Abstract
Lymphedema has traditionally been underappreciated by the healthcare community. Understanding of the underlying pathophysiology and treatments beyond compression have been limited until recently. Increased investigation has demonstrated the key role of inflammation and resultant fibrosis and adipose deposition leading to the clinical sequelae and associated reduction in quality of life with lymphedema. New imaging techniques including magnetic resonance imaging (MRI), indocyanine green lymphography, and high-frequency ultrasound offer improved resolution and understanding of lymphatic anatomy and flow. Nonsurgical therapy with compression, exercise, and weight loss remains the mainstay of therapy, but growing surgical options show promise. Physiologic procedures (lymphovenous anastomosis and vascularized lymph node transfers) improve lymphatic flow in the diseased limb and may reduce edema and the burden of compression. Debulking, primarily with liposuction to remove the adipose deposition that has accumulated, results in a dramatic decrease in limb girth in appropriately selected patients. Though early, there are also exciting developments of potential therapeutic targets tackling the underlying drivers of the disease. Multidisciplinary teams have developed to offer the full breadth of evaluation and current management, but the development of a greater understanding and availability of therapies is needed to ensure patients with lymphedema have greater opportunity for optimal care.
Introduction
Lymphedema is the disease state in which excess lymph fluid accumulates in tissues due to dysfunction of the lymphatic system. The resultant fluid accumulation initiates a specific pathobiology marked by increased inflammation, fibrosis, and deposition of adipose tissue, leading to a myriad of clinical symptoms and decreased quality of life.1–3 Additionally, there is an associated financial burden due to decreased productivity, substantial time required for maintenance therapy, and cost of compression garments.4,5 It has been traditionally under-recognized and underappreciated by the healthcare system, though there is growing interest as of late. 6 There are significant limitations and variation in guideline recommendations and lack of clear consensus on appropriate management. 7 Nonetheless, there are exciting new developments in the evaluation of lymphedema, including advancements in imaging techniques, surgical treatments, and therapies targeting the underlying mechanisms driving lymphedema. An outline for this review was developed and a search was performed in June through August 2023, utilizing PubMed, targeted for the specific topic areas related to lymphedema.
Epidemiology and classification
Lymphedema is generally categorized as primary, due to an intrinsic dysfunction of the lymphatic system, or as a secondary dysfunction from an acquired process, most commonly cancer-related treatment in Western countries. Though filarial infections are believed to be the most common etiology of lymphedema worldwide, the incidence is decreasing with the widespread administration of anthelmintic drugs. 8 Secondary lymphedema is much more common than primary lymphedema. An accurate determination of its prevalence is difficult, but it has been estimated that one in seven patients with cancer will develop some form of edema. Nearly 20% of those with advanced cancer will develop lymphedema.9,10 Estimates based on cancer survival rates and other etiologies of secondary lymphedema currently suggest that over a million individuals in the United States have secondary lymphedema. 11 Among patients with breast cancer, one in three who undergo axillary lymph node dissection and regional lymph node radiation will develop lymphedema. In patients who undergo axillary lymph node dissection, concomitant regional nodal irradiation more than doubles the incidence of breast cancer-related lymphedema (BCRL). 12 Although breast cancer-related lymphedema is the most common form of lymphedema in developed countries due to the high incidence of breast cancer, nodal dissections for the management of other malignancies also portend a high risk of lymphedema development, particularly in gynecologic malignancies and sarcoma. 10 Upwards of 90% of those who develop lymphedema following breast cancer treatment do so within 2 years of therapy, but for some it can occur several years later, often following an additional provoking event. 13
An additional frequent cause of secondary lymphedema is chronic venous disease. 14 In a study of patients presenting with lower-extremity lymphedema to a specialized clinic, phlebolymphedema was the most common etiology (41.8%). 15 Reported rates of secondary lymphedema are likely underestimated due to the high rate of venous disease in the general population. Other common noncancer-related secondary causes include obesity, recurrent cellulitis, and trauma. ‘Senile’ lymphedema, the development of lymphatic dysfunction at an advanced age, has received growing recognition in recent years. Though the pathophysiology is not fully understood, it is suspected to be related to aging of the lymphatic system and deterioration of lymphatic pump function. 16
Primary lymphedema is overall more common in female patients (3.5:1), though it more frequently occurs in boys during infancy and girls during adolescence.17,18 Primary lymphedema has traditionally been classified by three categories based on the age of onset, but such differentiation does not reflect the underlying pathogenesis, associated syndrome features, or guide appropriate therapeutic interventions. There are a variety of newer proposed classification systems, but none has been widely accepted yet. 19 Most patients with primary lymphedema will have continued disease progression; however, a subset stagnate or even regress. 18 The underlying mechanism of lymphatic dysfunction in primary lymphedema is rarely identified for a specific individual. Potential mechanisms include smooth muscle pump dysfunction, valvular incompetence, and hypoplasia of the lymphatic vessels. Over 30 genes and been implicated in primary lymphedema, but 70% of patients with primary lymphedema do not have an identifiable genetic defect. The detection rate varies based on timing of disease onset and severity.19,20 There is likely an overlap in primary and secondary lymphedema among patients who develop persistent edema following only a mild insult (orthopedic surgery, ankle sprain). Clinically, we often see such patients and suspect there was subclinical, mild intrinsic lymphatic dysfunction that only became clinically evident after a ‘second hit’. Additional classifications like ‘constitutional lymphatic weakness’ have been suggested to better define these presentations that do not clearly fit into the traditional primary or secondary lymphedema schema. 21
Pathophysiology
Lymphatics play an integral role in immune response and fat absorption and transport, but primary to lymphedema is its role in fluid hemostasis. Classic teaching has underrepresented the essential nature of the lymphatic system to maintain appropriate fluid hemostasis. Venules have little role in reabsorption and it is the lymphatics that are the primary avenue of reabsorption and transport of interstitial fluid. 22 It is estimated that one-sixth of total body fluid is contained within the interstitial space. 23 The amount of interstitial fluid present is driven by the balance of Starling forces – the hydrostatic pressure in the capillaries and oncotic pressure of the interstitial space. Based on experimental data, it is believed that, in a normal state, there is a low-level excess of filtration into the interstitial space that is not equivalently balanced by reabsorption via the venules. Lymphatic capillaries, with a discontinuous basal lamina, absorb fluid to precollectors which drain fluid to superficial and eventually deep lymphatic collecting vessels. In normal lymphatic vessels, there are smooth muscle cells and valves to maintain unidirectional flow toward the central circulation.24,25 Lymph is transported to lymph node beds where it is concentrated and surveilled for foreign antigens by circulating immune cells. Additionally, it is possible that a proportion of fluid is filtered to the venous system via lymphatico-venous anastomoses. 26 Lymph nodes express high levels of aquaporine-1, a water channel, at the surface of endothelial cells. 27 Water is reabsorbed and the lymph becomes more concentrated, as water is shunted to the venous system via these lymphatico-venous anastomoses (important in understanding the potential mechanism of a lymph node transfer below). 26 Fluid maintained in the lymphatic circulation travels to the central lymphatic system and ultimately back to the venous system via the thoracic ducts.
