Abstract
Although angiography has been the primary imaging modality used in peripheral vascular intervention, this technique has major limitations due to the evaluation of three-dimensional vessels in two dimensions. Intravascular ultrasound (IVUS) is an important adjunctive tool that can address some of these limitations. This systematic review assesses the appropriateness of IVUS as an imaging modality for guiding peripheral intervention through evidence collection and clinical appraisal of studies. Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, a cohort of 48 studies (29 arterial; 19 venous) detailing IVUS use in peripheral vascular intervention were extracted. Qualitative assessment of the studies evaluated pre- and postprocedure efficacy of IVUS and revealed that IVUS-guided peripheral intervention in arterial and venous diagnosis and treatment was superior to other imaging techniques alone. Each study in the cohort was further assessed for reliability and validity using the Oxford Centre for Evidence Based Medicine (CEBM) level of evidence scale. The majority of both arterial (79.3%) and venous (73.7%) studies received a 2b rating, the second highest level of evidence rating. The evidence to date indicates that IVUS results in better clinical outcomes overall and should be more widely adopted as an adjunctive imaging modality during peripheral intervention.
Keywords
Background
Peripheral intravascular ultrasound (IVUS) is a novel imaging modality that can be used adjunctively with angiography during peripheral vascular intervention. Although angiography remains the mainstay in image guidance during peripheral intervention, it has limitations. For instance, angiography alone produces two-dimensional projections of three-dimensional vessels, which can be challenging to interpret in the setting of vascular tortuosity and may underrepresent the extent of complex and eccentric lesions. Conversely, IVUS produces cross-sectional images of the vascular lumen, providing detailed insight into plaque and thrombus morphology. These images can be used to aid in evaluating the adequacy of vessel preparation prior to percutaneous intervention and assessing optimization of procedures including stent implants postprocedure.1,2
There are numerous randomized trials assessing the utility of IVUS in percutaneous coronary interventions. The benefit of IVUS in these trials has been driven by decreased rates of target vessel revascularization and stent-related events.3,4 The use of IVUS to guide peripheral interventions, both arterial and venous, is less well studied. To date there are limited prospective studies or trials assessing head-to-head outcomes between angiography-guided and IVUS-guided peripheral vascular intervention (PVI). Most of the current evidence evaluating IVUS exists in the form of retrospective observational cohort studies.
Although randomized controlled trial (RCT) data concerning the efficacy of IVUS in peripheral vascular interventions compared to the current standard of care is needed, we sought to summarize and critically appraise the existing studies comparing IVUS to angiography and alternative imaging modalities during peripheral intervention. The purpose of this contemporary systematic review is to summarize the existing evidence and independently grade the quality of existing evidence.
Methods
Our methods (including literature search and corresponding analyses) followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines.
5
The study was registered with
Inclusion/exclusion criteria
We comprehensively searched (online Supplemental Tables 1 and 2) PubMed, Cochrane, and Embase libraries for human-based studies that evaluated the utility of IVUS in diagnosing and treating lower extremity peripheral artery disease and iliofemoral venous disease. Only studies which compared IVUS to other imaging modalities, such as angiography or venography, in peripheral lower extremity locations (iliac, femoral, popliteal, and tibial arteries; iliofemoral veins) were eligible. Our search criteria, as described in online Supplemental Table 1 for arterial and online Supplemental Table 2 for venous, included various combinations of peripheral intervention and disease-based medical terms with ‘intravascular ultrasound’, such as ‘claudication/critical limb ischemia’ in the arterial circulation and ‘deep vein thrombosis’ in the venous circulation. In addition, Medical Subject Headings (MeSH) terms were constructed for querying PubMed.
All databases were searched from 01/01/1995 to 04/30/2021 and studies were restricted to human subjects only. Single-case studies, or case reports which discussed the success of IVUS in only one patient, were not eligible. Reviews and editorial publications that did not directly discuss new research but rather commented on existing studies were also not eligible. In addition, we excluded all existing systematic reviews and meta-analyses, but conducted an extensive search of their references to ensure that our selection of eligible studies was thorough. Furthermore, we omitted studies that were in a language other than English to minimize translation and/or comprehension errors.
