Abstract
Background:
The placement of inferior vena cava (IVC) filters often emerges as an alternative preventative measure against pulmonary embolism in patients with upper gastrointestinal (GI) bleeding and isolated distal deep vein thrombosis (DVT). We aimed to investigate the association of IVC filter placement and the incidence of venous thromboembolism (VTE) recurrence in this patient population.
Methods:
We performed a retrospective cohort study including 450 patients with upper GI bleeding and isolated distal DVT. Propensity score matching using logistic regression was conducted to mitigate potential selection bias. Logistic regression models and additional sensitivity analyses were conducted to estimate the association between IVC filter implantation and VTE recurrence. Interaction and stratified analyses were also performed according to the background covariates.
Results:
Patients who underwent IVC filter placement were significantly younger than patients in the surveillance group (55.8 ± 9.0 vs 58.4 ± 11.2 years, p = 0.034). Patients in the IVC filter group demonstrated a higher distal thrombus burden. The VTE recurrence composite was significantly higher in patients who underwent IVC filter placement (44.1% [45/102] vs 25% [87/348], p < 0.001). Unmatched crude logistic regression analysis identified a significant association between IVC filter placement and VTE recurrence composite (OR = 2.37; 95% CI, 1.50–3.75). Sensitivity analyses yielded congruent outcomes.
Conclusion:
This study revealed an increased risk of VTE recurrence among patients receiving IVC filter placement, suggesting that IVC filter placement may not be suitable as a primary treatment for patients with upper GI bleeding and isolated distal DVT.
Keywords
See Commentary by Houghton and Carman. Caution: Inferior vena cava filters in distal deep vein thrombosis. Vasc Med 2024;29:435–437.
Background
The standard management of proximal deep vein thrombosis (DVT) with therapeutic anticoagulation to prevent thrombus propagation and pulmonary embolism (PE) has been established. 1 However, the strategies for handling isolated distal DVT, colloquially referred to as calf vein thrombosis, are still subjects of ongoing debates.2 –4 Isolated distal DVT is defined by the presence of thrombosis limited to either the axial (comprising posterior tibial, peroneal calf, and anterior tibial veins) or muscular (soleal and gastrocnemius veins) vessels within the calf. Despite the relatively low risk of embolization in the absence of anticoagulant therapy, the propagation of isolated distal DVT augments the threat of PE and postthrombotic syndrome. The guidelines recommend serial imaging of the deep veins for 2 weeks over anticoagulation if patients presented without severe symptoms or risk factors for extension. 1 Otherwise, anticoagulation is recommended over serial imaging of the deep veins.
Upper gastrointestinal (GI) bleeding, defined as hemorrhage emanating from a source between the esophagus and the ligament of Treitz, is an absolute contraindication to anticoagulant therapy. Whereas isolated distal DVTs are commonly diagnosed in patients presenting with upper GI bleeding. Complicated factors that might tip the scale in favor of anticoagulation, like positive D-dimer, active cancer, and history of venous thromboembolism (VTE), are also commonly observed in these patients.5,6 As a result, the placement of inferior vena cava (IVC) filters often emerges as an alternative preventative measure against PE.
Evidence from studies regarding the efficacy of IVC filters in patients with isolated distal DVT are inconclusive in the general population.7–9 Similar study on the management of isolated distal DVT in patients with upper GI bleeding is lacking. Given this, we performed this study to investigate the association of IVC filter placement and the incidence of VTE recurrence in patients with upper GI bleeding and concomitant isolated distal DVT.
Methods
Study design and patients
This single-center retrospective cohort study was app-roved by the institutional review board of the Second Affiliated Hospital of Chongqing Medical University. In consideration of the retrospective observational nature of the study, the necessity for informed consent was waived. This study was performed in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 10
We identified consecutive patients presenting with upper GI bleeding between January 1, 2013 and December 31, 2022 by scrutinizing discharge records. All patients who experienced upper GI bleeding and were subsequently diagnosed with isolated distal DVT, impacting either the axial or muscular veins or both, through whole-leg compression ultrasonography, were included in this study (Figure 1). Exclusion criteria included: (1) the lack of at least one duplex ultrasound follow-up within 3 months of the initial examination; (2) previous or concurrent more proximal DVTs; and (3) previous or concurrent PE. Patients were bifurcated into two groups based on their treatment methodology: those who underwent IVC filter placement and those who were subject to imaging surveillance. Treatment decisions were made by a multidisciplinary team of gastroenterologists, emergency physicians, and vascular surgeons, all of whom remained independent of the study. All lower-extremity duplex ultrasound scans were performed by specialized vascular ultrasound technologists who were independent to this study. Whole-leg duplex ultrasound was performed in each patient as a combination of color Doppler imaging, compression ultrasound, and duplex sonography from the distal external iliac vein to the calf veins by using a EPIQ 7C ultrasound system (Philips; Amsterdam, Netherlands).
