Abstract
Keywords
Atherosclerosis has significant inflammatory and lipid components that lead to adverse cardiovascular events, driven in part by high-risk, vulnerable coronary plaque rupture.1,2 Lipid-rich necrotic core (LRNC) is a rupture-prone plaque feature predictive of future cardiovascular events and linked with myocardial infarction. 3 We previously showed that psoriasis, a chronic inflammatory disease predisposing to accelerated atherosclerosis, had increased LRNC prevalence that attenuated with biologic therapy over 1 year. 4 These findings relied on statistical models that could only assess a few variables to predict complex outcomes. However, preventative treatment centers on the ability to correctly assess cardiovascular risk and clinical outcomes given a multitude of data variables from a patient. Machine learning (ML) algorithms provide such assessment by analyzing multiple variables from complex datasets with minimal statistical bias. We previously leveraged ML algorithms to identify top predictors of noncalcified coronary plaque, a marker of subclinical atherosclerosis, in psoriasis. 5
Therefore, we aimed to utilize similar ML algorithms to (a) determine top predictors of LRNC modulation over 1 year and (b) highlight those that associate with progression, or worsening, of LRNC and regression, or improvement, of LRNC over 1 year. Understanding these predictors may elucidate important therapeutic interventions that decrease cardiovascular risk longitudinally, especially for high-risk populations including patients with psoriasis.
The ML algorithm based on random forest was developed using 213 consecutive patient records from the Psoriasis Atherosclerosis Cardiometabolic Disease Initiative (January 2013 through November 2022). LRNC was quantified by previously published methods 4 (Figure 1A) and 110 phenotypic variables were measured at baseline and 1-year follow-up visits (online Supplemental Table 1). Top predictors were selected using variable importance based on fivefold cross-validation. Data were reported as mean ± SD for parametric, median (IQR) for nonparametric, and n (%) for categorical variables. Statistical analysis and machine learning were conducted using STATA 17 (StataCorp, College Station, TX, USA) and R version 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria), respectively. All participants provided written, informed consent, and study protocols were approved by the institutional review board at the National Institutes of Health in accordance with the Declaration of Helsinki.