Edema results when there is an overload of filtrate surpassing the ability of the lymphatic system to maintain appropriate homeostasis to reabsorb the excess flux (e.g., nephrotic syndrome), the lymphatic system is not functioning appropriately to remove a normal level of filtrate (lymphedema), or a combination of both excess flux and insufficient lymphatic function (e.g., phlebolymphedema). Understanding the underlying pathophysiology of lymphedema is important in order to appreciate current and future treatment paradigms. 28 Whether the underlying etiology is primary or secondary, the resultant development of interstitial fluid overload and lymphatic congestion leads to a similar sequela. Disruption to the normal transport of interstitial fluid and macromolecular proteins results in a localized inflammatory response that ultimately leads to adipose deposition and fibrosis of both skin and lymph vasculature. 29 There appears to be a complex inflammatory response mediated by increased expression of cytokines and activation of immune cells. For example, CD4+ T cell levels are significantly increased in lymphedema, and depletion of CD4+ T cells increased lymphangiogenesis in a mouse tail model of lymphedema. 30 Th2 cell differentiation leads to the production of cytokines like interleukin (IL)-13 and are critical in the development of fibrosis and lymphatic dysfunction. 31 Numerous inflammatory cytokines are also elevated in both the affected lymphedematous tissue and in the circulation, including tumor necrosis factor (TNF) alpha, IL-6, IL-8, and monocyte chemoattractant protein-1 (MCP-1).32,33
In addition to a significant inflammatory response, lymphatic fluid stasis leads to localized fibrosis and deposition of adipose tissue. It is this process that leads to the significant increase in limb girth with relatively mild amounts of edema and has resulted in the misrepresentation of lymphedema presenting as ‘nonpitting’ edema. Some degree of pitting is present in almost all patients, but pitting may be mild relative to the severity of increased limb girth depending on the amount of adipose deposition that has occurred. 29 This adipose deposition occurs independently of caloric intake or weight, but may be exacerbated in patients with obesity.34,35 The underlying mechanism of this process is not fully elucidated, but likely is related to the localized inflammatory state, particularly Th2 cytokines and macrophages.30,31,36 Obesity is a known risk factor for the development of lymphedema and has deleterious effects on lymphatic function. Localized adipose tissue deposition then further exacerbates lymphatic dysfunction in the effected limb. 37
There is also a growing understanding of the underlying genetic defects for a subset of patients with primary lymphedema. This has led to a greater understanding of the mechanisms that result in lymphatic dysfunction and as potential targets for future therapeutics. For example, patients with lymphedema-distichiasis syndrome have a defect in the FOXC2 mutation which disrupts regulation of lymphatic endothelial cell differentiation, smooth muscle cell layer formation, and lymphatic valve morphology. 38
Determining the genetic defect in patients with primary lymphedema usually has little clinical implication on lymphedema management currently. However, identification of a specific defect can assist patients with selective fertilization for those of reproductive age or identify risk for associated nonlymphedema-related processes (e.g., increased risk of myelodysplastic syndromes with a GATA2 mutation or congenital heart defects with FOXC2 mutation).
Diagnosis and imaging
A thorough history and exam remain invaluable to the evaluation of patients with limb edema and suspected lymphedema. Identifying risk factors for the development of lymphedema and performing a physical exam to differentiate features of lymphedema from other common etiologies of edema, such as chronic venous insufficiency, are essential. A systemic evaluation is also necessary, particularly for patients with bilateral lower-extremity edema, to exclude heart failure, disease processes leading to hypoalbuminemia, or concomitant medications that can lead to edema like taxanes and dihydropyridine calcium channel blockers. There is a broad differential diagnosis for limb edema with specific exam features for lymphedema; this is beyond the scope of the present review, but a few characteristic findings are shown in Figure 1.23,39
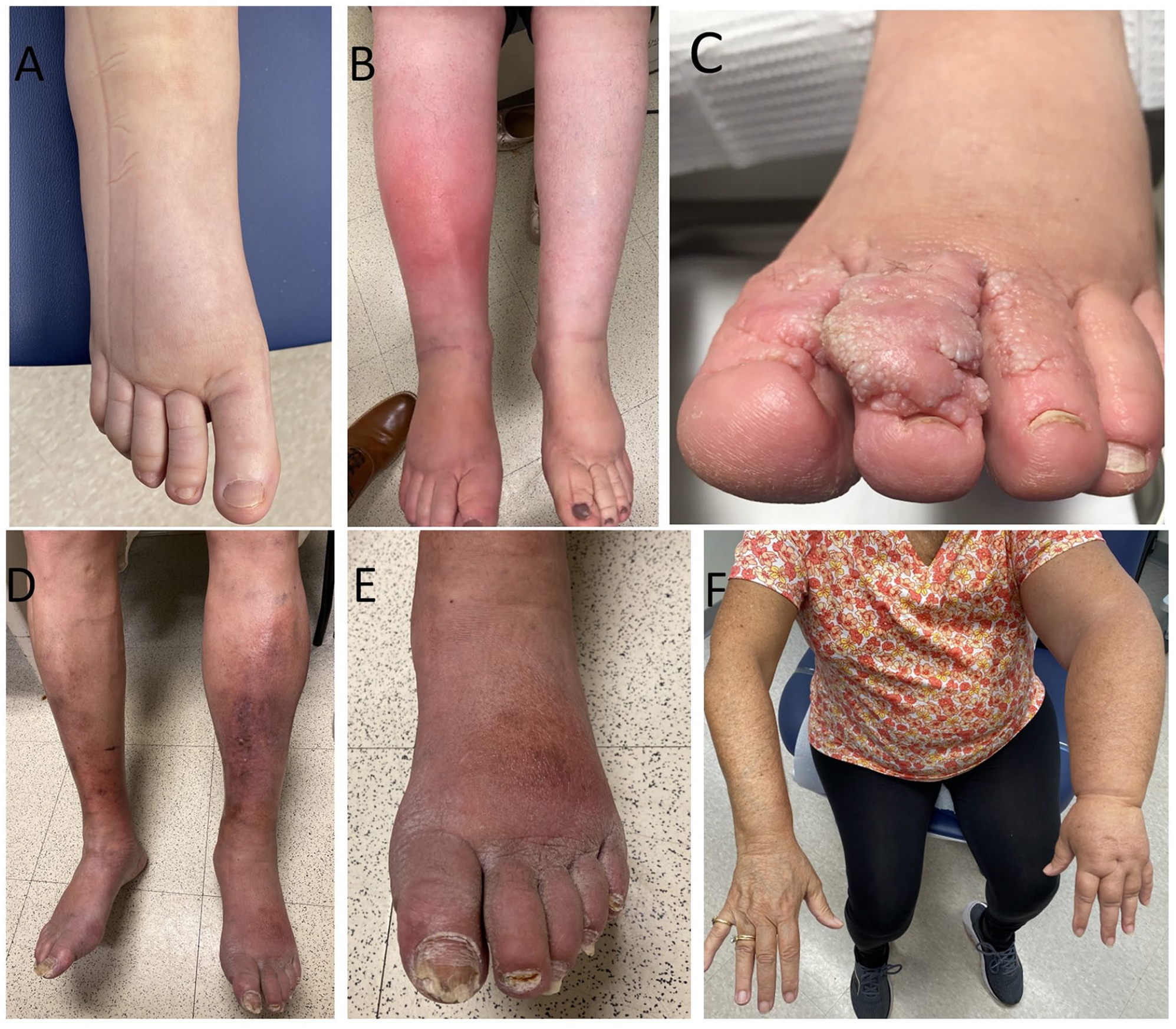
Thorough clinical exam is essential in the evaluation of patients with limb edema as there are characteristics consistent with lymphedema.
