Abstract
Background:
Despite strong association of elevated lipoprotein (a) (Lp(a)) levels with incident coronary and cerebrovascular disease, data for incident peripheral artery disease (PAD) are less robust. The main objective of the present systematic review was to analyze the association between elevated Lp(a) levels and PAD outcomes.
Methods:
This systematic review was performed according to PRISMA guidelines. A literature search was performed to detect randomized clinical trials or observational studies with a cohort design that evaluated the association between Lp(a) levels and PAD outcomes.
Results:
Fifteen studies including 493,650 subjects were identified and considered eligible for this systematic review. This systematic review showed that the vast majority of the studies reported a significant association between elevated Lp(a) levels and the risk of PAD outcomes. The elevated Lp(a) levels were associated with a higher risk of incident claudication (RR: 1.20), PAD progression (HR: 1.41), restenosis (HR: 6.10), death and hospitalization related to PAD (HR: 1.37), limb amputation (HR: 22.75), and lower limb revascularization (HR: 1.29 and 2.90). In addition, the presence of elevated Lp(a) values were associated with a higher risk of combined PAD outcomes, with HRs in a range between 1.14 and 2.80, despite adjusting for traditional risk factors. Heterogeneity of results can be explained by different patient populations studied and varying Lp(a) cut-off points of Lp(a) analyzed.
Conclusion:
This systematic review suggests that evidence is available to support an independent positive association between Lp(a) levels and the risk of future PAD outcomes.
Introduction
Lipoprotein (a) (Lp(a)) consists of a low-density lipoprotein (LDL)-like core lipoprotein molecule. This lipid particle contains apolipoprotein B, to which a glycoprotein of variable molecular weight, apolipoprotein (a), is covalently bound via a cysteine-cysteine disulfide bond. 1 High levels of Lp(a) are an independent risk factor for atherosclerotic cardiovascular diseases through mechanisms associated with increased atherogenesis, inflammation, and thrombosis. 2
Despite strong and consistent prospective associations of elevated Lp(a) levels with incident coronary and cerebrovascular disease, data for incident peripheral artery disease (PAD) are less robust. 3 PAD is the clinical manifestation of chronic progressive atherosclerotic disease that typically affects the abdominal aorta, iliac arteries, and lower limbs. 4 The most significant risk factors for PAD are hyperlipidemia, hypertension, diabetes mellitus, chronic kidney disease, and smoking. Likewise, a variety of other potential risk factors for PAD, such as high Lp(a) levels, have been examined. 5
Lp(a) concentrations vary widely between individuals within the same population, as well as between groups of different ethnicities. 6 This variation complicates the determination of a clinical cardiovascular risk threshold, which is currently considered to be > 50 mg/dL.7,8 Since levels above 50 mg/dL are observed in roughly 20% of the caucasian population and at an even higher frequency in African American and Asian-Indian ethnicities, it can be assumed that Lp(a) is one of the most important genetically determined risk factors for cardiovascular disease. 9
Many epidemiological studies have evaluated the association between Lp(a) levels and PAD outcomes.10 –24 However, the populations included, the cut-off points for Lp(a) analyzed, the methodologies to measure Lp(a) used, the follow-up times, and the vascular outcomes evaluated were heterogeneous in the comparison of the studies.
Therefore, the main objective of the present systematic review was to analyze the association between elevated Lp(a) levels and PAD-related outcomes.
Materials and methods
Data sources
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
25
This systematic review was registered in
A literature search was performed to detect studies that have evaluated the association between Lp(a) levels and PAD outcomes. Two independent reviewers searched the electronic PubMed/MEDLINE, Embase, Science Direct, Scopus, Google Scholar, and Cochrane Controlled Trials databases using the ‘lipoprotein (a)’ term combined with the following terms: ‘cardiovascular disease’, ‘major adverse limb events’, ‘critical limb ischemia’, ‘limb revascularization’, ‘peripheral revascularization’, ‘amputation’, and ‘peripheral artery disease’. The final article search ended on September 30, 2021.
The following inclusion criteria were used to select eligible studies: (1) Randomized clinical trials or observational studies with a cohort design (prospective or retrospective). (2) Studies evaluating two groups of patients: with elevated Lp(a) levels or not. We did not set a specific cut-off point for Lp(a). Some studies used the highest quintile/quartile of Lp(a), whereas other studies analyzed preestablished cut-off points (e.g., 50 mg/dL). (3) Studies that evaluated the relationship between Lp(a) levels and the risk of PAD outcomes. (4) Studies with a follow-up of at least 6 months.
There were no idiomatic, geographical or publication restrictions. Excluded studies included expert opinions, reviews, case-series, cross-sectional and case–control studies.
Data analysis
Data were extracted on the baseline population and effect sizes for the relationship between Lp(a) and PAD outcomes. Effect sizes and 95% CIs for Lp(a) were reported as odds ratios (ORs), relative risks (RRs) or hazard ratios (HRs) according to the reports of the original publications.
The primary endpoint of the study was the PAD outcomes incidence. PAD-related outcomes were defined according to the reported events within the selected studies, being a combination of clinically relevant outcomes such as significant decrease in the value of the ankle–brachial index (ABI), incident claudication, critical limb ischemia (CLI), peripheral revascularization, amputation, or mortality related to PAD.