Resulting studies from the three database searches were imported into Covidence (Melbourne, Australia), which was used to manage the review process of studies for eligibility assessments. Two reviewers (SN and JS) independently examined the titles, abstracts, and full texts of the studies for inclusion, and a third reviewer (ES) was available for any necessary tie-breaks. All reviewers in this study were in agreement of the inclusion criteria. Following this, all three investigators (SN, JS, and ES) reviewed all potentially eligible studies to confirm that all criteria were met. For each included study, study-level data were exported into tables, including the indication for the study, study design, sample size, endpoints, and findings.
Following detailed review of the included studies, it was decided not to perform an outcomes-based meta-analysis due to concern about consistency in measured endpoints across studies and heterogeneity in study populations and disease locations included in the studies.
Grading the level of evidence
The final cohort of studies were further assessed for reliability and validity using the Oxford Centre for Evidence- Based Medicine (CEBM) level of evidence scale (online Supplemental Table 3). A widely accepted tool for clinical appraisal, the CEBM scale ranges from level 1-5, with level 1 representing the strongest level of evidence. In addition, CEBM categorizes studies as prognostic, diagnostic, or therapeutic. Each investigator (SN, JS, and ES) was trained on applying these criteria using online material and independently evaluated each study’s level of evidence using the full text. For any disagreement, the majority vote decided the final rating.