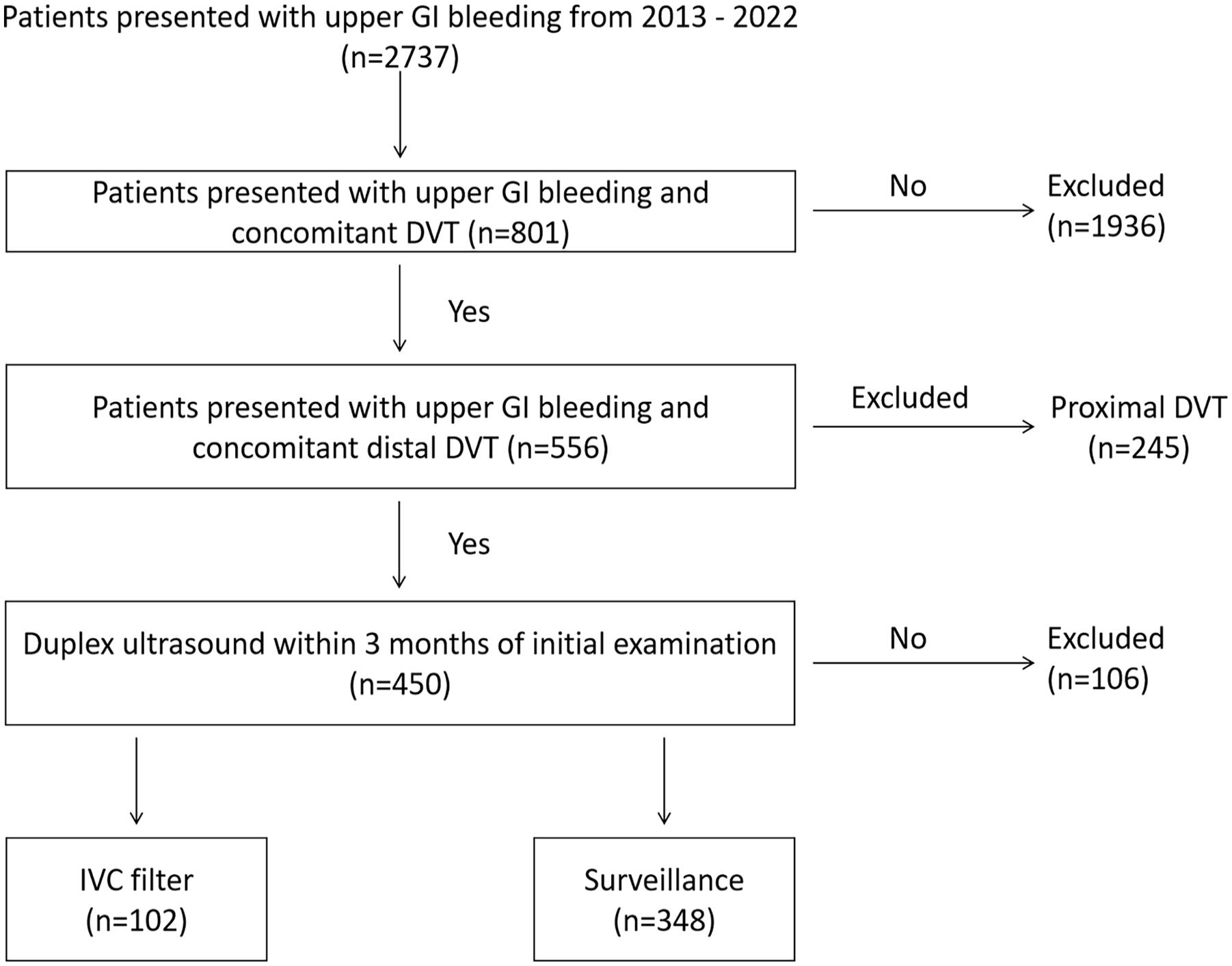
Flowchart of patient enrollment.
Data collection
Demographic characteristics, medical history, comorbidities, laboratory results, radiology images, and reports were reviewed and collected from the electronic medical record. The diagnosis of DVT was determined by the duplex ultrasound performed at both the initial admission and during follow up. An ultrasound scan was routinely performed within 24 hours of initial admission, and blood samples were collected within the first 2 hours postadmission. Laboratory tests were performed at the clinical laboratory of the Second Affiliated Hospital of Chongqing Medical University. To ensure the accuracy of ultrasound image interpretations, two independent researchers (YH and HL) reviewed these images, thereby validating the conclusions drawn from the reports.