There are proposed stages for lymphedema, though we do not find these to be particularly helpful in guiding management and they have limited clinical implications, as also noted by the authors of the International Society of Lymphology consensus statement. 40 Essential to the physical exam is an appreciation of the degree of edema present. In most patients, there is both adipose tissue deposition, which can occur just months after onset of symptoms, and concomitant edema. A patient’s degree of residual pitting determines the adequacy of their current level of compression along with the degree of adipose deposition present in the affected limb (e.g., significant increased girth with minimal pitting suggests a large component of adipose tissue). Clinicians’ skill and experience are variable; prior studies have demonstrated that a physical exam alone may be insufficient to diagnose lymphedema and diagnostic imaging is often appropriate. 41
There is no currently available imaging modality that is all-encompassing for the evaluation of lymphedema, but there are a variety of techniques that are complementary to obtain a comprehensive evaluation (Table 1). Though imaging is not required in many patients with lymphedema, it can be vital in patients with undifferentiated edema or in the preoperative setting. In discussion with patients, we generally offer imaging with a new diagnosis of suspected primary lymphedema and for patients without risk factors for the development of lymphedema who lack the classic exam features of lymphedema. Much of the difficulty in lymphatic imaging is due to the small size of lymphatic vessels, with diameters of 0.4–0.8 mm, and the relatively slow flow of lymph through these vessels in comparison to blood flow through veins and arteries. The thoracic duct is the largest lymphatic vessel and only measures around 2–4 mm in diameter. Direct cannulation of peripheral lymphatic vessels has been attempted with traditional lymphography, but more recently developed techniques rely on use of a tracer or contrast agent taken up by the lymphatic system through intradermal injection.
Lymphatic imaging options.

ICG, indocyanine green; LVA, lymphovenous anastomosis; MRI, magnetic resonance imaging; MRL, magnetic resonance lymphangiography.
Nuclear lymphoscintigraphy
Nuclear lymphoscintigraphy has been considered the gold standard for lymphatic imaging evaluation (Figure 2A). Though there are variations in protocols and the specific tracers utilized, lymphoscintigraphy offers a noninvasive evaluation of lymphatic function. Nuclear tracers bound to protein are injected intradermally and taken up by the lymphatic system. A series of images are acquired over 2–4 hours to evaluate transit within the lymphatics; gross visualization of lymphatic channels and uptake in lymph nodes can be seen with SPECT imaging. Increasingly, concomitant CT is performed to assess the location of lymph node uptake more accurately. Delayed transit or decreased uptake in the affected limb is consistent with lymphatic dysfunction. Dermal backflow, the accumulation of tracer underneath the skin and in the soft tissues due to congestion and lymphatic reflux, is believed to be diagnostic for lymphedema. The diagnostic accuracy is high – with a reported sensitivity of 92–96% and specificity of 93–100%; however, existing studies are biased by various study populations, clinical assessments, and diagnostic criteria at high-volume centers.42,43
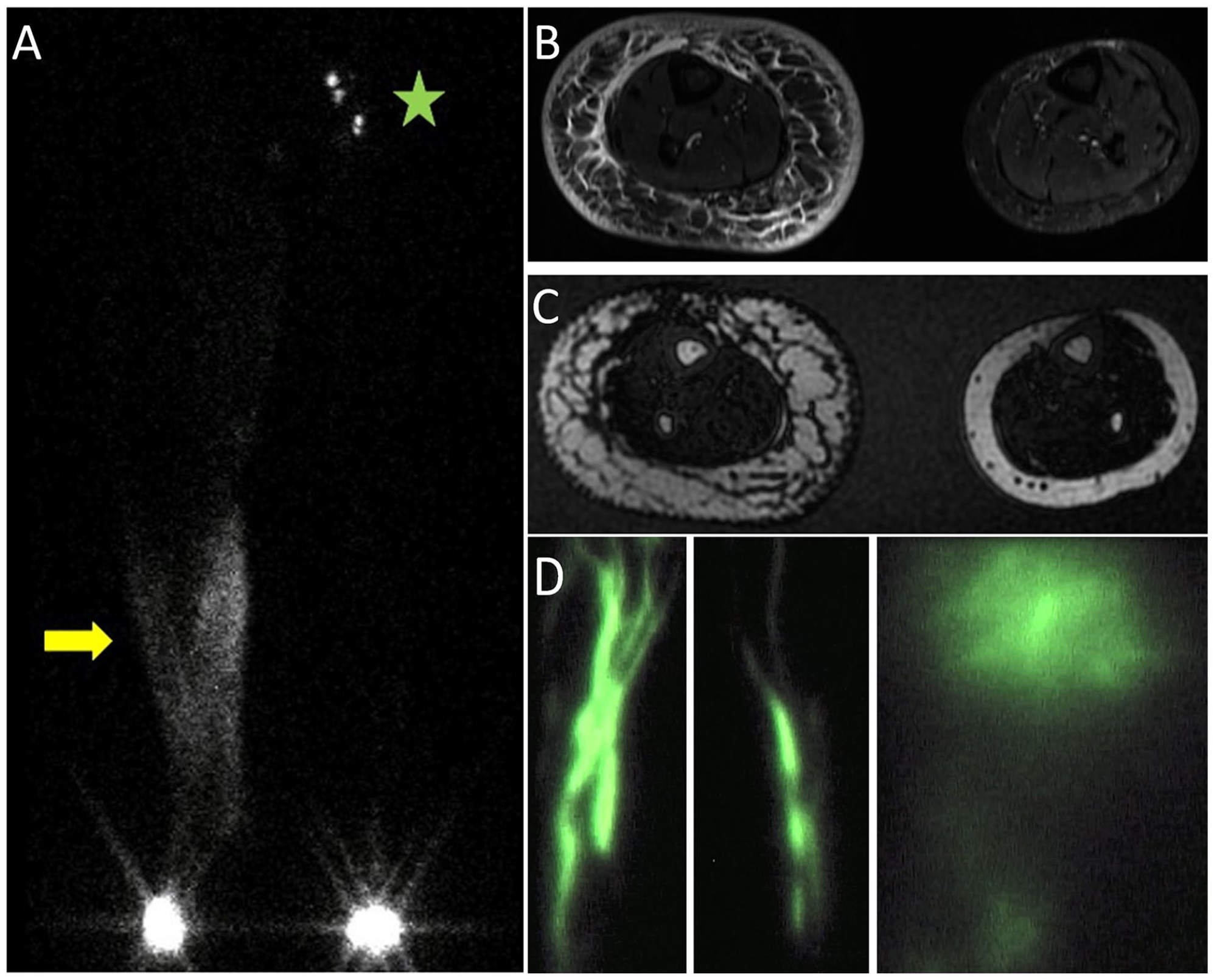
A middle-aged woman developed significant right lower-extremity edema following treatment for endometrial cancer.
