Abstract
Introduction:
Racial disparities exist in patients with peripheral artery disease (PAD), with Black individuals having worse PAD-specific outcomes. However, mortality risk in this population has been mixed. As such, we sought to evaluate all-cause mortality by race among individuals with PAD.
Methods:
We analyzed data from the National Health and Nutrition Examination Survey (NHANES). Baseline data were obtained from 1999 to 2004. Patients with PAD were grouped according to self-reported race. Multivariable Cox proportional hazards regression was performed to calculate adjusted hazard ratios (HR) by race. A separate analysis was performed to study the effect of burden of social determinants of health (SDoH) on all-cause mortality.
Results:
Of 647 individuals identified, 130 were Black and 323 were White. Black individuals had more premature PAD (30% vs 20%, p < 0.001) and a higher burden of SDoH compared to White individuals. Crude mortality rates were higher in Black individuals in the 40–49-year and 50–69-year age groups compared to White individuals (6.7% vs 6.1% and 8.8% vs 7.8%, respectively). Multivariable analysis demonstrated that Black individuals with both PAD and coronary artery disease (CAD) had a 30% higher hazard of death over 20 years compared to White individuals (HR = 1.3, 95% CI: 1.0–2.1). The cumulative burden of SDoH marginally (10–20%) increased the risk of all-cause mortality.
Conclusions:
In a nationally representative sample, Black individuals with PAD and CAD had higher rates of mortality compared to their White counterparts. These findings add further proof to the ongoing racial disparities among Black individuals with PAD and highlight the necessity to identify ways to mitigate these differences.
Keywords
Introduction
Peripheral artery disease (PAD) is a progressive, atherosclerotic disease involving the medium to large-sized arteries of the lower extremities. It is estimated that more than 200 million people worldwide and 8–9 million people in the United States (US) have PAD.1,2 PAD is associated with atherosclerosis in other arterial beds (i.e., polyvascular disease), and has been linked with a higher risk of cardiovascular events. In fact, among those with chronic limb-threatening ischemia (CLTI), the most severe form of PAD, 1-year mortality rates as high as 25% have been reported.3,4
In the US, data suggest that the prevalence of PAD differs by race; the prevalence for Black individuals is about twice as high as for White individuals in adults older than 40 years. 1 Furthermore, PAD is historically underdiagnosed in the general population, and as such, these numbers might under-represent the true burden of PAD among the Black population. 5 In addition to higher prevalence, numerous studies have demonstrated that Black individuals are less likely to receive guideline-directed medical therapy (GDMT), and are also less likely to undergo revascularization procedures.6–8 This has resulted in worse PAD-specific outcomes in this patient population, including higher incidence of major adverse limb events (MALE) and major amputations when compared to White individuals. 9 Given these facts, it is only reasonable to assume that mortality among Black individuals with PAD will be higher than in their White counterparts.
Nevertheless, studies have not consistently shown an increased mortality among Black individuals compared to other races. In fact, prior studies have demonstrated mixed results, with some reporting lower and others suggesting higher mortality rates within this patient population.10–13 As such, we sought to evaluate the all-cause mortality among community-dwelling Black and White individuals with PAD over a period of 20 years by using the National Health and Nutrition Examination Survey (NHANES). The United States Centers for Disease Control and Prevention (CDC) recently released 2019 mortality files linked to NHANES participants, making this analysis possible.
Methods
Data source
NHANES is a cross-sectional survey of US civilian, noninstitutionalized individuals that uses a stratified and multistage probability sampling scheme to create a nationally representative data set. The data set consists of data on general health, household interviews, nutrition, lifestyle factors, and environmental exposures. The NHANES study protocol was approved by the National Center for Health Statistics Institutional Review Board, and written informed consent was obtained from all participants.
