Abstract
Background:
Multiple frailty screening tools are implemented; however, it is unclear whether they perform in a comparable way for both frailty detection and prediction of perioperative outcomes in patients undergoing lower-extremity revascularization.
Methods:
Patients undergoing lower-extremity revascularization were identified from the Vascular Quality Initiative (VQI) national database. Two cohorts were established based on the revascularization type (percutaneous vascular interventions (PVI) or lower-extremity bypass). Frailty was assessed by the 5-item modified frailty index (mFI-5) and the VQI-derived risk analysis index (RAI).
Results:
Out of 134,081 patients undergoing PVI, frailty was identified in 67% by mFI-5 and 28% by RAI. Similarly, out of 41,316 patients in the bypass cohort, frailty was identified in 69% by mFI-5 and 16% by RAI. There was little agreement between the two frailty tools for both vascular cohorts (PVI: kappa: 0.17; bypass: kappa: 0.13). In an adjusted analysis, frailty as assessed by mFI-5 and RAI was associated with higher odds of mortality in both cohorts (p < 0.001). A significant association between frailty and unplanned amputations was only noted in the bypass cohort when RAI was applied (OR: 1.50, p < 0.01). The addition of frailty to traditional PAD risk factors marginally improved model performance to predict mortality and unplanned major amputations.
Conclusion:
There was significant variation in frailty detection by mFI-5 and RAI. Although frailty was associated with mortality, the predictive value of these tools in predicting outcomes in PAD was limited. Future research should focus on designing new frailty screening tools specific to the PAD population.
See Commentary: Strom JB, Secemsky EA. Seeing the entire elephant: The challenges of frailty assessment for peripheral artery disease. Vasc Med. 2022; 27: 259-261.
Introduction
In carefully selected patients, revascularization strategies are indicated for the management of lower-extremity peripheral artery disease (PAD) alongside guideline-directed medical therapy.1–4 With increasing prevalence of PAD among aging populations, revascularization procedures are increasingly offered to this high-risk group who often present with multiple comorbidities.2–4 In an effort to weigh the risk–benefit for vascular procedures and to improve clinical outcomes following these procedures, more attention is being given to the identification of PAD patients who may be vulnerable to adverse clinical outcomes.2,3,5,6 One such factor that could aid in the understanding of patients’ perioperative risk following revascularization procedures is frailty.
In December 2012, a consensus statement of six international geriatric societies produced an operational definition of frailty – a state of enhanced vulnerability to physiological stressors and adverse clinical events. 7 However, frailty assessment remains cumbersome and lacks standardization.8–10 Two approaches to frailty assessment have become popular: the deficit accumulation model, which relies on the impairment summation across different domains of physical health; 11 and the frailty phenotype model, based on five predetermined criteria (weight loss, exhaustion, weakness, slowness, and reduced physical activity). 12 Based on these approaches, numerous frailty screening tools were developed and evidenced some validity in predicting adverse outcomes in heterogeneous surgical populations.7,8,13–15 Though frailty is an independent risk factor for adverse outcome in PAD,16,17 frailty assessment remains challenging in this cohort. None of the commonly used frailty screening tools are specific to PAD. Therefore, it is unknown whether these tools will adequately represent frailty status in this population or augment the prediction of mortality and amputations over traditional PAD risk factors such as advancing age, diabetes status, smoking status, or chronic kidney disease.
To address these knowledge gaps, we used the Vascular Quality Initiative (VQI) database to establish two cohorts of lower-extremity PAD patients by the revascularization procedure received: a peripheral vascular interventions (PVI) cohort and a lower-extremity bypass surgery cohort. We described the proportion of PAD patients undergoing vascular interventions who would be labeled as frail based on two cumulative deficit frailty screening tools (the 5-item modified frailty index (mFI-5) and the VQI-derived risk analysis index (RAI)). We examined the concordance of these tools in identifying frail patients in both cohorts. We also assessed whether these frailty tools augment the prediction of perioperative adverse outcomes (mortality, unplanned major amputation) over traditional PAD risk factors.
Methods
Study population
We used the VQI national database to identify our population of interest. The methodology of this registry is available online at www.vqi.org. In brief, VQI is a multicenter registry that collects demographic, clinical, procedural, and outcome variables for patients undergoing vascular procedures. As of 12/2019, there were approximately 600 participating centers in the Society for Vascular Surgery (SVS) VQI. These centers are distributed over 18 regional vascular groups throughout the United States. The VQI registry follows the American Heart Association/American College of Cardiology (AHA/ACC) guidelines’ definition of PAD, which includes history and/or physical examination suggestive of PAD confirmed with diagnostic testing (generally ankle–brachial index (ABI)). We created two cohorts of lower-extremity PAD using two VQI modules: PVI cohort and surgical bypass cohort.
