Abstract
Over a third of patients surviving acute pulmonary embolism (PE) will experience long-term cardiopulmonary limitations. Persistent thrombi, impaired gas exchange, and altered hemodynamics account for aspects of the postpulmonary embolism syndrome that spans mild functional limitations to debilitating chronic thromboembolic pulmonary hypertension (CTEPH), the most worrisome long-term consequence. Though pulmonary endarterectomy is potentially curative for the latter, less is understood surrounding chronic thromboembolic disease (CTED) and post-PE dyspnea. Advances in pulmonary vasodilator therapies and growing expertise in balloon pulmonary angioplasty provide options for a large group of patients ineligible for surgery, or those with persistent postoperative pulmonary hypertension. In this clinical review, we discuss epidemiology and pathophysiology as well as advances in diagnostics and therapeutics surrounding the spectrum of disease that may follow months after acute PE.
Introduction
The postpulmonary embolism (PE) syndrome is a spectrum of persistent cardiopulmonary limitations in the chronic phase of PE.1–4 Chronic thromboembolic pulmonary hypertension (CTEPH) is the most severe manifestation, but those with persistent dyspnea, measurable exercise intolerance, and residual pulmonary vascular obstruction without pulmonary hypertension (PH) comprise a larger group (Figure 1).1,2 Mechanisms of thrombus nonresolution, a common pathophysiologic link, are partially understood though prevention remains elusive. 4 However, established and developing therapies offer a potential cure and symptomatic relief to patients on the continuum of the post-PE syndrome. In this review, we discuss the epidemiology, pathophysiology, diagnosis, and treatment options of the conditions encompassing the post-PE syndrome.
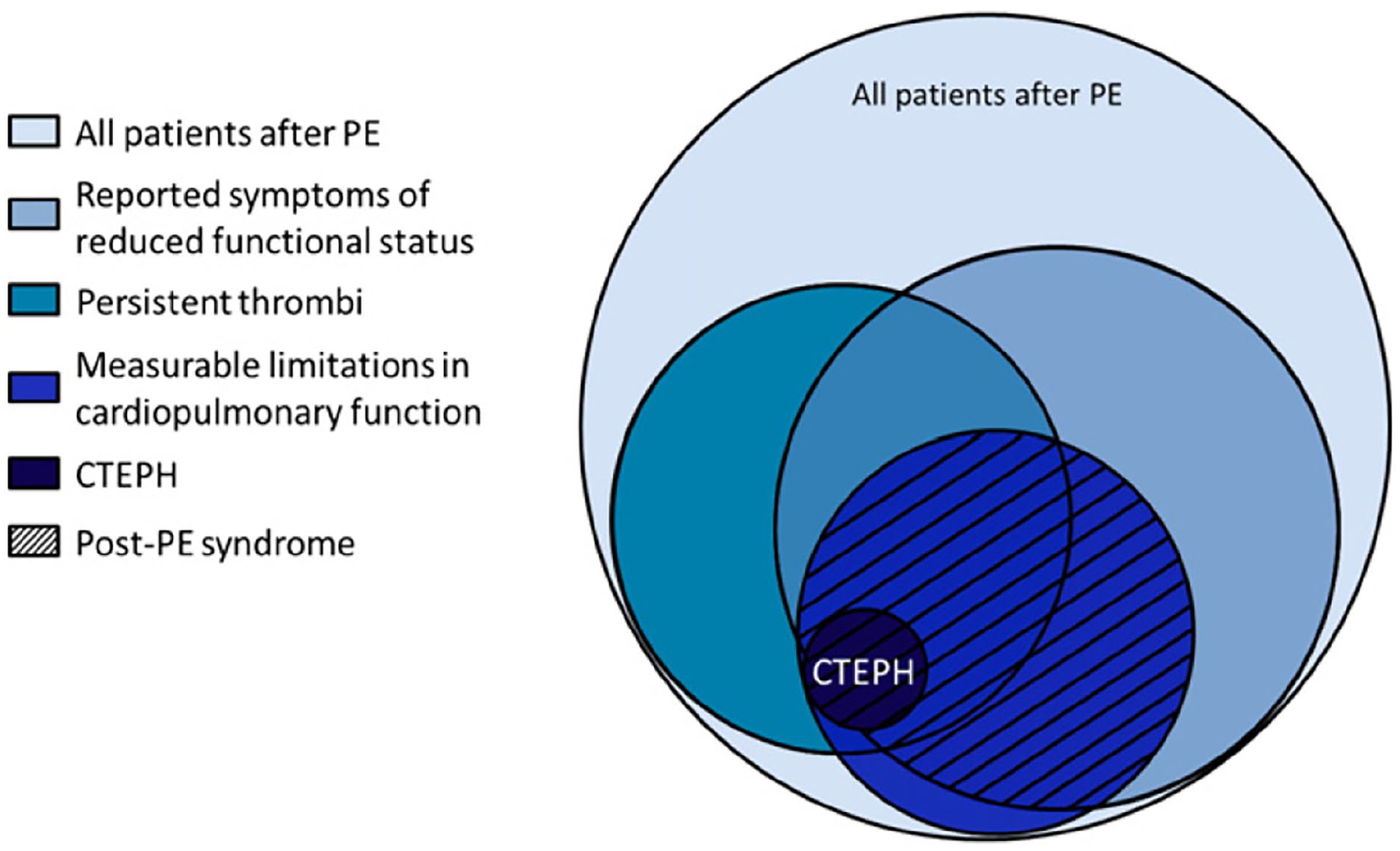
Overview of the chronic consequences of pulmonary embolism.