Exposure variable, covariates, and outcome
An ankle–brachial pressure index (ABI) was calculated for all participants aged ⩾ 40 years in the three NHANES surveys from the years 1999–2000, 2001–2002, and 2003–2004. The ABIs were performed in mobile centers which traveled throughout the country to obtain these measurements. Individuals were identified as having PAD based on an ABI of less than or equal to 0.9 in either lower extremity. Our analysis only included individuals with PAD (i.e., ABI ⩽ 0.9). 14 Our exclusion criteria were individuals < 40 years of age, if an ABI was not available, or if the ABI was greater than 0.9. We categorized individuals into three groups by self-reported race: White (non-Hispanic), Black (non-Hispanic), and others. The ‘others’ group included Mexican Americans, other Hispanics, and all other races including multiracial individuals. Information on covariates such as sex, age, comorbid conditions including coronary artery disease (CAD), smoking status, and use of medication were collected in NHANES by self-reported questionnaires. Participants were categorized by age at time of PAD diagnosis into three groups: 40–49 years, 50–69 years, and 70 years or greater. Participants in the 40–49 years group were considered to have premature PAD as previously defined. 15 Participants aged 70 years or greater were grouped together due to higher prevalence of PAD within this population. 16 Social determinants of health (SDoH) have been associated with cardiovascular outcomes. 17 Given that different racial groups in the US may carry a differing burden of SDoH, we identified the various SDoH categories and compared them within our studied population. Specifically, we identified 10 SDoH from NHANES representing five domains, as specified by the Healthy People 2030 objectives of the US Department of Health and Human Services (HHS). 18 Economic stability (domain-1) was measured by annual household income less than $35,000, unavailability of economic support when needed, or the family income to poverty ratio of < 2. The family income to poverty ratio was calculated by dividing the family income by the poverty guideline of the department of US HHS specific to family size and appropriate year and state. Less than high-school education was domain-2. Healthcare access (domain-3) was measured by the lack of health insurance or unavailability of a healthcare place to go when needed. Neighborhood and environment (domain-4) was measured by food insecurity. Social and community context (domain-5) was defined as social isolation measured by the lack of partner/spousal presence, unavailability of emotional support when needed, or self-reported non-White race/ethnicity. The primary outcome of interest was all-cause mortality. The all-cause mortality was determined using the Linked Mortality File (that links all NHANES participants to 2019 all-cause mortality), which was provided by the CDC and thus provides a total follow-up period of up to 20 years for participants depending on time of enrollment.
Statistical analysis
