Abstract
Lymphedema, a chronic debilitating disease, is not always easily diagnosed. A total of 254 new patients ((217 women, 37 men), median (Q1–Q3) age 61 (46–72) years) were referred for suspected limb lymphedema to an exclusively lymphedema-dedicated department for a first consultation (January – March 2015) were included; among 118 with upper limb involvement, 100 (84.7%) were diagnosed with post-breast cancer therapy and four with primary lymphedemas; among 136 with lower limb involvement, 31 (22.8%) were diagnosed with primary lymphedemas and 35 (25.7%) with post-cancer lymphedemas. The main alternative diagnoses were: 32 (45.7%) lipedemas/lipo-lymphedemas and 21 (30%) chronic venous insufficiencies. Age at symptom onset, body mass index, referral origins and first-symptom-to-specialized-consultation intervals differed between primary, post-cancer lymphedema and alternative diagnosis patients. Among the entire cohort, 57 (22.4%) had cellulitis. For all 135 (53.1%) upper or lower limb post-cancer lymphedemas and the 119 (46.9%) others, the median (Q1–Q3) first-symptom-to-specialized-consultation intervals were 1.4 (0.7–3.8) and 4 (1.1–18.8) years, respectively (p < 0.0001). Specialized consultations confirmed primary and post-cancer lymphedema diagnoses and identified others, especially for patients with suspected lower limb lymphedema.
Introduction
Lymphedema is a chronic and debilitating disorder most often involving upper or lower limb(s). It is classically divided into distinct primary and secondary forms. Primary lymphedemas develop more frequently in young females, sometimes with onset in children or newborns. 1 They can be isolated, with congenital and familial (Milroy disease), or syndromic forms accompanied by other clinical malformations associated with genetic mutations or chromosomal abnormalities.2,3 Secondary lymphedemas are the most frequent in industrialized countries, mostly occurring after cancer treatment, including lymph-node excision, sometimes associated with radiotherapy (breast cancer for the upper extremity, pelvic cancer or melanoma for the lower limb), while filariasis is the first cause worldwide.4,5 Lymphedema diagnosis is usually clinical. For the upper limb, unilateral volume increase is easy to confirm after breast cancer treatment. Increased lower limb volume, a frequent clinical situation and reason for medical consultation, may have various causes and it is important to determine its precise etiology for each patient because each entity’s treatment is specific and adapted. Lymphedema, poorly recognized by the medical community, specialists or patients themselves, is often misdiagnosed. 6 Its misdiagnosis may lead to inappropriate management, prolong the time to specific therapy and enhance the patient’s detrimental anxiety/depression. We describe new patients referred to our center, which is exclusively dedicated to lymphedema diagnosis and treatment.
Materials and methods
Department of Lymphology
Since 2006, our unit has been labeled as a member of the National Referral Center for Rare Vascular Diseases dedicated to primary lymphedema. It also receives patients with post-cancer lymphedema or suspected lymphedema. It oversees consultations and has a multidisciplinary team trained in lymphedema management comprised of physiotherapists, nurses, a psychologist, podiatrists, a dietician and an orthopedic specialist to obtain the detailed measurements needed to make the individually tailored elastic garments. Other specialists are available in collaborating centers for explorations, such as lymphoscintigraphy, non-contrast magnetic resonance lymphography and treatment (e.g. plastic surgery). Lymphedema management is based on complete decongestive therapy, also called complex or multimodal decongestive physiotherapy, a term proposed by Michael Földi in the 1980s to define lymphedema treatment. His approach is used in our Department of Lymphology and has several components: low-stretch bandage, manual lymph drainage, meticulous skin/nail care, exercises, and a patient-education program to learn self-bandaging and elastic compression.7,8 Lymphedema patients require regular and long-term follow-up.
Patients and methods
Every month, our specialized lymphedema consultation team sees about 370 patients, followed for known limb lymphedema or newly suspected lymphedema. From January to March 2015, we collected all new patients (first consultation) referred to our tertiary care Department of Lymphology with a suspected diagnosis of upper or lower limb lymphedema. According to French law, Ethics Committee approval is not required because we conducted an observational and descriptive – not interventional – study, with patients managed according to our standard procedures.