Epidemiology
In the United States, incidence of acute PE is approximately 115 per 100,000 population; approximately 60,000–100,000 individuals will die from this disease yearly. 5 Among survivors, morbidity is considerable. Between 30% and 50% of patients experience chronic dyspnea months to years after the initial event.1–3,6 The incidence of CTEPH, confirmed PH by right heart catheterization (RHC) in conjunction with imaging criteria, is significantly lower but represents the most severe form of the post-PE syndrome. In a large meta-analysis including 4047 patients evaluated at 3–6 months following diagnosis of acute PE, CTEPH developed in 0.56% of ‘all-comers’, 3.22% of survivors, and 2.79% of survivors without major co-morbidities.7,8 Therefore, by the most conservative estimates, 3000 cases of CTEPH would be diagnosed yearly. Among operated and nonoperated patients, 3-year survival is 89% and 70%, respectively. 9
Although in most cases CTEPH is associated with prior documented PE, approximately 25% of patients do not carry such history.3,10 By contrast, only 15–33% of patients with CTEPH had a history of antecedent PE in a Japanese cohort. 11 Several retrospective studies identified multiple radiographic markers of CTEPH to be present on initial CT scans of patients ultimately diagnosed with the disease; the first presentation of CTEPH may simply mimic acute PE.12,13 Despite these findings, diagnostic delay may be significant. An international prospective study of 679 patients with CTEPH found the median time from symptom onset to diagnosis to be 14.1 months. 10 In this registry, over 80% were classified as having New York Heart Association (NYHA) class III/IV symptoms with a reduced median 6-minute walk distance (6MWD) of 329 feet at diagnosis suggesting advanced disease. 10
Patients with chronic thromboembolic disease (CTED) have imaging findings similar to those with CTEPH but do not meet criteria for PH at rest.2,4 Exercise limitation in this group may be related to exercise-induced PH or dead-space ventilation.1,7,14 Though the true incidence of CTED is unknown, the revised definition of PH to a mean pulmonary artery pressure of > 20 mmHg from > 25 mmHg by the 6th World Health Symposium on Pulmonary Hypertension may lead to reclassification of a proportion of patients with CTED to CTEPH. 15 Unresolved thrombi contribute to both entities, with CTED representing an intermediate phenotype.1,11,16,17 In one study, residual pulmonary thromboemboli were detected in 29% of patients at 12 months following acute PE and were associated with functional limitations. 18 Whether residual obstruction directly influenced symptoms was not established. 18
Whereas a minority of patients develop CTED and CTEPH following acute PE, persistent dyspnea with NYHA class ⩾ II symptoms is common, affecting up to 50% of individuals, with 10–20% experiencing class ⩾ III symptoms.3,6,19,20 The nature of post-PE dyspnea is controversial. In one study of patients 12 months after acute PE, 46.5% had reduced peak VO2 to < 80% predicted on cardiopulmonary exercise testing (CPET). The limitation was mostly attributable to deconditioning rather than circulatory or ventilatory impairment, and findings did not correlate with residual pulmonary vascular obstruction nor perfusion imaging.2,21 Plausibly, underlying comorbid conditions in conjunction with deconditioning contribute to this manifestation of the post-PE syndrome.2,20
Risk factors
Of patients who go on to develop CTEPH, 75% have had a prior documented PE and 33% have had recurrent PE.10,22,23 Multiple retrospective studies identified risk factors associated with the development of CTEPH.22,24 In a large analysis of 772 patients, unprovoked PE, hypothyroidism, symptom onset > 2 weeks before PE diagnosis, and right ventricular (RV) dysfunction were factors most predictive for the development of CTEPH. In a multivariable model, the first three factors carried the most weight. 23 Compared to patients with other forms of PH, ventriculoatrial shunts and infected pacemaker leads, history of splenectomy, non-O blood groups, thyroid replacement therapy, and history of malignancy were associated with CTEPH.22,24,25 Postulated mechanisms include increased thromboembolic potential. For instance, thyroid replacement therapy increases von Willebrand factor (vWF) levels which are up to 30% lower in individuals with blood group O. 26
Pathophysiology
The pathophysiology of CTEPH and CTED rests on the embolic hypothesis – unresolved pulmonary thromboemboli lead to increased pulmonary vascular resistance with downstream small vessel remodeling.3,26,27 Complete thrombus resolution may only occur in 70–85% of patients 12 months following acute PE, though smaller studies have reported rates as low as 30%.17,18,28 Historically, an alternative theory postulated a primary arteriopathy and endothelial dysfunction leading to in situ thrombosis. Thrombi were viewed as sequalae rather than inciting events. 27 Though the former theory has greater acceptance, it remains unclear as to why thromboemboli fail to resolve in a subset of patients. Importantly, these chronic sequelae of PE are not only related to organized thrombi, but to resultant small vessel arteriopathy.26,29
Percutaneous pharmacochemical therapies in acute PE are an area of active research. 30 Whether such interventions alter the natural history and long-term PE sequela is unknown. Recent data highlight that thrombus resolution occurs within a restricted window following restoration of blood flow; benefits may be limited if reperfusion occurs not only too late, but also too early. 31 An understanding of the mechanisms of thrombus nonresolution may inform our future approach to medical and interventional therapies.
Thrombus nonresolution
Thrombus nonresolution has been attributed to abnormalities of coagulation, fibrinogen, fibrinolysis, and impaired angiogenesis.26,29,32,33 Chronic thromboembolic diseases may be associated with a ‘nonclassical’ thrombophilia. 32 Typical thromboembolic risk factors such as factor V Leiden and protein C and S deficiency are not increased in patients with CTEPH. However, higher levels of factor VIII, vWF, and antiphospholipid antibodies have been identified.26,29,32,34 Morris and colleagues demonstrated that fibrinogen purified from patients with CTEPH is resistant to plasmin-mediated lysis compared to fibrin from healthy controls; fibrinogen genotype and allele frequencies also differ between patients with CTEPH and control subjects.32,35 Interestingly, abnormal levels and reduced activity of tissue plasminogen activator (tPA) and plasminogen activator inhibitor (PAI)-1 have not been shown to result in thrombus persistence. 26
Inflammation is thought to contribute to CTEPH. C-reactive protein (CRP), interleukin (IL)-10, and matrix metalloproteinase (MMP)-9 levels are higher in patients with CTEPH and surgical samples following pulmonary endarterectomy (PEA) reveal high levels of neutrophils and macrophages correlating with inflammatory makers.29,36 Neovascularization is known to enhance thrombus resolution and animal models of defective angiogenesis demonstrate increased thrombus volume. Injection of vascular endothelial growth factor (VEGF) augments thrombus resolution in a rat model to further support the hypothesis. 26 Overall, the fibrotic transformation of thrombi is thought to resemble the natural process of wound healing. Over time, the fibrin-rich thrombus dense with erythrocytes transforms to one that is acellular, endothelialized, and predominated by connective tissue. 26
Recent studies have highlighted the role of transforming growth factor (TGF)-β, a growth factor involved in the processes of angiogenesis and fibrosis, in thrombus nonresolution.37,38 Thromboendarterectomy specimens demonstrate neutrophil-mediated inflammation, and neutrophil extracellular traps (NETs) enhance TGF-β signaling, leading to thrombus fibrosis. 39 In an animal model, targeting NETs with DNase1 diminishes fibrosis and promotes thrombus resolution. 39 Bochenek and colleagues demonstrated that platelet-specific deletion of TGF-β1 in mice accelerated venous thrombus resolution; activated TGF-β signaling was noted in specimens obtained from patients with CTEPH. 37 Additionally, endothelin-1 (ET-1) antagonization (a therapeutic target in PH) may reverse overactivated TGF-β signaling, providing a mechanistic link behind this therapy beyond its vasoactive effects. 37
Splenectomy and CTEPH
An increased prevalence of asplenia among patients with unexplained PH and CTEPH has been established.40,41 The risk of PE 90 days following splenectomy is over 30-fold that of the general population. 42 Beyond underlying hematologic disorders, splenectomy is thought to predispose patients to PH as a result of increased thrombus formation in conjunction with delayed thrombus resolution.40,43 Enhanced thrombogenicity may be related to platelet-derived microparticles and anionic phospholipids on red cell membranes occurring following the loss of splenic filtration. Phospholipids may inhibit neovascularization leading to thrombus nonresolution.40,44 Thrombocytosis following splenectomy can lead to downstream platelet activation and endothelial damage. Nitric oxide depletion by senescent red cells may further promote vasculopathy. Although guidance is limited, patients who have undergone splenectomy may benefit from close monitoring for the development of PH. 44
Secondary arteriopathy
Progression from unresolved thrombi to CTEPH is in part the result of a secondary pulmonary vascular remodeling. Pathologic studies reveal plexiform lesions and intimal fibromuscular proliferation akin to patients with idiopathic PH.29,45 Increased resistance in obstructed arteries diverts blood flow to nonobstructed segments. Hyperdynamic flow promotes endothelial proliferation and pathologic vasoconstriction; massive microvascular remodeling has been identified in lungs of patients following PEA.32,46
Systemic blood flow to the lungs via bronchial artery collaterals can be up to 30% higher in patients in CTEPH compared to healthy individuals, representing 3% of total cardiac output. Resultant fistulous anastomoses between bronchial arteries and pulmonary veins produces arterialization and vessel wall thickening of the latter.32,47 On a molecular level, nitric oxide (NO) levels are reduced leading to a reduction in soluble guanylate cyclase (sGC) and cyclic guanosine monophosphate (cGMP) levels with resultant loss of smooth muscle relaxation.29,48 Molecular pathways leading to vascular remodeling have been exploited for the purpose of drug development. Clinically, small vessel disease is not amenable to PEA and may result in persistent PH postoperatively.