Univariate analysis was conducted to compare the differences between Black and White participants using Student’s t-test for continuous variables and chi-squared test for categorical variables. We performed multivariable Cox proportional hazards regression to calculate adjusted hazard (mortality) ratios (HR) with 95% CI for Black individuals comparing with the reference population of White individuals. We identified potential confounders from Table 1 as those variables with p < 0.05, including SDoH, and these were included in the stepwise Cox proportional hazards regression. The p-values were not adjusted for multiple comparisons. Since CAD is an effect modifier, we decided to stratify our analysis by CAD. We also conducted a separate analysis to study the effect of burden of SDoH on all-cause mortality in individuals with PAD. We calculated mortality rates stratified by age group, race, and sex. Statistical analyses were performed using SAS software, Version 9.1 (SAS Institute, Inc., Cary, NC, USA). To determine statistical significance, α < 0.05 was set.
Baseline characteristics of NHANES participants with peripheral artery disease by race.
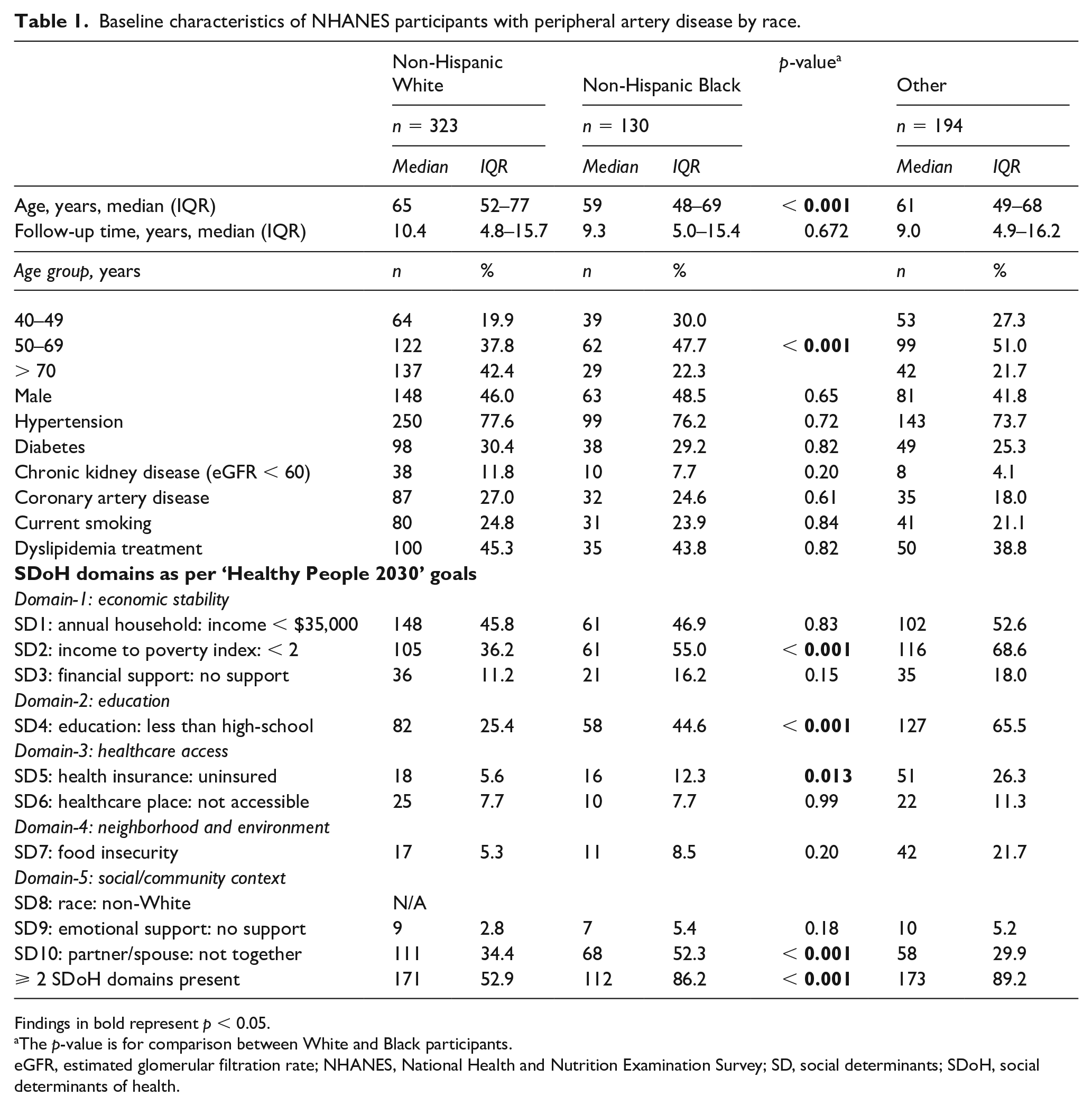
Findings in bold represent p < 0.05.
The p-value is for comparison between White and Black participants.
eGFR, estimated glomerular filtration rate; NHANES, National Health and Nutrition Examination Survey; SD, social determinants; SDoH, social determinants of health.
Results
A total of 31,126 participants were included in the NHANES cohort from 1999 to 2004 (Figure 1). Of these, 7639 underwent ABI measurements. A total of 647 individuals were identified as having an ABI of 0.9 or less. Of these, 323 were White, 130 were Black, and 194 were categorized as other race (Table 1). Black participants were younger at the time of PAD diagnosis compared to White participants (online Supplemental Figure 1). The median age at PAD diagnosis for Black participants was 59 years compared to the median age of 64 years in White participants (p < 0.001). Additionally, premature PAD was more prevalent among Black individuals (30.0% vs 19.9%). All domains of SDoH as determined by the Healthy People 2030 objectives were worse in Black compared with White individuals (Table 1). Specifically, Black individuals had a lower level of education compared with White individuals; 45% of Black participants had less than high-school education compared to 25% of White participants (p < 0.001). Black individuals had a higher proportion of uninsured individuals compared with their White counterparts (12.3% vs 5.6%, p = 0.013). Economic stability as a domain of SDoH was defined by a family income to poverty index of < 2, which was worse in Black compared with White individuals (55% vs 36%, p < 0.001). Finally, a higher proportion of Black individuals experienced social isolation, evident by lack of presence and support of a partner/spouse, compared to White individuals (52% vs 34%, p < 0.001). When combined, 86% of Black individuals had a higher SDoH burden, with two or more SDoH present, compared to only 53% for White individuals (p < 0.001). There was no difference in sex when stratified by race (46% White men vs 49% Black men, p = 0.65). There were no significant differences in health-related baseline characteristics between groups, including similar proportions of hypertension, diabetes, CAD, chronic kidney disease (eGFR < 60), current smoking status, and use of dyslipidemia treatment (Table 1).