We recorded the following characteristics at the first consultation: sex, age, age at first symptom, previous cancer and its treatment, first-symptom-to-specialized-consultation interval, lower or upper limb localization, uni- or bilateral involvement, previous cellulitis (erysipelas), body mass index (BMI = body weight (kg)/height2 (m2)), and referral origin (oncologists, surgeons, radiotherapists, dermatologists, internists, cardiologists, primary care physicians, physiotherapists, patient’s initiative).
Upper limb lymphedema was diagnosed when the affected limb was 2 cm larger than the contralateral side. Lower limb lymphedema was diagnosed clinically based on increased limb volume, with pathognomonic Stemmer’s sign (inability to pinch the dorsal side of the skin of the second toe (or its base)) confirming the diagnosis (Figures 1 and 2). Other clinical anomalies, particularly signs of chronic venous insufficiency (varicose veins, skin and subcutaneous tissue changes: pigmentation, eczema, lipo-dermatosclerosis, atrophie blanche and healed or active venous ulcer(s)) were also recorded according to the clinical, etiologic, anatomic and pathophysiologic (CEAP) classification (Figure 3). 9 Wold et al.’s criteria 10 were applied to define lipedema as bilateral abnormal accumulation of fat from hips to ankles, essentially (but not exclusively) seen in obese women, associated with superficial pain, easy bruising, little improved by weight loss, and especially sparing the feet, with no Stemmer’s sign and a distinct step-off at the ankle (Figure 4). Chronic venous insufficiency signs are present in 20% of lipedema cases. 10
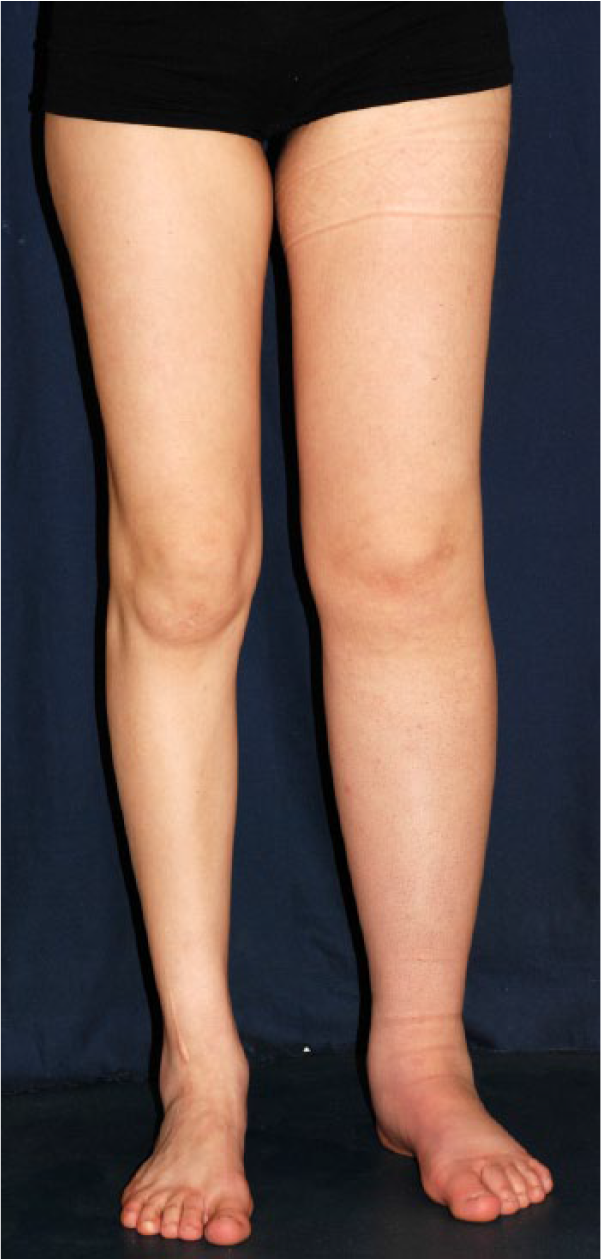
Left lower limb primary lymphedema in a 16-year-old female, with unilateral involvement and positive Stemmer’s sign, sparing the right limb.