For each cohort, we included the index procedure for patients undergoing a revascularization procedure between 1/2010 and 12/2019 (Figure 1). We excluded patients presenting with acute limb ischemia and patients with nonhealing amputation. We also excluded subjects with missing data for items included in the frailty indices, unknown procedural lower-extremity side, or missing data for study outcomes. The study protocol was approved by the Institutional Review Board at Yale University School of Medicine.
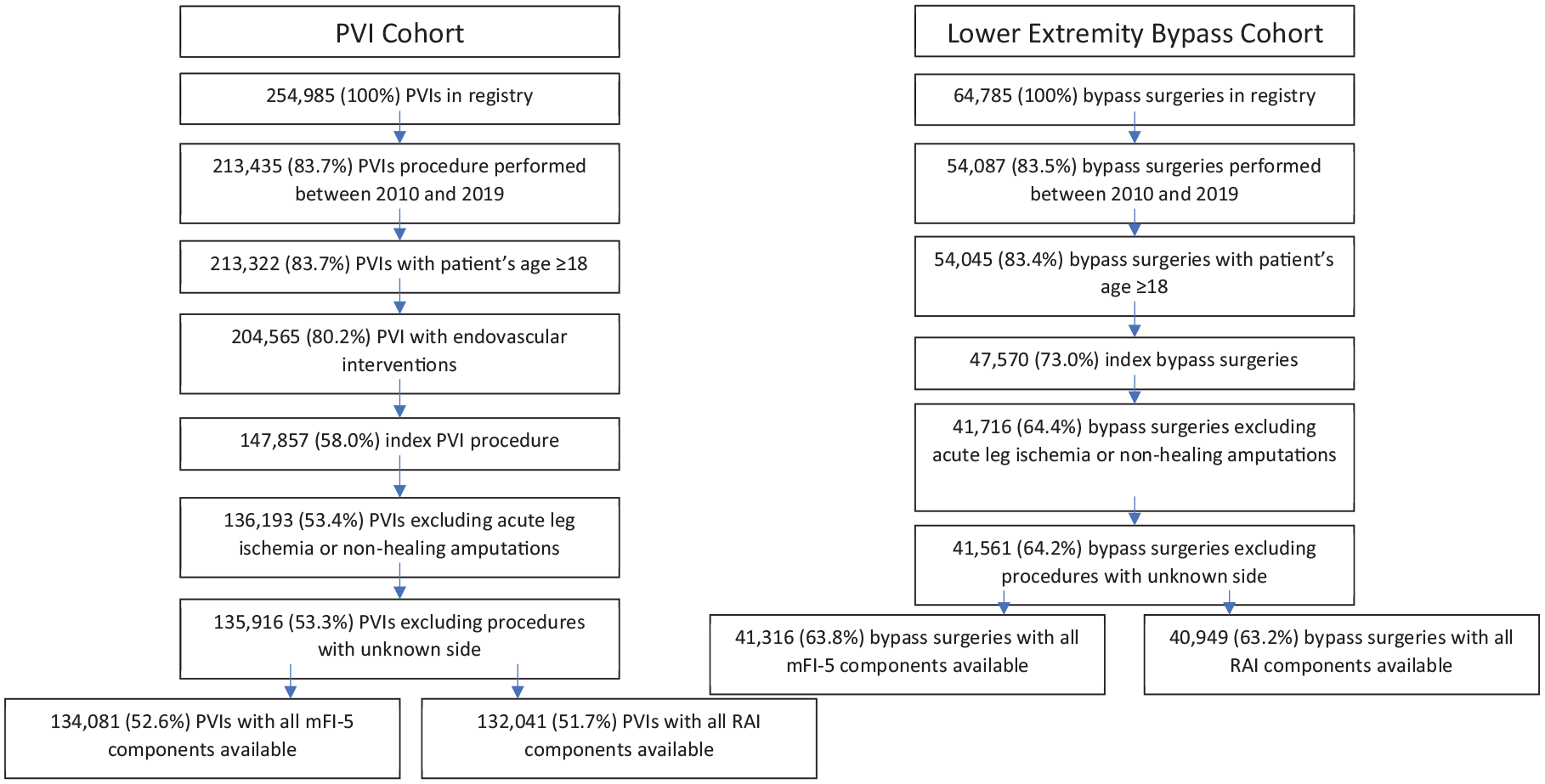
Study design establishing the PVI cohort and the lower-extremity bypass surgery cohort.