Results
Lower extremity peripheral artery studies
Twenty-nine studies detailing the use of IVUS in the management of lower extremity peripheral artery studies met the inclusion criteria (Figure 1). Of the included studies, 25 were cohort studies and four were case series. The total number of patients studied was 95,192, and the majority of studies evaluated IVUS in a cohort of > 10 patients.
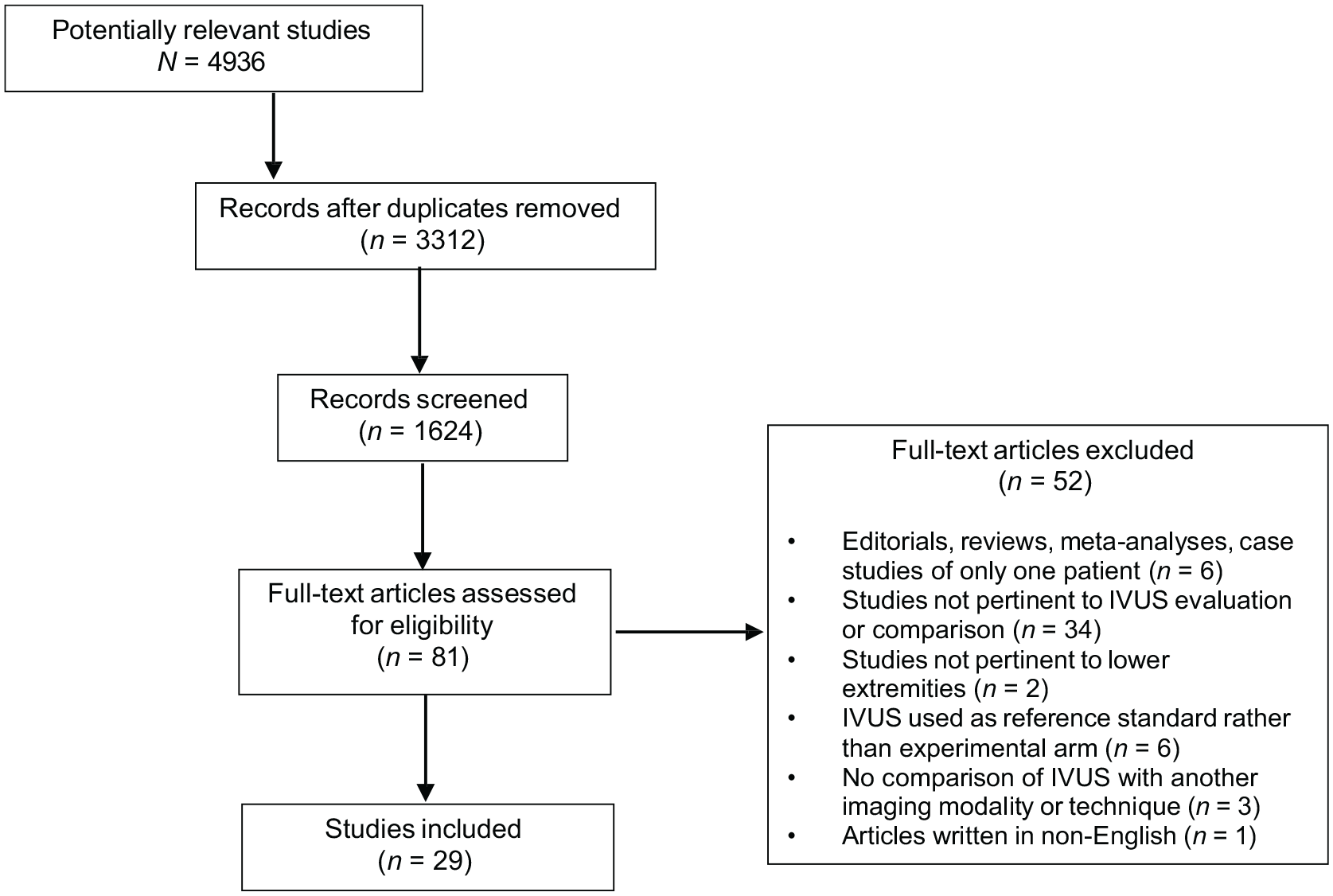
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram for determining inclusion of lower extremity peripheral arterial studies, which shows the total number of studies captured in the literature search, the exclusion criteria, and the final number of studies (n = 29) deemed eligible to assess the use of intravascular ultrasound (IVUS) in lower extremity peripheral arterial interventions. Eligibility of studies was determined through a systematic approach as outlined in the PRISMA guidelines.
A sample of arterial studies can be found in Table 1, with the complete list of eligible arterial studies organized by clinical scenario in online Supplemental Table 4. The majority of studies (18/29) examined the role of IVUS during revascularization for device sizing, placement, and optimization. Of the remaining studies, 6/29 examined the utility of IVUS in the evaluation of lesion characteristics and