Abstract
Background:
To date, PCSK9 inhibitors are well known for eliminating cardiac and cerebral artery ischemia events by lowering the serum lipid level. However, the pathophysiological value of in-plaque PCSK9 expression is still unclear.
Methods:
Advanced plaques removed by carotid endarterectomy were sectioned and stained to identify the PCSK9 expression pattern and its co-expression with rupture-relevant markers. To investigate the correlation of PCSK9 expression with regional blood shear flow, hemodynamic characteristics were analyzed using computational fluid dynamics, and representative parameters were compared between PCSK9 positive and negative staining plaques. To explore this phenomenon in vitro, human aortic vascular smooth muscle cells were used to overexpress and knock down PCSK9. The impacts of PCSK9 modulations on mechanical sensor activity were testified by western blot and immunofluorescence. Real-time polymerase chain reaction was used to evaluate the transcription levels of downstream rupture-prone effectors.
Results:
PCSK9 distribution in plaque preferred cap and shoulder regions, residing predominantly in smooth muscle actin-positive cells. Cap PCSK9 expression correlated with fibrous cap thickness negatively and co-expressed with MMP-9, both pointing to the direction of plaque rupture. A hemodynamic profile indicated a rupture-prone feature of cap PCSK9 expression. In vitro, overexpression and knockdown of PCSK9 in human aortic vascular smooth muscle cells has positive modulation on mechanical sensor Yes-associated protein 1 (YAP) activity and transcription levels of its downstream rupture-prone effectors. Serial section staining verified in situ colocalization among PCSK9, YAP, and downstream effectors.
Conclusions:
Cap PCSK9 possesses a biomarker for rupture risk, and its modulation may lead to a novel biomechanical angle for plaque interventions.
Keywords
Background
Circulating proprotein convertase subtilisin/kexin type 9 (PCSK9) was known for its role in targeting low-density lipoprotein (LDL) receptors by degradation and thus increasing the LDL-cholesterol level. 1 Randomized clinical trials have sealed the status of PCSK9 inhibitors as an established lipid-lowering therapy benefiting the prevention of cardio-cerebrovascular diseases.2–4 PCSK9 was also detected in atherosclerotic plaques, and has been reported for eliminating LDL receptors on the surface of macrophages, its crosstalk with ROS/NF-κB/LOX-1/oxLDL, and its promotion of platelet activation and thrombosis.5–7 However, the specific distribution mode of PCSK9 and relevant functional impact on plaque structure have not been fully evaluated.
The histological classification of atherosclerotic plaque was based on reports from the Council on Arteriosclerosis of the American Heart Association, with a type VI lesion being the most dangerous. 8 Among all the criteria, a thinner fibrous cap and/or surface defect are of the most importance to define type VI plaques. Under hemodynamic stress from blood flow and other stimulations, the fibrous cap becomes more and more vulnerable and eventually ruptures, followed by clinical ischemic events and lesion progression. We sought to investigate the molecular biology, with specific distribution in fibrous cap to provide insight into the mechanism of cap rupture.
On the other hand, the fibrous cap is the region sensing hemodynamic stress from blood flow. In the realm of atherosclerosis research, computational fluid dynamics (CFD) has emerged as a pivotal tool for elucidating the complex hemodynamic forces at play within arterial plaques. CFD simulations enable the detailed analysis of blood flow patterns and shear stress distributions, which are critical in understanding the biomechanical environment of atherosclerotic lesions. 9 These hemodynamic parameters, particularly wall shear stress, have been implicated in the modulation of plaque composition and stability,10,11 influencing both endothelial function and the behavior of vascular smooth muscle cells (VSMCs).12,13 Furthermore, CFD studies have provided insights into the mechano-transduction pathways that govern cellular responses to hemodynamic forces, highlighting the interplay between fluid dynamics and molecular signaling in plaque progression and vulnerability. 14
In this study, we investigate the PCSK9 distribution pattern and its impacts on plaque vulnerability, focusing on cap rupture. Using histologic analyses, methods of CFD simulation, and a molecular biological technique, we aim to provide a unique hemodynamic profile of cap PCSK9 in advanced plaques, and its rupture-prone nature, for the first time.
Methods
Tissue processing and immunohistochemistry
This study was approved and performed under guidelines of the Institutional Ethics Committee of Changhai Hospital. Patients with severe atherosclerotic carotid artery stenosis who underwent carotid endarterectomy (CEA) in Changhai Hospital were included in this study after giving written informed consent. To investigate the distribution of PCSK9 in carotid plaque and its association with Yes-associated protein 1 (YAP) and downstream effectors, plaque tissues were collected and prepared in 4-μm thick formalin-fixed, paraffin-embedded (FFPE) serial sections and the segment with most stenosis was scanned for the purpose of detecting PCSK9 expression. Goat antibody against PCSK9 (Abcam, Cambridge, MA, USA) was used at a dilution of 1:100, with appropriate negative controls stained in parallel. Then the tissue sections were incubated with 1:1000 dilution of secondary donkey anti-goat antibody for 1 hour (Abcam), followed by incubation of tissue sections with DAB for 10 minutes. The immunohistochemistry distribution of PCSK9 was then recorded in atherosclerotic lesions.
Rabbit antibodies against YAP1 (Abcam), connective tissue growth factor (CTGF) (Abclonal, Wuhan, China), and Snail family zinc finger 2 (SLUG) (Abclonal) were all used at the same dilution of 1:500, then incubated with 1:1000 dilution of secondary donkey anti-rabbit antibody. Plaques were dichotomized into the PCSK9 positive staining group and the PCSK9 negative staining group. The clinical data and lipid level were compared between two groups.
Cell culture and treatment
Human aortic vascular smooth muscle cells (HAVSMCs) (Beiao, Guangzhou, China) were cultivated on the cell culture flasks in DMEM/F12 medium (Hyclone, Logan, UT, USA) containing 10% bovine serum (Yuanmu, Shanghai, China) at 37°C and 5% CO2. All experiments involved HAVSMCs at no more than a total passage of five. Cells were grown to 75% confluence before stimulation. For statistical reasons, all the experiment that received stimulation was repeated three times.
PCSK9 knockdown and overexpression
To explore the impacts of PCSK9 on cell morphology, function, and YAP activation, we modulate PCSK9 expression in HAVSMC. Small interfering RNAs against human PCSK9 (si-PCSK9 sequence 1, sense: 5’-GGC AGA GAC UGA UCC ACU UTT-3’, anti-sense: 5’-AAG UGG AUC AGU CUC UGC CTT-3’; si-PCSK9 sequence 2, sense: 5’-GGG UCA UGG UCA CCG ACU UTT-3’, anti-sense: 5’-AAG UCG GUG ACC AUG ACC CTT-3’; si-PCSK9 sequence 3, sense: 5’- GAA UGA CUU UUA UUG AGC UTT-3’, anti-sense: 5’- AGC UCA AUA AAA GUC AUU CTT-3’) and scrambled si-RNA were purchased from Genomeditech (Shanghai, China) and utilized according to the manufacturer’s protocol. The generation of plasmid DNA (transcript NM_174936.3), transfection with HAVSMC, as well as the lentiviral transduction to target cells were performed. All the modulation of PCSK9 proteins were confirmed using Western blot.