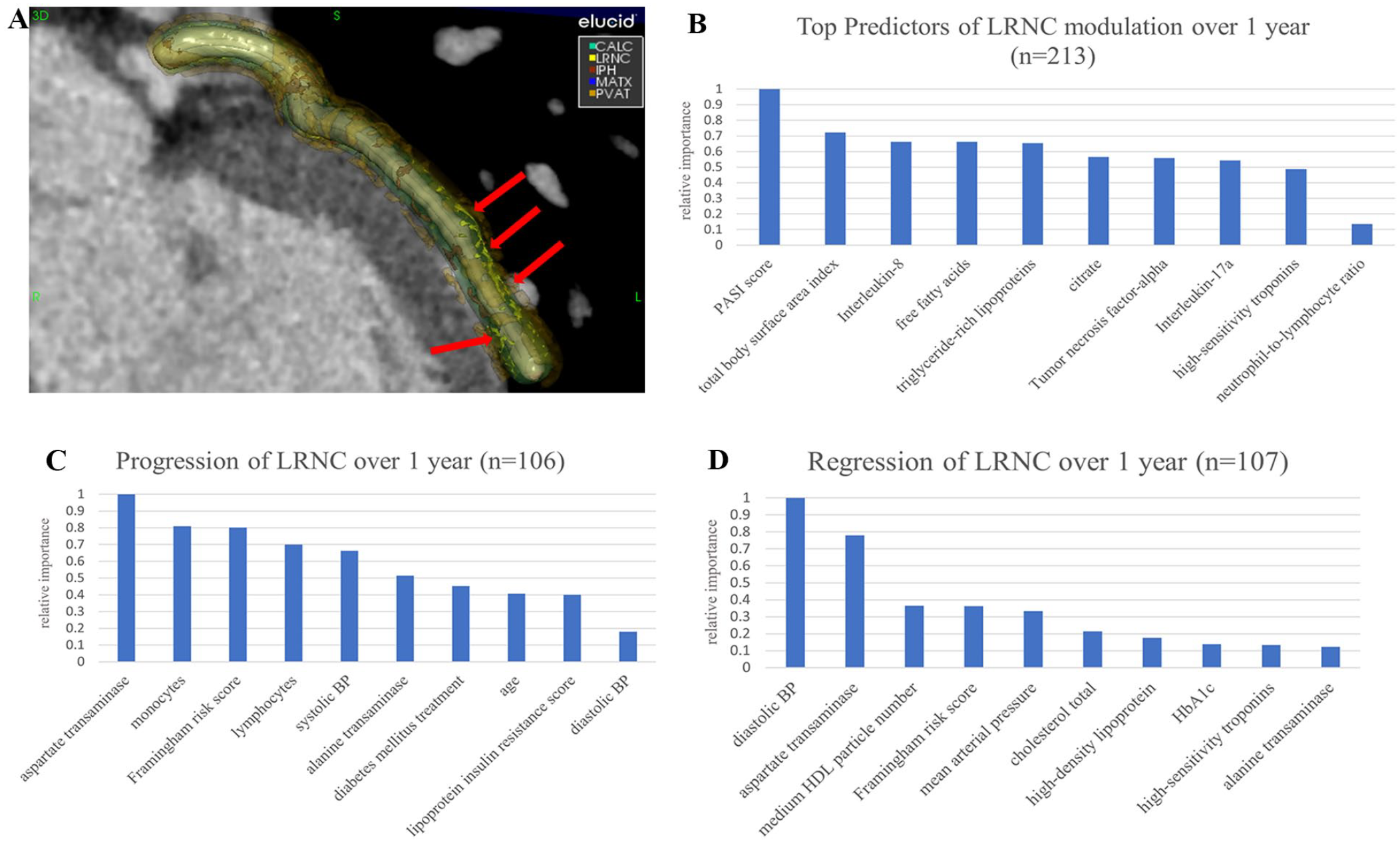
Top 10 predictors in order of relative importance of LRNC modulation over 1 year. (
At baseline, patients with psoriasis were middle-aged (50 ± 12.5 years), predominantly men (62%), overweight (body mass index: 28.55 [25.15–32.15]), and had mild-to-moderate skin disease as measured by the Psoriasis Area Severity Index (PASI) score (5.4 [2.8–9.75]). Based on the target variable, delta LRNC, the remaining variables were ranked for importance using the random forest algorithm, with a value of 1.0 indicating highest importance in predicting delta LRNC over 1 year. The top 10 predictors are listed by importance for the complete cohort (n = 213) in Figure 1B and both subgroups at 1 year (LRNC progression, n = 106; LRNC regression, n = 107) in Figures 1C and 1D. For the complete cohort, top predictors were PASI score, total body surface area index (TBSAI), interleukin (IL)-8, free fatty acids, triglyceride-rich lipoproteins, citrate, tumor necrosis factor (TNF)-alpha, IL-17a, high-sensitivity troponins, and neutrophil-to-lymphocyte ratio (online Supplemental Table 2). For the LRNC progressor cohort, top predictors were aspartate transaminase (AST), monocytes, Framingham risk score (FRS), lymphocytes, systolic blood pressure (BP), alanine transaminase (ALT), diabetes mellitus treatment, age, lipoprotein insulin resistance score, and diastolic BP (online Supplemental Table 3). For the regressor cohort, top predictors were diastolic BP, AST, medium high-density lipoprotein (HDL) cholesterol particle number, FRS, mean arterial pressure, cholesterol total, HDL, HbA1c, high-sensitivity troponins, and ALT (online Supplemental Table 4).
Through this study, we demonstrate the clinical applicability of machine learning in assessing complex cardiometabolic variables to identify pathophysiological pathways potentially implicated in LRNC modulation. We utilized ML to assess the whole population and LRNC progressors and regressors, providing a nuanced understanding of specific predictors associated with LRNC modulation over 1 year. Analyzing these predictors over time provides understanding of the short-term effects of atherosclerotic-related changes that will have robust effects on future cardiovascular outcomes.