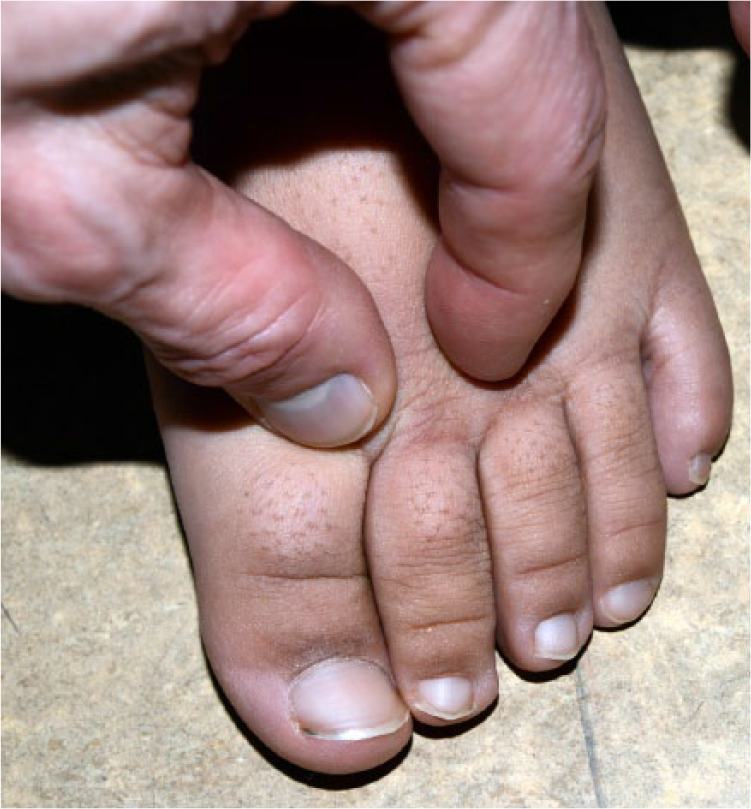
Stemmer’s sign: inability to pinch the dorsal side of the skin of the second toe (or its base).
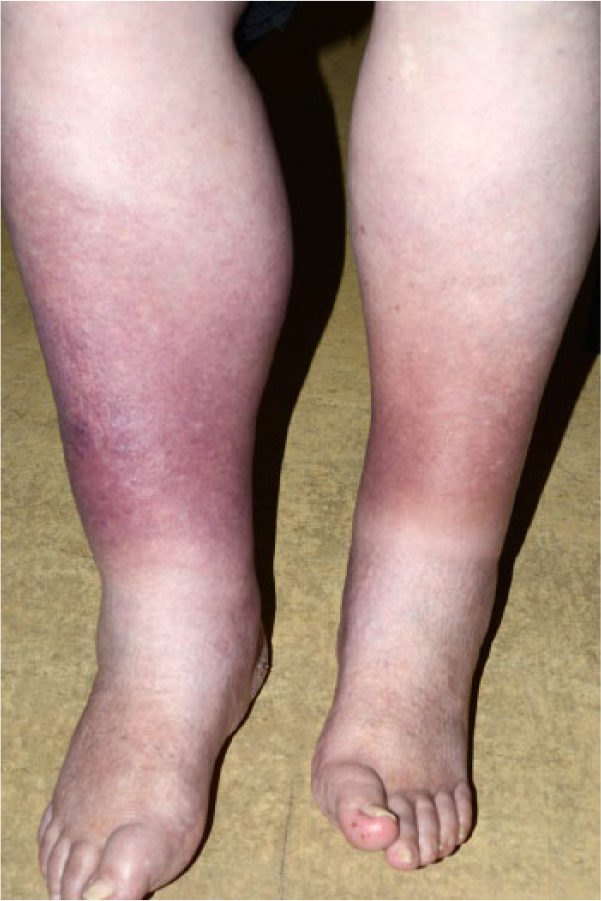
Chronic venous insufficiency with pigmentation.
