Abstract
Background:
Arterial stiffness represents an established cardiovascular risk marker. Sodium-glucose co-transporter-2 (SGLT-2) inhibitors have significant cardio-protective effects. Herein we sought to determine the effect of SGLT-2 inhibitors on pulse wave velocity (PWV).
Methods:
We searched PubMed, Cochrane Library, and grey literature from inception to 7th February 2022 for randomized controlled trials (RCTs) enrolling adult subjects with or without type 2 diabetes mellitus (T2DM), assigned to a SGLT-2 inhibitor versus control and addressing their effect on PWV. We set as primary efficacy outcome the change in PWV with SGLT-2 inhibitors versus placebo or control.
Results:
We pooled data from six trials in a total of 452 enrolled participants assigned either to SGLT-2 inhibitor or control. Overall, SGLT-2 inhibitor treatment compared to control resulted in a nonsignificant decrease in PWV. Exclusion of a trial utilizing cardiac magnetic resonance imaging for the assessment of PWV demonstrated that SGLT-2 inhibitors induce a significant reduction in PWV by 0.21 m/s. When we restricted our analysis to RCTs enrolling subjects with T2DM, we observed that SGLT-2 inhibitor compared to control resulted in a significant decrease in PWV by 0.17 m/s.
Conclusion:
SGLT-2 inhibitors do not decrease PWV in patients with established cardiovascular disease or cardiovascular risk factors. However, we have shown that SGLT-2 inhibitors lead to a slight, but significant decrease in PWV in patients with T2DM. The latter finding is of great value, based on the significant correlation between PWV and micro- and macro-vascular complications of T2DM.
Introduction
Arterial stiffness is an established and well-defined cardiovascular risk marker, able to provide significant prognostic information regarding cardiovascular morbidity and mortality, and all-cause mortality as well. 1 Pulse-wave velocity (PWV) is considered the ‘gold-standard’ for quantification of arterial stiffness in clinical practice. Recent guidelines recommend that PWV can be measured in the context of a thorough assessment of a patient with hypertension for the potential identification of target organ damage. 2 Carotid-femoral PWV is the marker of interest for the evaluation of aortic stiffness, which is of particular interest for specific populations of patients.
The significant, prognostic role of arterial stiffness in patients with hypertension without any other comorbidities was first documented two decades earlier. 3 Later, it was shown that arterial stiffness might also have prognostic implications in patients with end-stage renal disease. 4 More recent evidence has implied that arterial stiffness might also have an important prognostic impact in patients with type 2 diabetes mellitus (T2DM), as it is strongly associated with cardiovascular morbidity and mortality and all-cause mortality.5–7 Besides surrogate cardiovascular outcomes, arterial stiffness has also been shown to significantly correlate with microvascular complications of T2DM, namely diabetic nephropathy, neuropathy, and retinopathy.8–11 Therefore, it has been previously questioned whether arterial stiffness should be incorporated in the routine management of patients with T2DM. 12
In clinical practice, tonometry performed in the office is the most widely used method for the assessment of PWV. Recently, oscillometric methods, which permit the 24-hour, ambulatory measurement of PWV, have been introduced. Even though oscillometric assessment seems to slightly underestimate PWV, the agreement between the two methods is satisfactory. 13 During the last decade, cardiac magnetic resonance imaging (cMRI) has also been introduced as a novel method for the measurement of PWV, which enables the measurement of regional PWV and distensibility, in contrast to tonometric methods. 14 However, there is no certain evidence regarding the prognostic implications of PWV generated by cMRI.
Sodium-glucose co-transporter-2 (SGLT-2) inhibitors represent a novel class of antidiabetic drugs, with proven cardiovascular efficacy, even in subjects without diabetes mellitus.15,16 However, there is still a vivid and ongoing discussion on the mechanisms underlying the observed cardiovascular benefits with SGLT-2 inhibitors. 17 Their potential favorable effect on arterial stiffness has been early hypothesized, leading to the conduction of several relevant studies, some of which have provided contradictory results, especially in the real-world setting. 18
Therefore, we sought to perform the first meta-analysis of randomized controlled trials (RCTs) in the field, to determine whether SGLT-2 inhibitors produce a significant effect on PWV, in patients with and without T2DM.