Covariates
The selection of covariates was informed by relevant literature and includes the following: age, sex, height, weight, hypertension, diabetes mellitus, cirrhosis, history of previous upper GI bleeding, coagulopathy, use of NSAIDs (nonsteroidal antiinflammatory drugs), alcohol consumption, recent surgery or trauma, immobility, hormonal therapy, pregnancy, active cancer, personal and family history of VTE, known thrombophilia, tobacco history, hemoglobin, platelets, international normalized ratio (INR), prothrombin time, activated partial thromboplastin time, D-dimer, treatment for upper GI bleeding, limb involvement, and location of distal DVT.
Upper GI bleeding was defined as any acute episode of hematemesis or melena in the 24 hours prior to admission to the hospital. Isolated distal DVT was determined by the presence of noncompressible thrombus limited to infrapopliteal veins of the calf on ultrasound images. Hypertension was defined as a systolic pressure exceeding 140 mmHg or a diastolic pressure over 90 mmHg, recorded on at least two occasions postadmission, or through the usage of antihypertensive medications. Coagulopathy was defined as platelet count ⩽ 50 × 109/L, and/or INR ⩾ 1.5. The determination of cirrhosis relied on ultrasound or magnetic resonance imaging reports. Histories of chronic diseases and medications were collected from the medical records.
Outcomes
The primary outcome was the VTE recurrence within 3 months, encompassing both thrombus propagation and the development of new VTEs in regions not initially affected at the time of admission. Acute thrombosis was defined as the presence of an incompressible segment, the absence of color flow, or echogenic material in the lumen. Thrombus propagation was defined as extension of calf thrombus into a proximal vein, including popliteal, femoral, or iliac veins of the ipsilateral limb. Confirmation of thrombus propagation required ultrasonography showing continuous extension into a proximal vein not visible in the initial examination. The development of a new VTE included recurrent DVT involving the opposite lower-extremity and upper extremities or a new PE. Recurrent symptomatic PE was confirmed by computed tomography pulmonary angiography. The secondary outcome was all-cause mortality.
Follow up
In alignment with the guideline-recommended standard duration of anticoagulation treatment being 3 months, and IVC filters also are usually removed within 3 months if no thrombus propagation was found, 1 only follow-up data ranging from 0 to 3 months were collected. Inpatients routinely underwent serial ultrasound examinations once or twice a week. For outpatients, these serial ultrasound examinations were typically conducted biweekly or once weekly. Graduated compression stockings were routinely used for all inpatients and suggested for all outpatients. Intermittent pneumatic compression was used for immobile inpatients in intensive care units. Anticoagulation (5000 IU dalteparin or 4100 IU nadroparin subcutaneous injection once daily or rivaroxaban 10 mg orally once daily) was used for patients with: (1) stable hemoglobin levels over 9 g/dL for more than one week; and (2) thrombus propagation to the femoropopliteal or iliac vein. Furthermore, during any instances of readmission, medical history collection and laboratory tests were performed.
Statistical analysis
Multiple imputations were used for missing data on history of cirrhosis and other clinical risk factors. In total, five data sets were imputed, and results were pooled according to Rubin’s rules. 11 Continuous variables were presented as means ± SDs or median with first and third quartiles and categorical variables as frequencies and percentages. Distributions of demographics and clinical characteristics were compared between two groups using the Mann–Whitney test for continuous variables and the chi-squared test for categorical variables.
To mitigate potential selection bias owing to the retrospective nature of this study, propensity score matching (PSM; 1:1 match, caliper 0.2) using logistic regression was conducted. The PSM were performed by the nearest neighbor matching algorithm without replacement and based on background covariates: age, sex, height, weight, hypertension, diabetes mellitus, cirrhosis, history of previous upper GI bleeding, coagulopathy, NSAID use, alcohol intake, recent surgery and trauma, immobility, hormonal therapy, pregnancy, active cancer, personal and family history of VTE, known thrombophilia, tobacco history, hemoglobin, platelets, INR, prothrombin time, activated partial thromboplastin time, D-dimer, treatment of upper GI bleeding, limb involvement, and location of distal DVT.
The odds ratios associated with the effect of IVC filter implantation on VTE recurrence was estimated using logistic regression models. In addition to a crude logistic model, multiple additional sensitivity analyses were also undertaken, including multivariable adjusted, propensity score adjusted, propensity score matched inverse probability of treatment weighting (IPTW), standardized mortality ratio weighting (SMRW), pairwise algorithmic (PA), and overlap weight (OW) models. Interaction and stratified analyses were also performed according to the background covariates. The continuous variables were categorized using median as category boundaries in the stratified analysis.
All analyses were performed with the statistical software packages R (www.R-project.org; The R Foundation) and EmpowerStats (www.empowerstats.com; X&Y Solutions, Inc.). A two-tailed p < 0.05 was considered to reflect statistical significance.