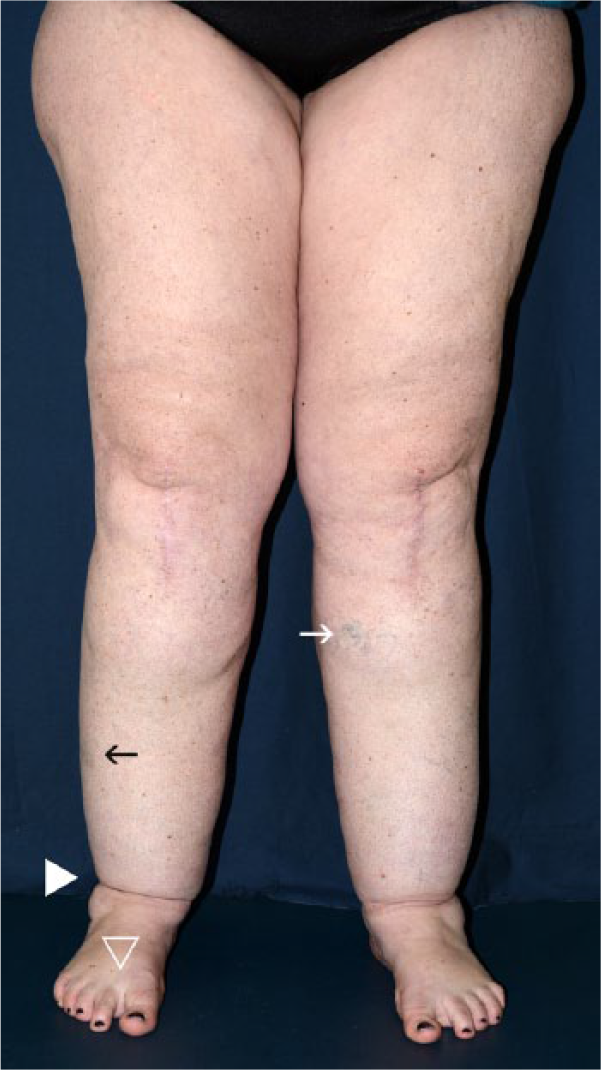
Bilateral lipedema in a 55-year-old woman with adipose tissue accumulation from hips to ankles, with step-off at the ankle (solid white arrowhead) and sparing the feet (without Stemmer’s sign, arrowhead), associated with easy bruising and varicosities (black and white arrows, respectively).
Statistical analyses
Data are expressed as numbers (%) for categorical variables and medians (interquartile range, Q1–Q3) or means (± standard deviation, SD) for quantitative variables. Student’s t-test, ANOVA, Fisher’s exact or χ2 test were used for comparisons. A p-value < 0.05 defined significance.
Results
From January to March 2015, 254 new patients were addressed to our center for suspected lymphedema (136 lower limb; 118 upper limb). The main patient characteristics and referral origins for consultation are given in Table 1.
Characteristics of the 254 patients referred for suspected limb lymphedema.
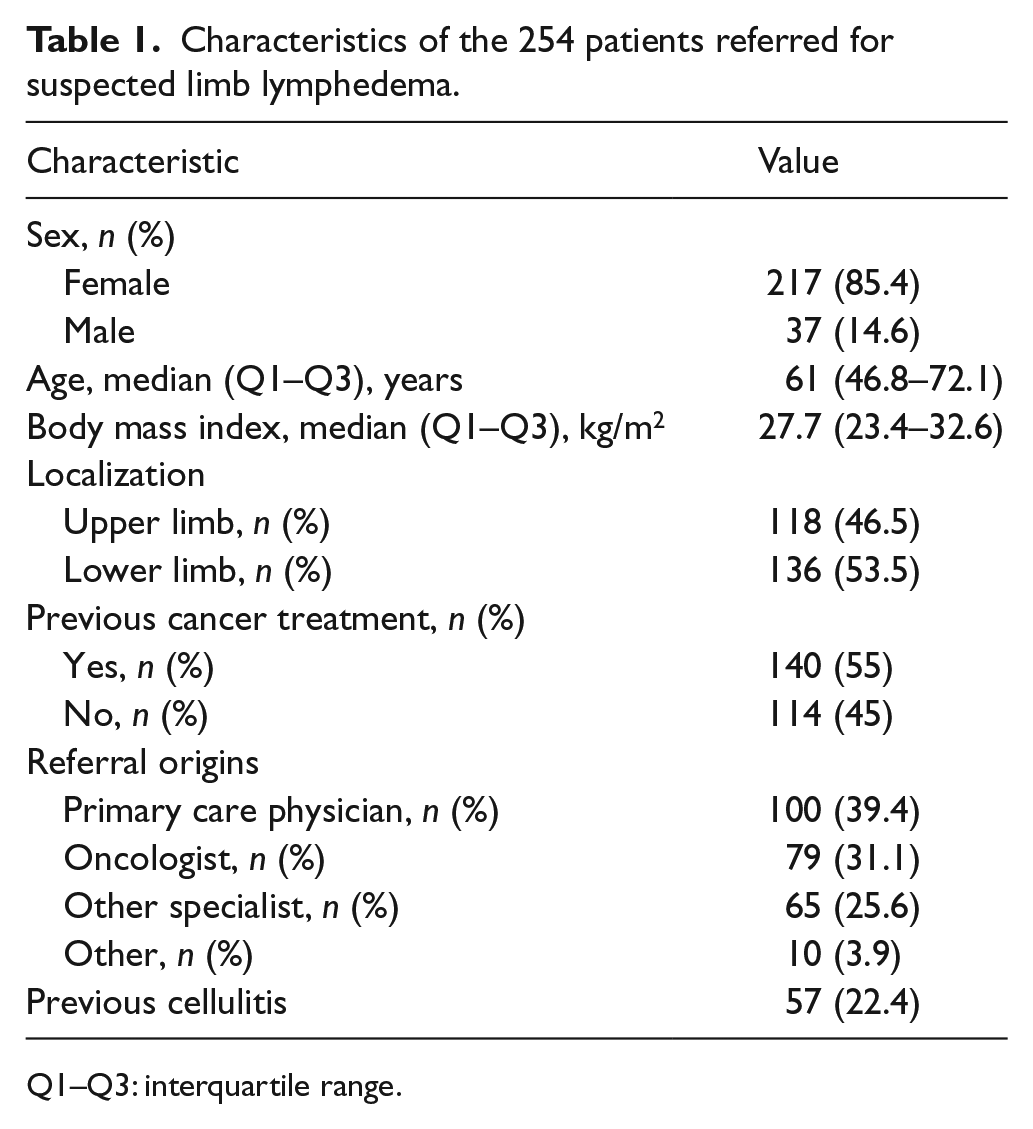