Methods
This systematic review and meta-analysis was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guidelines. 19
Eligibility
We searched for parallel groups or cross-over RCTs enrolling adult subjects with or without T2DM, assigned to a SGLT-2 inhibitor versus placebo or active comparator and addressing their effect on PWV under static or ambulatory conditions. We excluded trials enrolling subjects with type 1 diabetes mellitus (T1DM) or studies performed in the pediatric population, if any.
Search
We used the following search strategy implemented on 7 February 2022 both in the PubMed and Cochrane Library databases: “SGLT-2 inhibitor” OR “dapagliflozin” OR “empagliflozin” OR “canagliflozin” OR “sotagliflozin” OR “ertugliflozin” OR “ipragliflozin” OR “tofogliflozin” OR “luseogliflozin” OR “bexagliflozin” OR “licogliflozin” OR “remogliflozin” AND “arterial stiffness” OR “aortic stiffness” OR “pulse wave velocity” OR “PWV”. We searched the clinicaltrials.gov and clinicaltrialsregister.eu registries. We did not impose any filter regarding sample size, study setting or publication language.
Outcomes
We set as primary efficacy outcome the change in PWV with SGLT-2 inhibitors versus placebo or control.
Data extraction
After deduplication, two independent reviewers (DP, AK) screened all records at title and abstract level and then assessed the full text of eligible records. Any disagreements were resolved by consultation of a third reviewer (MD).
Three independent reviewers (DP, NF, and CP) extracted the data from the eligible reports. Extracted information were: first author, year of study conduction, study setting, study sample size, country of origin, type of administered SGLT-2 inhibitor, type of control (placebo or active comparator), measurement method of PWV, change in PWV in each treatment arm, follow-up duration, mean age of participants, male to female ratio, major comorbidities, and concomitant treatment of interest with established beneficial effect on PWV (e.g., renin-angiotensin-aldosterone system blockers).
Statistical analysis
Differences were calculated with the use of mean difference (MD), with standard error (SE), after implementation of the inverse variance (IV) random effects formula. In those studies not reporting SE, we calculated SE from sample size and SD.
Statistical heterogeneity among studies was assessed by using I2 statistics. 20 I2 ranging between 0 and 40% is considered as low, I2 ranging between 50% and 90% may represent substantial heterogeneity, and I2 ranging between 75% and 100% may be indicative of considerable heterogeneity. 20 All analyses were performed at the 0.05 significance level, with the RevMan 5.3. software. 21
Risk of bias assessment
Two independent reviewers (DP and CP) assessed the quality of the included RCTs, by using the revised Cochrane risk-of-bias tool for randomized trials (RoB 2.0) for the primary efficacy outcome. 22 Discrepancies between reviewers were solved by discussion, consensus or arbitration by a third senior reviewer (MD).
Grading of evidence
Finally, grading of retrieved evidence for the primary efficacy outcome was performed according to GRADE framework. 23
Results
Results of search and trial characteristics
We retrieved a total of 184 results from the PubMed and the Cochrane Library electronic databases; no additional results were retrieved from grey literature sources.
We excluded one trial performed in subjects with T1DM, 24 according to our prespecified eligibility criteria, and one trial addressing the effect of tofogliflozin on brachial–ankle PWV (baPWV). 25 The study selection process is provided in the corresponding flow diagram (online supplementary Figure 1).