Frailty definitions
Frailty was assessed by the mFI-5 and the VQI-derived RAI. The mFI is derived from patients undergoing myriad of surgical interventions included in the National Surgical Quality Improvement Program (NSQIP) database using 16 variables cross-walked to the Canadian Study of Health and Aging Frailty Index. 15 By 2015, and as part of a quality improvement project, only five variables out of the previous 16 were present in the NSQIP and, using the available variables, the mFI-5 was derived. 14 These five variables included hypertension, diabetes, congestive heart failure, chronic obstructive pulmonary disease, and functional status deficit. Frail patients were identified using a score of ⩾ 2.14,18
The original RAI is a validated frailty tool derived from the veteran population and includes: age, sex, cancer, unintentional weight loss, poor appetite, renal failure, congestive heart failure, shortness of breath, residence other than independent living, cognitive deterioration, and activities of daily living.11,13 This tool was further adapted to the VQI dataset in the form of the VQI-derived RAI with the nutritional domain (unintentional weight loss and poor appetite) represented by extremes of body mass index (BMI) (underweight: < 20 kg/m2; morbidly obese: > 35 kg/m2) (online Supplemental Table 1).19,20 Furthermore, shortness of breath was replaced with chronic obstructive pulmonary disease. Since the VQI dataset did not have variables for cancer, and cognitive status, these variables were not included in the VQI-derived RAI.19,20 Consequently, this tool included the following variables: age, sex, BMI, renal failure, congestive heart failure, chronic obstructive pulmonary disease, living status, and functional status. For the PVI cohort, we used the functional status and ambulatory status to cross-walk patient dependency in terms of the activities of daily living (online Supplemental Table 2). Three of the authors (MA, KS, CM) independently assessed these two variables to create three levels of dependency: totally dependent, partially dependent, and independent. For the bypass cohort, the functional status was not available and only the ambulatory status was used to define patients’ dependency in activities of daily living. Frailty was defined as a VQI-derived RAI of ⩾ 35 points.13,19,20
Study outcomes
Our primary outcomes were perioperative mortality and unplanned major amputation. Mortality was defined as the last known mortality status before hospital discharge. Unplanned major amputation was defined as unplanned amputation performed after a revascularization procedure and before hospital discharge at a level above the metatarsals.
Statistical analyses
We used the mFI-5 and VQI-derived RAI to identify frail patients, as described above. To assess the interrater agreement between frailty status derived by mFI-5 and VQI-derived RAI, we calculated Cohen’s kappa statistic in each of the two cohorts (PVI and bypass). In general, kappa values of 0.01–0.20 denote slight agreement; 0.21–0.40 fair agreement; 0.41–0.60 moderate agreement; 0.61–0.80 substantial agreement; and 0.81–0.99 almost perfect agreement. 21 We used descriptive statistics to illustrate patients’ baseline characteristics by frailty status for the PVI and bypass cohorts. Continuous variables were reported as means and SDs. Differences were compared with Student’s t-tests for normally distributed variables, and Wilcoxon rank sum test for non-normally distributed variables. Categorical variables were presented as frequencies and proportions, and differences were assessed using chi-squared or Fischer’s exact tests.
To evaluate the predictive value of the frailty variable in detecting clinical outcomes, we fitted and compared three logistic regression models. For each cohort (PVI cohort, bypass cohort) and for each frailty index (mFI-5, VQI-derived RAI), we fitted a simple logistic regression model which included frailty only (model 1); the second model was a multivariable logistic regression model which included PAD risk factors only (age, female sex, smoking status, coronary artery disease (CAD), chronic kidney disease (CKD), history of amputations, Rutherford classification, and procedure urgency) (model 2); and the third model was a multivariable logistic regression model which included both frailty and PAD risk factors (model 3). In order to assess the goodness-of-fit of these models, we calculated the C statistic. 21 To compare the C statistic across the three models, receiver operator curves (ROC) were compared using the nonparametric DeLong method. 22
Rates of missingness among frailty tool variables were low (< 3% in the PVI cohort and < 2% in the bypass cohort) and as such these observations were censored. Sensitivity analyses were conducted to assess baseline differences between patients with complete and missing frailty tool variables yielding small and not clinically meaningful differences between the two groups. We used SAS software, version 9.4 (SAS Institute, Cary, NC, USA) for statistical analyses.