The most appropriate utilization of nuclear lymphoscintigraphy is debated and clinical indications are primarily based on expert opinion.14,44 In patients with a clear history and exam consistent with lymphedema, imaging is unlikely to change management; however, it can be beneficial to patients without a definitive clinical diagnosis. It also can be helpful in younger patients, given that earlier diagnosis can improve prognosis or differentiate lymphedema from other potential etiologies of edema (e.g., a patient with left lower-extremity edema and concomitant venous outflow obstruction). There are a variety of objective parameters utilized, including transit time of the tracer or clearance rate of the tracer from the injection site, though no specific measurement has been widely accepted. Notably, there does not appear to be a correlation between the severity of abnormalities seen on nuclear lymphoscintigraphy and the severity of disease. Some argue that imaging reports should be limited to differentiating normal versus abnormal lymphatics, with limited additional use for diagnosis, management, or treatment. 45
Magnetic resonance imaging
MRI has become an increasingly utilized modality for the evaluation of lymphedema – both in diagnosis and in presurgical planning. MRI can image edema, adipose tissue, and lymphatic anatomy (Figures 2B and 2C). As discussed above, excess adipose deposition is a unique pathophysiologic property of lymphedema and can be diagnostic. Although MRI cannot distinguish normal adipose tissue from adipose deposition in lymphedema, it can detect excess adipose compared to the unaffected limb in unilateral lymphedema. Additionally, certain patterns of edema are characteristic of lymphedema, such as honeycombing, epifascial fluid, and the presence of dermal thickening. Interestingly, the areas of dermal thickening and subcutaneous honeycombing, termed a dermal rim sign, correspond to dermal backflow visualized on nuclear lymphoscintigraphy. 46
MR lymphangiography (MRL) allows for direct visualization of the deep and superficial lymphatic vessels. MR contrast agents are injected subcutaneously or intradermally and enter lymphatic capillaries. Patients with impaired lymphatic function will have delayed opacification of the lymphatic collecting vessels and lymph nodes. Pooling or congestion of contrast can also be seen in a similar pattern to dermal backflow on nuclear lymphoscintigraphy. 47 Diseased lymphatic vessels, particularly in some forms of primary lymphedema, will also be tortuous and dilated. 48 MR lymphangiography offers the advantage of superior spatial resolution and detail of lymphatic anatomy compared to nuclear lymphoscintigraphy. For central lymphatic imaging, intranodal injection can be performed as well. Though not the focus of this review, MRL can assess central lymphatic integrity, lymphatic leaks, or thoracic duct stenosis, and is increasingly utilized in patients with refractory chylous ascites or chylothorax. 49 Though a variety of MRI staging criteria have been proposed, there are currently no uniformly accepted criteria. Additionally, there are no uniform protocols, but MRI does allow the radiologist to tailor scans to the particular questions a clinician may have.
Indocyanine green lymphography
Near-infrared fluorescence lymphography utilizing indocyanine green (ICG) is increasingly being utilized in both the diagnostic and the presurgical setting (Figure 2D). ICG is injected intradermally, bound by albumin and lipoproteins, and then taken up by superficial lymphatic vessels. It offers improved visualization of the superficial lymphatic channels and greater characterization of patterns of dermal backflow compared to nuclear lymphoscintigraphy. Various staging systems have been described, but there are no uniformly accepted criteria. ICG lymphography may provide more sensitive diagnostic assessment of lymphatic function compared to nuclear lymphoscintigraphy and nonimaging parameters, which are more often used in surveillance programs (e.g., circumferential measurements, percent water content ratio).50,51 Earlier detection offers potential for the initiation of therapy in the preclinical state, which may be important with growing prophylactic surgeries and potential biochemical interventions in the future.
Additional assessment modalities
Ultra-high-frequency ultrasound (e.g., 48 and 70 MHz) enables direct visualization of lymphatic vessels and is increasingly used for both assessment and presurgical planning. Visualization allows for characterization of the lymphatic channel, description of the degree of fibrosis and obstruction, and localization to clarify its adequacy for use in a potential lymphovenous anastomosis (LVA).52,53
Invasive lymphangiography is now more frequently utilized for assessment of central lymphatic abnormalities and can be done with concomitant intervention in select patients with a central lymphatic obstruction; however, the role for invasive lymphography is limited for most patients with lymphedema. Bioimpedance spectroscopy utilizes the flow of electric currents to determine the degree of fluid present in a limb. It is important in the surveillance of patients at high risk of developing secondary lymphedema and can be used to guide adequacy of a particular compression strategy. 54
Nonsurgical management
It is important to understand the goals and expectations of patients living with lymphedema, as they can vary significantly between individuals. 55 A multidisciplinary team approach may offer patients a more comprehensive evaluation and understanding of the growing treatment options available (Figure 3).56,57 There have not been significant changes in the nonsurgical management of lymphedema over the past few decades; however, it remains the cornerstone of therapy for most patients and is essential for preoperative optimization and postoperative maintenance of results in the select few patients who undergo surgery.40,58 It is imperative that patients understand the chronic nature of lymphedema, appreciate their role in stagnating the progression and sequelae of the disease, and maintain consistency in ongoing nonsurgical care.