We pooled data from six trials in a total of 452 enrolled participants assigned either to SGLT-2 inhibitor or control.26–31 All trials enrolled subjects with T2DM except for two trials performed by Requena-Ibáñez et al., 27 in which none of the participants had a prior diagnosis of T2DM, and by Kolwelter et al., 28 who enrolled subjects with a prior diagnosis of heart failure with reduced ejection fraction (HFrEF), from whom less than one-fourth suffered from T2DM prior to inclusion in the trial. A thorough description of participants’ baseline characteristics of interest along with PWV analysis methods is provided in Table 1. In short, four trials utilized empagliflozin, one trial dapagliflozin, and one trial canagliflozin. Requena-Ibáñez et al. 27 assessed PWV utilizing cMRI, Striepe et al. 26 and Papadopoulou et al. 29 assessed 24-hour, ambulatory PWV, and in the remaining eligible trials, office PWV was quantified before and after the initiation of a SGLT-2 inhibitor or control (placebo or active comparator).
Summary of included studies addressing the primary efficacy outcome.
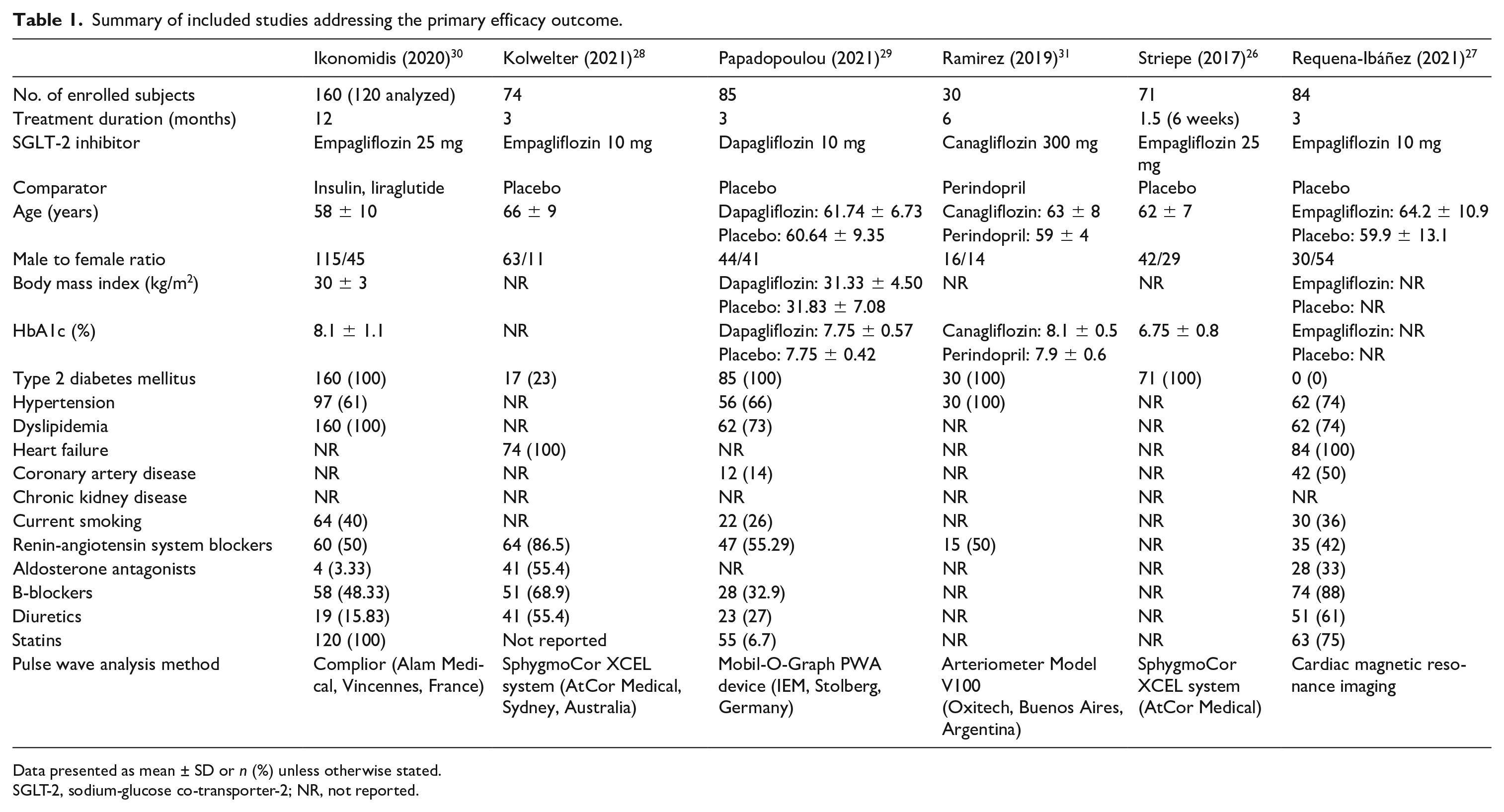
Data presented as mean ± SD or n (%) unless otherwise stated.SGLT-2, sodium-glucose co-transporter-2; NR, not reported.