Results
Study population and baseline characteristics
Among the PVI cohort, 134,081 had all the mFI-5 components available, of which 67.0% were identified as frail and 132,041 had all components of VQI-derived RAI available, of which 27.1% were identified as frail (Figure 1, Supplemental Table 3). Among the bypass patients, all mFI-5 components were available in 41,316, of which 69.2% were identified as frail and all VQI-derived RAI components were available in 40,949, of which 15.7% were identified as frail (Figure 1, Supplemental Table 4). There was slight to no agreement between the two tools of frailty for both vascular cohorts (PVI: kappa 0.17 (0.16–0.17), p < 0.0001; bypass: kappa 0.13 (0.12–0.14), p < 0.0001) (Figure 2).
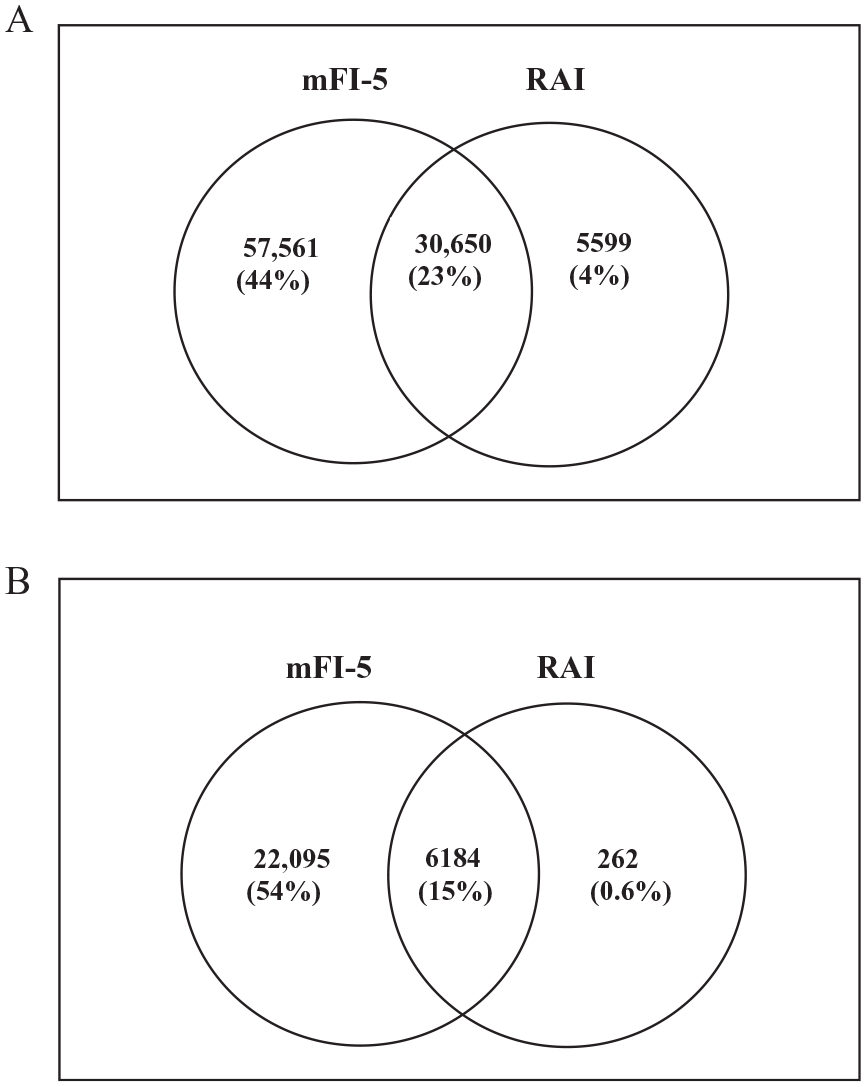
The overlap of frailty status as assessed by the mFI-5 and the RAI.