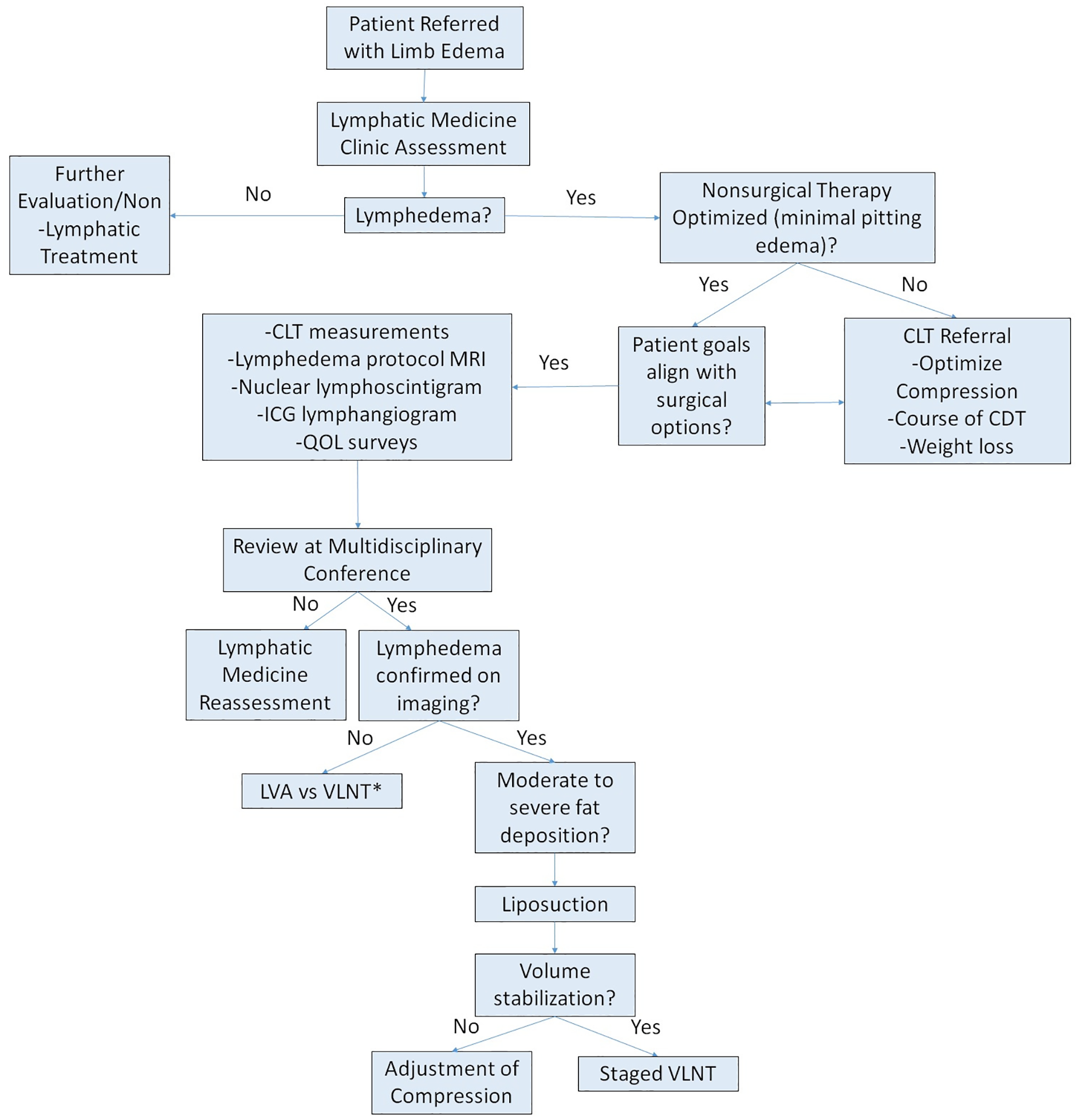
Our approach to patients presenting for evaluation of possible lymphedema.
Complete decongestive therapy (CDT) with multilayer, low-stretch bandaging, manual lymphatic drainage (MLD), and skin care is the mainstay for volume reduction prior to initiation of compression. 59 Velcro wraps can also be effective for volume reduction, particularly in the lower extremities, and may be more feasible for some patients. 60 MLD and pneumatic compression have a role, though in our experience they are limited. There is a misconception that intermittent MLD is helpful in persistent reduction of edema (e.g., going to a certified lymphatic therapist a couple of times a week or a month for MLD). MLD benefits are usually transient, in the region of hours, and though it can assist with decongestion when paired with high-quality compression, it does not appear to be particularly helpful in patients undergoing CDT. 61 A recent systematic review noted the poor quality of data for MLD, but suggests there may be a benefit in early-stage breast cancer-related lymphedema with a limited benefit for more moderate-to-severe lymphedema. 62 There may be a role for ICG-guided MLD to assist in the identification of outflow pathways given the variation in drainage patterns among individuals. 63 Our focus in patients with significant residual edema is CDT – which begins with intensive wrapping for several weeks and once reduction has occurred and plateaued, patients are fitted for flat-knit custom garments to maintain the reduction that was achieved. CDT results in a greater reduction in volume compared to MLD or compression alone, and can achieve a significant reduction in limb volume (> 1 L volume reduction with a median of 11 days of CDT).64–66 Ongoing compression is essential to maintain the reduction achieved during CDT. 67 CDT is not required for all patients, particularly those with mild edema or those with fat hypertrophy with minimal pitting edema.
Optimal compression is the backbone of lymphedema therapy, but what specifically is optimal can be variable based on a patient’s needs, goals, and tolerance. Patients who utilize compression report less pain and improved range of motion. 68 A flat-knit custom garment can offer the highest quality of compression by maintaining superior limb fit, compression strength, and associated comfort and compliance. In our experience, custom fitting garments are particularly helpful in lymphedema patients as the degree of limb size and shape distortion can be variable. It is important for patients to understand the goal of compression: to maintain the least amount of change in edema from when they wake in the morning and don the garment, to when they go to bed. There are a wide variety of materials, sizes, and degree of compression available. The two most common forms of compression are circular-knit garments and flat-knit garments. 69 Circular-knit garments are ‘ready to wear’ off the shelf and are widely available in standardized sizes and strengths. Owing to their more elastic nature, circular-knit garments tend to stretch more through the day and the standardized sizes may not fit a particularly deformed limb. A flat-knit garment has greater integrity than a circular-knit garment and thus there is better edema containment. Most flat-knit garments require custom fitting, which allows for a better fit in a wide range of limb shapes and sizes and are easier to don. For some patients, wearing a custom flat-knit garment overnight is also an option. However, flat-knit garments are more expensive and require access to a garment fitter. Custom garments may not be necessary for all patients; circurcular-knit garments can provide adequate control at a more affordable cost, particularly for those with milder symptoms. For patients with limitations in donning a more traditional garment, an adjustable Velcro compression wrap can offer good control. 70 Garments should be replaced every 3–6 months as they tend to lose their integrity with multiple wears and cleaning. Unfortunately, for many patients in the United States, poor insurance coverage of compression garments makes optimal compression financially unachievable. 71
Pneumatic intermittent compression can be helpful for some patients with residual edema, and in small studies has demonstrated modest reduction in volume and improvement in quality of life.72,73 Studies have demonstrated that use of pneumatic pumping is associated with a reduction in episodes of cellulitis.74,75 Other data have not shown a benefit when utilized with routine lymphedema management. 76 Similar to MLD, isolated pneumatic compression offers short duration benefit and is best utilized with high-quality compression as an adjunct to maintain any reductions in edema. Additional downsides include requirements to be sedentary and, for some patients, to become dependent on the pump without clear benefit. Fortunately, newer pumps may allow more mobility.