Characterization of post-PE dyspnea and CTED
Most research into the post-PE syndrome concerns CTEPH whereas CTED and long-term cardiopulmonary limitations following PE are less well characterized. In the 6 months following acute PE, 20–70% of patients report worse health status than at the time of diagnosis. 1 In a meta-analysis of 3671 patients at a median of 18 months post-PE, 33.2% had at least mild functional impairment, with an 18.1% prevalence of RV dysfunction. Pooled 6MWD was in the 5th percentile and quality of life scores were in the 40th. 20 In a study of 607 patients with post-PE dyspnea, a predefined alternative diagnosis was present in all patients, suggesting this manifestation of post-PE syndrome may be unrelated to the thromboembolic event. 6 CPET 6–12 months following acute PE identifies objective exercise impairment occurs in 46–70% of patients, as evidenced by peak VO2 < 80% predicted.2,21,49 However, these limitations do not seem to be attributable to RV-pulmonary coupling, ventilatory impairment, nor residual pulmonary vascular obstruction.21,49
In the Evaluation of Long-term Outcomes after PE (ELOPE) study, despite a high prevalence of functional impairment, < 6% had residual pulmonary vascular obstruction and most patients had low-risk PE. 21 Such limited perfusion defects unlikely fully explain symptomatology. Further, the degree of residual pulmonary vascular obstruction at 6 and 12 months did not correlate to percent predicted VO2. 50 In a more recent prospective study of 880 patients following acute PE, post-PE impairment (a combination of functional, imaging, and laboratory parameters) had a 16% 2-year incidence. 51 Identifying those with post-PE impairment allowed for the selection of most individuals who would develop CTEPH. To better understand post-PE dyspnea, functional studies would be required to define abnormal RV-pulmonary artery coupling and vascular limitations to exercise. 2 In our current model, co-morbid diagnoses and deconditioning are thought to play a substantial role.
CTED is thought to represent an intermediate clinical phenotype between normal and CTEPH – patients do not develop RV failure and have lesser functional limitations.2,14,52,53 By contrast to post-PE dyspnea, CPET in this population provides objective evidence of functional impairment that is related to ineffective ventilation and reduced pulmonary perfusion. 54 Compared to healthy controls, RV contractile reserve is diminished, as is pulmonary compliance with resultant mismatch of contractility and afterload. 14 In a small study of patients with CTED undergoing exercise RHC, the majority demonstrated PH with exercise (mean pulmonary artery pressure (mPAP) ⩾ 30 mmHg with mPAP/cardiac output slope of > 3 Wood units). 52 Hemodynamic parameters and symptoms are known to improve following PEA in this cohort.52,53 Several studies identified ‘borderline elevated’ resting mPAP in patients with CTED just above 20 mmHg; by the most recent definition of PH, these patients would be reclassified as having CTEPH.14,15,52 Presently, the natural history of CTED is unknown and there is no evidence to suggests it evolves into CTEPH. 7 Application of CTEPH guidelines to this population is not recommended.
Screening and diagnostic testing
Screening asymptomatic survivors of acute PE for chronic thromboembolic disease is not currently recommended.55,56 In a cohort of 866 patients followed for an average of 34 months after acute PE, a structured screening program was not superior to routine clinical care. 57 By contrast, in a prospective study of patients with persistent symptoms ⩾ 3 months following acute PE, 8.5% were diagnosed with CTED or CTEPH. 56 As most thrombus resolution should be achieved at 3 months, persistent symptoms thereafter merit diagnostic testing.16,58 Further, Klok and colleagues recommend additional evaluation for those with evidence of CTEPH already at the time of PE diagnosis as well as for those with significant risk factors.16,55,59 Importantly, the absence of prior PE by history does not exclude the possibility of CTEPH as 25% of those ultimately diagnosed do not carry such a history. 55
Dyspnea is universally present but is nonspecific as many patients with persistent dyspnea can typically have alternative diagnoses. 10 With advanced disease, signs and symptoms of RV dysfunction such as jugular venous distension, right-sided S3, exertional angina, and syncope may be present. Proposed diagnostic algorithms combine imaging and functional or hemodynamic studies with transthoracic echocardiography (TTE) as the initial test of choice (Figure 2). Among patients with prior PE, an electrocardiogram (ECG) and N-terminal (NT)-pro hormone-BNP levels can effectively rule out CTEPH; normal levels without ECG findings of RV dysfunction carry a negative predictive value of 99%. 60 This approach was not prospectively validated and does not exclude CTED.
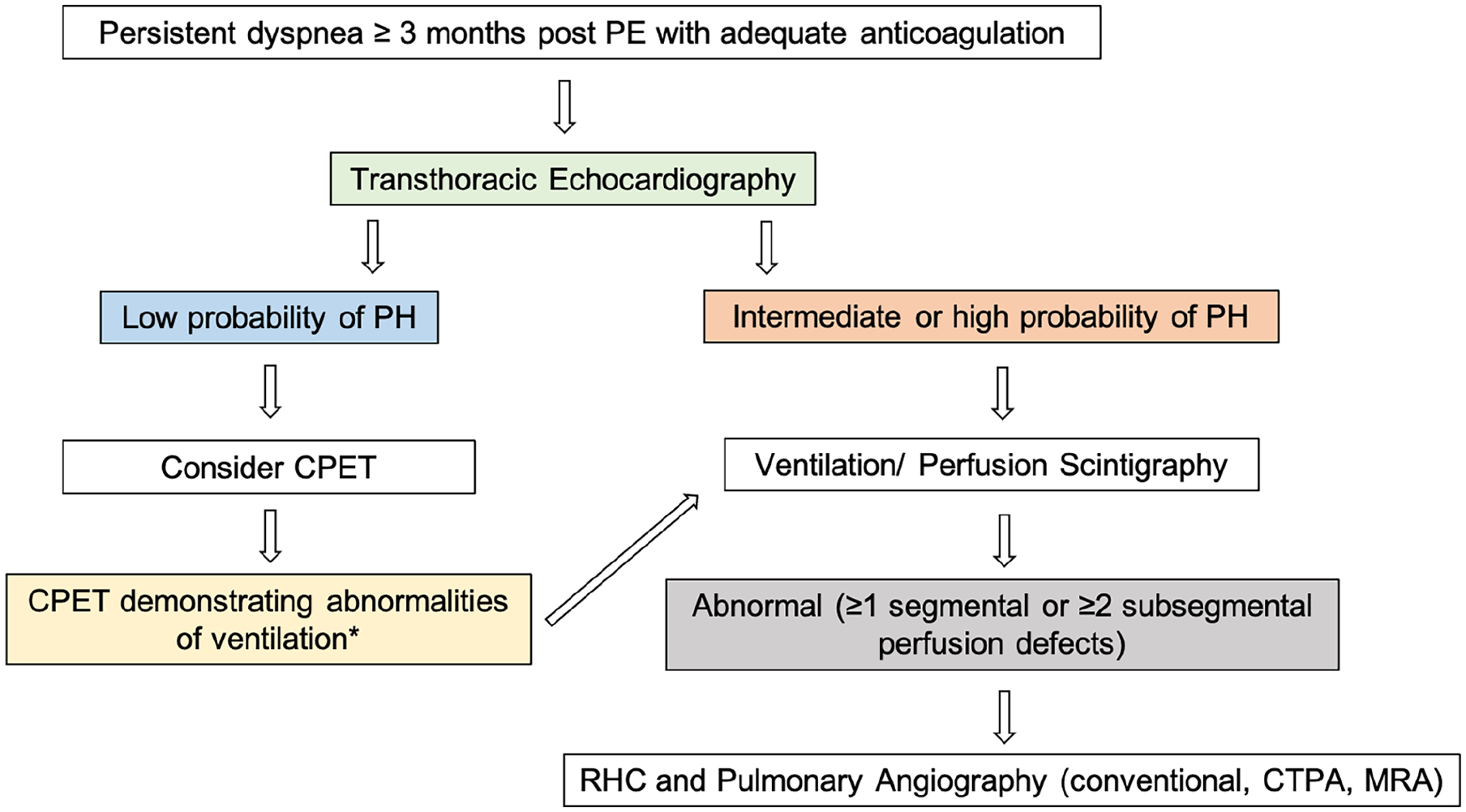
Chronic thromboembolic disease diagnostic algorithm. Patients should be anticoagulated for at least 3 months following acute PE prior to workup of chronic thromboembolic disease. Probability of PH by echocardiography is based on estimated pulmonary artery systolic pressure with additional imaging findings, as described in the text.