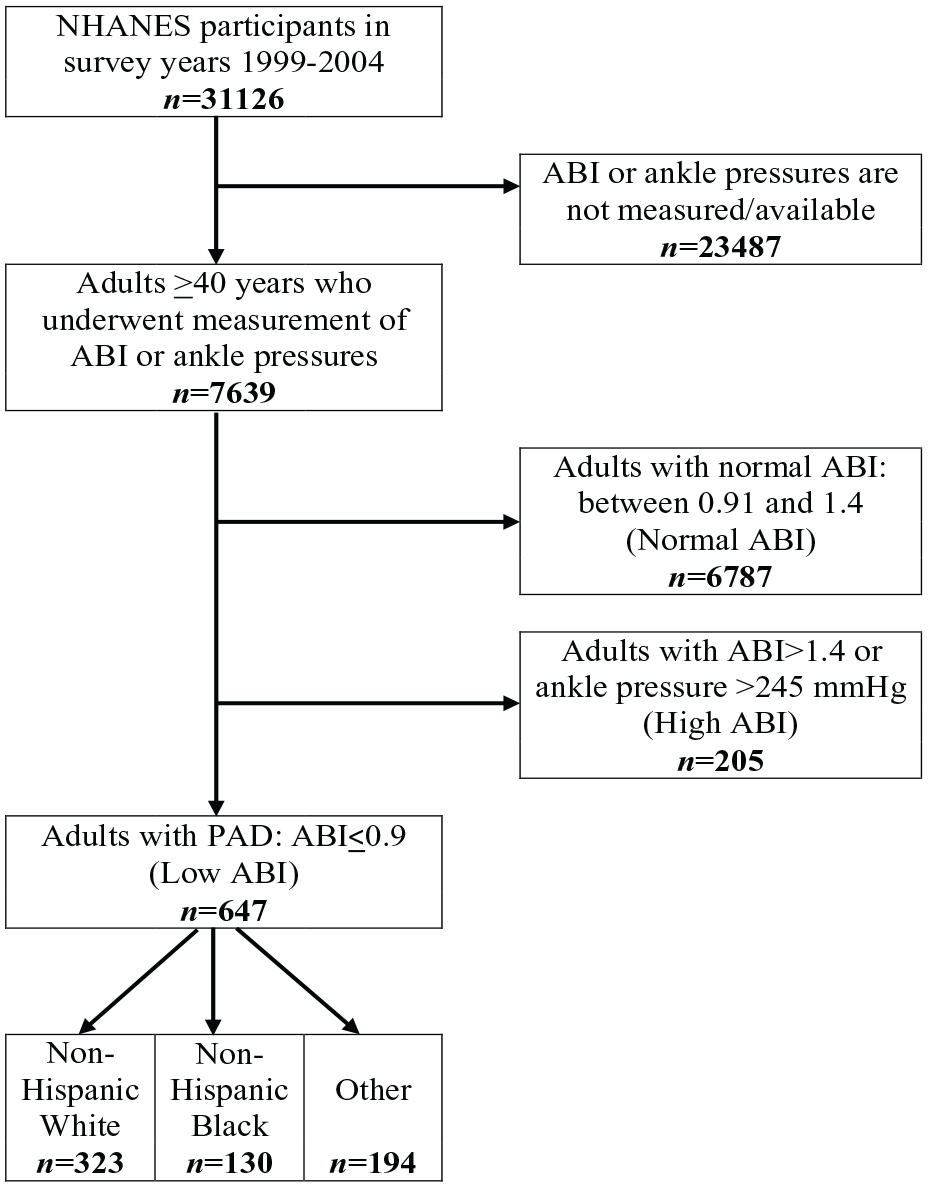
Flow diagram for selection of eligible participants.
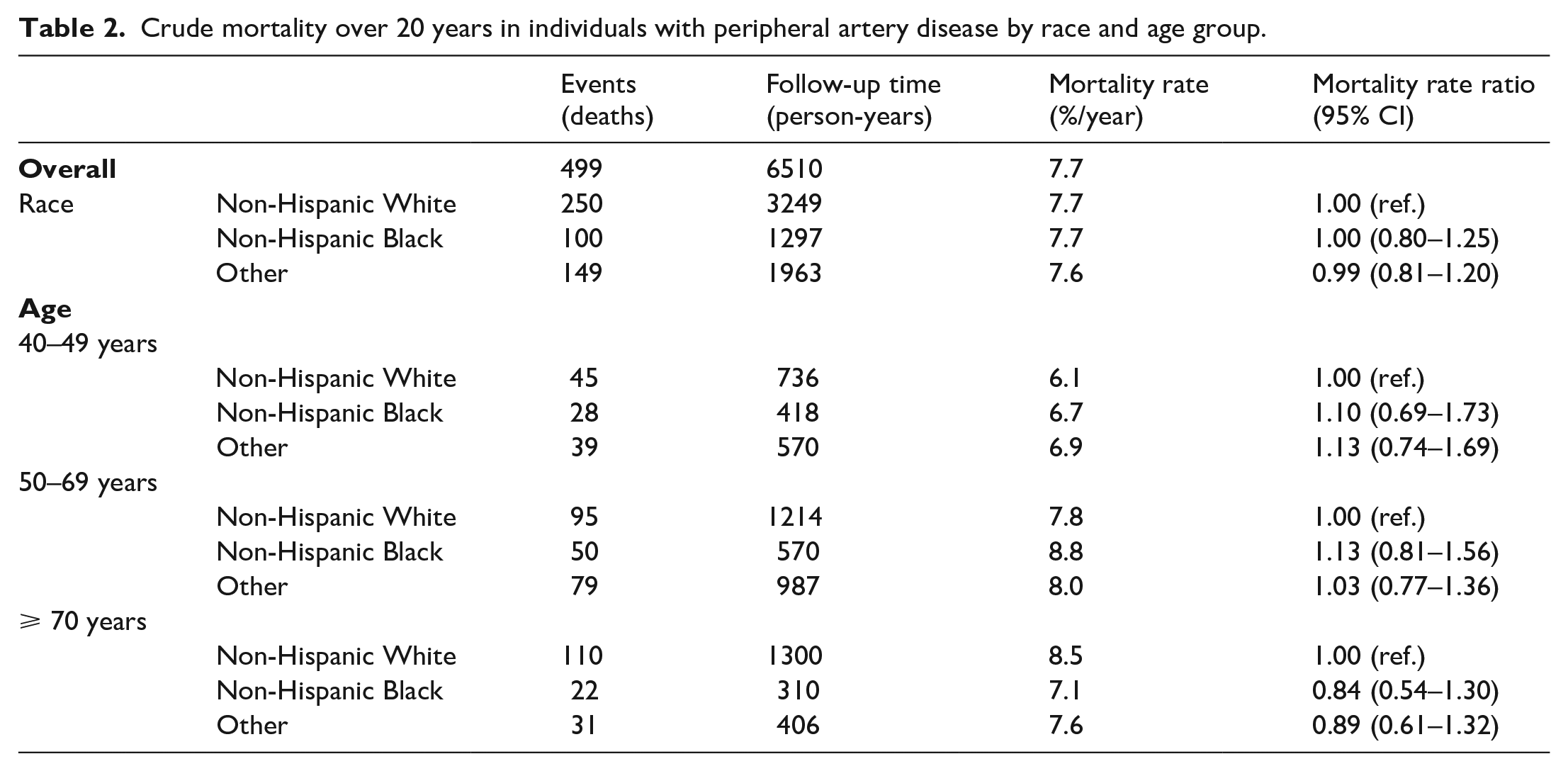
All-cause mortality rates are shown in Table 2, stratified by age and race. Overall mortality rates were similar for the Black, White, and other races (~7.7% per year) for a follow-up of up to 20 years (online Supplemental Figure 2). The mortality rates for Black individuals diagnosed with PAD in the 40–49-year and 50–69-year age groups were 6.7% per year and 8.8% per year compared with 6.1% per year and 7.8% per year among White individuals, respectively, whereas the mortality rate was 7.1% per year for Black compared with 8.5% for White individuals in the 70 years and above age group.
Crude mortality over 20 years in individuals with peripheral artery disease by race and age group.
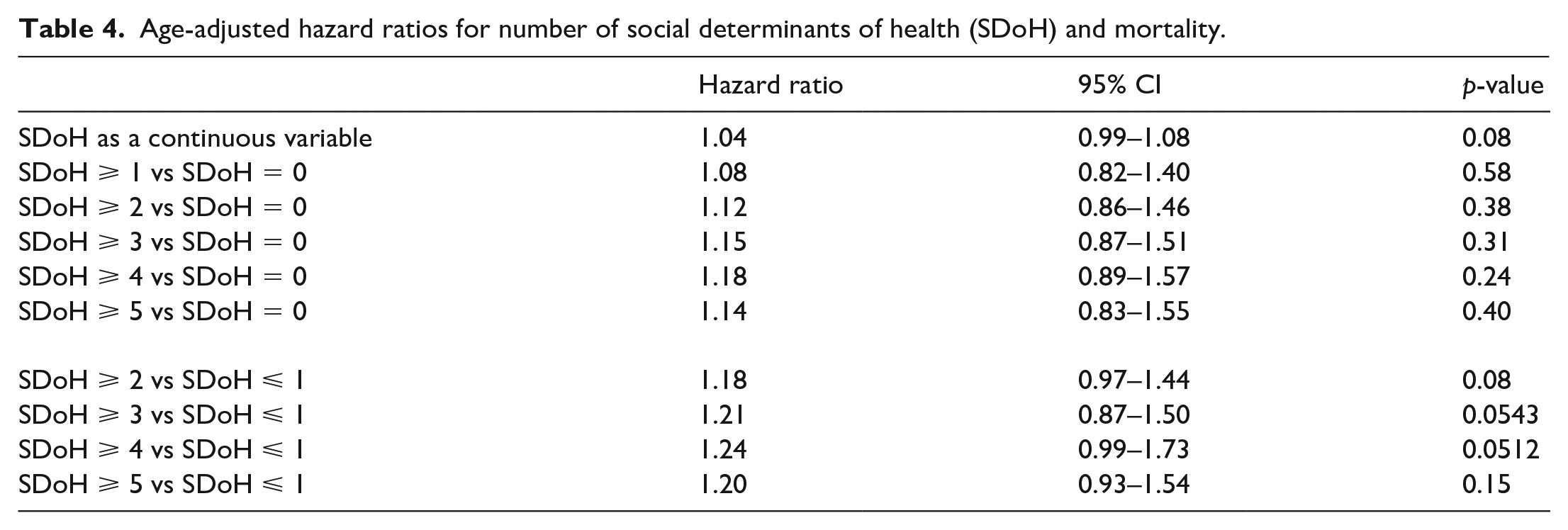
Multivariable adjusted HRs with 95% CIs for all-cause mortality stratified by CAD are presented in Table 3. Black individuals had a 30% higher hazard of death over 20 years in the presence of CAD and PAD when compared to White individuals (HR: 1.3, 95% CI: 1.0–2.1). This association was not present for individuals without CAD (HR: 0.9, 95% CI: 0.7–1.3). Also, lack of health insurance was independently associated with all-cause mortality in individuals with and without CAD at baseline (HR: 1.9, 95% CI: 1.0–3.5 and HR: 1.3, 95% CI: 1.0–1.8, respectively). Nonetheless, cumulative burden of SDoH, defined as the summation of each SDoH present, only marginally (10–20%) increased the risk of all-cause mortality in individuals with PAD as shown in Table 4. There was a weak signal for a dose–response relationship between increasing number of SDoH and worse outcome.