Results
Demographic characteristics
A total of 450 patients were included for statistical analysis after exclusions, in which 102 patients received IVC filter placement and 348 patients received imaging surveillance. Demographics and clinical characteristics are summarized in Table 1. Patients who underwent IVC filter placement were significantly younger than patients in the surveillance group (55.8 ± 9.0 vs 58.4 ± 11.2 years, p = 0.034). Moreover, patients in the IVC filter group were more likely to have undergone surgery for upper GI bleeding treatment (15.7% [16/102] vs 4.9% [17/348], p < 0.001). Medical history and clinical risk factors were evenly distributed between the two groups.
Baseline demographics and clinical characteristics.
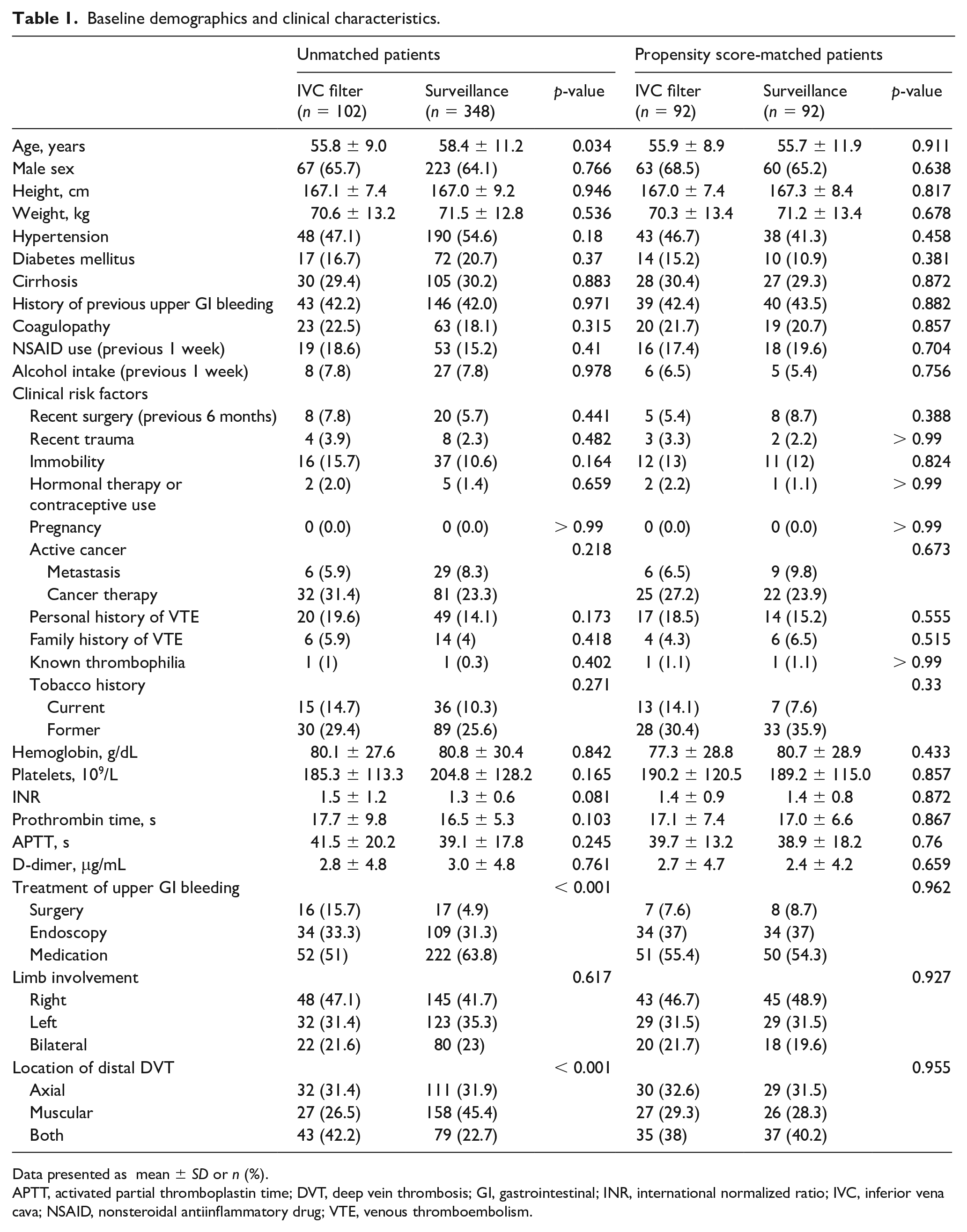
Data presented as mean ± SD or n (%).
APTT, activated partial thromboplastin time; DVT, deep vein thrombosis; GI, gastrointestinal; INR, international normalized ratio; IVC, inferior vena cava; NSAID, nonsteroidal antiinflammatory drug; VTE, venous thromboembolism.