Imaging
TTE should be the primary imaging modality used in the evaluation of PH. In addition to imaging of right heart structure and function, pulmonary artery systolic pressure (PASP) can be estimated via spectral Doppler. A tricuspid regurgitant maximum velocity of ⩾ 2.8 m/s, corresponding to an estimated PASP of ⩾ 36 mmHg, raises the suspicion of PH with values ⩾ 3.4, making the diagnosis likely.16,59,61 PASP in conjunction with additional imaging and Doppler findings determines echocardiographic probability of PH. 59 Specific echocardiographic criteria are described in the 2015 European Society of Cardiology/European Respiratory Society (ESC/ERS) guidelines for the diagnosis and treatment of PH. 59 In a large retrospective study, Doppler echocardiography carried a Pearson’s correlation coefficient of 0.87 when compared to RHC; however, other studies provided less optimistic agreement.61,62 Notably, as RV function should be preserved in CTED and resting pulmonary artery pressures are not elevated, a low-probability TTE does not conclude the workup of post-PE syndrome.
Ventilation/perfusion (V̇/Q̇) scintigraphy remains the cornerstone screening test for CTED/CTEPH (Figure 3). In a study of 227 patients, sensitivity and specificity were 96–97% and 90–95%, respectively, though slightly higher values have been reported for both.63,64 A normal V̇/Q̇ scan effectively excludes the disease with a negative predictive value ⩾ 98%. 64 Planar scintigraphy is the current diagnostic standard, yet single-photon emission computed tomography (SPECT) V̇/Q̇ may have improved diagnostic performance. 65 However, V̇/Q̇ scintigraphy may underestimate the degree of vascular obstruction and is unable to distinguish between causes of mismatch (i.e., CTEPH vs extrinsic pulmonary artery obstruction, fibrosing mediastinitis, vascular tumor).63,66 Partial vascular recanalization may lead to tracer redistribution despite significant pathology. At least one segmental or two or more subsegmental perfusion defects are required to make the diagnosis; guidelines recommend a binary interpretation rather than the familiar probabilistic scores.56,65
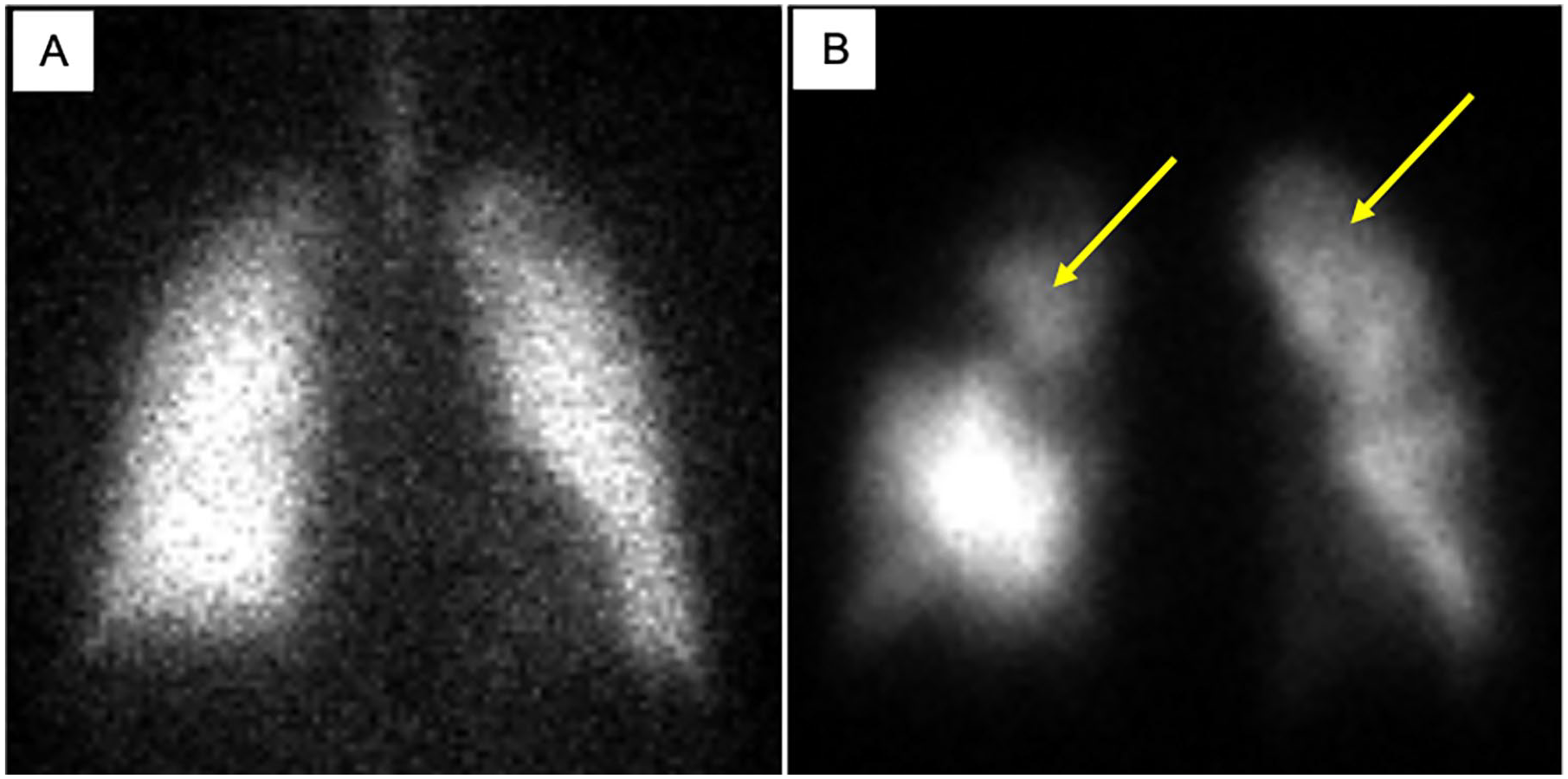
Ventilation/perfusion scintigraphy in chronic thromboembolic disease.