Immunofluorescent staining
To verify the expression of PCSK9 from VSMC in the cap region, we performed tissue slice immunofluorescent staining. The paraffin-embedded sections were deparaffinized and rehydrated, and incubated in citrate buffer for antigen retrieval. Goat antibody against PCSK9 (Abcam) and mouse antibody against alpha-smooth muscle actin (α-SMA) (Abcam) were used at a dilution of 1:100 and 1:500, respectively. Appropriate negative controls were stained in parallel. After washing, the tissue sections were incubated with a 1:2000 dilution of secondary donkey anti-goat antibody (Yeasen, Shanghai, China) and donkey anti-mouse antibody (Abcam) for 1 hour. To investigate PCSK9 influence on YAP nuclear localization, we performed cell immunofluorescent staining. Goat antibody against PCSK9 (Abcam) was used at a dilution of 1:200, then incubated with a 1:1000 dilution of secondary donkey anti-goat antibody. Rabbit antibodies against YAP1 (Abcam) and pYAP1Ser127 (Abcam) were used at the same dilution of 1:250, then incubated with a 1:1000 dilution of secondary donkey anti-rabbit antibody. Nuclear staining was done by using 4’,6-diamidino-2-phenylindole (DAPI; dihydrochloride). To assemble the images, the staining was evaluated using a Leica microscope (Olympus BX60). To calculate an integrated fluorescence density of PCSK9 in Figure 1D, a 250 × 250-μm rectangular region of interest (ROI) was manually drawn on the PCSK9 positive region directly under the cap surface using ImageJ software (National Institutes of Health [NIH], Bethesda, MD, USA), then cap thickness (ROI to surface distance) was correlated with PCSK9 integrated density using GraphPad Prism (Treestar, Boston, MA, USA). Similarly, to calculate cell morphological features in tissue VSMCs, the ROI of an individual cell was established based on SMA staining, and the PCSK9 expression level was stratified based on its integrated intensity into high (higher than 2000 of intensity) and low (lower than 2000) cells (Figure 4F). To analyze colocalization of PCSK9 and MMP-9, digital slides were acquired (3DHistech, Budapest, Hungary), then images were saved and processed by ImageJ as well as JACoP to generate Pearson’s coefficient (NIH).
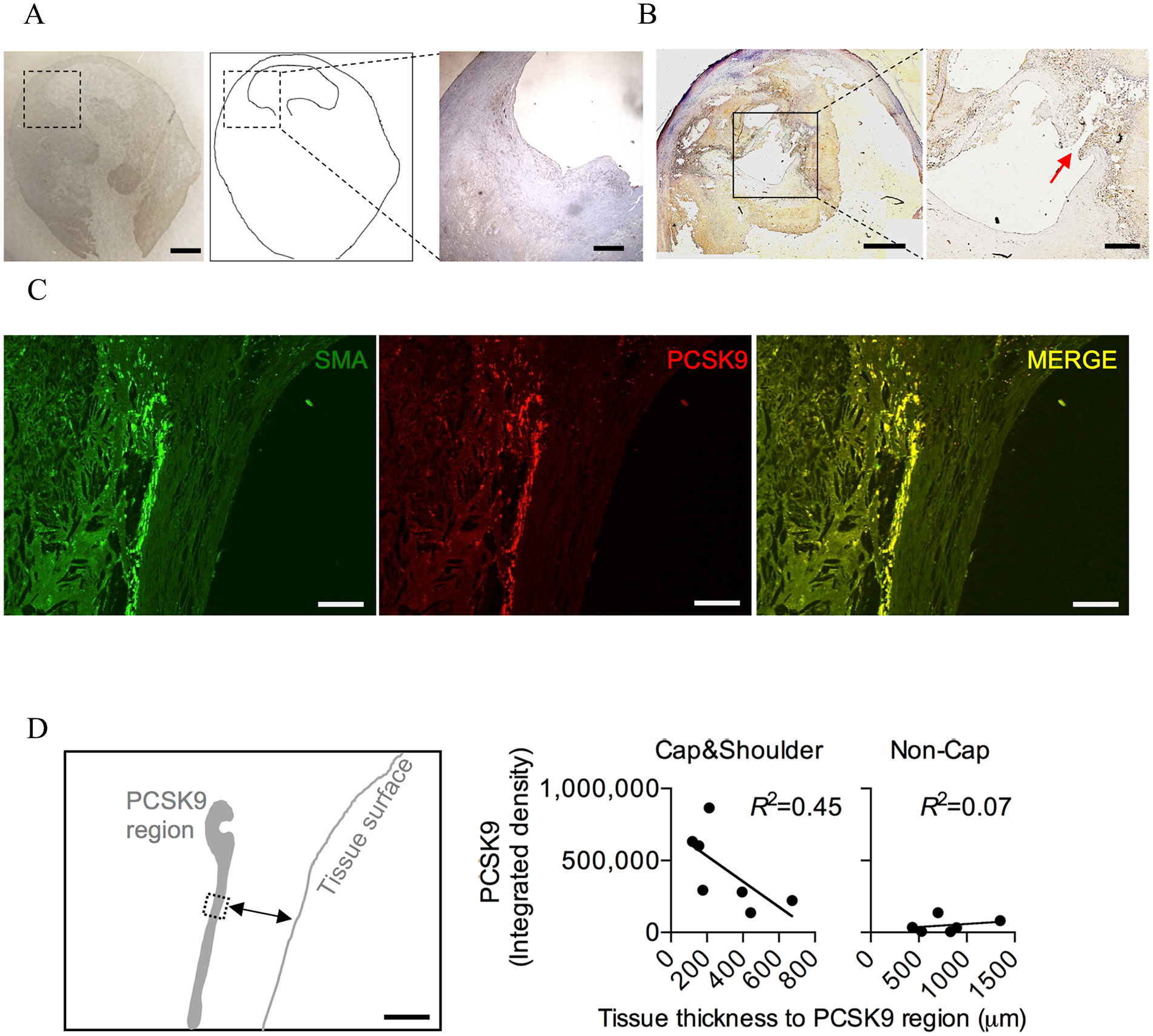
PCSK9 expression in cap and shoulder region in advanced plaques. (
Western blot
To verify the modulation of PCSK9 on YAP phosphorylation level, we performed Western blot. HAVSMCs were collected and lysed in RIPA lysis buffer, followed by incubation on ice for 30 minutes. After the centrifugation, the supernatant was collected and protein concentration quantified using a BCA protein assay kit (Yeasen, Shanghai, China). Then, the proteins were separated by SDS-polyacrylamide gels and transferred to polyvinylidene difluoride (PVDF) membranes. We used 5% nonfat milk to block the blots and primary antibodies were then used for incubation overnight (goat antibody against PCSK9 [Abcam], 1:100, rabbit antibody against YAP1 [Abcam], 1:500, rabbit antibody against pYAP1Ser127 [Abcam], 1:500). The secondary antibodies were used the next day and detected using an enhanced chemiluminescence detection kit. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Abcam) was used as the internal reference. ImageJ software was used to measure the concentration value.
Migration assays
An independent assay for migration was the Boyden chamber assay. HAVSMCs were treated with either PCSK9 siRNA or control siRNA and then seeded into a 24-well Boyden chamber plate (3 × 103 cells). After 4 hours, cells were cultured in a serum-deprived medium overnight and treated with platelet-derived growth factor-BB (PDGF-BB; 20 ng/mL) for 8 hours at 37°C. Cells were then fixed, stained with crystal violet, and counted using a microscope. The cells were then imaged, and the result was statistically evaluated.
Proliferation assays
The viability of HAVSMCs was then measured by a CCK8 proliferation assay using a Cell Counting Kit (CCK-8)(Beyotime, Shanghai, China). Briefly, HAVSMCs were seeded into 96-well plates after transfecting with either PCSK9 siRNA or control siRNA, followed by the addition of CCK-8 solution (10 μL per well). Cells were incubated in the dark for 1 hour and then tested using a microplate absorbance reader at 450 nm.