severity, 3/29 studies explored IVUS use in the management of arterial dissections, and 2/29 studies studied IVUS guidance for reentry of chronic total occlusions (CTOs). The general consensus across all 29 studies was that IVUS-guided intervention resulted in improved clinical outcomes compared with angiography alone.
Sample of lower extremity peripheral artery studies.
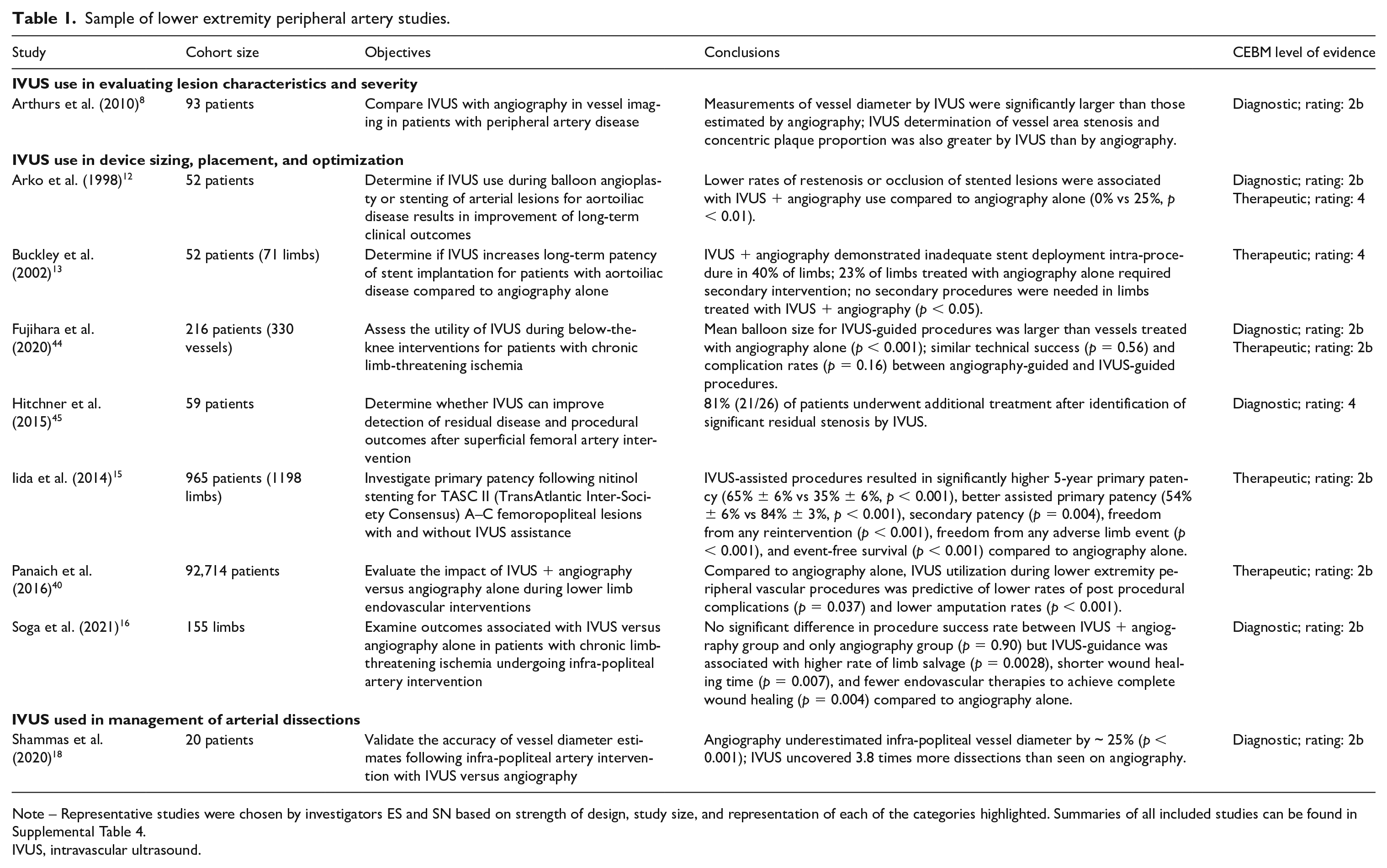
Note – Representative studies were chosen by investigators ES and SN based on strength of design, study size, and representation of each of the categories highlighted. Summaries of all included studies can be found in Supplemental Table 4.
IVUS, intravascular ultrasound.
Of all the studies included, 23 (79.3%) received a level 2b rating, the second highest level of evidence rating. All of the studies evaluating lesion characteristics and severity were level 2b (6/6, 100%). Similarly, for the studies evaluating device-based interventions, the majority (13/18, 72.2%) were level 2b, and the remaining studies were level 4. For studies assessing IVUS use for CTO reentry, 50% (1/2) of studies were level 2b and for those assessing IVUS use in the management of arterial dissections, 100% (3/3) of the studies were level 2b. The majority 2b rating indicates that the studies evaluating IVUS use compared to imaging modalities such as angiography were robust and of good quality.
Iliofemoral venous studies