Analysis of primary efficacy outcome
Overall, after pooling relevant data of interest from the six eligible trials, it was shown that SGLT-2 inhibitor treatment compared to control resulted in a nonsignificant decrease in PWV by 0.14 m/s (MD = −0.14, 95% CI −0.31 to 0.03, I2 = 63%, p = 0.11), as shown in Figure 1.
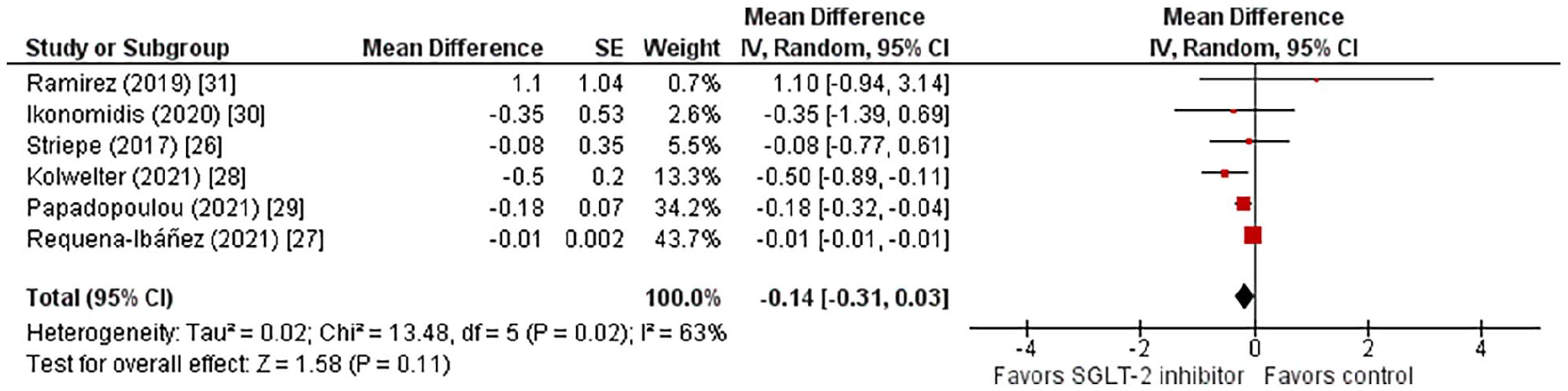
Effect of SGLT-2 inhibitors compared to control on PWV.
Sensitivity and subgroup analyses
In order to investigate the sources of the observed substantial heterogeneity, we decided to perform relevant sensitivity analyses to check the robustness of the generated results. Therefore, we first excluded the trial by Requena-Ibáñez et al. 27 , which utilized cMRI, a not widely adopted method for the assessment of PWV, although this technique has evolved as an important method for estimating regional PWV and distensibility. 14
Exclusion of the aforementioned trial demonstrated that SGLT-2 inhibitors induced a significant reduction in PWV by 0.21 m/s (MD = −0.21, 95% CI −0.35 to −0.07, I2 = 2%, p = 0.003). Therefore, the trial conducted by Requena-Ibáñez et al. 27 was recognized as the major source of heterogeneity in this meta-analysis. However, at present, we cannot deduce that the observed heterogeneity can be attributed to methodological differences between the various techniques for the assessment of PWV.