Generally, psoriatic disease severity, inflammatory cytokines, and lipoprotein dysfunction were the most important predictors of LRNC. When examining those with worsening LRNC, inflammatory mediators, and traditional cardiovascular risk factors were top predictors. Interestingly, several common predictors were identified for LRNC progression and regression, including diastolic BP and AST. Because our ML algorithms are optimized for identifying the predictive capacity of risk factors, the nature of the relationships between predictors and LRNC modulation are better elucidated through partial dependency plots (PDP), which capture linear, monotonic, and complex relationships (online Supplemental Figures 1 and 2). Indeed, a PDP of HbA1c, a known predictor of diabetes, and LRNC regression showed that a decrease in HbA1c conferred a slight increase in LRNC disease improvement in patients with normal and prediabetic HbA1c values (online Supplemental Figure 2). These findings underscore the importance of early cardiovascular risk prevention and control of cutaneous disease at baseline to prevent high-risk coronary plaque formation and progression.
The main limitation of our study was its observational nature, subjecting to potential unmeasured confounders. Recorded hard cardiovascular events were not studied. Finally, larger studies with longer follow-up are necessary to understand whether ML is effective for longitudinal analyses.
Our findings highlight the importance of assessing cardiovascular risk factors, inflammation, and lipid dysfunction to prevent LRNC modulation over 1 year in patients with psoriasis. We show that ML can be leveraged to identify potential novel markers and disease pathways that may not be overtly detected by traditional analytical methods. Thus, ML can potentially provide a more individualized risk assessment of a patient’s subclinical disease and future cardiovascular event risk, given the patient’s baseline clinical features. Further exploration into these top predictors of LRNC longitudinally may yield insight into the treatment of inflammation and prevention of comorbidities and adverse cardiovascular events in psoriasis.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231171948 – Supplemental material for Machine learning demonstrates top predictors of lipid-rich necrotic core modulation over 1 year in psoriasis
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231171948 for Machine learning demonstrates top predictors of lipid-rich necrotic core modulation over 1 year in psoriasis by Christin G Hong, Haiou Li, Philip M Parel, Alexander R Berg, Nidhi Patel, Harry Choi, Heather L Teague, Eric Munger, Andrew J Buckler, Alexander V Sorokin and Nehal N Mehta in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X231171948 – Supplemental material for Machine learning demonstrates top predictors of lipid-rich necrotic core modulation over 1 year in psoriasis
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X231171948 for Machine learning demonstrates top predictors of lipid-rich necrotic core modulation over 1 year in psoriasis by Christin G Hong, Haiou Li, Philip M Parel, Alexander R Berg, Nidhi Patel, Harry Choi, Heather L Teague, Eric Munger, Andrew J Buckler, Alexander V Sorokin and Nehal N Mehta in Vascular Medicine
Footnotes
Acknowledgements
We thank our participants for their generous contribution to research endeavors. We also thank the clinical care team of the NIH Clinical Center Outpatient Clinic-7 for their care of our participants.
Declaration of conflicting interests
Dr Mehta has served as a consultant for Amgen, Eli Lilly, and Leo Pharma, receiving grants/other payments; as a principal investigator and/or investigator for AbbVie, Celgene, AstraZeneca, Janssen Pharmaceuticals, Inc., Abcentra, and Novartis, receiving grants and/or research funding; and as a principal investigator for the National Institutes of Health, receiving grants and/or research funding. Dr Buckler is an employee and shareholder of Elucid Bioimaging Inc. All other authors have no disclosures to report.
Funding
This study was funded by the National Heart, Lung, and Blood Institute Intramural Research Program in Bethesda, Maryland (HL006193-07). This research was made possible through the NIH Medical Research Scholars Program, a public–private partnership supported jointly by the NIH and contributions to the Foundation for the NIH from the Doris Duke Charitable Foundation, Genentech, the American Association for Dental Research, the Colgate-Palmolive Company, and other private donors. The funding sources had no impact or role in the study design or conduct; collection, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Data availability
The study data are available from the corresponding author upon reasonable request.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