Though computed tomography pulmonary angiography (CTPA) is 99% specific, a sensitivity of 51% limits its utility as a screening test. 64 A more recent study suggested a narrower difference with reported sensitivity of 92.2% possibly related to more proximal lesions, expert centers, and newer generation scanners. 63 CTPA offers important data not provided by V̇/Q̇ scintigraphy and is a reasonable follow-up test when perfusion defects are encountered. Notable findings in CTEPH include dilatation of the main pulmonary artery along with asymmetric dilatation of proximal vessels in addition to intraluminal webs and bands or abrupt vessel narrowing. Chronic thrombi are distinct from acute PE; organization can mimic vascular wall thickening at times with calcification (Figure 4).63,67 Dual-energy CT (DECT) allows for the simultaneous generation of a CT angiogram with an iodine map of relative regional perfusion to the parenchyma (Figure 4C).63,68 This modality may allow for the detection of more distal disease and reflect microvascular circulation. In a small study, Masy and colleagues found excellent agreement between DECT and V̇/Q̇ scintigraphy. 69 As 43% of patients with PH never undergo V̇/Q̇ scanning, DECT is appealing as a ‘one-stop’ approach. 63
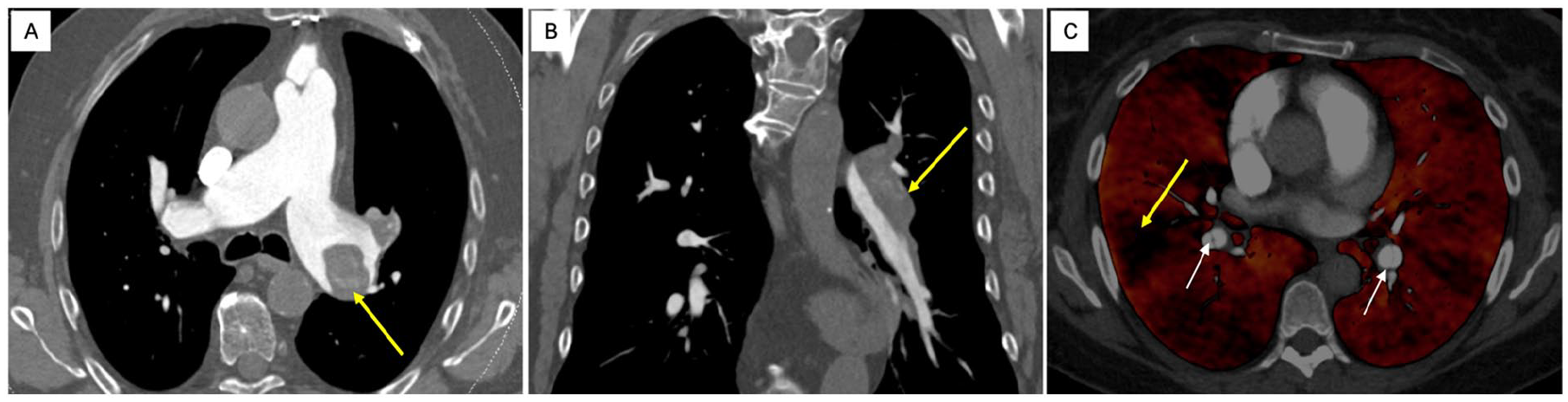
CTPA in chronic thromboembolic disease.
Despite advances in imaging modalities, conventional pulmonary angiography remains the gold standard test for vascular characterization in CTEPH and for the purpose of surgical planning.11,59,63 Angiographic findings differ from the distinct filling defects encountered in acute PE and are reflective of organization and recanalization. 70 Findings include abrupt vessel narrowing, webs, pouch defects, and complete pulmonary artery obstruction (Figure 5).70,71 Additionally, angiography allows for the assessment of subpleural perfusion, which is associated with distal disease and surgical risk. 56 Despite its invasive nature, angiography can be combined with RHC in the assessment of the patient’s hemodynamic profile. Whether CTPA with newer generation scanners can substitute for invasive angiography in the evaluation of lesions for surgical consideration remains unknown.
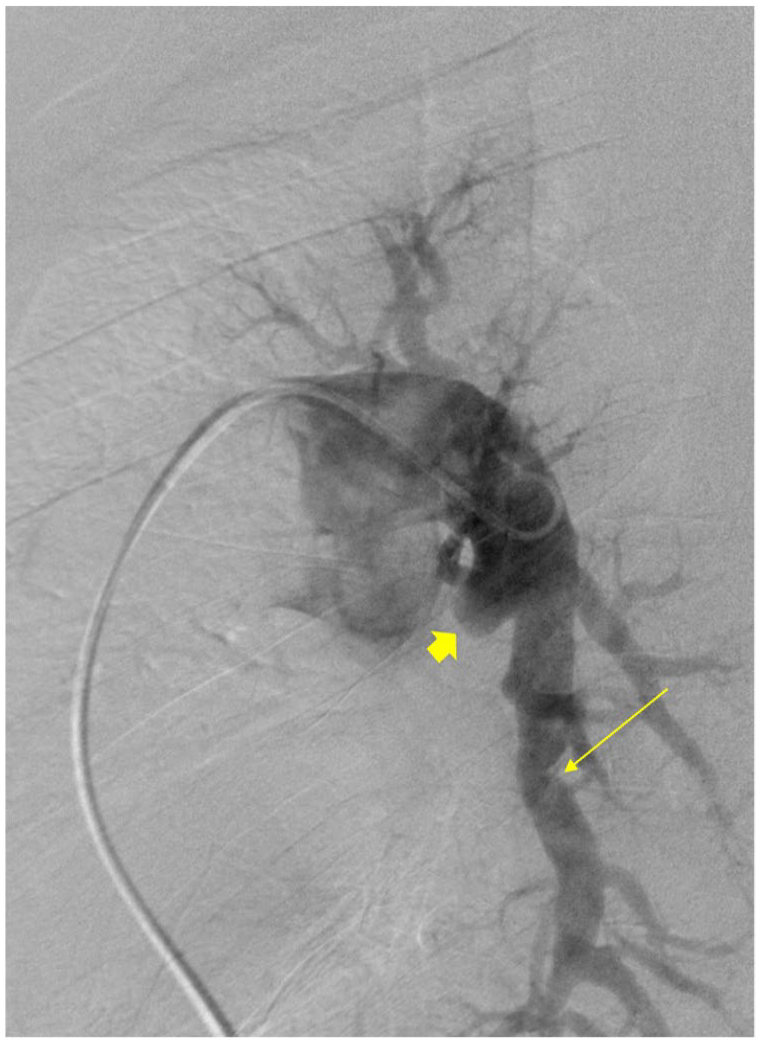
Pulmonary angiography in chronic thromboembolic pulmonary hypertension. Conventional angiography with findings consistent with chronic thromboembolic pulmonary hypertension. Diffuse tortuosity of the segmental and subsegmental vessels. Pouch defect/abrupt cutoff of the anteromedial basal segment of the left lower lobe (short arrow). Web demonstrated in the posterior basal branch of the left lower lobe (long arrow).