When we also excluded the other trial performed in the HFrEF population, 28 we observed that SGLT-2 inhibitor compared to control led to a significant decrease in PWV by 0.17 m/s (MD = −0.17, 95% CI −0.31 to −0.04, I2 = 0%, p = 0.01), with complete elimination of the observed heterogeneity. Of note, this analysis corresponds to the effect of SGLT-2 inhibitors on PWV in subjects with T2DM at background, showing a favorable effect on that cardiovascular risk marker. The latter finding is of great value, since arterial stiffness has been shown to be an independent predictor of both micro- and macro-vascular complications of T2DM.5–12
In addition, we decided to perform a subgroup analysis according to duration of follow-up period (> 6 or < 6 months). Long-term administration of SGLT-2 inhibitors results in a significant reduction in PWV by 0.01 m/s (p < 0.0001, I2 = 0%), according to analysis of data from the two relevant trials.26,31 Similarly, administration of SGLT-2 inhibitors for a shorter duration results in a marginally nonsignificant reduction in PWV by 0.23 m/s (p = 0.05, I2 = 25%).
Finally, we performed another subgroup analysis according to the type of control (placebo or active comparator). When SGLT-2 inhibitors were compared with active comparator,30,31 they did not have a significant effect on PWV (MD = −0.1, 95% CI −1.21 to 1.41, I2 = 35%, p = 0.88), and when SGLT-2 inhibitors were compared with placebo, they similarly did not confer a significant reduction in PWV (MD = −0.14, 95% CI −0.32 to 0.04, I2 = 75%, p = 0.12).26–29
Risk of bias assessment and publication bias
Overall risk of bias was considered as low, as demonstrated in online supplementary Table 1. Generated evidence was rated as of low certainty.
Finally, inspection of the corresponding funnel plot did not reveal the presence of publication bias (online supplementary Figure 2).
Discussion
A previous meta-analysis showed that incretin mimetics produce a significant decrease in PWV, with the greater effect seen with glucagon-like peptide-1 receptor agonists (GLP-1RAs). 32 In this meta-analysis, Batzias et al. 32 included only one trial assessing the effect of SGLT-2 inhibitors on PWV. Therefore, in fact, our meta-analysis of available RCTs is the first to evaluate the effect of SGLT-2 inhibitors versus placebo or active control on PWV.
In our meta-analysis, SGLT-2 inhibitors did not produce a significant decrease in PWV in patients with established cardiovascular disease or cardiovascular risk factors, such as T2DM. However, sensitivity analysis by exclusion of the trial performed by Requena-Ibáñez et al., 27 which utilized cMRI for the assessment of PWV, demonstrated a slight, however significant, reduction in PWV with SGLT-2 inhibitors. In addition, when we restricted our analysis to studies enrolling adults with T2DM, we documented a sustained, significant decrease in PWV. Finally, we have shown in the present meta-analysis that treatment with SGLT-2 inhibitors > 6 months might indeed have a beneficial effect on arterial stiffness, yet the result is nonsignificant for shorter treatment duration.