In the PVI cohort, there were significant differences in the baseline characteristics between the frail and the nonfrail (Supplemental Table 3). As identified by mFI-5, frail patients were older, more likely to have CAD, CKD, prior amputations, and higher Rutherford classification. Similarly, these baseline characteristic differences were noted when frailty status was assessed by VQI-derived RAI. In the surgical bypass cohort, baseline characteristics also differed significantly between the frail and the nonfrail (Supplemental Table 4). When frailty was assessed by mFI-5, frail patients also were noted to be older and have a higher proportion of CAD, CKD, prior amputation, and higher Rutherford classification. These differences were also noted when VQI-derived RAI was used to identify the frail.
Frailty and clinical outcomes
In unadjusted analyses, among PAD patients undergoing PVI, frailty was associated with higher mortality rate as assessed by mFI-5 (OR: 3.13, CI: 2.59–3.77, p < 0.0001) and as assessed by VQI-derived RAI (OR: 5.22, CI: 4.54–5.99, p < 0.0001); frailty was also associated with unplanned major amputations as assessed by both indices (mFI-5: OR: 2.24, CI: 1.74–2.89, p < 0.0001; VQI-derived RAI: OR: 2.48, CI: 2.04–3.03, p < 0.0001) (Figure 3). In multivariable analyses adjusting for traditional PAD risk factors, frailty was associated with mortality (mFI-5: OR: 1.59, CI: 1.30–1.93, p < 0.0001; VQI-derived RAI: OR: 2.23, CI: 1.84–2.70, p < 0.0001) but not with the unplanned major amputations in the PVI cohort (Figure 3).
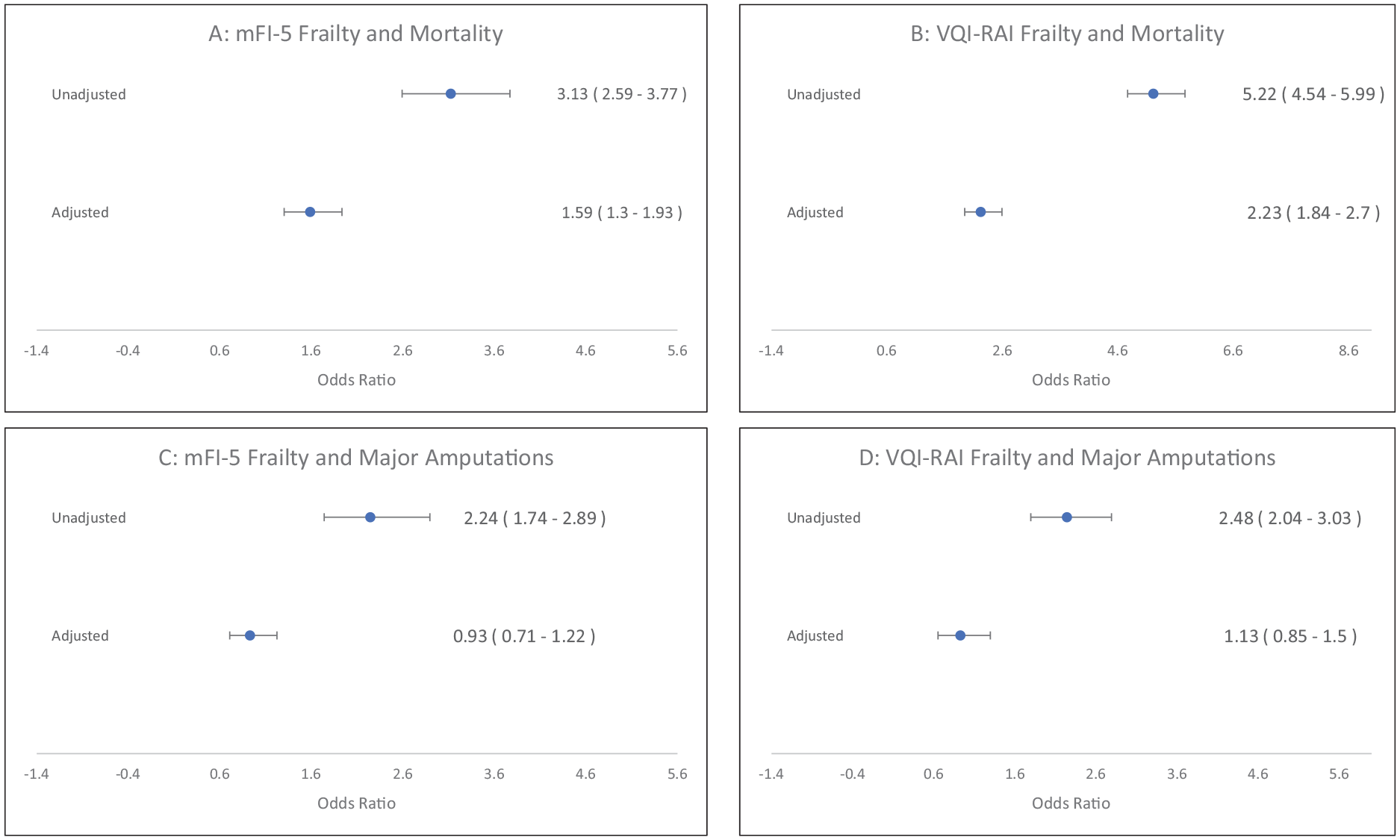
Univariate and multivariate association of frailty and clinical outcome in the PVI cohort.