Nineteen studies examining the utility of IVUS during iliofemoral venous procedures met the criteria and were included in the review (Figure 2). Of the 19 included studies, 18 were cohort studies, one of which was a prospective multicenter cohort study, and one was a case series. The total number of patients studied was 2630, and the majority of studies evaluated IVUS in a cohort of > 10 patients.
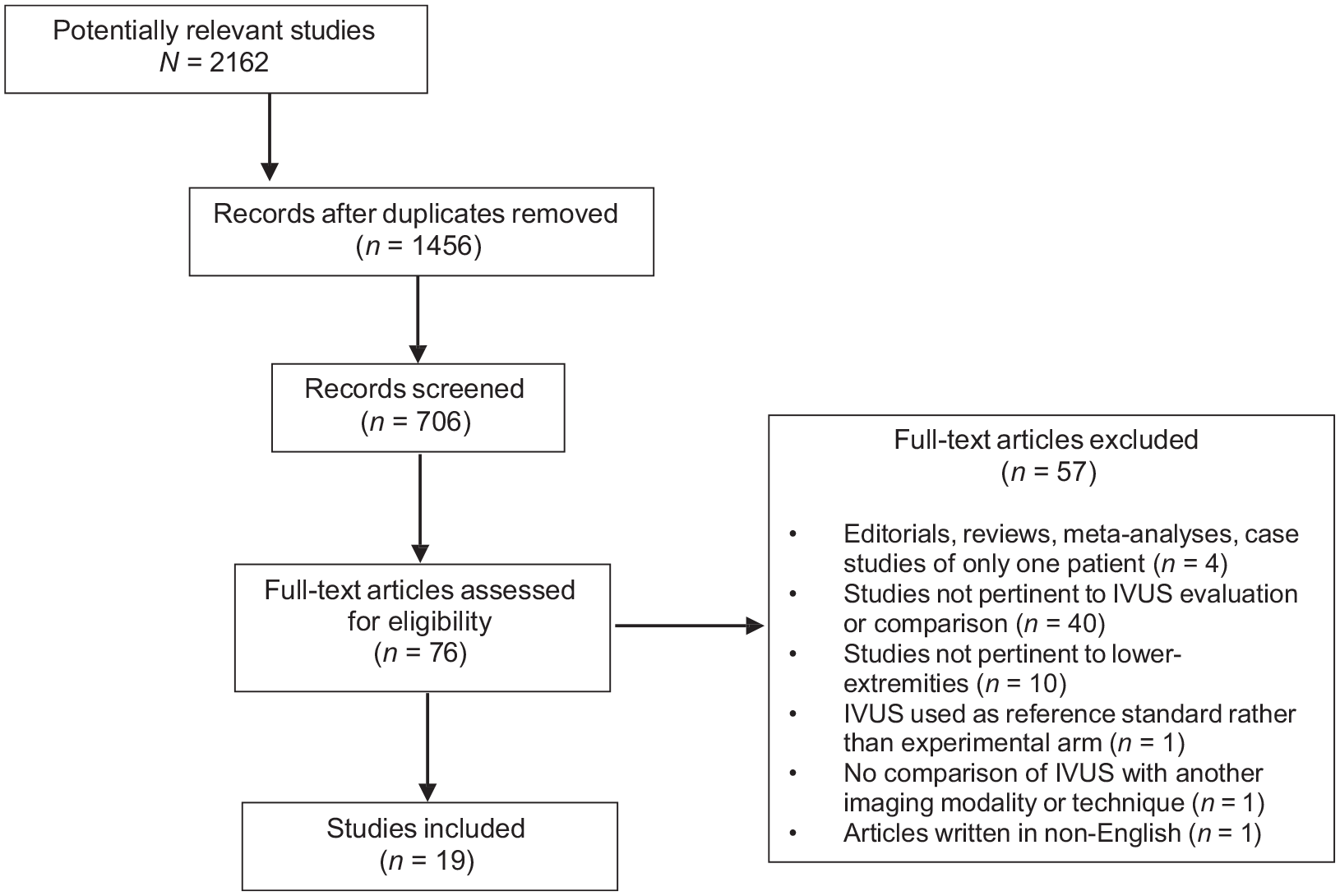
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram for determining inclusion of iliofemoral venous studies, which shows the total number of studies captured in the literature search, the exclusion criteria, and final number of studies (n = 19) deemed eligible to assess the use of intravascular ultrasound (IVUS) in iliofemoral venous interventions. Eligibility of studies was determined through a systematic approach as outlined in the PRISMA guidelines.
As described in the sample of iliofemoral venous studies (Table 2) and further detailed in online Supplemental Table 5, IVUS was compared to other imaging modalities to evaluate lesion characteristics and to evaluate lesion severity in the majority of studies (11/19). Other studies assessed the utility of IVUS to assist in stent sizing, placement, and optimization (5/19), and to evaluate thrombotic-related venous disease (3/19). Comparator venous imaging techniques included venography, duplex ultrasound, and computed tomography. The general consensus across all 19 studies was that IVUS-guided intervention resulted in improved clinical outcomes compared with other imaging modalities.
Sample of iliofemoral venous studies.
