Abstract

Statins are effective lipid-lowering agents that reduce the risk of cardiovascular events and are widely used for primary or secondary prevention of cardiovascular diseases.1,2 In cardiovascular trials, statin therapy has been extremely safe and the two best-known clinical adverse effects of statins—myopathy and liver injury—have been rare. In a recent individual patient data analysis of 19 randomized trials, the excess risk of myopathy with statin therapy versus placebo was only eight events per 100,000 person-years. 1 An American Heart Association Scientific Statement estimates serious statin-associated hepatotoxicity in only 0.001% of patients. 3 A systematic review and meta-analysis of randomized trials estimated that over an average trial follow-up period of 24 weeks, elevation of transaminases occurred in three more per 1000 patients receiving rosuvastatin than placebo. 4
Patients with cancer may frequently be on statin therapy, due to joint associations of cardiovascular disease and cancer with age or other shared risk factors. 5 Additionally, statins have been tried in clinical trials for patients with cancer with the intent to exploit their pleiotropic effect to improve cancer outcomes. However, the safety of statin therapy in patients with cancer receiving chemotherapy has not been rigorously investigated. Observational studies may be flawed by selective reporting, confounding, and other limitations. A meta-analysis of 11 randomized controlled trials (RCTs) included patients with solid tumors and their investigation was defined by proportions of grades 3–5 and myopathy, not by reporting a significant difference between patients receiving statins versus control. 6 However, that study included a relatively small number of patients. Since publication of that study, additional RCTs have been performed. Therefore, we conducted an updated systematic review of RCTs of patients with cancer who were assigned to statin therapy versus placebo or no treatment to assess the risk of clinically relevant myopathy and hepatotoxicity.
We searched MEDLINE with the PubMed interface for RCTs that randomized adult patients (aged ⩾ 18 years) with cancer to any statin therapy versus placebo or no treatment. A confirmatory search was performed with EMBASE along with a manual review of the reference list of relevant articles. We set no restriction on language, type or dose of statin, or type of cancer, although only studies with available full-text publication were considered for review, unless the abstract explicitly reported the safety outcomes. No restriction was set for the type or dose of the statins or the type of cancer.
The main adverse hepatic outcomes were defined as a significant elevation in transaminases requiring medical attention, including stopping the drug or an unscheduled visit according to the definition in the individual studies, although any reported increase in hepatic enzymes was also noted. The main muscle-related adverse outcome was reported as, and objectively confirmed, myopathy, and elevation of creatine kinase as defined by individual studies was also explored. Myalgia and other subjective symptoms were not considered among the main outcomes since recent evidence indicates a nocebo effect, with most of these symptoms also being reproducible with placebo. 7 Although meta-analysis of events was deliberated, prior to data extraction it was considered unfeasible because of the low number of events for myopathy and insufficient clarity of definition and reporting for hepatotoxicity.
The results are summarized in Figure 1. We searched PubMed and EMBASE (date of last search August 4, 2023), and identified 1750 records. After removing duplicates, we screened 1545 records. After careful review of the titles and abstracts, 1522 records were excluded (online Supplemental Figure 1). The excluded records mostly included publications that were not RCTs. RCTs that did not specifically address the study population of interest (i.e., human adults with cancers) or the intervention of interest were also excluded. Ultimately, 23 RCTs underwent full-text review, six of which were excluded due to not reporting the safety outcome of interest (i.e., hepatic adverse events and myopathy) or the comparison group being another dose of the same statin or another statin.