Similarly, in unadjusted analyses, frailty as assessed by both mFI-5 and VQI-derived RAI was associated with mortality (mFI-5: OR: 3.03, CI: 2.32–3.96, p < 0.0001; VQI-derived RAI: OR: 5.18, CI: 4.32–6.21, p < 0.0001) and with unplanned major amputations (mFI-5: OR: 1.80, CI: 1.47–2.19, p < 0.0001; VQI-derived RAI: OR: 2.31, CI: 1.92–2.77, p < 0.0001) among PAD patients undergoing bypass surgery (Figure 4). In multivariable analyses, frailty was associated with mortality among the bypass cohort patients as assessed by both tools (mFI-5: OR: 1.79, CI: 1.35–2.38, p < 0.0001; VQI-derived RAI: OR: 1.97, CI: 1.53–2.54, p < 0.0001) (Figure 4). In the bypass cohort, there was no significant association between frailty status as assessed by mFI-5 and major amputations. However, there was a significant association between frailty as assessed by the VQI-derived RAI and major amputation in the bypass cohort (OR: 1.48, CI: 1.16–1.90, p = 0.0016) (Figure 4).
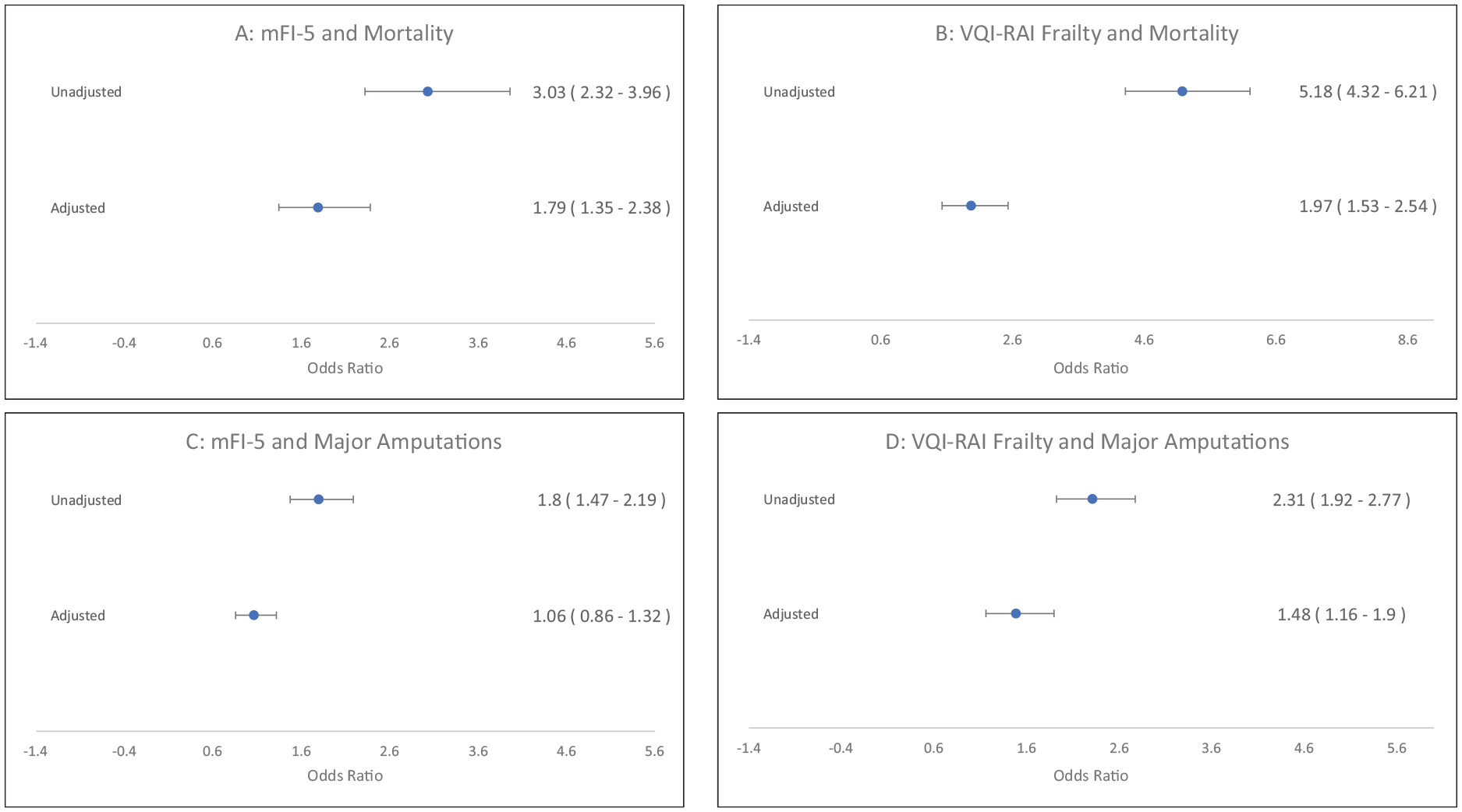
Univariate and multivariate association of frailty and clinical outcome in the lower-extremity bypass surgery cohort.
Frailty and prediction model performance
To assess the predictive value of the frailty variable in detecting clinical outcomes, we compared the C-statistic of the three logistic models. In the PVI cohort when assessing mortality, the C-statistic of model 1 was significantly lower than model 2 regardless of frailty tool used (mFI-5: model 1: 0.596, model 2: 0.823; C-stat difference = 0.23, p < 0.0001; VQI-derived RAI: model 1: 0.696, model 2: 0.825; C-stat difference = 0.13, p < 0.0001) (online Supplemental Tables 5 and 6). The addition of frailty to the PAD risk factors in model 3 had slight improvement in the C-statistic over the PAD only model 2 (model 3 vs model 2: mFI-5: C-stat difference = 0.003, p = 0.004; VQI-derived RAI: C-stat difference = 0.007, p = 0.0001) (online Supplemental Table 6). Similar results were noted when major amputation was assessed.
Likewise, when assessing mortality in the bypass cohort, model 1 had a lower C-statistic than model 2 (mFI-5: model 1: 0.592, model 2: 0.776; C-stat difference = 0.18, p < 0.0001; VQI-derived RAI: model 1: 0.666, model 2: 0.776; C-stat difference = 0.11, p < 0.0001) (online Supplemental Tables 7 and 8). A slight difference in the C-statistic was noted between model 2 and model 3 (mode 3 vs model 2: mFI-5: C-stat difference = 0.0035, p = 0.19; VQI-derived RAI: C-stat difference = 0.0073, p = 0.0099) (online Supplemental Table 8). Similar results were noted when major amputation was assessed.