Real-time polymerase chain reaction (RT-PCR)
To investigate PCSK9 influence on YAP downstream rupture-prone effectors, we performed a RT-PCR test. Total RNAs were extracted from cultured HAVSMCs using TRIzol Reagent (Beyotime). To detect mRNA, we then synthesized the cDNA from 1 μg of total RNA. (a) h-SLUG (5-3) ACGCCTCCAAAAAGCCAAACACTCACTCGCCCCAAAGATG; (b) h-CTGF (5-3) GTGGAGTATGTACCGACGGCGCAGGCACAGGTCTTGATGA; (c) h-GAPDH (5-3) GCACCGTCAAGGCTGAGAACTGGTGAAGACGCCAGTGGA. Then, RT-PCR analysis was performed with SYBR Premix Ex Taq (Thermo Fisher Scientific, USA). We then used a relative standard curve method (2-△△CT) to detect the relative mRNA expression. The RT-PCR was performed using SYBR Green Master Mix (Thermo Fisher Scientific). And GAPDH was used as the internal control unless specified otherwise.
Vessel model reconstruction
To conduct a computational fluid dynamics simulation, we have to reconstruct the vessel model first. Raw data were generated from computed tomography angiography (CTA) (Siemens, Germany; 1.0 mm per slice). The acquired CTA raw data were reconstructed in Mimics 19.0 software (Materialise, Leuven, Vlaams-Brabant, Belgium) to generate STL files for 3D carotid artery models subsequently imported into Geomagic 2015 software (3D Systems Inc., Rock Hill, SC, USA) for model repair, trimming, and smoothing. Based on the reconstructed 3D arterial models, angles between the internal carotid artery (ICA) and common carotid artery (CCA), as well as between the ICA and external carotid artery (ECA), were calculated along the vessel centerlines. The degree of stenosis was determined using the North American Symptomatic Carotid Endarterectomy Trial (NASCET) method. 15
Computational fluid dynamics (CFD) imulation
The trimmed artery model was imported and meshed using Star-CCM+ (CD Adapco, Melville, NY, USA) to create around 1 million polyhedral elements with a maximum mesh size of 0.1 mm and four layers of wall prism elements for accurate boundary layer resolution. Blood was considered as Newtonian fluid without regard to energy equations and gravity. The basic equation governing the flow was the incompressible Navier–Stokes equation and the laminar simulation was carried out in transient-state using Star-CCM+. The viscosity coefficient of blood was constant, which was 0.0035 Pa‧s, and the density was 1056 kg/m3. The pulsed mean flow rate was used as the inlet and outlet boundary conditions. 16 Traction-free boundary conditions were implemented at each outlet. The patient-specific mean flow rate of the inlet and outlet boundary was calculated by combining digital subtraction angiography (DSA) images and a 3D model, referring to the Thrombolysis in Myocardial Infarction (TIMI) frame count method commonly used in coronary stenosis research. 17 A rigid-wall and no-slip boundary condition were implemented at each vessel wall. Three pulsatile cycles were simulated to ensure that numerical stability was achieved, and the simulation results of the last cycle were taken as the output. All data presented are the time averages over the third pulsatile cycle of flow simulation. Postprocessing of the CFD simulation results was conducted using Tecplot 360 EX 2022 software (Tecplot, Inc., Bellevue, WA, USA).
Hemodynamic analysis
From the simulated flow fields, we calculated the distribution of the following hemodynamic parameters: wall shear stress (WSS), wall shear stress gradient (WSSG), and oscillatory shear index (OSI). For the plaque stenosis site, we calculated the average and maximum values of WSS, WSSG, and OSI, as well as the maximum values of normalized WSS (NWSS) and normalized WSSG (NWSSG), which were normalized by the value of the reference vessel segment. The reference vascular segment was a segment without plaques on the stenotic vessel.
Statistical analysis
Patients who underwent CEA were divided into two groups based on the presence of in-plaque PCSK9 expression. Baseline characteristics, vessel geometries, and hemodynamic parameters were compared using independent samples t-tests for continuous variables and chi-squared or Fisher’s exact tests for categorical variables. Logistic regression analysis was employed to identify independent risk factors for in-plaque PCSK9 expression, with odds ratios (ORs) and 95% CI calculated and presented. For cell line study, at least three independent replications were performed. Continuous variables were presented as mean ± SD, and Student’s t-test was used for comparisons between two groups; p < 0.05 was deemed statistically significant. Data were analyzed with R software, version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria) and statistical significance was calculated as p < 0.05, 0.01, and 0.001.
Results
In-plaque PCSK9 expression by VSMCs in cap and shoulder region
As PCSK9 has been reported for its production by VSMCs to eliminate LDL receptors on the surface of macrophages, 5 we were interested in its expression in advanced plaques. To our surprise, preliminary results showed its cap-biased expression in histology (Figure 1A), a well-known location for plaque rupture. To further confirm this finding, we specifically investigated ruptured plaque and found the same cap-biased PCSK9 expression (Figure 1B). To confirm its VSMC origin, we stained PCSK9 with α-SMA simultaneously, and both signals colocalized in the cap region (Figure 1C). Indeed, according to preliminary experiments, PCSK9 was expressed in 45% of advanced plaques tested, and more than 95% of positive plaques showed obvious cap-biased PCSK9 distribution, and this evidence gave us certain confidence of this neglected phenomenon.
Cap PCSK9 expression has a negative correlation with cap thickness
Next, we were intrigued by the clinical indication of this cap-biased distribution and hypothesized that local enriched PCSK9 may correlate with cap rupture. As fibrous cap thickness was a widely accepted measurement for plaque stability in vivo, we measured the cap thickness and in situ PCSK9 expression simultaneously. As shown in Figure 1D, we were able to identify a significant negative correlation between PCSK9 intensity and cap thickness, whereas PCSK9 was rarely expressed in noncap regions and had poor correlation with tissue thickness. Together, we found that PCSK9 dominantly expressed by α-SMA positive VSMC cells in the cap region, and cap PCSK9, may possess an indication for plaque rupture.
MMP-9 colocalization with cap PCSK9
Next, we were looking for a well-established marker for plaque rupture and chose matrix metalloproteinase-9 (MMP-9) as the major one. MMP-9 expression in the shoulder and cap region is an arrogant marker for plaque rupture. 18 To investigate the association of PCSK9 expression with cap rupture, we co-stained PCSK9 with MMP-9, which is used as a marker for cap rupture. We were able to identify MMP-9 colocalization with PCSK9 in the cap region of rupture plaques, whereas noncap PCSK9 rarely co-resided with MMP-9 (Figure 2). This evidence further suggested the possibility of cap PCSK9 association with rupture for advanced plaque.