Exercise, increased mobility, weight loss, and skin care are all important practices for patients in compression. Some providers call for an exercise program with both resistive and aerobic components as a mandatory component of a complete lymphatic therapy program. 9 Though certain activities may exacerbate edema in a particular individual, exercise is recommended as it is generally safe and often helpful. 77 Though the benefits of exercise have been mostly studied in patients with BCRL, a small study of patients with lower-extremity edema found that exercise in a well-fitted garment further reduced edema.78,79 Patients are often afraid or anxious about over-exertion of the affected extremity. However, a randomized trial of patients with BCRL found that weight-lifting did not worsen limb swelling and instead decreased exacerbations of edema, reduced symptoms, and increased upper limb strength. 80 Though the data are mixed, some swimming and water-based exercises can be helpful. 81
Obesity-induced lymphedema is a well-established phenomenon and has a clear association with massive localized lymphedema, exclusively seen in patients with morbid obesity. 82 Milder forms of obesity have also demonstrated deleterious effects on lymphatic dysfunction, and thus, even if not the primary etiology of lymphedema, obesity can be a significant contributor to more significant symptoms. 83 Obesity is also a clear risk factor for the development of BCRL. A dedicated effort should be made to manage weight, and in many lymphatic centers, there is a weight/body mass index (BMI) cutoff that patients must reach prior to consideration of surgical treatment. 84 We refer our patients with obesity to obesity medicine for a comprehensive assessment given the complexities of increasing medical and surgical weight loss options.
Surgical treatment options
Surgical treatment of lymphedema is becoming more widely available, though it is still generally offered only in a select number of dedicated centers. There remains significant debate around optimal patient selection, most appropriate surgery, and surgical technique. Our general approach is summarized in Figure 3. Much of the disagreement over appropriate surgical treatment is due to current limitations of data, which is fraught with poor quality, bias, and heterogeneity in reported outcomes. 85 There are two types of lymphatic surgeries: physiologic procedures to improve lymphatic function and debulking procedures to remove the accumulation of excess fibroadipose tissue.
Physiologic surgical options
Physiologic procedures include LVA and vascularized lymph node transfer (VLNT). Using a microsurgical approach, LVA entails identifying and then anastomosing a functioning lymphatic vessel with a vein to shunt lymphatic fluid to the central circulation via peripheral veins. The procedure requires a small incision and is an outpatient procedure, with infrequent complications. 86 The most appropriate technique and number of LVAs that should be performed is debated.
Although there is significant study heterogeneity, systematic reviews have demonstrated objective and subjective improvement in most patients following LVA.86,87 We believe reporting limb volume reduction as an outcome without also clearly reporting any changes in concomitant compression is suboptimal; however, concomitant compression is infrequently reported.88,89 If compression had been optimized prior to the procedure, thus limiting residual pitting edema, there would be little expected difference in volume postoperatively. The benefits of LVA tend to be modest but can have a significant impact on patients’ quality of life. It may be more effective in earlier stages of lymphedema prior to the onset of significant peripheral lymphatic vessel damage that can occur as lymphedema progresses.88,90
VLNT involves autologous transfer of a group of lymph nodes to the affected limb. There are various techniques and disagreement exists regarding the most appropriate location of lymph node harvest, lymph node transfer site (proximal vs distal limb), and mechanism of lymphedema improvement. Some believe that lymph nodes act as a bridge to connect areas of obstructed or impaired flow, and thus nodes are placed proximally in the limb. Others believe lymph nodes promote neo-lymphangiogenesis by local release of growth factors and cytokines that result in the development of afferent and efferent lymphatic vessels, thus establishing functional lymphatic flow to the transfer site. Lymph fluid is then drained from the transferred lymph nodes to the peripheral venous system via lymphatico-venous connections within the transferred lymph nodes, as was described above. 91 Donor sites vary and include axillary, groin, supraclavicular, and other omental and jejunal mesentery lymph node groups. There is a reported 10% risk of developing donor site lymphedema, but that risk can be entirely avoided by utilizing the omental donor site.92,93
Evidence for the efficacy of VLNTs for treatment of lymphedema remains limited to small observational studies with short-term follow up, but it appears to decrease limb volume, decrease recurrent cellulitis, and improve quality of life.94,95 In a meta-analysis of VLNT for the treatment of BCRL, there was a 40% reduction in limb volume. There is significant heterogeneity in the outcomes reported by existing studies and, as with LVA surgical treatment, understanding the degree to which VLNTs reduce limb volume requires reporting of concomitant compression. 96 For patients included in the small subset of studies that do report concomitant compression, 45% (27/60) were able to discontinue compression following VLNT.
Physiologic procedures may be more effective in secondary compared to primary lymphedema. 97 Some argue that LVA is unlikely to be beneficial in primary lymphedema, as lymphatic vessels are intrinsically dysfunctional or hypoplastic. Thus, creating a bypass is unlikely to aid in significant lymphatic outflow through the anastomosis. In contrast, patients with BCRL will develop progressive lymphatic vessel dysfunction as the disease course ensues. For this reason, LVA may be most beneficial at the time of axillary lymph node dissection (immediate lymphatic reconstruction) or in early lymphedema before vessels are irreversibly impaired by lymph outflow obstruction, adipose deposition, and fibrosis. However, there is limited support for LVA in primary lymphedema. 98 In a study of 70 patients (22 with primary and 48 with secondary lymphedema) who underwent LVA, patients with earlier stage disease responded better, and patients with secondary lymphedema (55% volume reduction) had a greater response than those with primary lymphedema (37% reduction). 99 The growing use of ICG lymphography allows for more specific evaluation of the anatomy and function of superficial vessels. Therefore, in patients with primary lymphedema, it is now possible to target functional vessels that may be more likely to facilitate flow through the anastomosis. 100 Our group generally favors VLNT for surgical treatment of primary lymphedema. However, given the lower risks of LVA in comparison to VLNT, we will evaluate patients for LVA and offer the procedure in select cases if there appears to be suitable anatomy and function on imaging.
