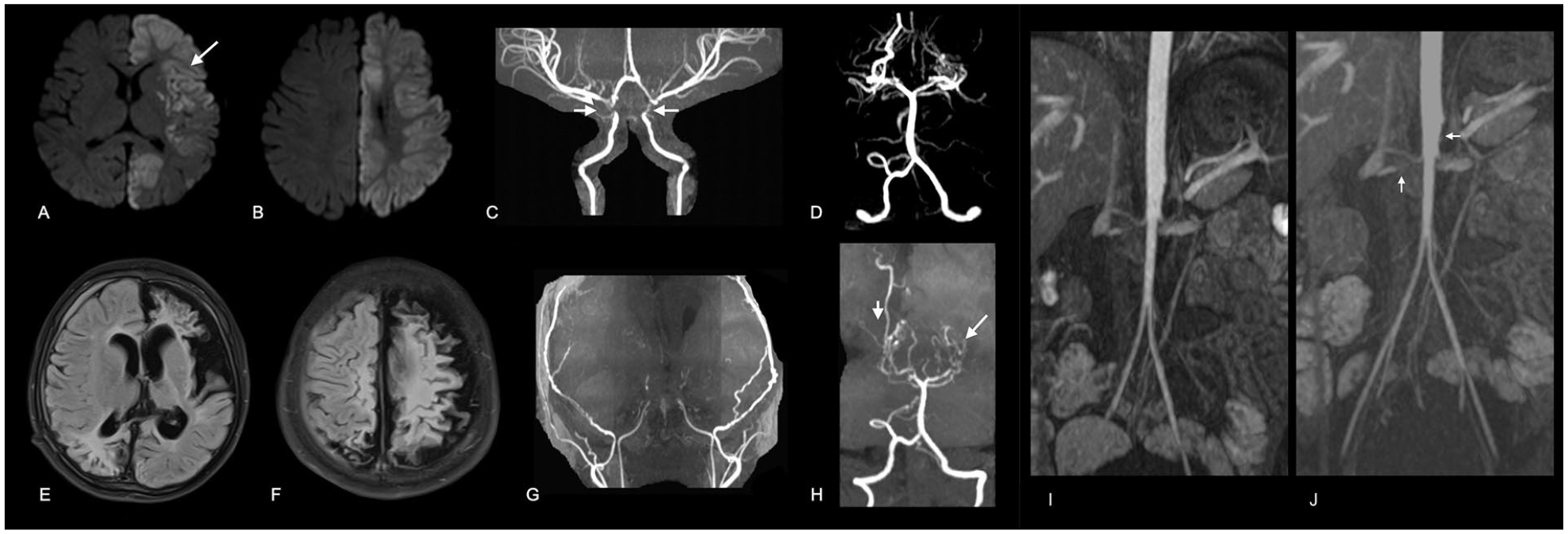
Illustrative images showing cerebral vasculopathy with systemic involvement and stroke. Case 3 is a boy aged 2 years and 8 months with global developmental delay, who presented at the age of 5 months with acute-onset weakness and was diagnosed with bilateral multifocal ischemic infarcts
Diagnosis of CSA-c
Evaluation of children with cerebrovascular disease should involve detailed clinical and radiographic examination of other vascular beds. Clinical clues for specific genetic syndromes include characteristic dysmorphic features in Down syndrome, Williams or Alagille syndromes, evidence of brachydactyly or syndactyly in Grange syndrome, joint hyperlaxity for connective tissue disorders, and other organ involvement (online supplementary Table 1). A thorough examination of the skin for evidence of café au lait in NF1 or other RASopathies, segmental hemangiomas in PHACES, chilblains in a SAMHD1-related disorder, or cutis marmorata. Basic vascular bed involvement assessment includes measuring blood pressure, including the use of ambulatory 24-hour blood pressure measurements if needed, echocardiography, and Doppler ultrasound of the kidneys.