Multivariable adjusted hazard (mortality) ratios with 95% CI in participants with PAD.
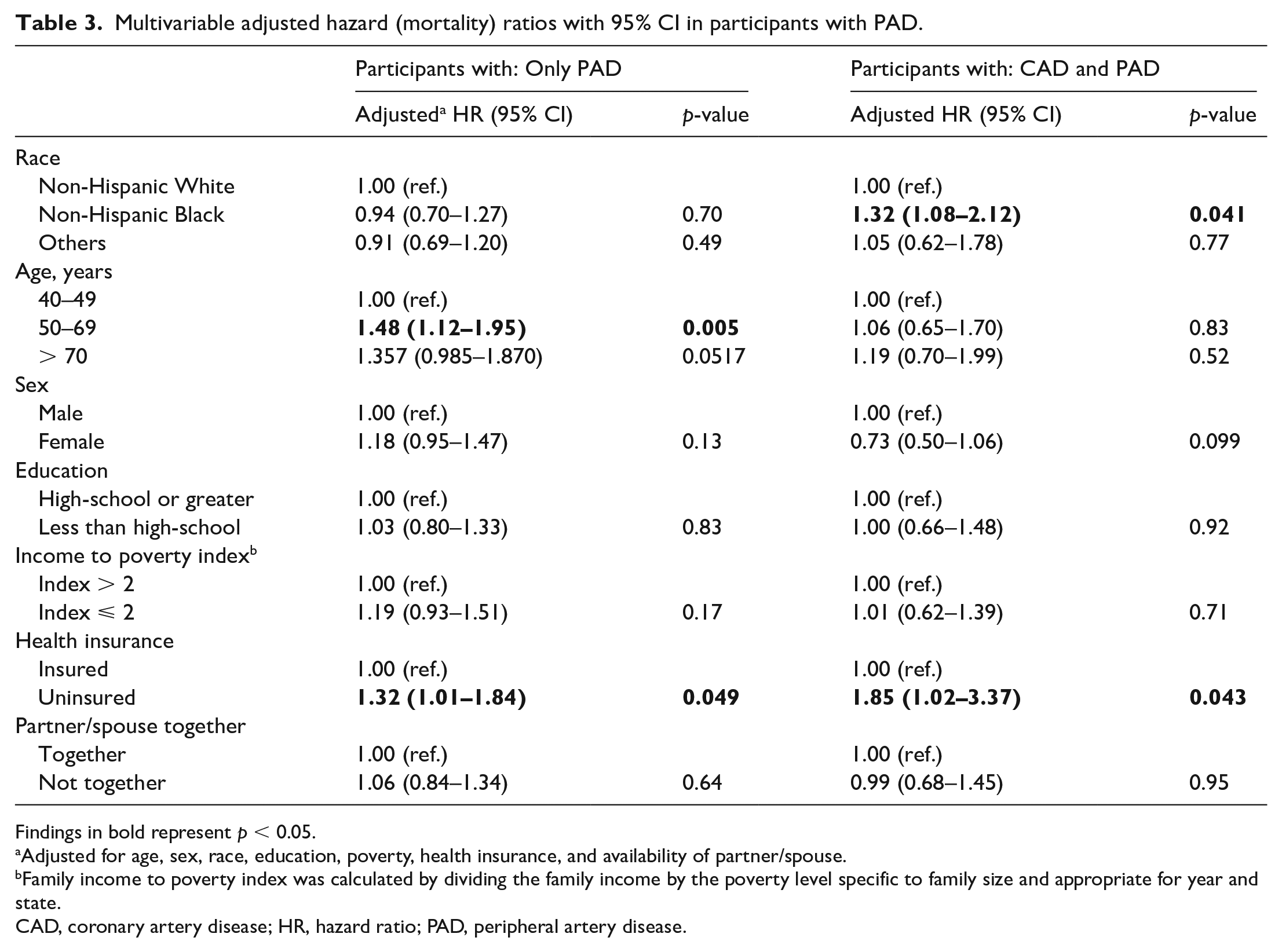
Findings in bold represent p < 0.05.