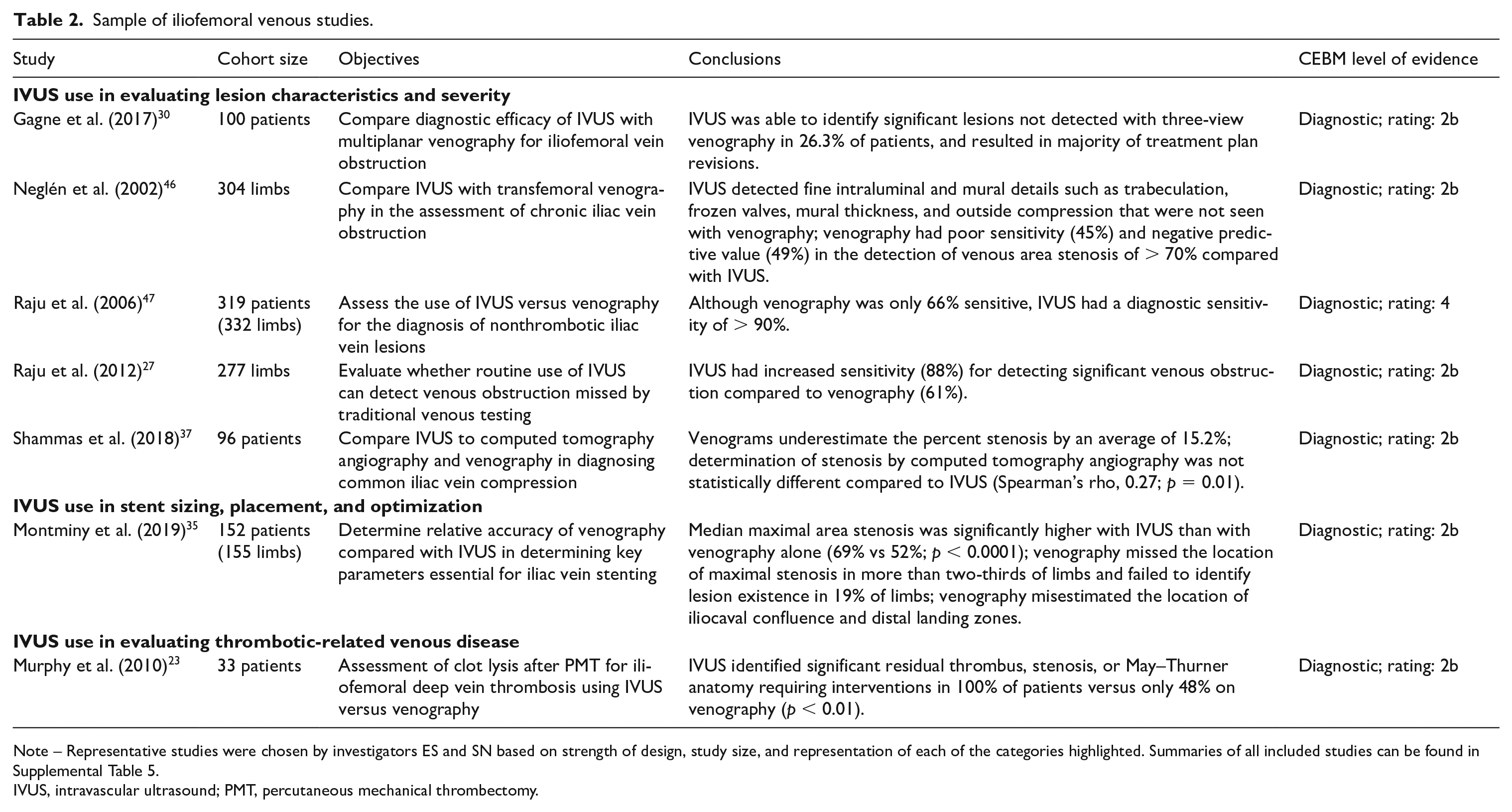
Note – Representative studies were chosen by investigators ES and SN based on strength of design, study size, and representation of each of the categories highlighted. Summaries of all included studies can be found in Supplemental Table 5.
IVUS, intravascular ultrasound; PMT, percutaneous mechanical thrombectomy.
Of all included studies, 14 (73.7%) received a 2b rating and five studies received a rating of 4. Among the 11 studies evaluating lesion characteristics and severity, the majority (9/11, 81.8%) were of high quality, with a level 2b rating. Of the five studies evaluating stent placement and optimization, the quality of studies varied, with 2/5 (40%) receiving a 2b rating. For studies evaluating thrombotic-related disease, all were of high quality, and 100% (3/3) received a rating of 2b.
Discussion
Although there are large-scale RCT data to support that IVUS use improves outcomes during percutaneous coronary intervention, there are limited prospective investigations assessing the efficacy of IVUS during peripheral venous and arterial intervention. Nonetheless, in this systematic review of studies in which the utility of adjunctive IVUS was evaluated during peripheral intervention, there was robust evidence consistently demonstrating a benefit of IVUS over comparative imaging modalities. In particular, these data suggest that IVUS can have a role in improving vessel characterization, vessel preparation, effective stent deployment, and monitoring for postprocedural complications during both arterial and venous peripheral interventions in appropriately selected patients. Furthermore, the majority of these studies were of high quality, with CEBM level of evidence ratings of 2b.