Q1–Q3: interquartile range.
Upper limb
For the 118 patients (111 women, 7 men) suspected of having upper limb lymphedema, the median age was 62.5 (Q1–Q3: 51.1–71.1) years and the median BMI 26.7 (Q1–Q3: 23.1–30.8) kg/m2. The median symptom-onset-to-the-specialized-consultation interval was 1.2 (Q1–Q3: 0.6–3.2) years. Oncologists referred 53% of the patients and primary care physicians 37%. Post-breast cancer lymphedema was diagnosed in 100 (84.7%) patients and primary lymphedema in four. The 14 (11.9%) others with no upper limb lymphedema, were diagnosed with lymphocele, limb heaviness, plexopathy/neuropathy, axillary web syndrome, post-thrombotic syndrome, hand paresthesias, arm burn or superior vena cava syndrome (Figure 5). One or more cellulitis episodes occurred in 22/104 (21.2%) lymphedema patients.
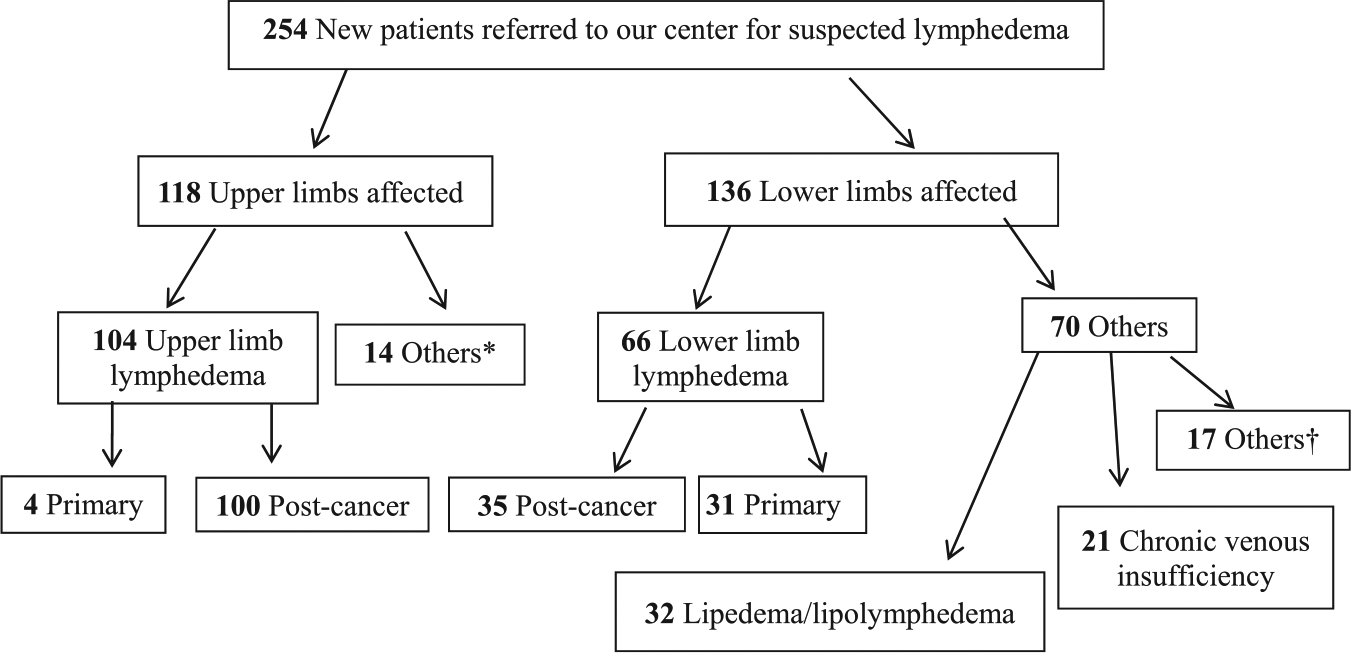
Flow chart of the patients addressed for the first consultation in our specialized lymphedema-dedicated center.