Patient selection varies among surgeons. Our group discusses surgical options in patients who are adherent to and optimized with nonsurgical therapy, have ongoing detriment to quality of life, and a BMI below 35. Though surgical treatment has typically been reserved for patients with significant symptoms, given the inflammatory cascade and resultant progression of disease, we will discuss an earlier intervention for select patients with mild disease to avoid later sequelae of fibroadipose deposition. This consideration is particularly relevant for patients who are candidates for LVA, given the low associated surgical risks.16,99,101
Debulking surgical options
Fibroadipose deposition is present in the overwhelming majority of patients; thus, those who hope for a significant reduction in limb girth will require debulking to achieve this goal (Figure 4). Debulking utilizing circumferential power-assisted liposuction was pioneered by Dr Hakan Brorson. Patients can achieve dramatic reductions in limb size, with existing studies reporting excess volume reductions of 101–108% in upper extremities and 86–115% in lower extremities.57,102–104 Additionally, there is great improvement in quality of life and a reduction in episodes of cellulitis.102,105 Debulking is most frequently utilized and reported for BCRL. For patients who subsequently use high-quality, long-term compression, the surgical benefits are sustained for at least a decade. 105 Custom, flat-knit compression garments worn effectively for 24 hours a day are required. For this reason, careful patient selection with demonstrated adherence to compression and minimal residual pitting prior to liposuction is essential. 106 Though more commonly performed in a staged fashion, some perform liposuction with concomitant VLNT. 107 Interestingly, changes in lymphatic flow have been noted following debulking surgery, though it remains unclear if this is due to improved lymphatic function or secondary to newly developed superficial lymphovenous connections formed by the surgical trauma of liposuction cannulation. 108 The Charles procedure, an excisional technique that requires removal of tissue to the muscle fascia with skin grafting, is now less frequently performed given its significant morbidity. However, it may be utilized in very select patients with no other surgical treatment options. 109
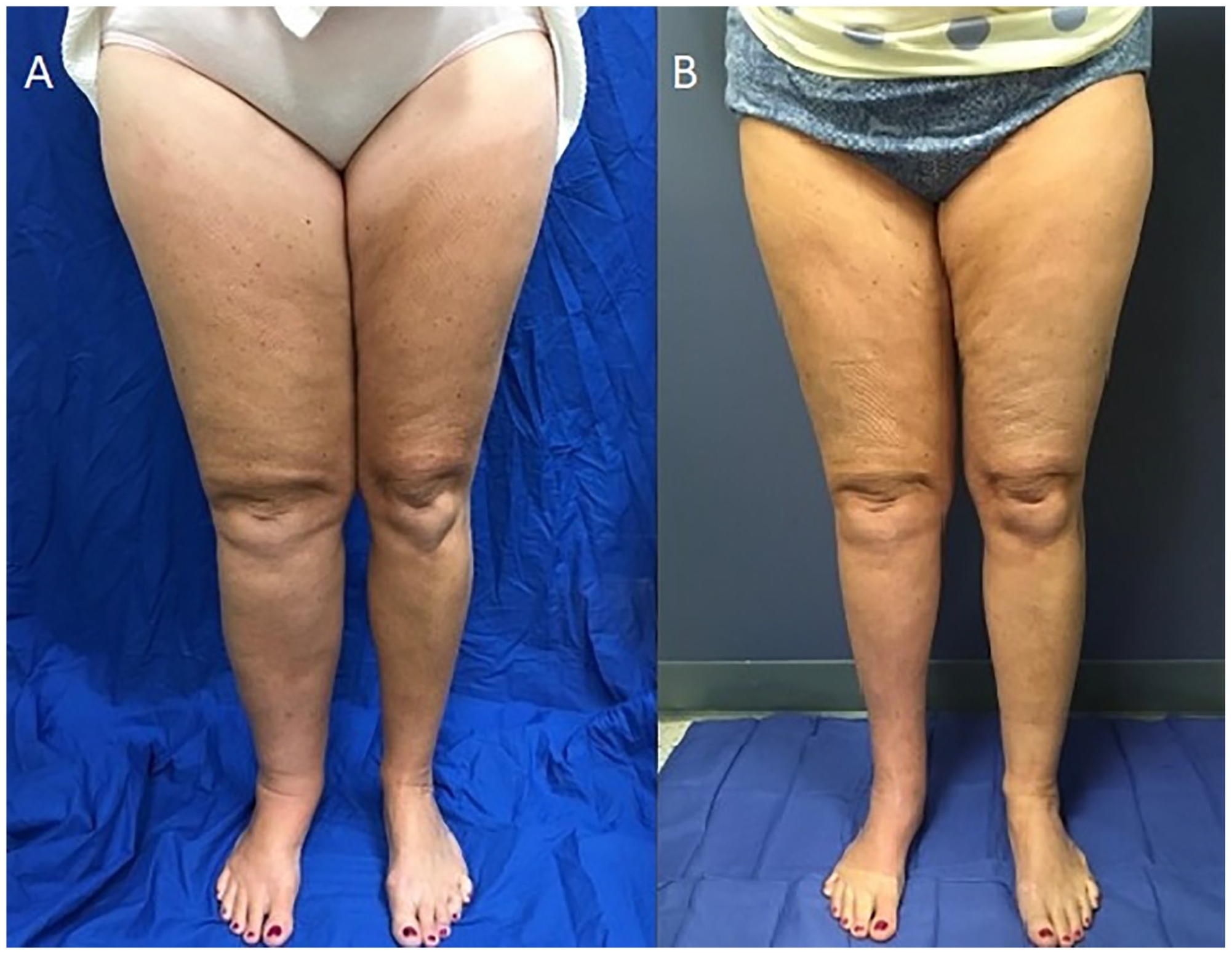
A middle-aged woman with secondary lymphedema of the right lower extremity (radiographic studies in Figure 2) with a 4.7-liter excess volume (52%) prior to debulking
Imaging in surgical management
Considering that nuclear lymphoscintigraphy remains the gold standard for functional lymphatic imaging assessment, it is appropriate to confirm the diagnosis prior to any surgical intervention. 44 Additionally, specific characteristics on nuclear lymphoscintigraphy may predict a patient’s response to lymphatic surgery. 110 ICG offers detailed anatomic assessment of the superficial lymphatics and can identify potential targets for LVA. MRI is most useful for determining the degree of adipose hypertrophy that is contributing to increased limb girth. In patients with unilateral edema, imaging of the contralateral limb is essential to determine the proportion of fat that is attributable to lymphedema and is useful in guiding debulking procedures. Postoperatively, MRI can evaluate the viability of lymph nodes after VLNT. ICG lymphography and nuclear lymphoscintigraphy can determine changes in lymphatic transport following both physiologic and debulking procedures.