Discussion
In this large observational study, we report that over two-thirds of the patients undergoing vascular interventions were labeled as frail using the mFI-5 tool. However, when the VQI-derived RAI tool was utilized, less than one-quarter of these patients was identified as frail. These findings highlight the lack of concordance between these cumulative deficit frailty screening tools. Regardless of the tool used, frailty status was significantly associated with perioperative mortality in both the PVI and bypass cohorts. In contrast, only frailty status measured by the VQI-derived RAI was associated with major amputations in the bypass cohort. More importantly, irrespective of the tool used and across the two PAD populations, the incremental contribution of frailty status to explain perioperative mortality and amputation over traditional PAD risk factors was modest.
Frailty assessment in PAD patients undergoing revascularization is challenging given the lack of reliable and validated frailty measures for this population. 23 Previous studies using the original mFI to identify frail PAD patients undergoing vascular procedures reported frailty rates ranging from 60% to 85%.16,17 In contrast, a study applying the VQI-derived RAI to the lower-extremity amputation population reported a frailty prevalence of just 32%. 24 In our study, we also observed discordance in frailty prevalence across PAD cohorts dependent upon the frailty screening tools used. The discordance in frailty prevalence may in part be explained by how these measures were originally derived. The phasing out of multiple components of the original mFI from the NSQIP database reduced the mFI-5 to a measure of comorbidities generally related to aging (i.e., hypertension, diabetes) and may no longer capture the specificity of frailty. 14 Likewise, the VQI-derived RAI does not include variables from the original RAI not present in the VQI database, such as cognitive status and malignancy, and uses BMI as a surrogate for weight loss.19,20 Furthermore, the performance of this measure lacks construct validation in assessing frailty as it has not been compared to the original RAI.19,20 Consequently, variability in frailty prevalence estimates in PAD patients may reflect measurement error given the modifications of frailty assessments to date.