Lower limb
For the 136 (106 women, 30 men) with affected lower limbs, the median age was 58.1 (Q1−Q3: 42−72) years and BMI 28.3 (Q1−Q3: 23.8−33.7) kg/m2. Primary lymphedema was confirmed for 31 (22.8%) patients (23 women, eight men; 16 unilateral, 15 bilateral), whose median age at onset was 16.7 (Q1–Q3: 8.5–30.3) years, including eight children <18 years. Post-cancer lymphedema diagnosis was confirmed in 35 (25.7%) patients (27 women, eight men, after pelvic cancers (10 ovarian, three cervical, four uterine, three melanomas, two bladder and 13 others)).
For the 70 (56 women, 14 men, 51.5%) patients with alternative diagnoses (non-post-cancer, non-primary lymphedema), the main diagnoses were: 32 (45.7%) lipedemas/lipo-lymphedemas and 21 (30%) chronic venous insufficiencies (13 CEAP C3, three C4, two C5 and three post-thrombotic syndromes). The main other diagnoses were: four with edema stasis (neurologic paralysis), three vascular malformations, two with hypoproteinemia, two had post-traumatic edema (Figure 5). One or more previous cellulitis episodes were observed in 35/136 (25.7%) patients (versus 21.2% with upper limb lymphedema, non-significant). Age at onset, first symptom(s), BMI, referral origins and symptom-onset-to-specialized-consultation interval differed between primary, post-cancer lymphedema and alternative diagnoses (Table 2). For the 135 (53.1%) upper or lower limb post-cancer lymphedemas and 119 (46.9%) others (primary lymphedemas and alternative diagnoses), the median first-symptom-to-specialized-consultation intervals were 1.4 (Q1–Q3: 0.7–3.8) years and 4 (Q1–Q3: 1.1–18.8) years (p < 0.0001), respectively.
Comparisons among lower limb primary, post-cancer lymphedemas and alternative diagnoses.