Prevention and surveillance
The development of secondary lymphedema is multifactorial with known risk factors such as obesity and the severity of lymphatic insult (e.g., number of lymph nodes removed and/or concomitant regional lymph node radiation in breast cancer treatment). However, there are likely underlying genetic risk factors and anatomic variations in lymphatic flow that have yet to be elucidated, but will later be used to identify those at highest risk for secondary lymphedema. 110 A variety of surveillance programs have been developed for those with an elevated risk, particularly following breast cancer treatment. These programs monitor patients with serial limb volume measurements and bioimpedance spectroscopy scores to assess for early-onset edema. Bioimpedance may offer a more sensitive assessment in early stages of lymphedema and enable a more proactive initiation of therapy. 111 Transient edema can occur early in the posttreatment course, and generally it is recommended to initiate therapy after demonstration of a 10% or more increase in relative arm volume. Guidelines regarding the most appropriate timing for initiation of therapy and the impact of early intervention on long-term outcomes are unclear due to limited data. 112 Of note, both Black race and Hispanic ethnicity are associated with increased risk of BCRL. 113 Further study is needed to better understand the mechanism of this finding, but particular vigilance and equal access to surveillance programs for these populations are vitally important.
There is growing interest in prophylactic LVA at the time of lymph node dissection, first termed lymphatic microsurgical prevention healing approach (LYMPHA) and now better known by its technical name, immediate lymphatic reconstruction (ILR). Early studies of ILR outcomes have demonstrated a two-thirds reduction in the incidence of lymphedema. 114 In an ongoing randomized trial of 144 patients undergoing axillary lymph node dissection, interim results have reported the rate of lymphedema was 9.5% after ILR compared to 32% without ILR. Patient follow up was variable but up to 24 months, the window in which most patients develop secondary lymphedema after cancer treatment. 115
Future directions
The understanding and management of lymphedema remains in its relative infancy. There are exciting, though early-stage, advancements in lymphedema management options. Investigation of pharmacologic targets to modulate the inflammatory and fibrotic cascade that drive much of the clinical manifestation of lymphedema is ongoing. Though surgery offers some promise, optimally there would be medications available to decrease or arrest disease progression in patients with early-stage lymphedema. Lymphangiogenic factors have also been explored, particularly vascular endothelial growth factor C (VEGF-C), which regulates lymphatic vessel development and lymphatic endothelial cell proliferation and migration. 116 In animal models, recombinant VEGF-C increased lymphangiogenesis and improved lymphatic function, resulting in decreased edema. 117 More recently, VEGF-C has been incorporated into a nanofibrillar collagen scaffold (BioBridgeTM Fibralign Corp, Union City, CA) utilized during physiologic surgeries to aid in the development of local lymphatics. This study demonstrated that patients receiving the VEGF-C collagen scaffold with surgery achieved a greater reduction in edema. 118
In a randomized trial of 34 patients, the nonsteroidal antiinflammatory drug ketoprofen was administered three times daily (75 mg/dose) for 4 months and demonstrated decreased dermal thickness, collagen deposition, and perivascular inflammation on skin biopsy. However, this treatment did not achieve reductions in limb size or degree of edema. 119 Given the long-term risks of ketoprofen, such as serious cardiovascular thrombotic events and gastrointestinal bleeding, the clinical utility is limited. However, it was noted that the benefits seen on skin biopsy were mediated by its inhibition of the 5-lipoxygenase metabolite, leukotriene B4 (LTB4). LTB4 promotes lymphedema through a variety of inflammatory and regulatory mechanisms, including inhibition of VEGFR3 mRNA expression. Inhibition with bestatin demonstrated promise in a mouse tail model of lymphedema. 120 The results of a randomized trial with 54 patients were inconclusive, but there remain plans for a larger clinical trial in the future (NCT02700529). Additional studies targeting Th2-related cytokine expression are also planned, based on early data demonstrating the potential benefits of inhibiting this pathway. Antibodies targeting IL-4 and IL-13 were demonstrated to reduce Th2 cytokines and epidermal thickness.116,121 Early data also suggest inhibition of the transforming growth factor beta-1 (TGF-β1) pathway may slow the fibrotic process and have shown promise in an animal model. There is now interest in exploring angiotensin-converting enzyme inhibitors as well.116,122
The focus of this review is peripheral lymphedema but as more is understood about lymphatics, it is becoming clear that they play an integral role in a variety of disease states beyond peripheral lymphedema. 26 The role of lymphatics in cardiovascular disease has just recently been explored. 123 Improved understanding and interventions directed at the central lymphatic system and thoracic duct may offer new insights and therapies for chylothorax, refractory ascites, and congestive heart failure.124,125
Given the continued expansion of treatment options, multidisciplinary teams offer patients the full spectrum of advanced imaging techniques, optimal nonsurgical management, and surgical treatment options for appropriate patients.56,57,126 A team-based approach with specialists from vascular medicine, radiology, lymphatic therapy, and plastic surgery offer patients a comprehensive approach to management. Through the Lymphatic Education and Research Network (LE&RN), specific Centers of Excellence have been identified to assist patients in finding institutions with particular interest in and experience with lymphedema. Increasing advocacy and recognition of lymphedema is providing the hope of more support for patients, researchers, and clinicians in the future. A new federal law coming into effect in 2024 ‘The Lymphedema Treatment Act,’ will improve insurance coverage for prescribed compression. In 2022, the NIH announced the National Commission on Lymphatic Research and in 2023, the Congressionally Directed Medical Research Program in the United States included funding for lymphatic disease for the first time. Ongoing efforts to increase awareness and education to medical students and experienced clinicians alike is important to further develop access to high-quality care for lymphedema patients.
Conclusion
The key points to remember are summarized in Table 2. There is growing interest in lymphedema and promise for more optimal care beyond the traditional nonsurgical measures. Integrating more advanced imaging techniques and team-based approaches to care offers patients the full breadth of options and personalized treatment options. Though surgical therapies are an exciting development, with further investigation there are potential medical targets to help tackle this traditionally underserved disease state.
Lymphedema: summary of key points.

Footnotes
Declaration of conflicting interests
Brett Carroll received research support (institutional) from Bristol Myers Squibb and serves on the scientific advisory board for Koya Medical. Dhruv Singhal reports no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
