Abstract
There is a growing body of evidence that diabetes represents a significant and largely modifiable risk factor for cardiovascular disease (CVD). It is known to markedly increase the risk of CVD—with CVD accounting for 2 of every 3 deaths in patients with diabetes. It is suggested that once patients with diabetes develop clinical coronary disease, they have a grim prognosis. In 2008, the Food and Drug Association mandated the evidence of CV safety in any new diabetic therapy, leading to a multitude of large CV outcome trials to assess CV risk from these medications. However, several of these outcome trials with novel antidiabetic therapies have demonstrated not only safety but a clear and definite CV advantage in patients with type 2 diabetes. In this review, we discuss 2 relatively newer classes of diabetic drugs, sodium glucose cotransport 2 inhibitors and glucagon-like peptide 1 agonists, evaluate their efficacy in improving CV outcomes, and discuss the future of CV prevention with these agents.
Introduction
Patients with type 2 diabetes have a significantly increased (2- to 4-fold) risk of developing cardiovascular (CV) disease (CVD) and carry, on average, a 6-year shorter life expectancy compared to individuals without diabetes. 1,2 Cardiovascular complications, extending from the micro- and macro-vasculature to major organs, are the overwhelming culprits of diabetes-related burden. Prior published literature attributes 40% of CVD-related deaths due to ischemic heart disease, 15% due to other forms of heart disease such as congestive heart failure (HF), and about 10% due to stroke. 1,3,4 Diabetes is now recognized as a coequal risk factor as smoking, hypertension, and cholesterol for CVD by the American Heart Association (AHA). However, despite diabetes being a major CVD risk factor, surveys indicate that approaches and therapies proven to reduce heart disease in patients with diabetes are most often neglected in clinical practice. 5
Abundant epidemiological data demonstrate strong associations between many metabolic derangements seen in diabetes and CVD, such as hyperglycemia, insulin resistance, and dyslipidemia. 6 -11 Furthermore, there are mechanisms at the molecular level that propagate atherosclerotic CVD in diabetes, including increased inflammation, endothelial dysfunction, and increased propensity for thrombosis. 1 With a rapidly aging population, the number of patients with diabetes across the globe is projected to reach approximately 600 million by 2035. The sheer magnitude of this chronic noncommunicable disease on global population health is immense. The rising burden of diabetes also underscores the necessity of innovative treatment options that may tackle these 2 major diseases concurrently. Since 2008, the US Food and Drug Administration requires evidence of CV safety for any new diabetic therapy. This has led to numerous randomized controlled trials investigating the CV risk from these medications. However, many of these outcome trials have not only validated safety of these therapies but also shown a clear and definite CV advantage in patients with type 2 diabetes. In this review, we discuss 2 relatively new classes of diabetic drugs, the sodium glucose cotransport 2 (SGLT-2) inhibitors and glucagon-like peptide 1 (GLP-1) agonists, in the context of their dedicated CV outcome trials and recent controversies and evaluate their potential in preventing and improving CV outcomes.
Sodium Glucose Cotransport 2 Inhibitors and CV Benefit
Sodium glucose cotransport 2 inhibitors act primarily through promoting glucosuria in diabetes treatment. They also positively modulate several CV parameters to reduce CVD risk: blood pressure, inflammatory and oxidative background reduction, and renal protection. These findings are likely related to the class’s glucouretic–natriuretic properties and metabolic effects in favor of consumption of ketones (instead of glucose and free fatty acids), which may provide an energy advantage for cardiomyocytes. 12 -15 (Figure 1). Ultimately, more longitudinal mechanistic studies are needed to understand the nature of these CV effects and to determine whether these cardioprotective effects are sustained. Still, evidence from pooled randomized control trials (RCTs) show a significant reduction in myocardial infarction, hospitalizations for HF, and CV mortality in patients with diabetes on SGLT-2 inhibitors. Despite this, the SGLT-2 inhibitors carry an increased risk of urinary tract infections and genital infections, most likely due to glucosuria induced by these agents. The effect on renal function is controversial, since there have been reports of acute kidney injury; however, RCTs suggest there may rather be some renal protective effects. We now turn to discuss a select few hallmark clinical trials using SGLT inhibitors on CV outcomes.