Studies evaluating the use of IVUS during lower extremity peripheral artery intervention reliably demonstrated greater ability to characterize targeted lesions prior to intervention compared with angiography alone. In the reviewed data, the use of preprocedural IVUS during lower extremity arterial intervention demonstrated improved accuracy in estimating vessel diameter6,7 and hemodynamic significance,8,9 which outperformed angiography alone. IVUS was also superior to angiography in assessing lesion properties, such as eccentricity, presence of thrombus, and calcification.8,10 Furthermore, IVUS had practical benefits intra-procedurally. Specifically, IVUS use in guiding stent placement has been shown to influence and inform clinical decisions. For instance, in the endovascular management of atherosclerotic aortoiliac occlusive disease, one study concluded that in roughly 40% of patients, IVUS had the capability to uncover limitations of previous stent placements (i.e., under-deployment) that angiography had deemed adequately expanded. 11 The evidence surrounding IVUS use as it relates to clinical outcomes in peripheral artery intervention is limited, but overall supportive. Several studies suggest that IVUS improves gain in vessel diameter and stent patency and resultant freedom from reintervention.12 –16 IVUS also enhances sensitivity for detecting procedural complications, including arterial dissections.17 –22 This may result in important changes to the treatment plan to minimize short-term peri-procedural complications.
This systematic review also found that IVUS is an effective tool in diagnosing, characterizing, and evaluating iliofemoral venous disease. When compared to venography alone, IVUS was found to uniformly improve procedural outcomes. Prior to peripheral venous intervention, IVUS was shown to be effective at assessing thrombus burden,23 –26 venous obstruction,27,28 and measuring lesion diameter29-31 with more precision than venography alone. IVUS was also shown in multiple studies to more effectively detect stenotic lesions that were missed or under-evaluated by venography alone.32,33 Thus, IVUS provides valuable information that can inform peripheral venous intervention and be used to make clinically relevant treatment decisions. Appropriate measures of lesion diameter or size result in more accurate stent placement24,34 and reduces the risk of migration and in-stent restenosis.35,36
This systematic review clearly demonstrated that IVUS is a much more valuable imaging modality than angiography or venography alone. Nonetheless, IVUS remains under-utilized during peripheral vascular interventions. This may be due to the lack of large-scale RCT data to adequately quantify the benefits of IVUS and evaluate long-term outcomes. Furthermore, IVUS might be rivaled by other nascent imaging techniques, such as computed tomography.37,38 In addition, there are little data regarding the cost effectiveness of IVUS compared to other, more historic and available imaging modalities 39 and thus it is possible that cost serves as a barrier to more widespread IVUS use in peripheral artery and venous disease.13,40 Although an RCT may improve adoption, IVUS has been demonstrated to be safe,41,42 can be implemented into the procedural workflow without substantial change in procedure length of time, and has been well-established in improving outcomes during coronary intervention. With this in mind, and the preponderance of data summarized in this review, more frequent use of IVUS may assist in improving outcomes during peripheral vascular procedures, which have been plagued with poor long-term results.12,13,43
Study limitations
This study has several limitations. While conducting the systematic search for potentially eligible studies to include in this review, the search criteria was designed to be as broad and to include as many applicable terms as possible. However, owing to differences in database sorting and algorithms between PubMed, Cochrane, and Embase, there was no feasible way to ensure that the search strategy was exactly the same across each platform. To mitigate this issue, the search strategy was vetted and examined by experts from the Harvard University library to ensure that there was continuity and accuracy between the various database searches. Furthermore, we specifically chose PubMed, Cochrane, and Embase as the three databases to search. Other databases, including MEDLINE and Web of Science, were omitted for redundancy or inability to maintain cohesiveness with search terms. Of note, all of the manuscripts captured in MEDLINE were present in the PubMed search. To address missing studies that may have been outside of the time frame of our search or excluded from the databases, we searched the references of a number of related papers (namely other systematic reviews) to ensure that our list of eligible studies was as comprehensive as possible. Further limitations include the size and heterogeneity of eligible studies, including differences in the adjudication of target endpoints, and inconsistent reporting of individual patient-level data and outcomes.