The current expert consensus on the diagnosis and management of FMD in adults recommends at least one computed tomography angiography (CTA) or MRA assessment of all vessels from the brain to the pelvis to identify other arterial bed involvement. 17 Although there are no specific guidelines for children, a diagnostic approach similar to that for adults is often employed. MRA or CTA can detect main RAS in children, whereas branch RAS, which are more common in children, are better diagnosed by renal digital subtraction angiography (DSA). 25 Often, cerebral DSA is performed in children with moyamoya as well as in those with suspected vasculitis or other steno-occlusive arteriopathies. Concurrent systemic angiography to evaluate renal, mesenteric, and aorto-iliac arteries should be considered in these instances. However, practical hurdles including coordination between two sets of operators for the cerebral and renal studies as well as calculation of the maximal amount of injected contrast are recognized. 70 Notably, renovascular hypertension may evolve years after presentation of cerebral vasculopathy, similar to patients with moyamoya; therefore these patients should be followed regularly.50,71 Assessment of the pulmonary and coronary arteries should be considered depending on the specific clinical and genetic phenotype (e.g., ACTA2, vasculitis or RNF213-related disorders).47,72 Laboratory tests including hemoglobin electrophoresis and an inflammatory work-up should be performed according to the clinical and radiological phenotype.
Genetic investigations for children with complex vascular diseases, including CSA-c, should be pursued in all patients following thorough clinical and radiological phenotyping. Genetic testing can include a single-gene panel for patients with clear clinical and radiological features (such as NF1), targeted gene panels, or whole-exome sequencing for patients with less specific phenotypes. The latter offers the advantage of testing all protein-coding genes, whereas panel diagnostics involve sequencing a limited selection of well-characterized genes, reducing the occurrence of unclear and difficult-to-interpret variants. Although data on the yield of each approach to genetic analysis is lacking, 73 a trio model of whole exome sequencing (WES) or whole genome sequencing (WGS) is preferred, if feasible, to enable the analysis of variants of unknown significance and a streamlined segregation model. Evaluation should consider the patient’s accurate clinical information, parental details, and a comprehensive analytic and clinical variant assessment. Re-evaluation analysis of all WES results should be conducted for negative cases. In addition, in case skin lesion are apparent, and histopathologic and genomic analysis from skin tissue may aid in identifying postzygotic mosaic conditions.
Therapeutic considerations of CSA-c
Children with CSA-c require comprehensive treatment that is best provided by a multidisciplinary team. Despite the high morbidity and mortality associated with systemic large-vessel disease, evidence-based guidelines for management are lacking.
Children with AIS are typically treated initially with antithrombotic agents to prevent recurrent stroke, either an antiplatelet (aspirin dosed at 3–5 mg·kg−1·d) or an anticoagulant (low-molecular weight heparin or unfractionated heparin), depending on stroke etiology and patient-specific factors. 74 Antiplatelet therapy for secondary prevention of ischemic stroke is a mainstay of stroke prevention in children with arteriopathies and are often prescribed to reduce the likelihood of thrombosis due to turbulent flow. 10 There is a scarcity of high-quality clinical trials assessing the superiority between antiplatelet or anticoagulant medication in children.75–77 The optimal duration of antithrombotic treatment remains uncertain, and there is insufficient data regarding the lifelong use of acetylsalicylic acid in renal, mesenteric, coronary, or peripheral arteriopathies. Thus, treatment should be tailored to individual patients. Other clinical considerations include education related to precautionary measures, such as advising patients at risk for cervical dissection to avoid activities associated with extreme hyperextension or lateral rotation of the neck.
Complexity related to involvement of the cerebrovascular and the peripheral vascular systems and associated comorbidities should be considered in therapeutic decision-making. Hypertension should be treated according to the clinical practice guidelines, 78 but should be individualized on a case-by-case basis. For example, treating patients with hypertension with antihypertensive drugs may predispose the patients to ischemic strokes, particularly in the presence of episodes of hypotension and/or hypoxia, due to the combination of cerebral vaso-occlusive disease and the dysfunction of the VSMCs, resulting in poor cerebrovascular autoregulation.56,72 Treatment should therefore be aimed at maintaining a balance between lowering the high blood pressure to preserve renal function while maintaining adequate cerebral perfusion.