Patients in the IVC filter group demonstrated a higher distal thrombus burden (Table 1, p < 0.001). The incidence of thrombus affecting only the axial veins was comparable between the two groups (31.4% [32/102] vs 31.9% [111/348]). The surveillance group had more patients with thrombus involving only muscular veins (26.5% [27/102] vs 45.4% [158/348]). Notably, thrombus involving both axial and muscular veins were more frequent in the IVC filter group (42.2% [43/102] vs 22.7% [79/348]). The baseline characteristics of the two groups demonstrated a well-balanced distribution after PSM (Table 1).
Outcomes
The outcomes of included patients are presented in Table 2. The VTE recurrence composite was significantly higher in patients who underwent IVC filter placement compared with those managed by ultrasound surveillance (44.1% [45/102] vs 25% [87/348], p < 0.001). Similarly, the rates of DVT propagation were also elevated in the IVC filter placement group (36.3% [37/102] vs 17.8% [62/348], p < 0.001). However, incidences of nonpropagation VTE recurrence were comparable between the IVC filter placement and surveillance groups (7.8% [8/102] vs 7.2% [25/348], p = 0.802). The frequencies of intermittent pneumatic compression (7.8% [8/102] vs 4.9% [17/348], p = 0.472), anticoagulation use (5.9% [6/102] vs 2.6% [9/348], p = 0.349) and death (4.9% [5/102] vs 4.9% [17/348], p = 1) were likewise similar in both groups. After using PSM to adjust the background covariates, the VTE recurrence composite was still significantly higher in the IVC filter placement group (42.4% [39/92] vs 19.6% [18/92], p < 0.001). Correspondingly, the DVT propagation rates in the IVC filter placement group were significantly higher (34.8% [32/92] vs 13.0% [12/92], p = 0.005).
Outcomes.
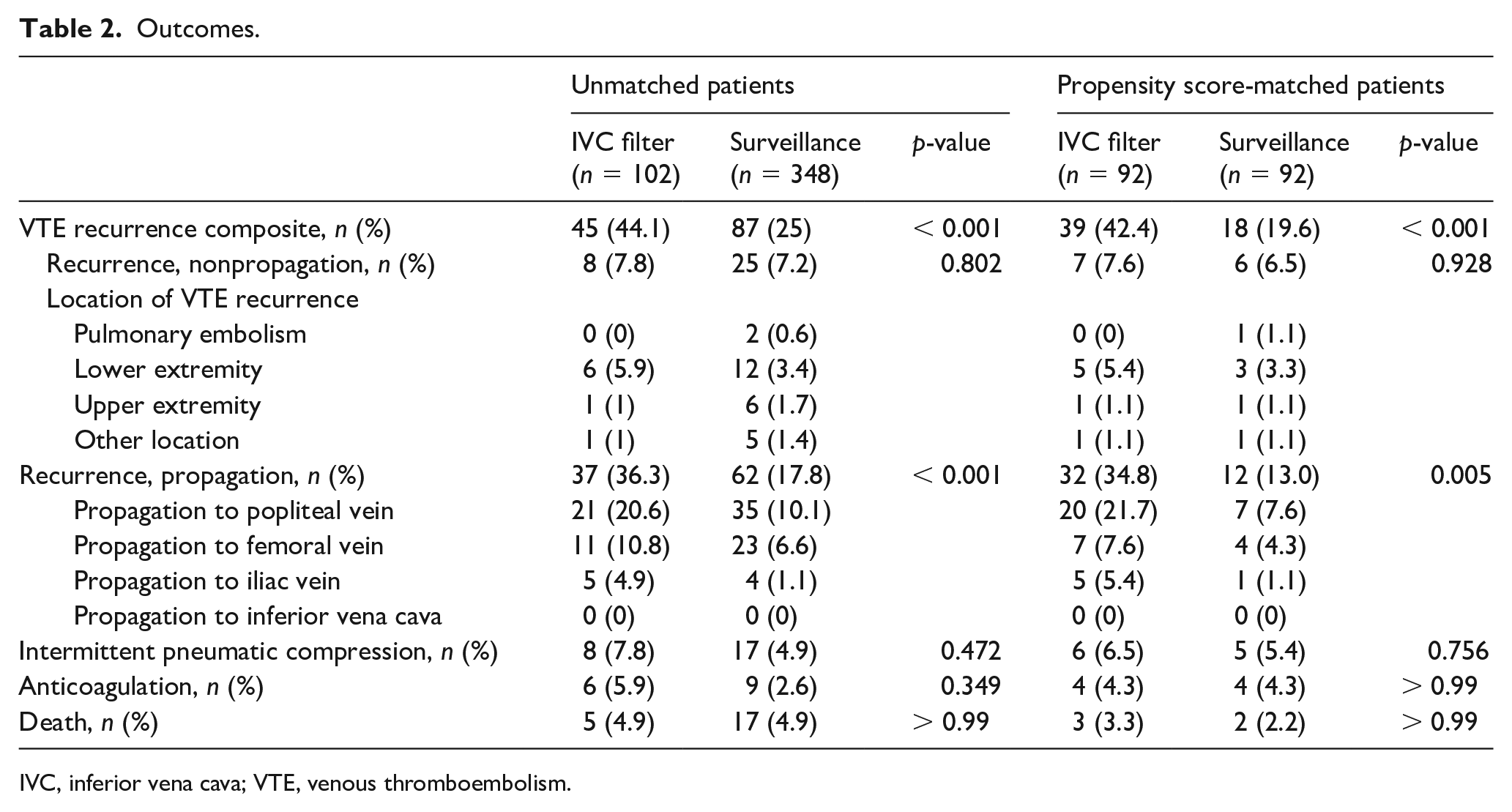
IVC, inferior vena cava; VTE, venous thromboembolism.