Hemodynamic and functional assessment
The diagnosis of CTEPH must be confirmed with RHC even in the presence of high probability TTE findings. 16 Notably, discrepancy between pulmonary capillary wedge pressure (PCWP) and left ventricular end-diastolic pressure (LVEDP) has been described. In a large registry, approximately half of the patients diagnosed with pulmonary arterial hypertension by PCWP criteria had elevated LVEDP and would need to be reclassified. 72 The 6th World Symposium on Pulmonary Hypertension defined precapillary PH as: (1) mPAP > 20 mmHg, (2) pulmonary artery wedge pressure ⩽ 15 mmHg, and (3) pulmonary vascular resistance (PVR) ⩾ 3 Wood units. Prior to 2018, guidelines and literature set the upper limit of normal for mPAP at ⩾ 25 mmHg. 59 Criteria were redefined as a mPAP range of 21–24 mmHg, which is > 2 SDs above the mean and is associated with decreased exercise tolerance and worse clinical outcomes.15,59 Prospective studies are required to determine whether this new population would benefit from specific therapies. 73 The presence of chronic thromboembolic lesions in conjunction with RHC criteria confirms the diagnosis of CTEPH. Patients with CTED may have normal resting hemodynamics with PH diagnosed during exercise RHC.52,54
Increased pulmonary arterial load is a key determinant of RV dysfunction and clinical outcomes. Despite normally being a low-pressure, high-compliance system, vascular remodeling in PH contributes to a rise in RV afterload. Abnormalities in pulmonary vascular resistance (‘static’ afterload) and compliance (pulsatile afterload) are associated with adverse outcomes in PH. Compliance is often estimated in clinical practice as SV/PApp, where SV is stroke volume and PApp is pulmonary arterial pulse pressure, and can be determined via RHC, echocardiography, or magnetic resonance imaging (MRI).74,75 Importantly, compliance changes may occur early, even in the setting of normal resting PA pressures and PVR; loss of PA compliance may in fact induce distal proliferative vasculopathy. 74 Plausibly, PA compliance may allow for disease screening and function as a therapeutic target. 74
In the workup of the post-PE syndrome, CPET provides objective evidence of functional limitations and defines the nature of exercise intolerance. Pugliese and colleagues propose CPET follows in the diagnostic investigation of the post-PE syndrome in the setting of a low-probability TTE but with perfusion abnormalities on V̇/Q̇ scintigraphy. 2 Patients with CTEPH and CTED demonstrate evidence of exercise intolerance related to disturbances in gas exchange and ventilation.2,54
Both in PAH and CTEPH, dead space ventilation is increased as a result of reduced perfusion to well-ventilated alveoli. Dead space is reflected as a high ratio of minute ventilation to CO2 production (V̇E/V̇CO2) which can be determined at the anaerobic threshold. 76 Though the hemodynamic profiles of PAH and CTEPH are similar, physiologic dead space and V̇E/V̇CO2 are higher among patients with CTEPH. 76 Further, a few studies have demonstrated improved peak oxygen consumption and markers of dead space among patients with CTEPH treated with medical and interventional therapies. 76 Held and colleagues compared CPET parameters among patients with CTEPH and normal TTEs to controls and demonstrated good diagnostic performance. The capillary to end-tidal carbon dioxide gradient (P[c – ET]CO2) during maximum exercise, also reflective of dead space ventilation, was the most sensitive and specific parameter to discriminate between the two entities at 86% and 88%, respectively. 54
In a small study of patients undergoing CPET, those with CTED demonstrated similar functional limitations to those with CTEPH. 54 Patients with post-PE syndrome not meeting criteria for the aforementioned diagnoses demonstrate decreased peak VO2 related to suboptimal effort. Individuals in this group often fail to achieve peak heart rate and do not demonstrate evidence of inefficient gas exchange. 21 Plausibly, this diagnostic modality can determine degree of exercise impairment and provide distinction between CTED and post-PE dyspnea.
Management
PEA is the treatment of choice in CTEPH and a potential cure.11,59,71,77 Presently, no objective preoperative assessment tool exists, although a patient should have sufficient accessible thromboembolic material in proportion to PVR to exclude extensive secondary vasculopathy. 78 Operability remains a source of debate; experts believe surgical treatment should be explored in all cases of CTEPH, assessed with a multidisciplinary team with second opinions at expert centers explored as needed.78,79 The increased use of medical therapies has led to delays in surgical referral, even in cases of clearly operative disease; medical therapy as a bridge to PEA is not recommented.78–80 Balloon pulmonary angioplasty (BPA) and pulmonary vasodilators are reserved for inoperable, exceedingly high-risk, or residual postoperative PH cases.7,71 In every scenario, lifelong anticoagulation is indicated, along with tailored supportive therapy. Bilateral lung transplantation remains a potential treatment option in cases of inoperable disease or significant residual PH.77,78 A proposed management algorithm is presented in Figure 6.