Adjusted for age, sex, race, education, poverty, health insurance, and availability of partner/spouse.
Family income to poverty index was calculated by dividing the family income by the poverty level specific to family size and appropriate for year and state.
CAD, coronary artery disease; HR, hazard ratio; PAD, peripheral artery disease.
Age-adjusted hazard ratios for number of social determinants of health (SDoH) and mortality.
Discussion
In this study of community-dwelling individuals with PAD who were followed for up to 20 years, we had the following notable findings: (1) Black individuals had more premature PAD and had higher burden of SDoH. (2) Black individuals with PAD had higher crude mortality rates at younger ages, specifically ages 40–69, compared to White counterparts. (3) The mortality of Black patients with PAD was dependent on CAD status: Black individuals with baseline CAD had a significantly higher adjusted mortality compared with White individuals. This association was not present when baseline CAD was not present. (4) Although the effect of burden of SDoH on all-cause mortality did not reach statistical significance, the overall trend was toward an increase in mortality. However, uninsured individuals with PAD, irrespective of CAD status, had higher mortality.
Prior studies have focused on PAD management and PAD-specific outcomes (e.g., MALE), but data have been conflicting regarding race-specific mortality in individuals with PAD. In an evaluation of over 800,000 patients with mortality attributed to peripheral vascular disease, Black patients were noted to have a 1.5 times higher mortality rate compared with White patients. 13 However, the definition of peripheral vascular disease within this study included both arterial and venous disease and there was no specific analysis for patients with PAD alone. Perhaps surprisingly, and contrary to these findings, two other studies using the Vascular Quality Initiative (VQI) registry have demonstrated lower mortality rates among Black and Hispanic patients with PAD compared to White patients, even after adjusting for age and CAD status.10,12 Of note, the follow-up period in both studies was approximately 1 year. Additionally, another study demonstrated lower in-hospital mortality in Black compared with White individuals, despite a higher incidence of CLTI and need of amputation. 11 In contrast, we used a specific and universally accepted definition of PAD (ABI ⩽ 0.9) and an extended follow-up period of up to 20 years, which is substantially longer than prior studies.
Our study demonstrated that Black individuals with PAD had nominally higher crude mortality rates compared with White individuals (Table 2). Even more so, when stratified for baseline CAD status, Black individuals with both CAD and PAD had significantly higher mortality rates compared with White individuals. These results are aligned with the numerous studies that reported poor PAD-specific outcomes in Black individuals. Furthermore, the differences in mortality rate among Black individuals as stratified by CAD status is notable and suggests that one of the drivers of PAD-related mortality is CAD. PAD is a known risk factor for cardiovascular disease and death, and approximately half of patients with PAD will have atherosclerotic disease in one or more vascular beds. 19 Among Black individuals, the prevalence of CAD is lower than among White individuals, yet the former have higher cardiovascular mortality rates. 5 In a retrospective review of cardiac death from 1968 to 2015, cardiac death rates were 21% higher among Black individuals compared to White counterparts. 20 Our study demonstrated similar findings of increased mortality among Black patients with CAD. The link between race and increased mortality in this population is uncertain but can be speculated to be due to systemic issues including poor access to care, lower use of GDMT prescription, and other barriers to receiving appropriate treatment. It is important for providers taking care of these individuals to be cognizant of the risks and poor outcomes associated with PAD, specifically when co-presenting with CAD.
In the current study, Black individuals had more premature PAD than their White counterparts. More specifically, in patients with PAD who did not have concomitant CAD, age was independently associated with higher mortality as expected. However, this relationship was attenuated in patients with PAD and CAD after adjusting for race. This is because White individuals followed the expected pattern of age–mortality relationship, with the oldest individuals (age 70 years and above) experiencing the highest mortality. In contrast, Black individuals aged 50–69 years experienced the highest mortality among all age groups. Moreover, younger Black individuals (aged 40–69 years) experienced higher mortality compared to their White counterparts. This finding is consistent with existing literature. In a retrospective analysis of over 27,000 patients, Black men aged 50–59 and 60–69 had a higher prevalence of PAD compared with White counterparts (5.0% vs 1.9% and 13.2% vs 5.4%, respectively). 21 Additionally, Black individuals appear to have an exponential increase in prevalence of PAD, from age 40 onwards, whereas other ethnic groups have a slow, linear increase in the prevalence of PAD from age 40 until the 8th decade, at which time the rate increased exponentially. 21 We found that the mortality rate was higher for Black individuals with PAD who were younger than 69 years (Table 2). Taken together with a younger age onset of PAD, one could postulate that other cardiovascular risk factors accumulate over a longer period, and thus manifest at a younger age compared to White individuals.