Relationship between IVC filter placement and VTE recurrence
The relationship between IVC filter placement and VTE recurrence composite is presented in Table 3. Unmatched crude logistic regression analysis identified a significant association between IVC filter placement and VTE recurrence composite (OR = 2.37; 95% CI, 1.50–3.75).
Association between inferior vena cava filter placement and venous thromboembolism recurrence.
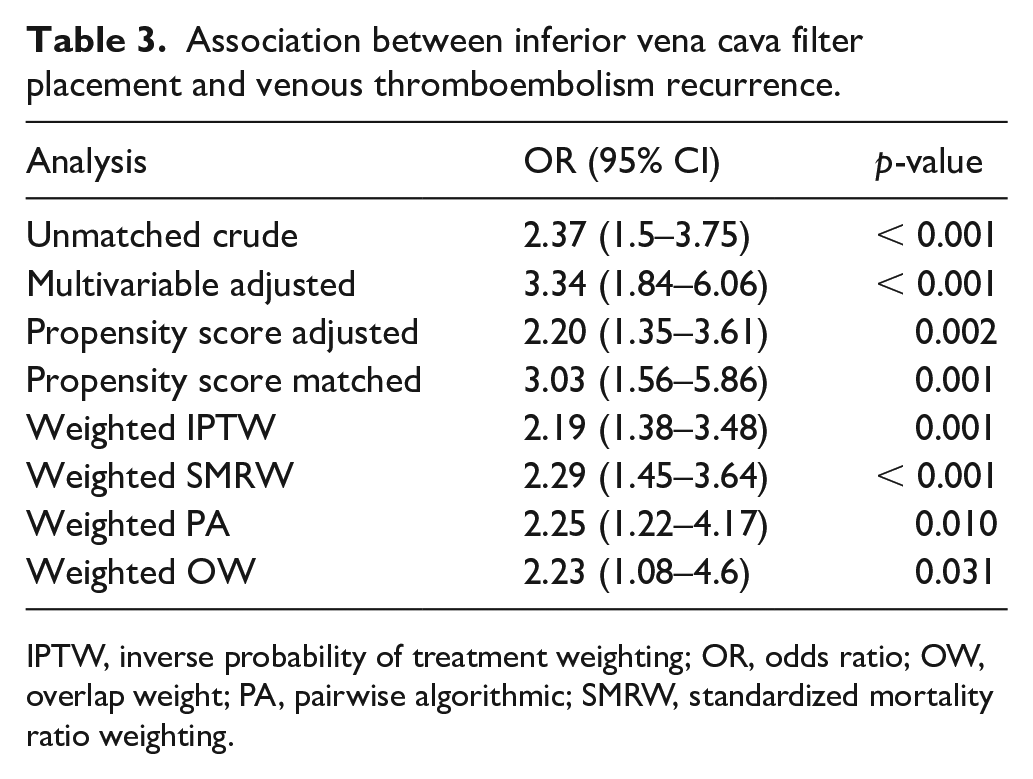
IPTW, inverse probability of treatment weighting; OR, odds ratio; OW, overlap weight; PA, pairwise algorithmic; SMRW, standardized mortality ratio weighting.
After adjustment for all covariates, multivariable logistic regression analysis yielded a congruent outcome (OR = 3.34; 95% CI, 1.84–6.06). Further adjustments made for the propensity score maintained the stability of this relationship (OR = 2.20; 95% CI, 1.35–3.61). The relationship was also revealed in the matched cohort (OR = 3.03; 95% CI, 1.56–5.86). Moreover, we weighted IPTW in univariable logistic regression analysis, and the OR remained consistent (OR = 2.19; 95% CI, 1.38–3.48). Similarly, the ORs of the weighted SMRW model, weighted PA model, and weighted OW model were also stable (Table 3).
The results of the stratified and interaction analyses of the association between IVC filter placement and VTE recurrence composite are presented in Table 4. The association between IVC filter placement and VTE recurrence composite in the stratified analysis was consistent with that in the logistic regression analyses. This relationship was stable across all subgroup analyses. Interaction analysis did not reveal any interactive role in the association between IVC filter placement and VTE recurrence.