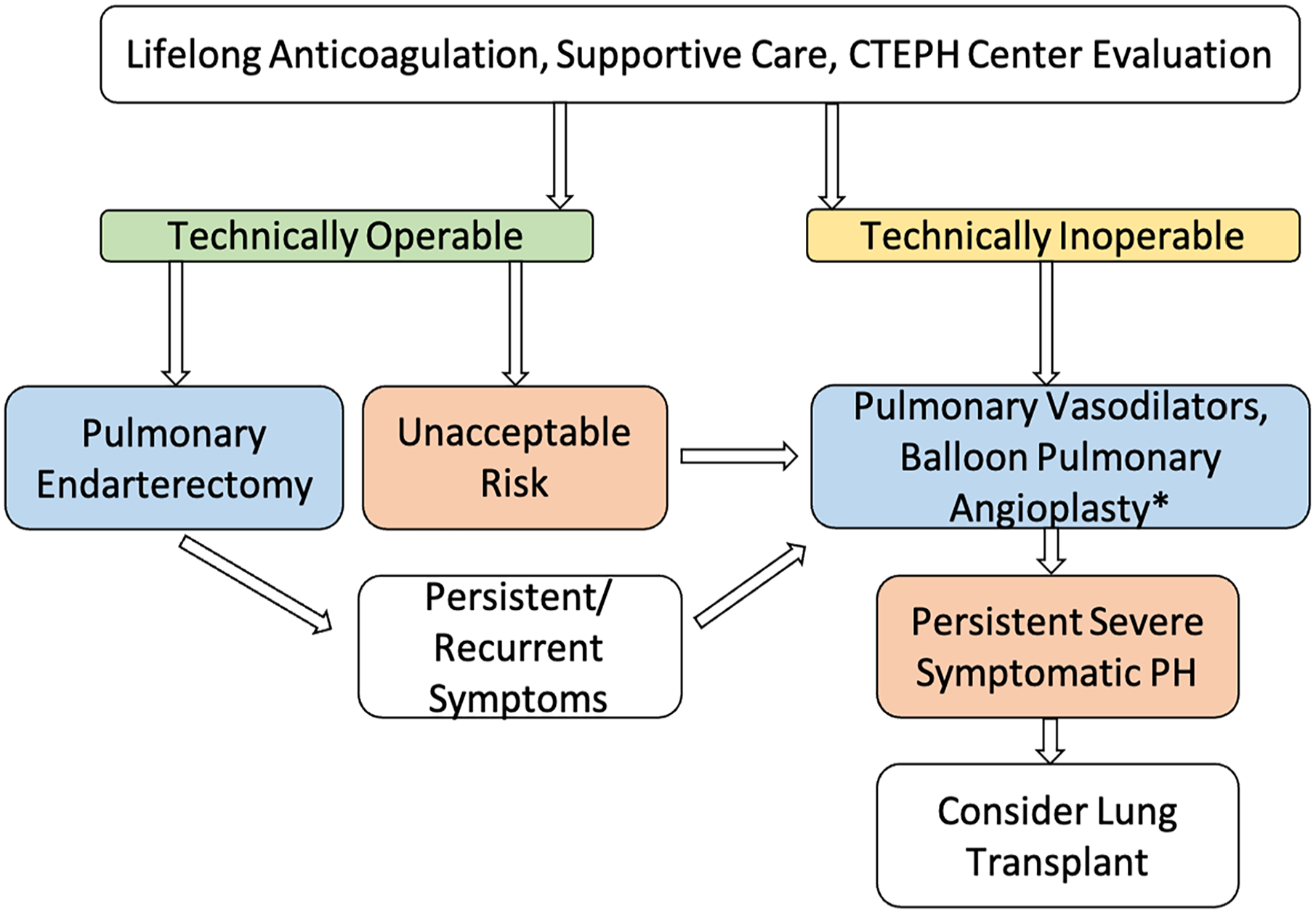
Management algorithm. All patients should be treated with lifelong anticoagulation and supportive therapies such as oxygen and diuretics as needed. Operability should be assessed at a referral center and a second opinion is recommended if a patient is deemed inoperable.
Pulmonary endarterectomy
The PEA technique was developed in the 1960s and standardized at the University of California – San Diego. Surgery is performed via median sternotomy with cardiopulmonary bypass and deep hypothermic circulatory arrest to 20°C as bronchial artery collateral flow otherwise precludes a bloodless field. 71 Obstructive thromboembolic material is cleared as a dissection plane, typically between intima and media, is advanced distally (Figure 7).78,79,81 In a large retrospective study, the mean cardiopulmonary bypass time of PEA was approximately 4 hours with a circulatory arrest time of 36 minutes; circulatory arrest is performed in limited intervals to decrease neurologic complications.71,79
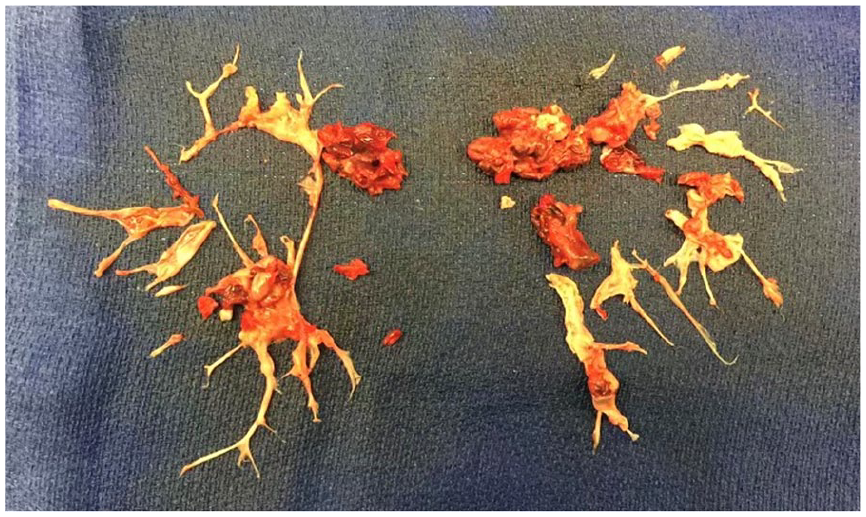
Gross surgical specimen from a bilateral pulmonary endarterectomy.
Operative mortality at experienced centers ranges from 2% to 5%. 79 Preoperative and postoperative hemodynamics influence outcomes. For instance, a residual PVR of > 500 dynes/sec/cm−5 is associated with a 10.3% mortality rate compared to 0.9% among those with a PVR < 500 dynes/sec/cm−5. 79 PEA leads to an immediate improvement and near-normalization of hemodynamic parameters. University of California – San Diego reported a mean postoperative PVR decline from 719 dynes/sec/cm−5 to 253 dynes/sec/cm−5, a 46 mmHg to 26 mmHg improvement in mPAP, and increase in cardiac index.77,79 Patients experience improved NYHA class, 6MWD, and quality-of-life scores as well. 77
Long-term survival rates exceed 80% at 5 years with a relative risk reduction of 63% compared to nonoperated patients.9,78 Mortality in hospitals with > 50 PEAs pear year, compared to 1–10 per year, was 3.4% and 8.8%, respectively, showing the importance of center and operator expertise. 9 More recent reports suggest successful PEA outcomes with very distal disease involving segmental and subsegmental vessels.77–79 Reperfusion lung injury occurs in approximately 10% of patients and can often be managed conservatively and, in severe cases, with veno-venous extracorporeal membrane oxygenation.71,78 Short intervals of deep hypothermic circulatory arrest of approximately 20 minutes are well tolerated without resultant neurologic impairment.77,78
CTEPH-targeted medical therapy
In a large international registry, nearly 40% of patients with CTEPH were deemed inoperable.80,82,83 The most common reason cited related to surgical inaccessibility of disease followed by medical co-morbidities. 84 After PEA, residual PH rates range from 11% to 35%.82,85,86 It is for these patient populations that drugs targeting key molecular pathways exist, although benefits tend to be modest. Importantly, pulmonary vasodilators prior to PEA do not impact outcomes, and may lead to delays in PEA referral. 78 Few randomized control trials have investigated pulmonary vasodilators in this population. Benefits are related to improved hemodynamics and symptoms; however, survival advantage has not been demonstrated.
Endothelial dysfunction as part of the secondary vasculopathy in CTEPH is related to derangement of the NO-sGC-cGMP pathway, and is the target of riociguat, a sGC stimulator approved for inoperable and persistent CTEPH.29,48,82 CHEST-1 was a phase III randomized clinical trial comparing riociguat to placebo for this indication. After 16 weeks, the WHO functional class improved, PVR declined, and the 6MWD increased 39 meters compared to a 6-meter decrease in the placebo arm. 87 Benefits were sustained in a follow-up study with a 2-year survival rate of 93% without significant adverse events compared to placebo. 88 Presently, riociguat is approved by the US Food and Drug Administration and is the preferred agent in this population.