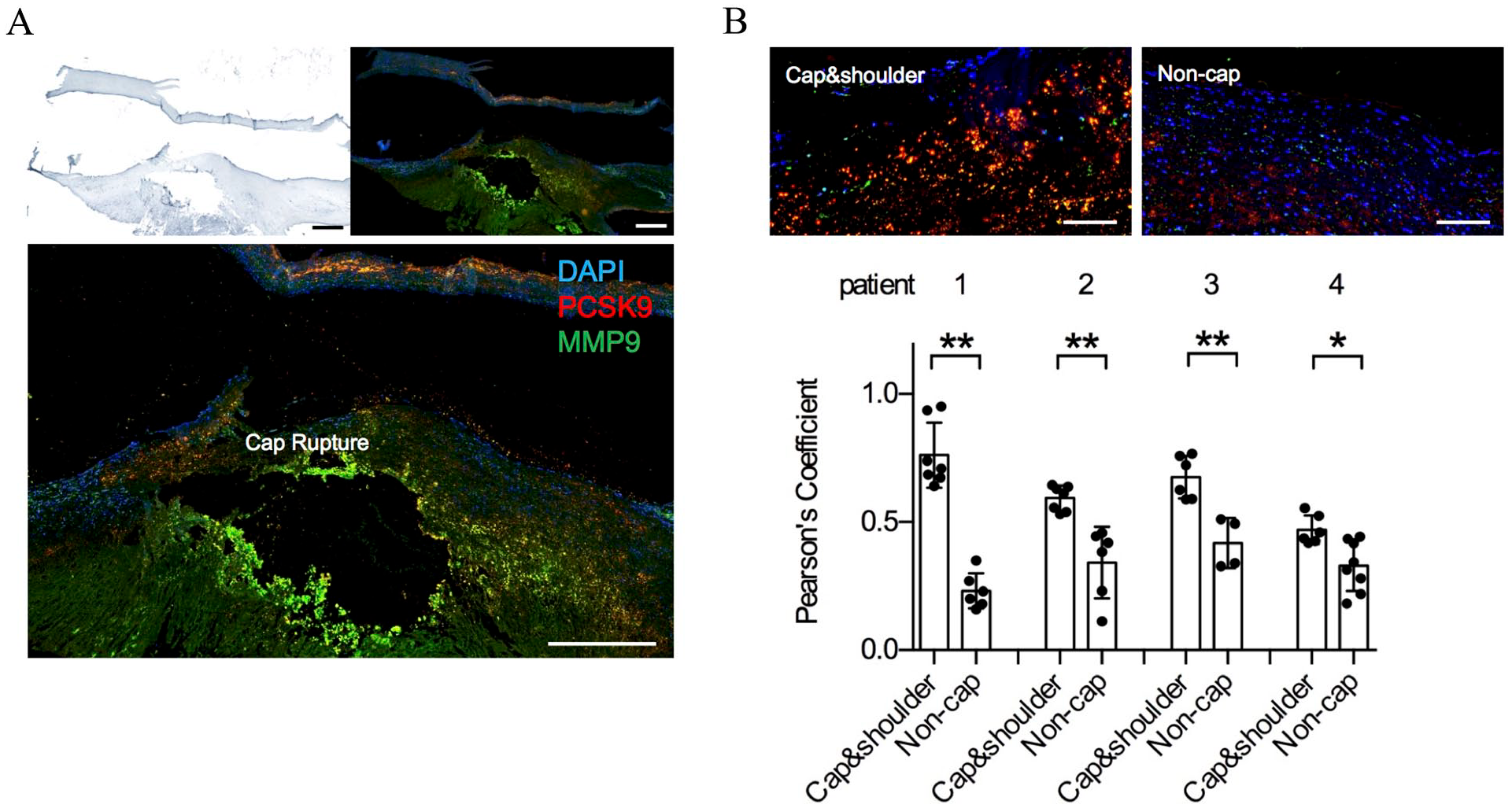
MMP-9 co-expression with PCSK9 in the fibrous cap region.
Hemodynamic characteristics featuring plaque rupture of cap PCSK9 expression
Cap regions are intensively impacting vessel sites where blood squeezed through. Specific hemodynamic parameters were reported to correlate with plaque vulnerability. To further explore cap PCSK9’s role in plaque vulnerability, we enrolled 20 CEA patients who had undergone a CTA scan 2 weeks before surgery. By using CTA reconstruction of vessels, we obtained hemodynamic parameters by CFD simulation, and then analyzed the differences in hemodynamic profiles between the cap PCSK9 positive staining group (n = 11) and the cap PCSK9 negative staining group (n = 9). Of them, 11 patients underwent carotid ultrasound (four soft plaques, five hard plaques, and two mixed plaques). The demographic and baseline data are presented in Table 1. Individual patient characteristics are presented in the online Supplementary Table 1.
Baseline data of enrolled patients undertaking carotid endarterectomy.
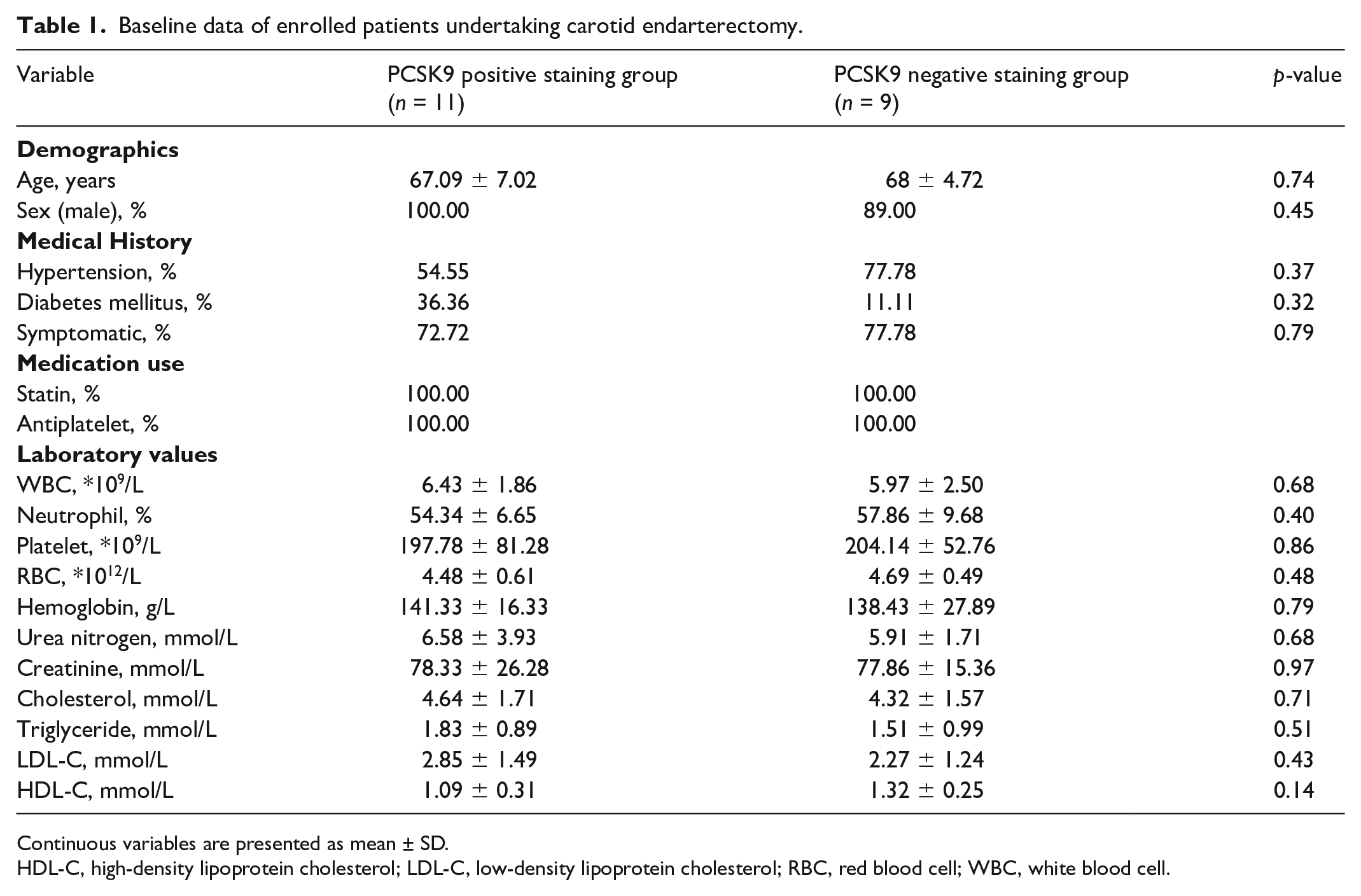
Continuous variables are presented as mean ± SD.
HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; RBC, red blood cell; WBC, white blood cell.
Vessel geometry, degree of stenosis, and hemodynamic parameters were compared between the cap PCSK9 positive staining group versus the negative group. We found that the PCSK9 positive staining group showed more severe stenosis, higher maximum WSS of plaque, higher average WSS, and higher average WSSG as compared with the PCSK9 negative staining group. Relevant data are presented in Table 2.