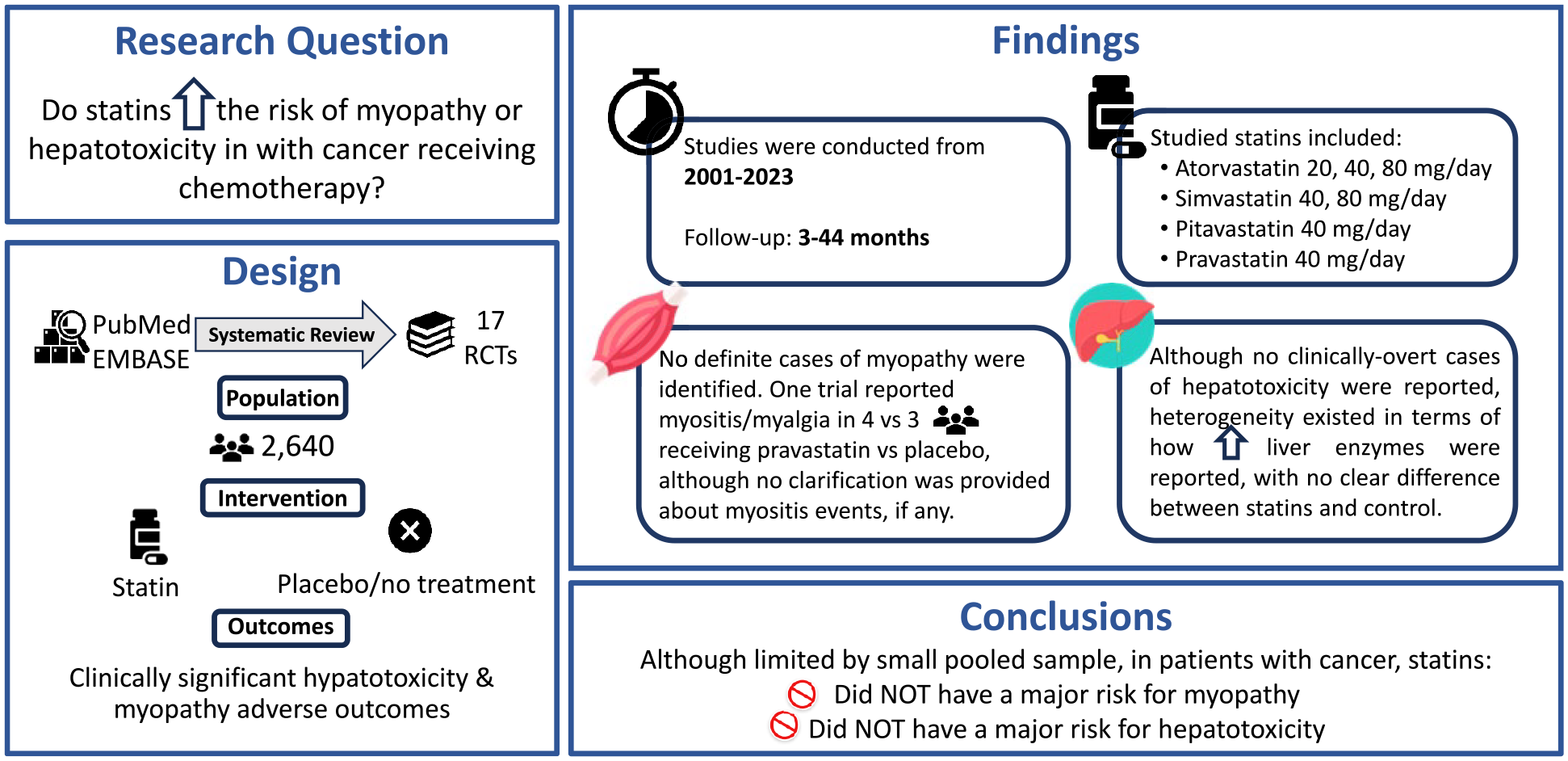
Graphical abstract of the study design and results.
Eventually, 16 RCTs inclusive of a total of 2640 patients were included. There were eight double-blind placebo-controlled trials, and the rest had no treatment (n = 7) or another active comparator (n = 1, rivaroxaban). One ongoing RCT13 was also identified (Supplemental Table 1). Details of the search strategy and a list of the trials are summarized in the for-peer-review online Supplemental Figure 2.
The average follow-up duration in the trials ranged between 1 and 44 months. The studied statins included atorvastatin 20, 40, and 80 mg, simvastatin 40 and 80 mg, pitavastatin 40 mg, and pravastatin 40 mg. No study used rosuvastatin. No definite cases of myopathy were identified. One trial reported myositis/myalgia in four versus three patients receiving pravastatin versus placebo, although no clarification was provided about myositis events, if any. 8 Though no clinically overt cases of hepatotoxicity were reported, heterogeneity existed in terms of how elevated liver enzymes were reported, without a clear difference between statins and control.
This systematic review of the literature, which included 16 RCTs of statin therapy compared with placebo or no treatment spanning from 2001 to 2023 in a diverse population of patients with cancer, including those with gastrointestinal and hepatocellular cancers, found low rates of either myopathy or hepatotoxicity.
The main limitations of this systematic review study are the small number of patients, short average follow-up period, and insufficient details about event definitions in most of the individual trials. However, review of the results across these trials demonstrates very few events—in many trials there were no events of either transaminitis or myositis, with rates consistent with those reported for trials of statins to prevent cardiovascular events in the general population. Though the data from this study may provide relevant insights, decision-making for individual patients in routine practice should also take into account other clinical conditions that may impact safety or effectiveness.
In conclusion, this systematic review of the literature indicates that use of statins in patients with cancer is associated with a very low number of severe transaminitis or clinically relevant myopathy events. Clinicians should be vigilant for these adverse events, but the existing data do not show a safety signal for a uniquely increased risk for these rare complications in patients with cancer.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241246471 – Supplemental material for Risk of myopathy and hepatotoxicity in patients with cancer receiving statin therapy: Systematic review of randomized controlled trials
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241246471 for Risk of myopathy and hepatotoxicity in patients with cancer receiving statin therapy: Systematic review of randomized controlled trials by Behnood Bikdeli, Farbod Zahedi Tajrishi and Jean M Connors in Vascular Medicine
Footnotes
Acknowledgements
We would like to thank Dr Paul M Ridker and Dr Robert J Glynn for reviewing a prior version of the manuscript and for their helpful feedback.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Outside the submitted work, Dr. Bikdeli is supported by the Scott Schoen and Nancy Adams IGNITE Award and is supported by the Mary Ann Tynan Research Scientist award from the Mary Horrigan Connors Center for Women’s Health and Gender Biology at Brigham and Women’s Hospital, and the Heart and Vascular Center Junior Faculty Award from Brigham and Women’s Hospital. Dr. Bikdeli is a member of the Medical Advisory Board for the North American Thrombosis Forum and serves in the Data Safety and Monitory Board of the NAIL-IT trial funded by the National Heart, Lung, and Blood Institute, and Translational Sciences. Dr Connors reports consulting fees or scientific advisory board honoraria from Abbott Laboratories, Anthos Pharmaceuticals, Bristol Myers Squibb, Pfizer, Roche, Sanofi SA, Werfen, and research funding to the institution from CSL Behring. Dr Zahedi Tajrishi has no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