Sodium glucose cotransport 2 (SGLT-2) inhibitors and glucagon-like peptide 1 (GLP-1) agonists.
EMPA-REG OUTCOME Study
The EMPA-REG OUTCOME study represented a giant leap in diabetes management: It was the first trial to mark success for secondary and tertiary CVD prevention with a glucose-lowering agent. The study randomly assigned more than 7000 patients with type 2 diabetes mellitus (T2DM) and preexisting CVD to receive either empagliflozin or matching placebo and found that after 3 years of active therapy, empagliflozin was associated with a marked reduction in mortality (1 in 3 CV-related deaths). 16 Heart failure hospitalization and progression of kidney disease also decreased significantly (35% and 39%, respectively) in patients treated with empagliflozin. Interestingly, the CV mortality and HF hospitalization curves diverged early, suggesting an abrupt, more hemodynamic, or metabolically driven effect suggestive of a predominant diuretic effect. Finally, there was no significant difference in the risk of stroke with empagliflozin versus placebo. The only adverse effect observed in the trial was an increase in genital infections, a previously well-known complication of glucosuria. Prior to EMPA-REG OUTCOME, no studies have showed successful reduction in CV outcomes with pharmacological intervention in patients with T2DM.
CANVAS Study
Results from the CANVAS trial further substantiated the findings from EMPA-REG OUTCOME for secondary prevention. This study included just over 10 000 patients randomized to canagliflozin or placebo and followed for 3.6 years. The primary prevention cohort had 34% of the study’s participants, who had 2 or more CV risk factors, while the secondary prevention cohort was comprised of the rest of the participants, who had a prior CV event. Treatment with canagliflozin resulted in a lower incidence of death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, and hospitalization for HF (latter, 16.3 vs placebo 20.8 per 1000 patient-years; hazard ratio, 0.78; 95% confidence interval, 0.67-0.91). 17 Moreover, patients with a prior history of HF appeared to benefit more than those without HF at baseline. Like the empagliflozin curves, the benefits of canagliflozin were evident early during follow-up, which can be attributable to the effects of osmotic diuresis and afterload reduction. 17,18 Overall, the reductions in risk for the primary end points, HF hospitalizations, and renal outcomes were similar between the 2 trials. However, empagliflozin boasted a 32% relative risk reduction in all-cause mortality compared to the 11% offered by canagliflozin.
Canagliflozin offered a huge success for secondary prevention, with (1) 18% reduction in the risk of the composite end point of CV death, nonfatal MI, or nonfatal stroke; (2) 41% reduction in renal outcome worsening; and (3) 32% reduction in HF hospitalization, compared to placebo. Notably, these CV and renal end points were at least 2 times higher in the secondary prevention cohort than in the primary prevention group. The primary prevention cohort did not offer significant benefit in the composite outcome of CV events over the placebo group.
CVD-REAL Trials
The CVD-REAL study was a large multinational program in which treatment with SGLT-2 inhibitors versus other diabetic agents was associated with lower risk of all-cause death and HF hospitalization. The results from the observational CVD-REAL studies support findings of the 2 aforementioned RCTs; however, this trial differs from the prior ones in that it was the first to study patients with T2DM who did not have preexisting CVD. These patients had a 51% lower risk of death and a 39% lower risk of hospitalization for HF than those starting another diabetic agent. 19 Better yet, these findings were consistent across different populations across the world. What does this mean? It suggested SGLT-2 inhibitors could be useful for patients in earlier stages of T2DM by offering a primary CVD prevention. Recently, the findings from CVD REAL 2 extend this CV benefit to outcomes in a relatively more Asian population. 20
However, this excitement may be guarded considering in light that the CVD-REAL is an observational study and not an RCT. There is also the possibility of confounding in CVD-REAL, with the SLGT2 group having been on more beneficial cardiac therapies (statins, angiotensin-converting enzyme inhibitors, and angiotensin receptor blockers). Another bias raised (with nearly all of these studies) is failing to account the duration of other therapies used immediately before initiation of the diabetic agent, with a possible washout effect.