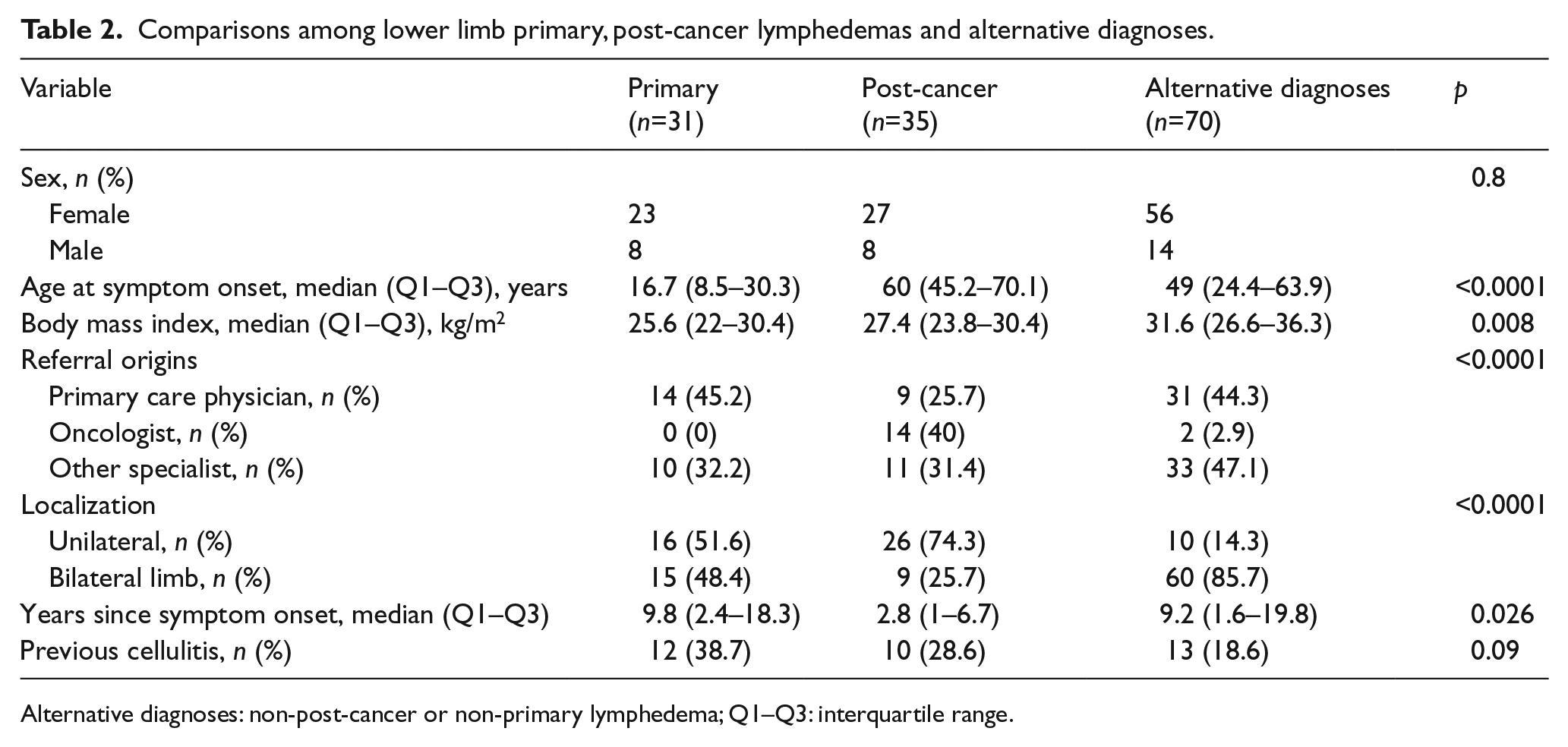
Alternative diagnoses: non-post-cancer or non-primary lymphedema; Q1–Q3: interquartile range.
Discussion
After a first consultation for suspected limb lymphedema in a specialized referral center, upper limb lymphedemas (88.1%) had mostly been correctly diagnosed, while half (51.5%) the patients with affected lower limb(s) had other diagnoses. Post-cancer upper or lower limb lymphedemas represented 53% of patients referred for suspected lymphedema. New patients referred to our specialized consultation were comparable for sex and age, as previously found in one Canadian center and 28 English treatment units.11,12 For the upper limb, lymphedema diagnosis was easily confirmed because the quasi-totality of patients had been treated for breast cancer, with surgery, axillary lymph-node excision and radiotherapy.