Comparative analysis of vascular geometry and hemodynamic parameters between PCSK9 positive versus PCSK9 negative staining groups.
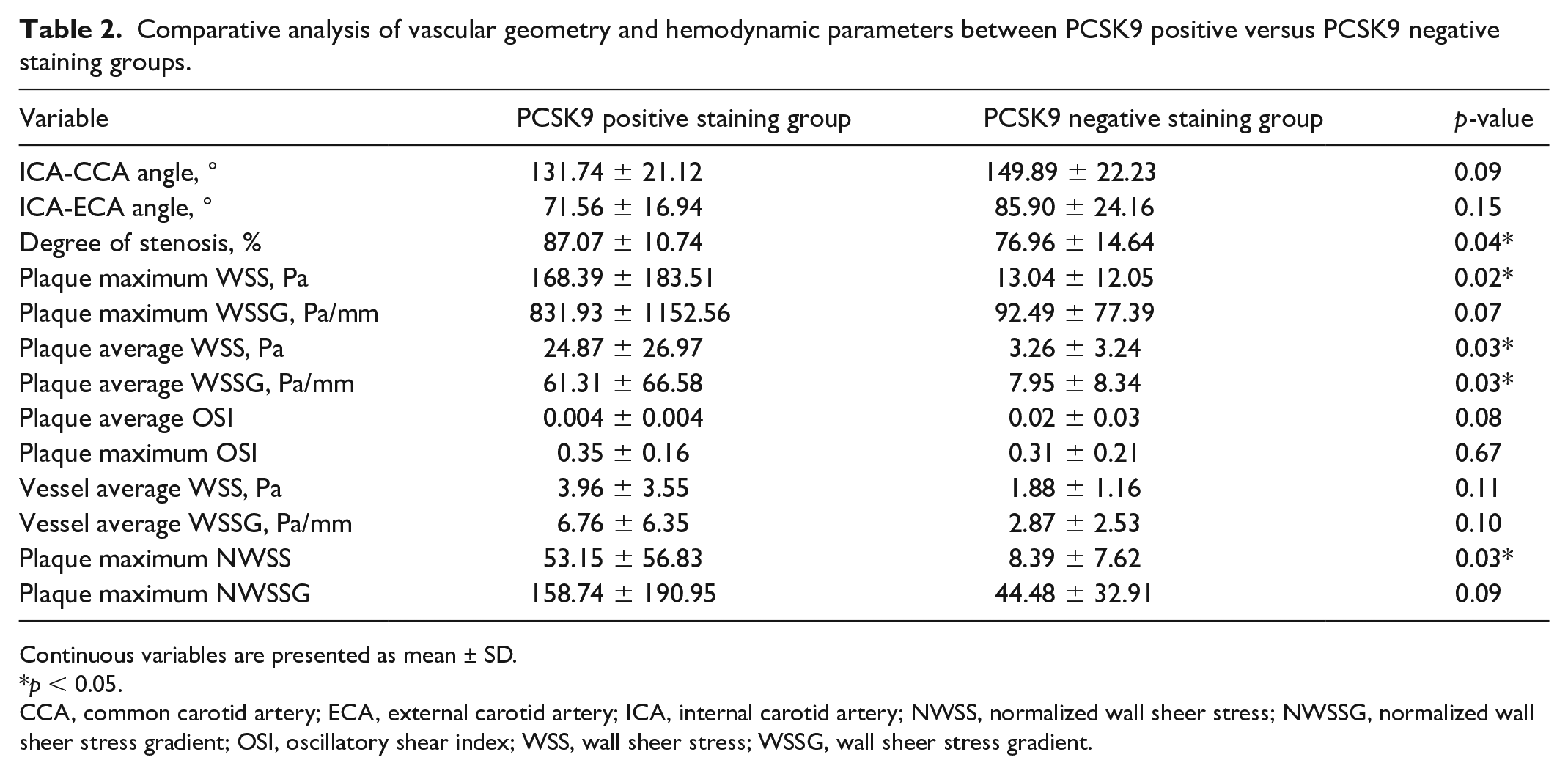
Continuous variables are presented as mean ± SD.
p < 0.05.
CCA, common carotid artery; ECA, external carotid artery; ICA, internal carotid artery; NWSS, normalized wall sheer stress; NWSSG, normalized wall sheer stress gradient; OSI, oscillatory shear index; WSS, wall sheer stress; WSSG, wall sheer stress gradient.
Based on logistic regression analysis, it was indicated that plaque maximum WSS, plaque average WSS, and plaque maximum NWSS were independent risk factors for cap PCSK9 expression. Plaques with cap PCSK9 expression exhibited a rupture-prone hemodynamic profile of higher plaque WSS levels. Relevant data are illustrated in Table 3.
Logistic regression analysis of hemodynamic parameters for in-cap PCSK9 expression.
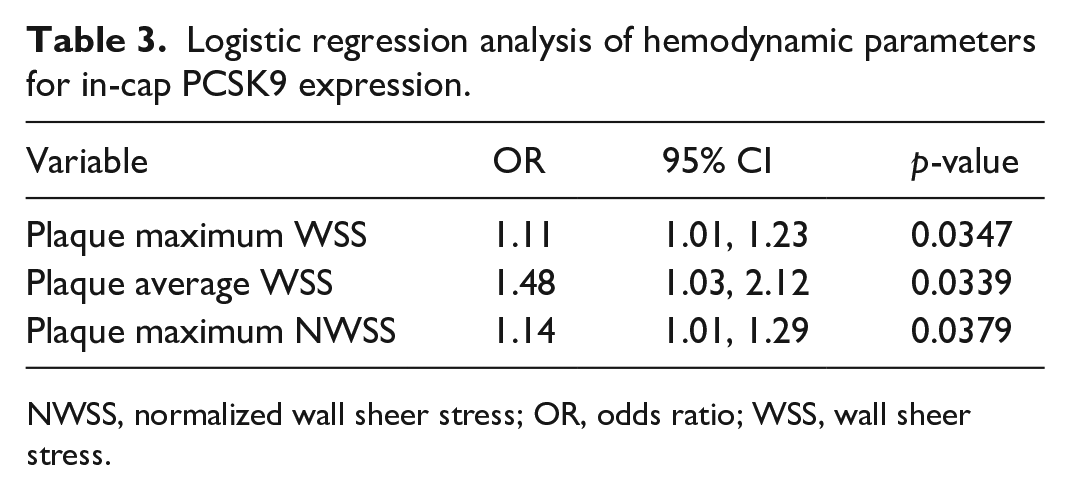
NWSS, normalized wall sheer stress; OR, odds ratio; WSS, wall sheer stress.
Case from each of cap PCSK9 positive staining group and PCSK9 negative staining group was depicted in Figure 3. To be specific, despite a similar degree of stenosis, the cap PCSK9 positive case exhibited higher WSS at the site of maximal luminal narrowing relative to the negative case, in terms of higher local pressure level in red. Together, we provided MMP-9 colocalization and local hemodynamic features with cap PCSK9 expression, which again added value to its clinical relevance.