Recent/Future Trials
Recently, preliminary results from DECLARE-TIMI-58 were released, with dapagliflozin showing reduction in composite end point of hospitalization for HF or CV death. The study examined the CV outcomes of dapagliflozin over 5 years in more than 17 000 patients with T2DM and elevated CV risk. This is the first CV outcome trial to have 2 co-primary efficacy outcomes, with the full results to be presented later this year. Researchers are also now looking ahead to VERTIS for how ertugliflozin affects CVD. Furthermore, there are several trials underway to assess the effects of SGLT-2 inhibitors specifically in patients with HF, including the EMPEROR (empagliflozin) and DAPA-HF (dapagliflozin) trials. Finally, CREDENCE (canagliflozin) and DAPA-CKD (dapagliflozin) will examine CV outcomes in patients with renal disease.
Glucagon-Like Peptide 1 Agonists and CV Benefit
Glucagon-like peptide 1 is an incretin hormone that stimulates insulin secretion, inhibits glucagon production, and delays gastric emptying. Preclinical and clinical studies suggest that the injectable GLP-1 agonists exert favorable effects in endothelial function and cardiac function (through blood pressure reduction and weight loss), as well as in reducing inflammatory markers. 21 Subsequently, we briefly discuss 4 major cardiovascular outcome trials for GLP-1 agonists and entertain a discussion on the future of clinical application of these agents in preventive cardiology.
Notably, these trials, as with the SGLT-2 inhibitor studies, show a significant risk reduction in major adverse CV events in patients with preexisting CVD and/or high risk for events. Despite this, much of the research and spotlight within cardiology has focused on SGLT-2 inhibitors. These 2 classes, however, may confer divergent CV benefits—where the SGLT-2 inhibitors show a stronger reduction in HF-related events and the GLP-1 agonists appear to improve CV outcomes mainly by reduction in atherosclerotic events. 22 The mechanisms are thought to be related to SGLT-2 inhibitors stimulating a diuretic effect and fat reduction, while GLP-1 agonists driving a more central effect. 22
LEADER Trial
The LEADER trial investigated CV outcomes in over 9000 patients with T2DM having preexisting CVD treated with liraglutide versus placebo who were followed for close to 4 years. In the patients who received liraglutide, there was a 13% significant reduction in composite of CV death, nonfatal MI, and nonfatal stroke compared to placebo, as well as all-cause mortality (8.2% vs placebo 9.6%; P = .02). There was no difference in HF hospitalization rates. As tempting it is to evaluate the results of LEADER and EMPA-REG OUTCOME head to head, as we consider below with additional trials, we must keep in mind the variations in study design for both. The authors of EMPA-REG OUTCOME show that empagliflozin has a faster effect on HF, while liraglutide plays a role in reducing atherosclerotic disease burden. 23 In addition to LEADER, there is an ongoing trial, Effects of Liraglutide in Young Adults with Type 2 Diabetes, 24 which assesses the effect of liraglutide on cardiac structure and function in younger obese patients with T2DM.
SUSTAIN-6 Trial
SUSTAIN-6, which studied semaglutide, had similar inclusion criteria as LEADER. The semaglutide group had significantly lower composite major adverse cardiovascular events (MACEs; comprising of CV death, nonfatal MI, and nonfatal stroke)—6.6% versus placebo 8.9%; P = .02, and reduction in events of stroke (1.6% vs placebo 2.7%; P = .04), in the study’s relatively short follow-up duration (median: 2.1 years). There was no reduction in CV death observed in this trial (2.7% in the semaglutide group vs 2.8% placebo). 25 Taken together, SUSTAIN-6 revealed a greater reduction in composite MACE score than EMPA-REG (26% vs 14%), a comparison often made between classes. However, neither liraglutide nor semaglutide significantly reduced HF admissions as seen with the SGLT-2 inhibitors.
ELIXA Trial
The ELIXA trial assessed the CV outcomes in patients with T2DM who had a recent coronary event. There were similar rates of major adverse CV events between placebo and the lixisenatide groups (13.2% vs 13.4%, respectively), with no significant differences in rate of hospitalization for HF or rate of death. Both LEADER and SUSTAIN-6 showed significant risk reduction in composite MACEs; however, ELIXA showed no change in composite MACEs during follow-up. However, compared to the former trials, ELIXA included patients with a recent event, potentially abrogating any benefit offered by short-term pharmacological diabetic intervention.