There has been a long discussion on the mechanisms underlying the cardiovascular and renal benefits observed with SGLT-2 inhibitors. Improvement in arterial stiffness parameters was proposed over 5 years ago now; however, initial reports were rather contradictory. 33 Effects such as osmotic diuresis, natriuresis, reduction in blood pressure, body weight and serum uric acid levels, increase in hematocrit and hemoglobin levels, along with effects against inflammation, oxidative stress and endothelial dysfunction, and alteration in myocardial substrate metabolism have been suggested during the last decade.33–35
Reduction in PWV with SGLT-2 inhibitors appears to be an interesting beneficial effect that cannot be directly translated into significant cardiovascular benefit. However, since an increase in PWV by 1 m/s has been shown to increase by 4%, 15%, and 15% the risk for major adverse cardiovascular events, cardiovascular mortality, and all-cause mortality, respectively, 1 it seems that the observed reduction in PWV with SGLT-2 inhibitors could ideally contribute to overall cardiovascular risk reduction. In addition, based on the fact that arterial stiffness strongly correlates with a steeper decline in renal function and development of incident chronic kidney disease (CKD), 36 and is also associated with an adverse cardiac remodeling leading to heart failure (HF) development, 37 it can therefore be deduced that arterial stiffness reduction could also contribute to the cardiovascular and renal benefits observed with SGLT-2 inhibitors in patients with advanced CKD or HF with preserved or reduced ejection fraction.
Unfortunately, owing to the unavailability of participants’ individual data, we were unable to perform subgroup analyses according to background treatment in subjects treated with SGLT-2 inhibitors. As we know, drug classes, such as renin-angiotensin-aldosterone system blockers,38–40 b-blockers, 41 and statins 42 exert beneficial effects on aortic stiffness. Future trials will determine whether the combination of SGLT-2 inhibitors with such drug classes provides a greater reduction in PWV. Recent data also suggest that GLP-1RAs lead to a significant decrease in PWV. 43 It would be of great interest if large-scale trials addressed the potential of a synergistic beneficial effect of a SGLT-2 inhibitor and GLP-1RA combination on PWV, 44 as shown in the trial performed by Ikonomidis and colleagues. 30
Of course, currently there is a shortage of long-term prognostic studies able to support the predictive value of 24-hour PWV, 45 and additionally PWV might be slightly underestimated under ambulatory conditions compared to office measurement (with devices, such as Complior (Alam Medical, Vincennes, France), SphygmoCor XCEL system (AtCor Medical, Sydney, Australia) and Arteriometer Model V100 (Oxitech, Buenos Aires, Argentina). 46 Therefore, more studies in this field are required in order to elucidate the effect of SGLT-2 inhibitors on 24-hour PWV and the prognostic significance of the latter risk marker.
Study limitations
We consider as a major limitation of the present meta-analysis the limited number of eligible RCTs for inclusion in quantitative synthesis. In addition, owing to inadequate reporting, we were not able to perform subgroup analyses according to prior treatment of interest that may have an impact on PWV, such as renin-angiotensin-aldosterone system blockers. Finally, we did not register our protocol in a publicly available depository. However, this is the first meta-analysis in the literature attempting to quantify the impact of SGLT-2 inhibitors on PWV, the gold-standard measure of arterial stiffness.
Conclusion
In conclusion, we have shown that SGLT-2 inhibitors do not lead to a significant decrease in PWV under static or ambulatory conditions. Low certainty of evidence, however, justifies the need for further, large-scale RCTs to establish this effect.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221101653 – Supplemental material for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221101653 for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials by Dimitrios Patoulias, Christodoulos Papadopoulos, George Kassimis, Nikolaos Fragakis, Vassilios Vassilikos, Asterios Karagiannis and Michael Doumas in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X221101653 – Supplemental material for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X221101653 for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials by Dimitrios Patoulias, Christodoulos Papadopoulos, George Kassimis, Nikolaos Fragakis, Vassilios Vassilikos, Asterios Karagiannis and Michael Doumas in Vascular Medicine
Supplemental Material
sj-png-3-vmj-10.1177_1358863X221101653 – Supplemental material for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-png-3-vmj-10.1177_1358863X221101653 for Effect of sodium-glucose co-transporter-2 inhibitors on arterial stiffness: A systematic review and meta-analysis of randomized controlled trials by Dimitrios Patoulias, Christodoulos Papadopoulos, George Kassimis, Nikolaos Fragakis, Vassilios Vassilikos, Asterios Karagiannis and Michael Doumas in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