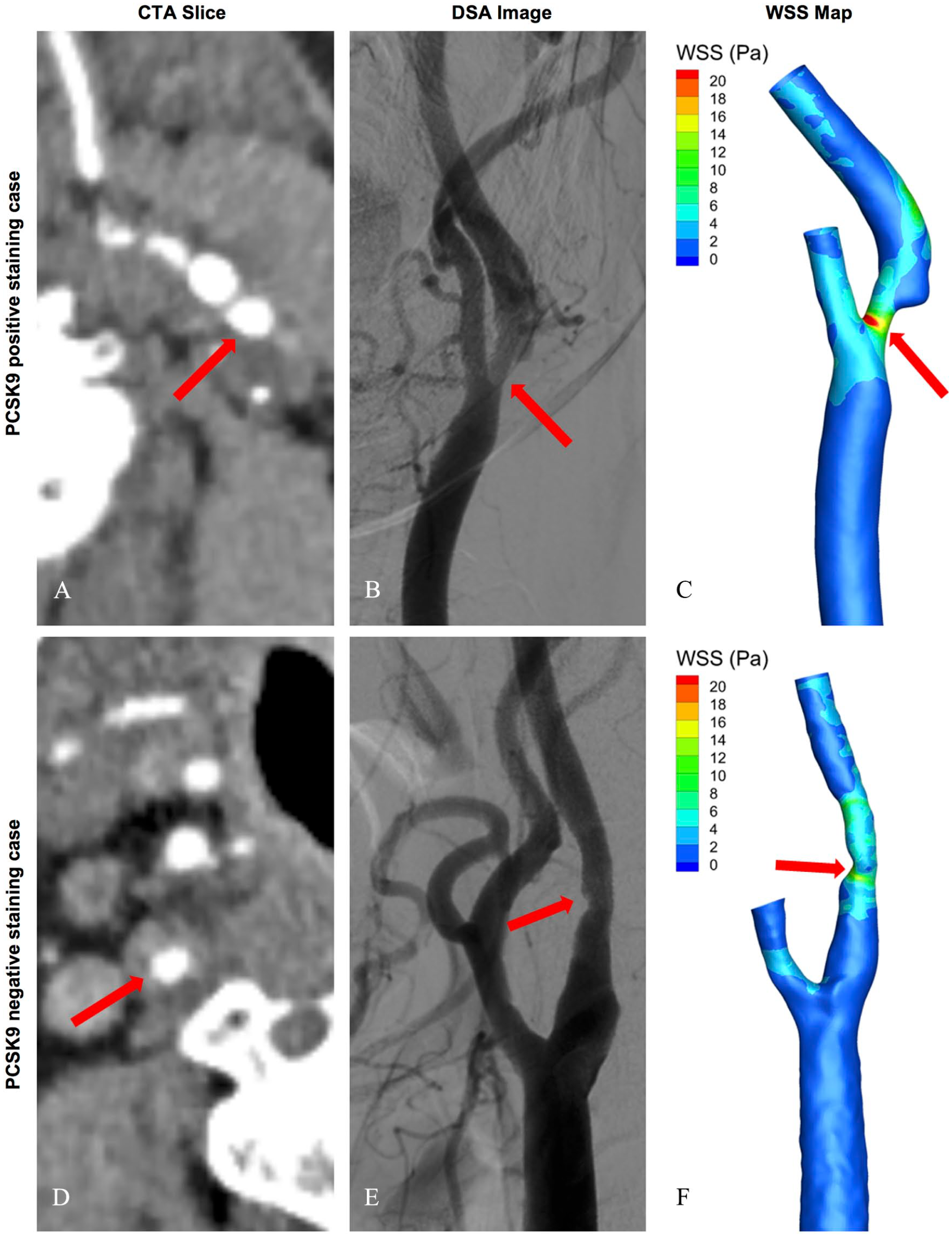
Typical cases from PCSK9 positive and negative staining groups. (
PCSK9-activated mechanical sensor YAP and its downstream rupture-prone effectors
Next, we manipulated PCSK9 expression in HAVSMC to identify its role on rupture-prone features. First, PCSK9 overexpression and knockdown showed cell morphology change. Cell migration and proliferation were both down-regulated by PCSK9 knockdown (Figure 4A–E). In section, PCSK9 highly expressed VSMCs showed an encircled shape, and PCSK9 highly expressed regions had significantly more cell counts than scarcely expressed regions (Figure 4F and G).
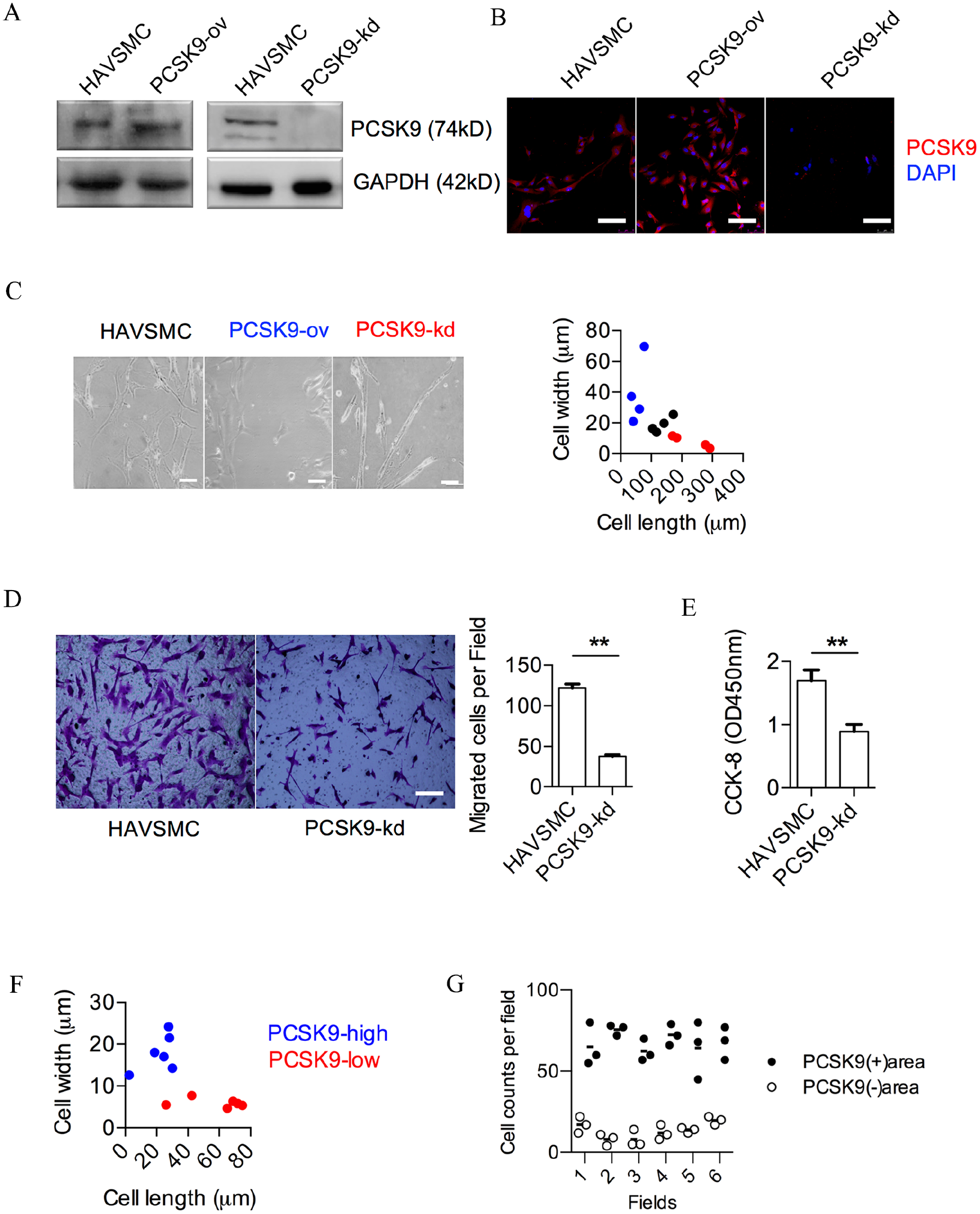
PCSK9-modulated HAVSMC morphology, migration, and proliferation. (
As both our histology and our CFD results showed that PCSK9 was enriched in the highly hemodynamic and pressure-sensitive cap region, we next determined to explore its impact on mechanical sensing. Mechanical sensing is a sophisticated regulating system, especially YAP, as a biomechanical sensor can be activated under flow stress and trigger atherosclerosis initiation with impacts on endothelial cells.19,20 It is also one of the major mechanical sensors and transcriptional activators in modulating the cell functions of VSMCs, and its expression was proved to be associated with cap vulnerability in advanced plaques.21,22 Thus, we next investigated the impact of PCSK9 on this mechanical sensor in VSMCs in vitro. First, PCSK9 overexpression in HAVSMCs reduced YAP phosphorylation at Ser127 and increased YAP nucleic localization. Conversely, PCSK9 knockdown increased YAP Ser127 phosphorylation and led to YAP cytoplasmic retention (Figure 5A and B). These results together suggest that sole manipulation of PCSK9 was sufficient to modulate the status of VSMC and the mechanical sensor YAP in vitro.