EXSCEL Trial
The latest results to emerge are those of EXSCEL, 26 which tested exenatide on CV outcomes. EXSCEL enrolled around 14 700 patients, out of which approximately three-fourth had a previous history of CV events. The EXSCEL trial demonstrated noninferiority for exenatide, with all-cause mortality statistically decreased by 14% and CV death decreased by 12% in the treatment group. While the treatment group had lower overall mortality, the data did not meet statistical significance required for superiority.
Despite the dissatisfaction that may have arisen with the findings from this trial, it fuels the question of appropriateness in comparisons among trials of the same and different classes. For instance, the EXSCEL was a much larger trial than SUSTAIN-6 and with high-risk patients (as ELIXA). Moreover, the trial duration was shorter for EXSCEL (2.4 years) compared to 3.5 years in LEADER. In other words, while EXSCEL may not have shown a statistically significant reduction in primary CV outcome, it resonates with the overwhelming findings of the earlier GLP-1 agonist trials with a major reservation being unable to claim a reduction in all-cause death due to reduction in CV mortality.
Recent/Upcoming Trials
Finally, this year, the HARMONY trial validated some of the guarded excitement over CV benefits of GLP-1 agonists. This study investigated almost 9500 patients treated with either albiglutide (which was removed from the market in 2017) or placebo for a median 1.6-year follow-up period. Albiglutide was shown to significantly reduce the risk of MACE by 22%; however, the study did not show a significant reduction in death from CV causes, as was demonstrated with liraglutide, another GLP-1 agonist in the LEADER trial. This finding may be related to the short duration of follow-up in the study. Still it is unclear whether albiglutide will enter the market again or branded by a different company after this study. PIONEER-6, FREEDOM CVO, and REWIND are ongoing trials in the GLP-1 agonist class (Table 1).
Outcomes Associated with SGLT-2 Inhibitors and GLP-1 Agonists.
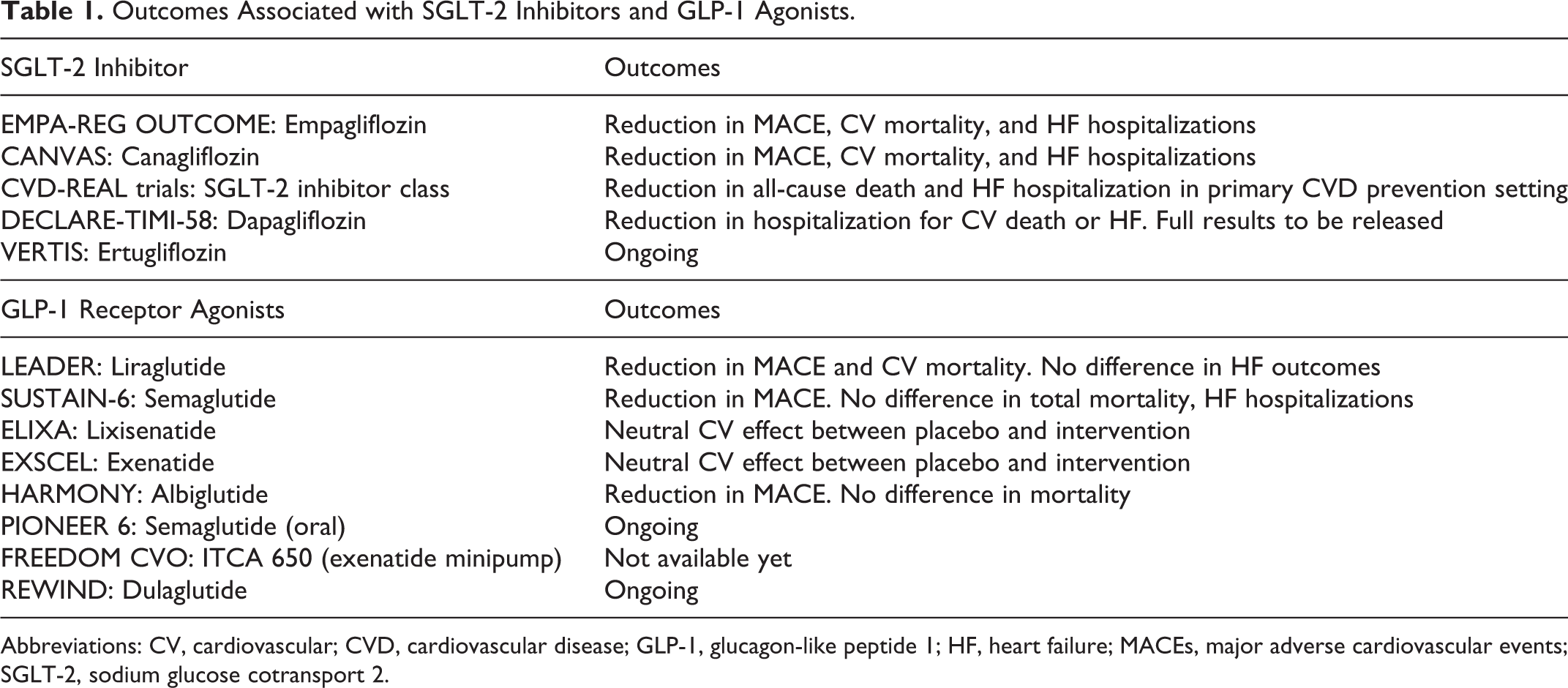
Abbreviations: CV, cardiovascular; CVD, cardiovascular disease; GLP-1, glucagon-like peptide 1; HF, heart failure; MACEs, major adverse cardiovascular events; SGLT-2, sodium glucose cotransport 2.
Conclusions
Cardiovascular risk reduction is an essential part of comprehensive care for patients with diabetes. Of recent, diabetes management has focused on macrovascular complications as a part of preventive cardiology—relying heavily on large RCTs and epidemiological data to guide clinical recommendations. What is fascinating about these new glucose-lowering agents is using them to step beyond the dogma of merely modulating conventional risk factors—blood pressure, weight, and lipids—to improve CV risk. We are now giving due attention to emerging mechanisms, namely, inflammatory pathways, the cardio–renal axis, and volume-driven metabolic changes.