Other than purely physical conditions, social factors may be relevant to PAD patients of different races. It is well documented that poor SDoH are associated with increased cardiovascular deaths. For example, a retrospective review of over 22,000 individuals demonstrated that age-adjusted incidence of fatal coronary heart disease increased with the number of SDoH. 17 Compared with individuals with no SDoH, those with three or greater determinants had a 67% higher hazard ratio for fatal coronary heart disease. Unfortunately, existing literature examining SDoH in patients with PAD is limited, and mainly revolves around PAD management and limb-specific outcomes.22–24 Thus, we examined those separately (e.g., education, income, social isolation, etc.) and in aggregate as the total burden of SDoH.
We found that all domains of SDoH as determined by the Healthy People 2030 objectives were worse in Black compared to White individuals, which has been demonstrated in prior studies. 25 However, as the number of SDoH increased irrespective of race, we found a marginal increase in the risk of mortality. This novel finding highlights the importance that SDoH has on outcomes for patients with PAD. However, it is possible that our study was underpowered to truly examine the effect of SDoH on mortality in PAD. Thus, larger studies are encouraged to determine the effect of these factors on patients with PAD. Nonetheless, individuals with PAD (irrespective of CAD status) who were uninsured had a higher mortality rate, as expected. This is in accordance with previous literature showing that the lack of health insurance has been associated with all-cause mortality and PAD-specific outcomes, including greater likelihood of amputation. A review of 7274 participants demonstrated that uninsured patients had an increased, adjusted all-cause mortality (HR = 1.57) compared to privately insured individuals. 26 Additionally, an evaluation of over 100,000 patients demonstrated that uninsured individuals were two to three times more likely to undergo a major leg amputation compared to individuals with private insurance. 27
Although a potential association of education level and mortality in PAD has not been previously examined, one prior study demonstrated that prevalence of PAD was higher among individuals with a lower education level. 28 A low education level has been associated with increased mortality in numerous disease states, including diabetes, 29 cancer, 30 heart failure, 31 and coronary artery disease. 32 Nevertheless, in our study, education level did not have an impact on mortality risk in individuals with PAD. Similarly, our study also did not demonstrate an association between income level and mortality in community-dwelling individuals with PAD. Prior data suggested that low-income patients with PAD had worse survival after surgical revascularization, 33 but this discrepancy likely relates to differences in the studied populations as our study includes community dwellers with PAD and the prior study included only patients with PAD undergoing surgical revascularization. Taken together, the findings related to SDoH paint a complex picture in which physical and social determinants may play differing roles affecting patient outcomes.
Study limitations and strengths
The NHANES registry is designed to be a representative sample of the United States population, yet there are limitations associated with this database that should be mentioned in the context of the current study. Many of the data points included in the analysis were participant self-reported and not verified with medical records. Additionally, there is a large percentage of individuals in the NHANES registry who did not undergo an ABI evaluation, likely as these were required to be obtained at a mobile center. As such, this could impact the generalizability of the study findings. Despite the large number of participants in NHANES from 1999 to 2004, the proportion of individuals with PAD is lower than the expected prevalence of PAD within the US, suggestive that the studied population might be healthier overall. Given the low proportion of individuals with PAD, the number of patients included in the study is small, thus potentially limiting the applicability of these findings to a larger population and the ability to detect certain associations. Additionally, we are unable to comment on the impact of PAD on races other than White and Black. Lastly, we do not know the symptomatic status of individuals with PAD in our cohort, and we cannot comment on the medication adherence and the prevalence of revascularization procedures during the follow-up period, which could also impact the outcomes. Despite these limitations, the study has strengths, including a follow-up period of up to two decades and the ability to examine multiple verified physical and social factors. Thus, it sheds light on racial determinants of mortality in these individuals.
Conclusions
In this nationally representative sample of patients with PAD, over a period of up to 20 years, Black individuals presented with more premature PAD than their White counterparts. Furthermore, they had a higher burden of SDoH. Black individuals with both PAD and CAD had higher rates of age-adjusted mortality compared to their White counterparts. These findings highlight the need for additional research to understand the root causes for these disparities and potential ways to mitigate them.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X231159947 – Supplemental material for Mortality differences by race over 20 years in individuals with peripheral artery disease
Supplemental material, sj-docx-1-vmj-10.1177_1358863X231159947 for Mortality differences by race over 20 years in individuals with peripheral artery disease by Andrew B Dicks, Vladimir Lakhter, Islam Y Elgendy, Robert M Schainfeld, Abhisekh Mohapatra, Jay Giri, Mitchell D Weinberg, Ido Weinberg and Gaurav Parmar in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