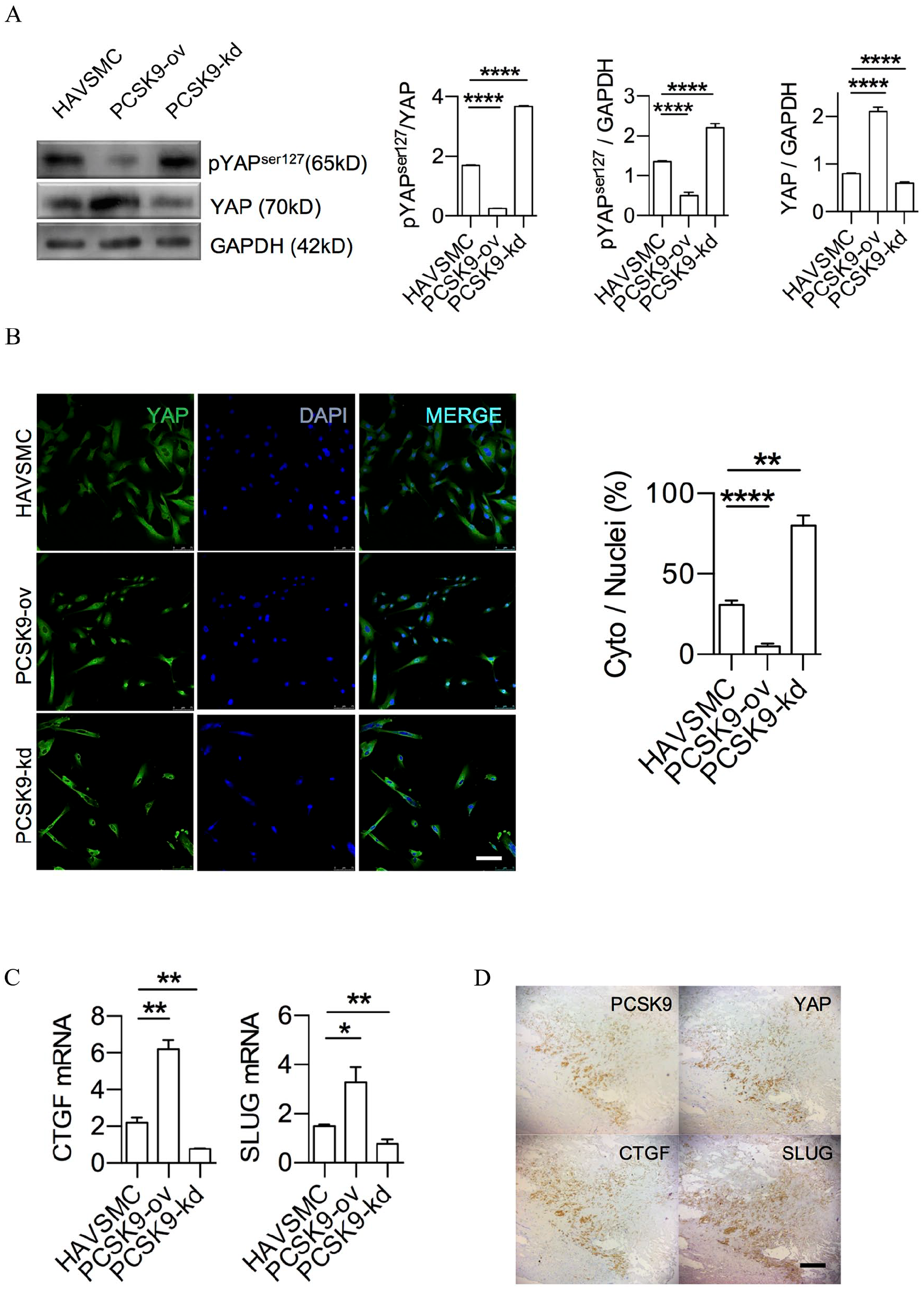
PCSK9-activated mechanical sensor YAP in vitro.
To further confirm this YAP signaling cascade under PCSK9 regulation, downstream effectors of YAP such as CTGF and SLUG (two molecules highly relevant to atherosclerosis progression and plaque rupture) were readily modulated by PCSK9 along with YAP activation (Figure 5C).23,24
Finally, to translate these cell line data back to pathology, we stained serial sections of plaques and found that PCSK9, YAP, CTGF, and SLUG were indeed colocalized at tissue level (Figure 5D). According to our results, in advanced plaque, PCSK9 from VSMC enriched in the stress-intensive cap region might activate YAP and downstream rupture-prone effectors to destroy cap stability (Figure 6).
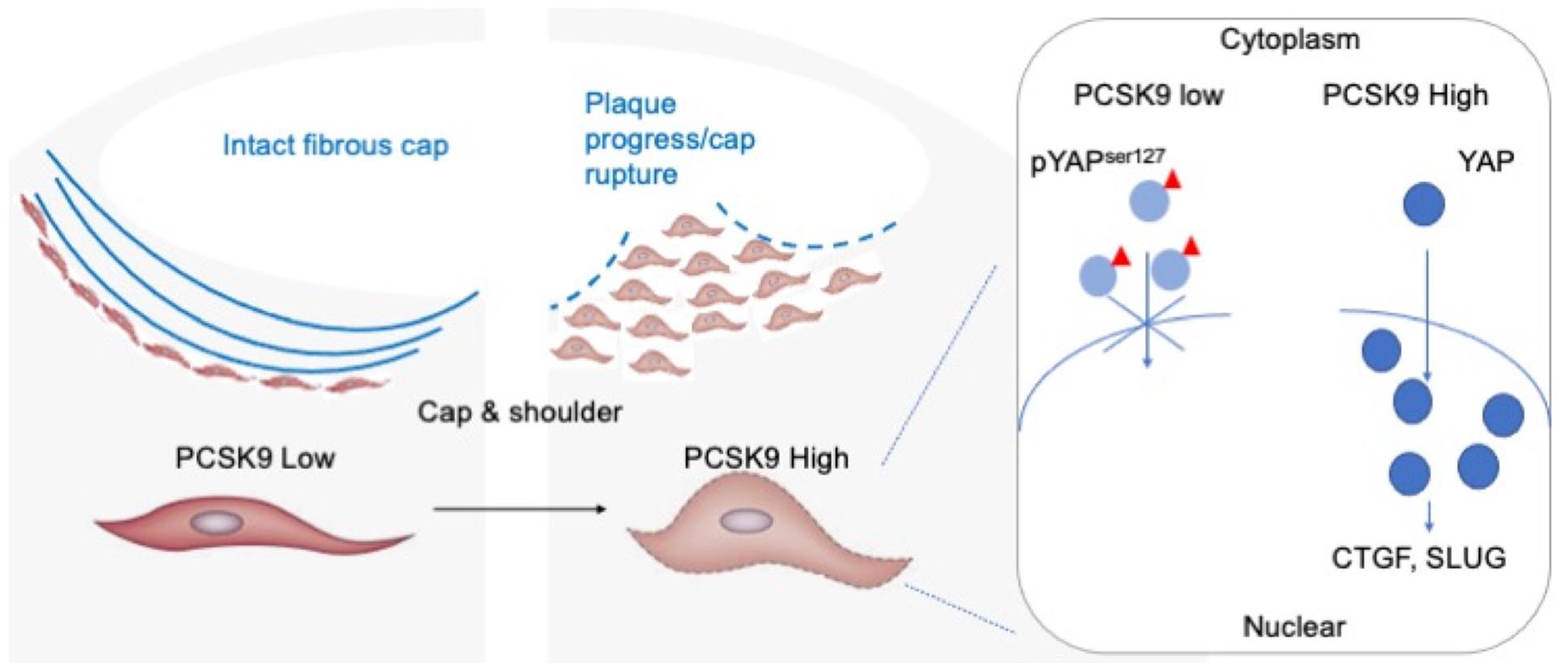
Proposed working model of cap PCSK9 from VSMCs.