Both SGLT-2 inhibitors and GLP-1 agonists may offer a degree of cardioprotection in patients with preexisting CVD. While providing specific recommendations for instances when to use an SGLT2 inhibitor over a GLP-1 agonist is beyond the scope of this article, a recent meta-analysis found SGLT2 inhibitors to be better for HF and myocardial infraction end points, while GLP-1 agonists ranked better for stroke outcomes. 27 Considering these trials have been short-term studies, prospective studies are needed to investigate the effects on long-term vascular end points and mortality.
Together, we are faced with many questions: Do these drug classes confer a benefit only when there is a background of cardiovascular derangement? Can the latter be prevented with earlier treatment? The results of the DECLARE trial (RCT with dapagliflozin as primary prevention) will allow us to come closer in answering some of these questions for SGLT-2 inhibitors. Furthermore, recent mechanistic data indicate that these medications may have synergistic effects on CV risk if administered concurrently.
We are seeing a new wave of CV prevention with more nuanced pathophysiological underpinnings and costly pharmacological therapies as elucidated by the CANTOS trial, 28 as well as all the other trials discussed in this review. A timely question that follows will be: Is it worth it? We cannot overlook cost going forward, as it is an influential player in prescribing practices. Which agent should be first-choice therapy for CV prevention in patients with T2DM in light of cost and benefit? Can the effect of these agents be achieved by a cheaper combination of pills—with simultaneous reduction in glucose, diuresis, and conferring CV benefit (ie, a combination of metformin and thiazide)? Diabetes care demands not merely glucose control but also long-term care strides for vascular health. The utilization of new diabetic agents in potentially reducing both risks may represent a radical paradigm shift in the way T2DM is managed.
Footnotes
Author Contributions
Aparna P. Sajja and Amit K. Dey have contributed equally to this study. Aparna P. Sajja contributed to conception and design and drafted the manuscript. Amit K. Dey contributed to conception and design, drafted the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. Avirup Guha drafted the manuscript and critically revised the manuscript. Youssef Elnabawi and Aditya A. Joshi drafted the manuscript and critically revised the manuscript. Ankur Kalra critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