Other PH-specific therapies have been subject to clinical trials in CTEPH. Macitentan, an endothelin receptor antagonist, resulted in lower PVR (73% baseline vs 87.2% baseline) compared to placebo in the MERIT-1 trial. 89 Whereas bosentan leads to a 24% decrease in PVR in the BENFiT trial, the 6MWD was not significantly altered.71,90 Presently, there are no head-to-head comparisons between different medical therapies. Hemodynamic effects of pulmonary vasodilators tend to be similar, although the impact on the 6MWD varies.71,82 Based on limited CPET data, vasodilator therapy may be effective for select patients with a predominant small vessel vasculopathy phenotype of CTEPH.82,91
Balloon pulmonary angioplasty (BPA)
BPA was first used for the management of CTEPH in 1988 with significantly increased uptake just over the last decade, and is a promising treatment option for those ineligible for PEA or those with recurrent PH.92–94 The procedure involves identification of treatable lesions and mechanical disruption with balloon dilations over a guidewire, usually involving three to six sessions.92,93 In a multicenter Japanese registry of 308 patients undergoing a total of 1408 procedures, significant hemodynamic improvement was reported (mean mPAP decline from 43 mmHg to 23 mmHg) with subsequent reduction in the use of pulmonary vasodilator therapy. These effects were sustained for over 3.5 years. Despite a high complication rate, 3-year survival was 94.5%, comparable to PEA data. 95 A large meta-analysis compared 755 patients undergoing BPA to 849 treated with pulmonary vasodilators. BPA resulted in greater improvement in mPAP, PVR, and 6MWD compared to medical therapy, at the expense of increased complications such as vascular injury and reperfusion pulmonary edema. 96 However, the rates of serious complications, notably severe reperfusion lung injury, have decreased from approximately 17% to under 4% with experience. 84
Recently, two multicenter randomized trials compared BPA to riociguat among patients with inoperable CTEPH. Of 61 patients in the Japanese MR BPA trial, 32 were randomized to BPA and underwent a mean of 4.7 procedures. At 12 months, mPAP was significantly reduced in the BPA group (−16.3 mmHg vs −7.0 mmHg). BPA-related complications (hemosputum, hemoptysis, pulmonary hemorrhage) occurred in 44% of patients; however, serious events were not observed. 97 Similarly, 105 patients with inoperable CTEPH were randomized to BPA versus riociguat in the RACE trial. Those undergoing BPA experienced more significant reductions in pulmonary vascular resistance; however, adverse events were more common (35% experienced lung injury). 98 The authors suggest a multimodality approach including BPA, and medical therapy may become the preferred strategy in this patient group. Several questions surrounding BPA remain unanswered including the ideal target population, lesion selection, and optimal combination of the procedure with medical therapies. Ongoing studies aim to address some of these unknowns. 98
Anticoagulation
Lifelong anticoagulation is recommended for all patients with CTEPH even following PEA and BPA. 56 Vitamin K antagonists (VKAs) are considered the standard due to decades of clinical experience; however, direct-acting oral anticoagulants (DOACs) may be appropriate on the basis of individual risk/benefit analysis. 56 Data surrounding DOACs is limited, but in a retrospective study of patients undergoing PEA, VTE recurrence was higher among those treated with DOACs compared to VKAs (4.6%/person-year vs 0.76%/person-year). 99 However, functional, hemodynamic, and survival outcomes were similar. In a separate study of patients undergoing PEA, those treated with DOACs were twice as likely to have associated acute or subacute thrombus compared to those on non-DOAC anticoagulant therapies. 100
In contrast, among 501 patients with CTEPH, major bleeding was more common in those treated with warfarin compared to rivaroxaban, though VTE recurrence was identical. 101 Despite the frequent use of DOACs in this population and several practical advantages over VKAs, important efficacy and safety concerns arise. Anticoagulant selection should be individualized and plausibly DOACs may have a role for those who are unable to adhere to VKAs. Prospective ongoing studies may better inform appropriate anticoagulant selection. 102
Management of CTED and post-PE dyspnea
Historically, a mPAP > 30 mmHg, PVR > 300 dynes/sec/cm−5, and NYHC class ⩾ III symptoms were requisite for PEA.71,79 Current selection criteria are more fluid, and those with less severe hemodynamic impairment may benefit from operative management. Taboada and colleagues first published on 42 patients with CTED treated with PEA. 53 In this cohort, all patients had NYHA class II or III symptoms with a mean mPAP of 21 mmHg and PVR of 164 dynes/sec/cm−5; functional limitations were out of proportion to hemodynamics. With surgery, 95% of patients were alive at 1 year with significant improvement in NYHA class and quality of life. However, perioperative complications were common. 53 Similarly, Pin and colleagues reported a 5-year survival of 97% in a cohort of 48 patients with CTED treated with PEA. 103 Although these studies lacked control groups, older natural history data on patients with mild, or ‘borderline’ PH suggest good prognosis with anticoagulation alone.53,104 Early referral of CTED patients to CTEPH centers for evaluation is recommended. As conservative management is likely a safe choice, the decision to proceed with surgery should consider the extent of the patient’s functional limitations. 53
BPA may serve as an option for inoperable CTED yet data on safety and efficacy is scant; outcomes were reported in two studies.105,106 Wiedenroth and colleagues noted improvement in functional class, 6MWD, and hemodynamics in 10 patients undergoing BPA with a single self-limiting procedural complication. 105 Inami and colleagues described significant improvement in mPAP and PVR on exercise RHC, as well as improved ventilatory efficiency on CPET in 15 patients treated with BPA. 106 With increased therapeutic options, experience with CTED patients will expand. Presently, expert evaluation of these patients at CTEPH centers is most appropriate.
Whereas procedural interventions and targeted medical therapies have been the focus in the management of CTED, cardiopulmonary rehabilitation has been explored in the treatment of post-PE dyspnea. 107 In a small, nonrandomized study, patients with chronic post-PE dyspnea experienced improved 6MWD, endurance, and self-reported health following a 6-week rehabilitation program. 107 Similarly, exercise training was shown to improve hemodynamic measures and quality of life in CTEPH patients in conjunction with other therapies.107,108 However, formal rehabilitation programs are not yet established in the care of the post-PE syndrome as they are implemented in other cardiopulmonary diseases. Future research into this field can help ameliorate functional limitations experienced by the large percentage of patients in the chronic phase of PE. 107
Conclusions
Key points regarding post-PE syndrome are summarized in Table 1. Although CTEPH is the most worrisome consequence of chronic PE, recent years have brought more attention to CTED and chronic cardiopulmonary limitations experienced by many well after the acute event. Likely, a spectrum exists between complete recovery and severe pulmonary vascular disease with resultant RV dysfunction. Disease mechanisms including thrombus persistence and secondary arteriopathy are better understood and have led to the development of novel therapies. Diagnostic imaging modalities are expanding and expertise in PEA and BPA procedures offer treatment and even cure to previously inoperable disease. Important questions into the prevention of post-PE complications with early intervention are of interest in patients with intermediate and high-risk PE, as are the optimal approaches to the management of the chronic thromboembolic disease.
Post-PE syndrome: Summary of key points.

CPET, cardiopulmonary exercise testing; CTED, chronic thromboembolic disease; CTEPH, chronic thromboembolic pulmonary hypertension; PE, pulmonary embolism; PH, pulmonary hypertension; RHC, right heart catheterization; RV, right ventricular; TTE, transthoracic echocardiography; V/Q, ventilation/perfusion.