Discussion
The results of this study included that: (1) in-plaque PCSK9 expression was mainly distributed in the hemodynamic stress-sensing region of the cap and shoulder; (2) cap PCSK9 was associated with cap thinning and PCSK9 co-expression with MMP-9 at rupture prone site; (3) PCSK9 positive staining plaques showed a significantly higher WSS level, which was linked to rupture risk; and (4) PCSK9 can modulate mechanical sensing factor YAP activation and downstream rupture-prone effectors. These results indicated that cap PCSK9 was involved in plaque rupture and provided insight into cap PCSK9 as a novel therapy target for vulnerable plaques.
Several studies have demonstrated the relationship between wall shear stress and plaque rupture. It was proved that high WSS segments develop a greater necrotic core and calcium progression, regression of fibrous and fibrofatty tissue, and excessive expansive remodeling, suggestive of transformation to a more vulnerable phenotype. 11 In another study, the high shear stress gradient is independently associated with plaque rupture, and the shear stress gradient is higher at rupture sites than at erosion sites. 25 The shear stress concentration was frequently correlated with the plaque rupture site and as a trigger of cap rupture, although the absolute value of shear stress is not sufficient to directly provoke mechanical destruction of the fibrous cap. 26 According to our results, PCSK9 expression correlated with higher WSS and higher NWSSG, and was independent of morphology of carotid bifurcation. 27 These data provided indirect supports for the in-cap PCSK9 as an indicator for plaque rupture. On the other hand, MMPs can catalyze the breakdown of the interstitial collagen, resulting in the thinning and weakening of the fibrous cap, thus compromising its tensile strength and making the plaque unstable. 28 Among several kinds of MMPs, MMP-9 both in circulation and in plaque can predict future events and serve as plaque rupture biomarkers.18,29 The co-expression of PCSK9 with MMP-9 in fibrous cap strongly support the rupture-prone nature of PCSK9 expression in the cap region. Whether and how PCSK9 influence the expression of MMP-9 is not the aim of the present study and needs further exploration.
Plaque rupture was not always symptomatic or events causing. 30 One in vivo study from coronary mapping compared the degree of stenosis with features of plaque vulnerability, and revealed that the prevalence of vulnerability features including plaque rupture were significantly highest in the tertile of greatest stenosis. 31 This is in line with our data, showing that the PCSK9 positive staining group harbors more severe stenosis compared with the PCSK9 negative staining group.
VSMCs play important roles in atherosclerosis pathogenesis and progression, in terms of proliferation, migration, and phenotype switch, especially in advanced plaques. 32 Fibrous cap has a critical role in the maintenance of the integrity of atherosclerotic plaque, with more differentiated VSMCs contributing increased plaque stability. 33 Animal models implied that different VSMC phenotypes may link in spatial plaque organization. 34 VSMCs might migrate and proliferate, and undergo phenotypic switch at the cap and shoulder region, where the hemodynamic stress was applied. Interestingly, PCSK9 itself was reported to express in VSMCs under hemodynamic stress in vitro and ex vivo.35,36 In this study, we found that silencing PCSK9 in HAVSMCs led to change in the cell function of proliferation and migration, which was in accordance with other literature. 37 Beyond that, we also detected changes in cell morphology as well as in SLUG, a marker for the epithelial mesenchymal transition signal.38,39 It would be of interest in the future to verify whether phenotypic switching of VSMCs under hemodynamic stress is introduced by PCSK9.
Study limitations
In our study, PCSK9 was found in only half of the atherosclerotic plaques analyzed. The reason for this phenomenon was unclear. We speculated that there were some possibilities as follows. First, as we used tissue pathology sampling from patients, we can only tell the characteristics of plaques at the time of endarterectomy. The evolution of atherosclerotic plaques under hemodynamic stress is a long-lasting process and is beyond the scope of single-time sampling. To solve this problem, plaque at different stages should be sampled and evaluated. However, due to ethical guidelines and clinical practical restraints, the patients undergoing CEA surgery are always a small subset of patients with carotid atherosclerosis. Second, in this study, we only scanned 20 samples at the most stenotic segment to detect PCSK9 expression. It was possible that we missed PCSK9 expression in other segments, and took these real positive plaques as false negative ones. Nonetheless, according to the vessel staining we performed at the long axis (e.g., in Figure 2), PCSK9 positive expression at the cap region of the most stenotic segment was indicated. Future studies will need to include a larger sample size. Third, according to clinical trial data from the coronary artery, some plaques revealed structure modulation after the use of statins or PCSK9 inhibitors. Whether statins or circulating PCSK9 inhibitors can influence cap PCSK9 expression has not been investigated. Meanwhile, all 20 patients in our cohort used statins and none of them used PCSK9 inhibitors. Thus, the likelihood of influence from lipid-lowing medication on cap PCSK9 expression difference is minimal.
Although we combined data from tissue markers, hemodynamic simulation, and in vitro experiments of HAVSMCs, our data demonstrate an association between cap PCSK9 expression and cap rupture with underlying mechanical sensor YAP activation in VSMC. Further studies will need to establish this as a causal relationship. There may be other mechanisms underlining cap PCSK9’s role in plaque stability. As PCSK9 was predominantly investigated with a lipid-dependent role in plaque initiation and growth, we speculate that cap PCSK9 might be a bi-input switch, sensing both lipid-dependent and mechanical-dependent cues, which warrants future investigation.
Conclusion
PCSK9 is distributed in the hemodynamic sensitive cap region of advanced plaques, and presence of PCSK9 correlates with cap vulnerability. We provide data to support that PCSK9 possesses a biomarker for plaque rupture risk, and cap PCSK9 modulation may lead to a novel biomechanical angle for plaque interventions.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241252370 – Supplemental material for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241252370 for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque by Yingying Zhang, Dongwei Dai, Shuang Geng, Chenbin Rong, Rong Zou, Xiaochang Leng, Jianping Xiang, Jianmin Liu and Jing Ding in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X241252370 – Supplemental material for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X241252370 for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque by Yingying Zhang, Dongwei Dai, Shuang Geng, Chenbin Rong, Rong Zou, Xiaochang Leng, Jianping Xiang, Jianmin Liu and Jing Ding in Vascular Medicine
Supplemental Material
sj-pdf-3-vmj-10.1177_1358863X241252370 – Supplemental material for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque
Supplemental material, sj-pdf-3-vmj-10.1177_1358863X241252370 for PCSK9 expression in fibrous cap possesses a marker for rupture in advanced plaque by Yingying Zhang, Dongwei Dai, Shuang Geng, Chenbin Rong, Rong Zou, Xiaochang Leng, Jianping Xiang, Jianmin Liu and Jing Ding in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