Previous studies have demonstrated frailty status is associated with higher mortality and amputation-free survival as assessed by the original mFI and RAI.16,17,24,25 Consistent with this, we noted significant associations between frailty and mortality in both the PVI and bypass cohorts. Moreover, we found a significant association between the VQI-derived RAI and amputations in the bypass cohort. However, our findings evidence that the performance of mFI-5 and VQI-derived RAI tools in the prediction of outcomes are limited compared to traditional PAD risk factors (e.g., age, CAD, CKD, and Rutherford classification). Nevertheless, it is worth noting that the original mFI-5 and RAI were both derived from elective surgery databases with greater heterogeneity than our current sample.13–15 Given the relatively uniform nature of our PAD cohort, most patients who experienced adverse outcomes shared risk factors often associated with PAD (i.e., hypertension, diabetes). Therefore, frailty screening tools such as mFI-5 and VQI-derived RAI applied to a PAD population did not show augmentation in predicting outcomes as compared to traditional PAD risk factors. Future research should recalibrate these tools to better serve PAD patients or attempt to design new tools specific to this population incorporating more social, functional, and nutritional assessments.
Study limitations
The findings of our study should be interpreted in the context of the following limitations. First, our study is observational in nature and is limited by selection biases and confounding. Second, the frailty tools utilized in this study use surrogates of frailty and should not replace formal geriatric assessments. Third, both the mFI-5 and VQI-derived RAI are based on the cumulative deficit approach and we did not assess the performance of a frailty tool related to the phenotype model. Fourth, the VQI national dataset does not have variables assessing patient cognitive status or the presence of malignancy – variables included in the original RAI tool. Moreover, for the bypass cohort, the functional status was not available and only the preoperative ambulatory status was used to define patients’ dependency in activities of daily living. Consequently, we may have underestimated frailty in the absence of these variables. Nevertheless, our approach is consistent with prior work examining frailty.19,20 Fifth, the correlation between the mFI-5 and mFI-11 in the original cohort was lower for vascular procedures compared to overall surgical procedures (Spearman’s coefficient: 0.90 vs 0.95). 14 Moreover, this tool has not been validated in the VQI cohort and may overestimate the presence of frailty. Finally, our outcomes were reported prior to discharge and may be affected by the length of stay.
Conclusion
In conclusion, identifying frailty among patients undergoing lower-extremity revascularization remains challenging. We report the largest cohort assessing the frailty status among patients undergoing lower-extremity revascularizations. Furthermore, our study is unique because we report our findings based on the revascularization strategy implemented. Application of two easily administered frailty screening tools showed significant discordance in identifying frail patients and only had modest incremental contribution over traditional PAD risk factors in predicting perioperative complications. Despite these limitations, frailty status was associated with higher rates of mortality and when assessed by RAI, frailty status was associated with higher rates of major amputations in the bypass cohort. In order to validate these tools in this specific population, it will be imperative to obtain more complete data that better represent frailty in addition to PAD factors associated with frailty.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X221083701 – Supplemental material for Frailty and outcomes following revascularization of lower-extremity peripheral artery disease: Insights from the Vascular Quality Initiative (VQI)
Supplemental material, sj-docx-1-vmj-10.1177_1358863X221083701 for Frailty and outcomes following revascularization of lower-extremity peripheral artery disease: Insights from the Vascular Quality Initiative (VQI) by Mohammed S Al-Damluji, Kim G Smolderen, Can Meng, Feng Dai, Michael G Nanna, Bauer Sumpio, Peter Henke and Carlos Mena-Hurtado in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr Smolderen reports unrelated grant funding from Johnson & Johnson, Shockwave, and Merck, and she is a consultant for Cook Medical, Abbott, and Optum Labs. Dr Mena-Hurtado reports unrelated grant funding from Shockwave, and he is a consultant for Abbott, COOK, and Optum Labs. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