In addition, our systematic review included studies that span a broad time frame (1996–2021), during which time the quality of imaging, ease of operation, and technology of IVUS has improved. Although some earlier studies may have assessed the clinical outcomes associated with using IVUS while it was still in a rudimentary form, our assertions about the utility of IVUS today are strengthened by the improvement in IVUS technology over time. Although aggregating data into a meta-analysis would have been useful to further evaluate the role of IVUS during peripheral vascular intervention, upon detailed review of the available studies, we felt that the specific endpoint definitions, timing of outcome adjudication and consistency in endpoint reporting were too heterogeneous to accurately meta-analyze outcome data. As such, we elected to focus on summarizing available data and evaluating the quality of evidence.
Conclusion
The systematic review of IVUS in peripheral artery and venous interventions resulted in a combined 48 eligible studies, the majority of which were retrospective cohort studies. Most studies were of a high level of evidence (2b, n = 37), suggesting that though most studies were non-RCTs, they represent evidence of high quality. This evidence base provides clear support demonstrating the superiority of IVUS when compared with angiography or venography during peripheral artery and venous intervention. Further effort is now needed to focus on improving adoption and utilization of this important adjunctive imaging device.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221092817 – Supplemental material for Intravascular ultrasound in peripheral venous and arterial interventions: A contemporary systematic review and grading of the quality of evidence
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221092817 for Intravascular ultrasound in peripheral venous and arterial interventions: A contemporary systematic review and grading of the quality of evidence by Sahana Natesan, Ramya C Mosarla, Sahil A Parikh, Kenneth Rosenfield, Joanna Suomi, David Chalyan, Michael Jaff and Eric A Secemsky in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Sahil Parikh is a member of the advisory board of Abbott, Boston Scientific, CSI, Janssen, Medtronic, and Philips; conducts research at Abbott, Boston Scientific (DSMB), Shockwave, Surmodics, TriReme, and Veryan Medical; and serves as a consultant for Abiomed, Inari, Penumbra, and Terumo. Dr Ken Rosenfield is a consultant for and serves on the scientific advisory board of Angiodynamics, Boston Scientific, Contego, InspireMD, Magneto, Mayo Clinic, Neptune Medical, Philips, Summa Therapeutics, Surmodics, Thrombolex, and Truvic; has grants from the NIH, Boston Scientific, Equity, Accolade, Access Vascular, Althea Medical, Contego, Cruzar Systems, Embolitech, Endospan, JanaCare, Magneto, Orchestra, PQ Bypass, Shockwave, Thrombolex, and Truvic; and is a board member at the National PERT Consortium, a not-for-profit 501c3 organization dedicated to advancing treatment and improving outcomes in pulmonary embolism. Dr David Chalyan is an employee at Philips IGT, an organization specializing in image-guided therapy for complex procedures using systems such as intravascular ultrasound. Dr Michael Jaff is a part-time employee at Boston Scientific Corporation, a manufacturer of interventional procedure medical devices including IVUS; is a consultant at Glide Healthcare; and has equity investments in R3 Vascular, Nectero, Vactronix, and Efemoral. Dr Eric Secemsky is a consultant for, a member of the speakers bureau, and on the advisory board at Bard, Cook, CSI, Janssen, Medtronic, and Philips; and has grants with AstraZeneca, Bard, Boston Scientific, Cook, CSI, Medtronic, and Philips. The remaining authors have nothing to disclose.
Funding
Research reported in this study is supported in part by the National Heart, Lung, and Blood Institute of the National Institutes of Health under award number K23HL150290. Joanna Suomi was compensated by the Smith Center for Outcomes Research in Cardiology for her contributions to this review. This review is also funded in part by unrestricted educational grants from Boston Scientific and Philips IGT, who manufacture IVUS catheters, to Beth Israel Deaconess Medical Center. Authors David Chalyan and Michael Jaff, who are employees of Philips and Boston Scientific, respectively, participated in editing and scientific feedback of the manuscript. Boston Scientific and Philips had no role in the decision to submit the manuscript, and, outside of contributions via David Chalyan and Michael Jaff, these companies had no access or participation with the creation of the manuscript.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