Association between IVC filter placement and VTE recurrence in subgroups.
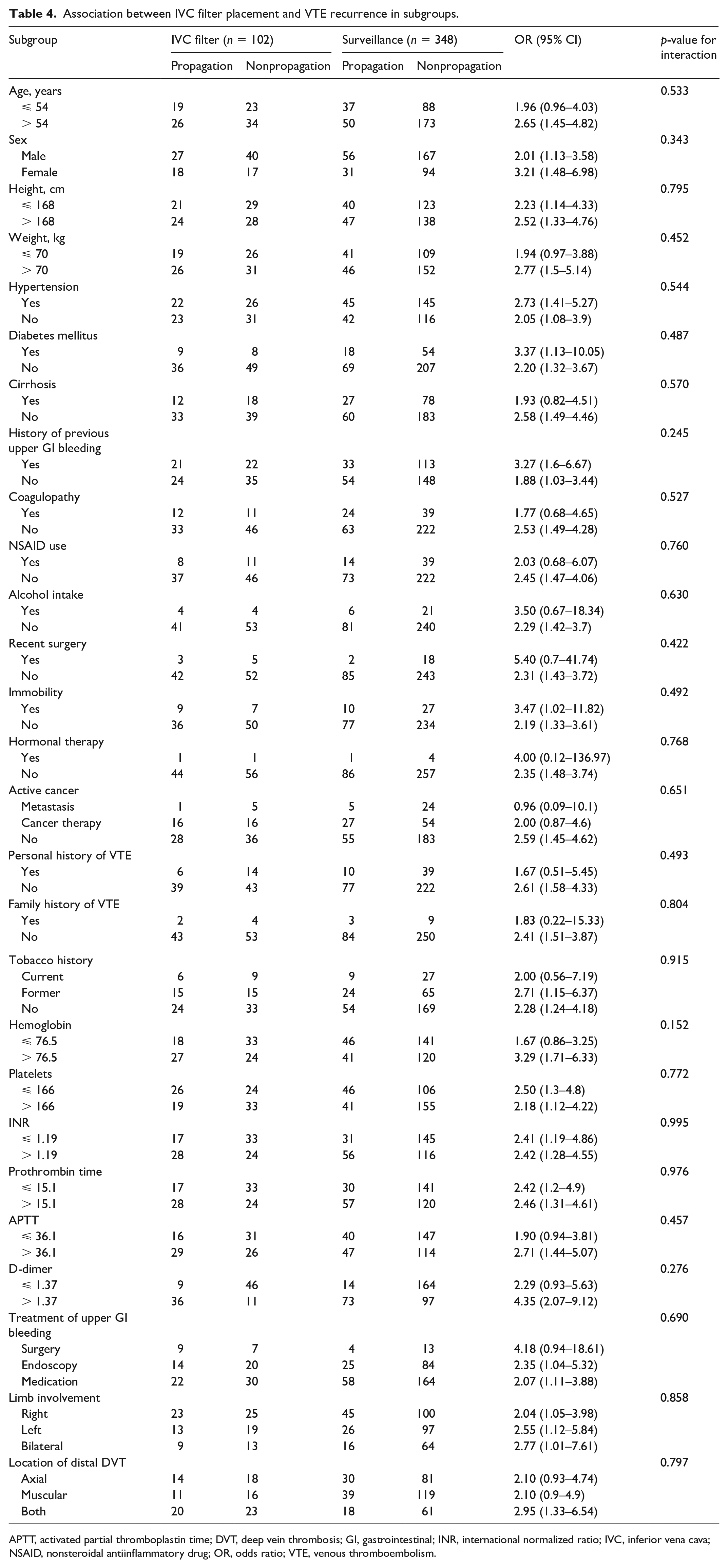
APTT, activated partial thromboplastin time; DVT, deep vein thrombosis; GI, gastrointestinal; INR, international normalized ratio; IVC, inferior vena cava; NSAID, nonsteroidal antiinflammatory drug; OR, odds ratio; VTE, venous thromboembolism.
Discussion
In this retrospective cohort study, we observed a significant association between IVC filter placement and VTE recurrence among patients presenting with upper GI bleeding and concomitant isolated distal DVT. Patients receiving IVC filter placement demonstrated a 2.37-fold higher risk of VTE recurrence as opposed to those managed via ultrasound surveillance. Both adjusted and weighted logistic regression analyses further confirmed the relationship between IVC filter placement and increased risk of VTE recurrence composite. Despite a higher prevalence of surgery to address upper GI bleeding and a greater distal thrombus burden in the IVC filter group, the relationship remained significant after adjustment for these confounding factors. The observed association remained stable across all subgroup analyses. Furthermore, we found no interactive role in the association between IVC filter placement and VTE recurrence composite, suggesting that IVC filter placement is associated with the increased risk of VTE recurrence composite independent of classical VTE risk factors.