Lower limb lymphedema was frequently misdiagnosed. Lipedema was the most common alternative diagnosis. Wold et al.’s criteria enabled confirmation of the lipedema diagnosis, almost exclusively affecting obese women, with the presence of bruising, being painful to touch (or unsolicited), first involving the thighs bilaterally (fat accumulation from hip to knee or ankle), while sparing the feet, but combined with edema primarily affecting the lower leg after orthostatism. 13 Two diagnostic criteria were also included in Wold’s criteria: a marked familial history of lipedema and a poor effect of weight loss on limb morphology. All those characteristics differ from lymphedema features and enable the two conditions to be distinguished. Pertinently, after long-standing lipedema, the lymphatic system may be altered, giving rise to lipo-lymphedema with the appearance of Stemmer’s sign and sometimes complications (e.g. papillomatosis and cellulitis). 13 When a lipedema diagnosis is uncertain or overlaps with lymphedema (with the presence of Stemmer’s sign), lymphoscintigraphy may be useful but is not essential. The lymphoscintigraphic images showed only decreased speed of radiocolloid displacement but no structural abnormality as in lymphedema, thereby suggesting a functional abnormality.14,15 Pertinently, those findings were also seen in obese patients with idiopathic edema. 16 Other complementary investigations may help distinguish lipedema from lymphedema with specific findings: body composition, high-resolution cutaneous ultrasonography or high-resolution unenhanced computed tomography.17–19
This distinction may be difficult to discern because lipedema/lipo-lymphedema is poorly recognized and is also often associated with chronic venous insufficiency. 10 Chronic venous insufficiency (without lipedema) was the second most frequent ‘misdiagnosis’ retained during the first consultation. It was diagnosed based on CEAP-classification clinical signs. For advanced disease (C4 or higher), lymphatic impairment secondary to venous disease is frequently seen, especially in patients with limb ulcer(s), but the initial dysfunction is venous, followed by lymphatic. 20
Cellulitis is frequent in our population, especially, but not only, in lymphedema patients. Our results are consistent with those of Shallwani et al. who observed cellulitis in 22% of their patients referred to a specialized Canadian center for suspected lymphedema. 12 Thus, limb lymphedema is a major risk factor for developing cellulitis, estimated at 6.8-fold. 21 The portal of entry may be a small cutaneous lesion not always found, especially for the upper limb. For the lower limb, toe-web intertrigo and hyperkeratosis with cracks are most often present. Moreover, chronic edema, including lipedema/lipo-lymphedema, may increase the risk of cellulitis, suggesting a lymphatic dysfunction, as Soo et al. showed with abnormal lymphoscintigraphic imaging after cellulitis.21,22 It remains to be determined whether lymphatic impairment precedes or follows the cellulitis.
In our population referred for suspected lower limb lymphedema, ages at symptom onset differed, being younger for primary lymphedema than post-cancer lymphedema or other diagnoses. Furthermore, the median first-symptom-to-specialized-consultation interval was significantly shorter for post-cancer lymphedema patients than other upper or lower limb diagnoses patients. These results confirmed that oncologists know the risk of lymphedema after some cancer treatments, are sensitive to it and promptly referred patients to specialized lymphedema-dedicated centers.11,12
Limitations and stengths
The main limitation of our study is the absence of validated criteria to diagnose the different pathologies, especially for patients with non-primary or non-post-cancer lower limb involvement. Post-cancer lymphedema is easily recognized based on the patient’s cancer history, including lymph-node excision and/or radiotherapy. Primary lymphedema may be more difficult to diagnose but first symptoms usually appear before 25 years, with frequent unilateral involvement. Bilateral primary lymphedema is located below the knees (feet, ankles, calves) and exceptionally involves thighs. The search for Stemmer’s sign is important because it is pathognomonic for lymphedema. Lipedema diagnosis associated bilateral thigh involvement sparing the feet, easy bruising, superficial pain, and almost exclusively in obese or overweight women. Lipedema management is different from that of lymphedema and may be based on tumescent liposuction. 23
The strengths of our study are its monocenter design in a specialized center exclusively dedicated to lymphedema diagnosis and management by a specifically trained multidisciplinary team following a large patient cohort (exceeding 4000 annually). Another strength is that our center receives patients with post-cancer lymphedema, the most frequent in France, and children, adolescents and adults, especially females, with primary lymphedema.
Conclusion
A specialized center dedicated to lymphedema is useful to confirm/exclude lymphedema diagnosis and help avoid lost opportunities for the patients. Lymphedema diagnosis, both primary and after cancer, is required to adapt therapy to international recommendations, stressing the importance of self-management for this chronic debilitating disease. The first-symptom-to-specialized-consultation interval is shorter for patients after cancer treatment, suggesting oncologists’ better awareness of lymphedema. Primary care physicians and specialists need to be sensitized to the existence of this entity so that it can be recognized more rapidly and the patients referred to a specialist for diagnosis confirmation and appropriate treatment. The lymphedema specialist can provide non-lymphedema patients with explanations about their disease (lipedema, chronic venous insufficiency) and then refer them to an angiologist/vascular disease specialist, nutritionist/dietician or plastic surgeon for specific management. Delayed referral may be prejudicial for patients with increased risk of complications and diminished quality of life. Specialized consultations are also useful to precisely answer a patients’ questions about various subjects affecting their everyday life. 24 Moreover, a specialized center can also be integrated into a multidisciplinary center to group all the competences required. 25
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