Endovascular therapy with angioplasty for RAS is typically restricted to cases with symptomatic stenosis despite optimal medical therapy or in cases with rupture of an intracranial aneurysm or dissection, as children have a high incidence of significant peri-procedural morbidity.17,79 Blood pressure may fall abruptly following renal angioplasty and should therefore be monitored closely, with concurrent medical interventions withdrawn during these procedures.
Surgical revascularization procedures for moyamoya are often challenging in patients with systemic involvement. Extracranial arterial involvement should always be sought prior to surgery as this may affect the peri-procedural and anesthetical management. For example, an NF1 patient with midaortic syndrome will require a tailored periprocedural and anesthetic approach. Management of blood pressure perioperatively is challenging. This is particularly true for patients undergoing direct or combined revascularization surgery, as a higher blood pressure level could lead to hyperperfusion syndrome and intracranial hemorrhage after revascularization, whereas lower blood pressure would increase the risk of cerebral infarction after revascularization. Hence, exploring an optimized equilibrium of perioperative blood pressure levels is essential in these patients. As blood pressure can fall after surgical revascularization of the cerebral circulation, due to improved cerebral perfusion, blood pressure should be monitored, and antihypertensive medications lowered if deemed necessary.
Future Perspective and Conclusion
Pediatric cerebral and systemic steno-occlusive vasculopathies are rare and heterogenous. However, they are associated with high morbidity and mortality and their medical management requires substantial medical resources. Health care providers should be alerted to potential systemic vascular involvement and its sequelae in children with complex cerebrovascular disease. FMD is a common terminology used for pediatric systemic vasculopathies that involve renal and cerebral vasculature, although current data reveal that pFMD shares clinical and pathological features with other cerebral and systemic monogenic vasculopathies, rather than with adult-type FMD. It is therefore more plausible that pFMD reflects a heterogenous group of heretofore undefined genetic systemic arteriopathies most likely related to genetic susceptibility, rather than a single disease. The relatively small-sized, single-center phenotypic and genetic studies continue to challenge our ability to uncover the genetic forces at play, further highlighting the need for an improved approach to diagnosis and classification that will facilitate the conduct of future trials.We offer a new term for multisystemic arteriopathies in the pediatric population, cerebral and systemic arteriopathies of childhood (CSA-c). This classification will provide a uniform terminology that can be applied by international collaborative registries of pediatric arteriopathies to facilitate consistent phenotypic characterization. We acknowledge that this terminology is primarily based on imaging, which lacks specificity, and likely reflects the combined effects of genetic, biomolecular, and environmental influences. Nevertheless, considering the widespread use of imaging in arteriopathy classification globally, we advocate for the adoption of this new terminology with the ultimate objective of integrating multimodal data, including advanced vascular neuroimaging techniques, biomarker discoveries, and high-throughput genomics, proteomics, and epigenomics in future research. This will facilitate a more comprehensive understanding of childhood cerebral arteriopathies with systemic involvement, and the discovery of genetic subtypes and provide new mechanistic insights into the pathophysiologic basis of this group of disorders, ultimately leading to a more mechanism-driven approach to classification, and the development of targeted interventions and preventive treatments.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241254796 – Supplemental material for Cerebral arteriopathies of childhood and stroke – A focus on systemic arteriopathies and pediatric fibromuscular dysplasia (FMD)
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241254796 for Cerebral arteriopathies of childhood and stroke – A focus on systemic arteriopathies and pediatric fibromuscular dysplasia (FMD) by Moran Hausman-Kedem, Pradeep Krishnan and Nomazulu Dlamini in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