The overall incidences of both the thrombus propagation and nonpropagation recurrence are higher in the present study than previously reported results.12–14 This might be attributed to the characteristics of this specific study population, as patients with upper GI bleeding often possess additional VTE risk factors, including immobility, a history of VTE, and a hypercoagulable state induced by cirrhosis or active cancer. Additionally, the rate of major bleeding observed in our study notably exceeds previously reported results, which may reflect the inherent heterogeneity of the study population.12–14
Patients with upper GI bleeding are at an increased risk for DVT, primarily due to the prevalence of multiple DVT clinical risk factors. Cirrhosis, a leading cause of upper GI bleeding, significantly elevates the risk of VTE events during hospitalization when compared with the general population.15–17 Additional comorbidities such as active cancer and immobility also amplify the VTE risk in patients with upper GI bleeding. The current guideline suggests that patients with acute isolated distal DVT and severe symptoms or risk factors for extension receive anticoagulation rather than serial imaging. 1 However, in the context of active upper GI bleeding, anticoagulation is contraindicated. Consequently, IVC filter placement is often the chosen intervention. Under the circumstances, the management decisions for patients with upper GI bleeding in whom isolated distal DVT occurs are determined on an empirical individual basis due to the lack of clinical data from this patient population. Our study findings suggest that the use of IVC filter placement as a primary, standalone treatment for isolated distal DVT in patients with upper GI bleeding may not be the most suitable approach.
The management strategy of VTE differs according to the patient population. In the general population with VTE, an 8-year follow-up randomized study concluded that systematic use of IVC filters is not recommended. 18 The study found that although IVC filters mitigated the risk of PE, they concurrently elevated the risk of DVT without improving survival rates. Another study in patients diagnosed with isolated distal DVT after intracranial hemorrhage also generated a similar conclusion that IVC filters are not appropriate as a primary treatment in this population. 19 However, a recent study involving patients with cancer with DVT and bleeding risk factors suggested that IVC filter placement increased the rate of PE-free survival. 20 Our study indicates that IVC filter placement, in patients with upper GI bleeding and isolated distal DVT, correlates with an increased risk of VTE recurrence and should not be utilized as a routine treatment strategy.
Despite the lack of evidence guiding the management decisions for patients with upper GI bleeding and isolated distal DVT, several studies have investigated the impact of anticoagulation in patients with upper GI bleeding.21,22 These studies consistently found no correlation between anticoagulation and increased mortality, irrespective of whether upper GI bleeding was variceal or nonvariceal in origin. Patients under anticoagulation therapy were more likely to present with severe bleeding, but such episodes can be effectively controlled by drugs, endoscopic treatment, or hemostatic support. The guidelines of the Japan Gastroenterological Endoscopy Society advise the reinstatement of antithrombotic therapy as soon as endoscopic hemostasis is confirmed. 23 Despite the lower rate of DVT propagation in the surveillance group observed in our study, approximately 20% of distal thrombus still extended to more proximal segments. The long-term risks of DVT propagation and postthrombotic syndrome, particularly in high-risk populations, remain of concern. To address these concerns, early anticoagulation after endoscopic hemostasis may be an advisable choice.
Study limitations
This study has several limitations. First, due to the observational design of this study, it is not possible to establish a causal relationship between IVC filter placement and VTE recurrence. However, the association between IVC filter placement and VTE recurrence remains significant even after adjusting for a multitude of covariates. Second, the follow-up data were collected only for a period ranging from 0 to 3 months. Patients in the surveillance group typically ended the imaging surveillance if no VTE recurrence was demonstrated by two to three once-weekly ultrasound examinations. Consequently, any VTE events occurring after ending the imaging surveillance may have been overlooked. Meanwhile, patients in the IVC filter group generally continued follow-up until filter removal, leading to a potentially longer follow-up period than that of the surveillance group. The potential difference of follow-up duration may lead to a discrepancy in the VTE recurrence between the two groups. Third, the potential for recall bias cannot be ruled out due to the self-reported medical history, which might impact the accuracy of disease distribution within the study population. Last, the recurrence of VTE in a long-term follow-up cannot be estimated based on the results of this study.
Conclusion
In conclusion, the optimal management of isolated distal DVT remains uncertain for patients who have contraindications to anticoagulation therapy and risk factors for VTE. Traditionally, the decision for IVC filter placement has been guided by individual clinician experience. However, this study has found that patients selected for IVC filter placement revealed an increased risk of VTE recurrence, suggesting that IVC filter placement may not be suitable as a primary treatment for patients with upper GI bleeding and isolated distal DVT. Further prospective interventional studies with long-term follow-up are needed to elucidate the potential causal relationship between IVC filter placement and VTE recurrence.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