Quality assessment
The quality of the included studies was assessed by two independent review authors using the criteria of the Quality in Prognostic Studies (QUIPS) tool. 26 This tool uses six important domains that should be critically appraised when evaluating validity and bias in studies of prognostic factors. A conclusive judgement of the risk of bias within each domain is made and expressed on a three-grade scale (high, moderate, or low). Any discrepancy between the two reviewers was resolved through discussion and by involving a third reviewer.
Meta-analysis was not possible due to heterogeneity of the populations included, the different cut-off points and measurements of Lp(a) used, and the type of PAD outcomes reported.
Results
The search included 840 potentially relevant articles after title screening, and 784 studies were excluded after abstract screening, as these were duplicate studies or did not assess the purpose of this study. After careful reading of the articles, 41 studies were removed, on the grounds that these were a case report, had a cross-sectional or case–control design, or did not report the exposure/event of interest. A flow diagram of the study’s screening process has been shown in Figure 1.
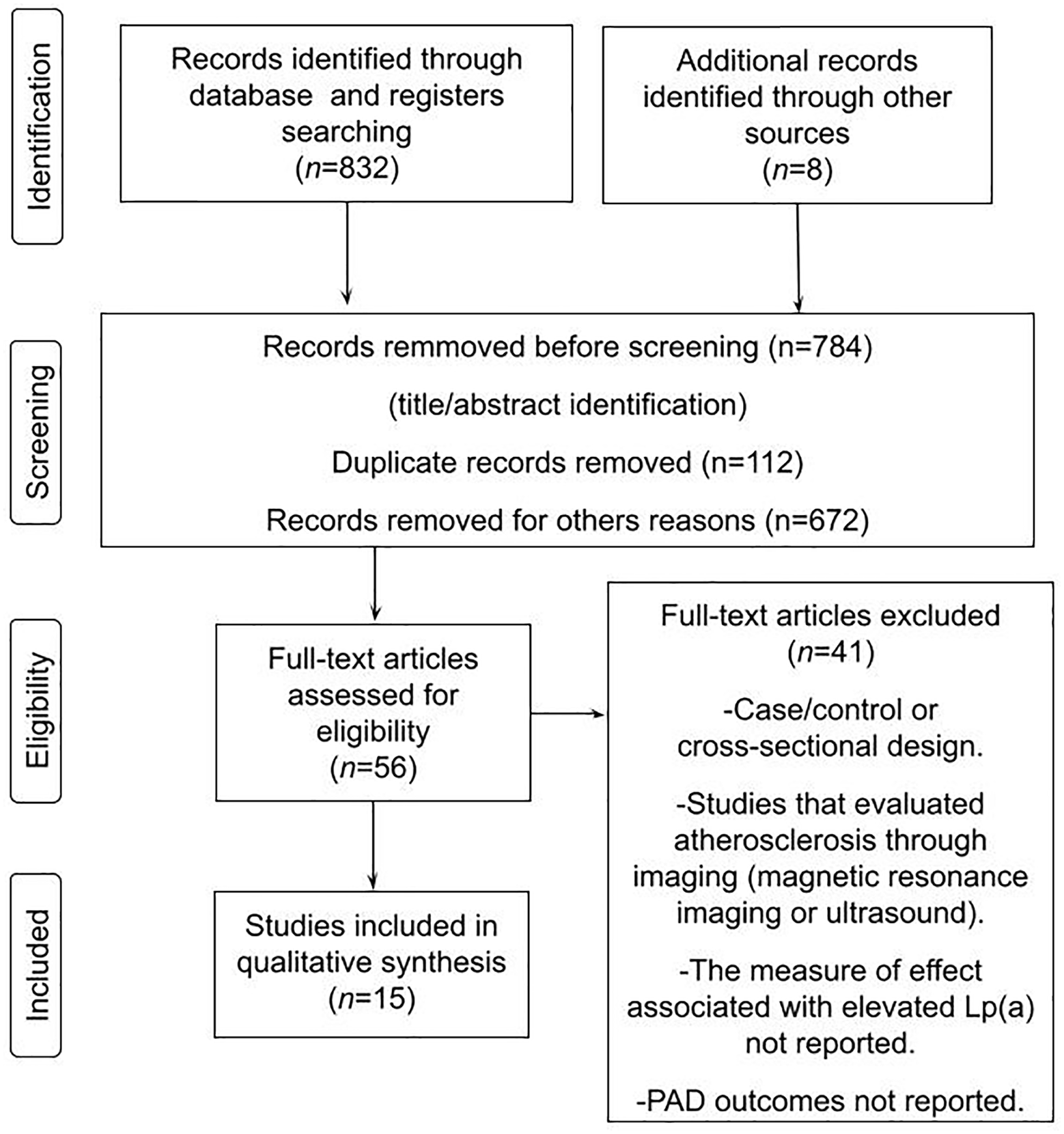
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram of the study screening process.
Fifteen studies including 493,650 subjects were identified and considered eligible for this systematic review. Seven studies evaluated general or primary prevention populations, and seven studies analyzed patients with established vascular disease (six of them with PAD and one of them with acute coronary syndrome). One study reported two cohorts, with and without a history of vascular disease, respectively.
In total, 14 cohort studies and one randomized clinical trial were analyzed. In all studies we assessed the level of bias. Three studies were assessed as being at high risk of bias and 10 studies as being at low risk of bias. In addition, two studies were assessed as being at moderate risk of bias. Those studies that were of high risk of bias had methodological issues within the domains concerning study attrition, outcome measurement, and statistical analysis or reporting, respectively. The quality of the studies evaluated can be seen in Figure 2.
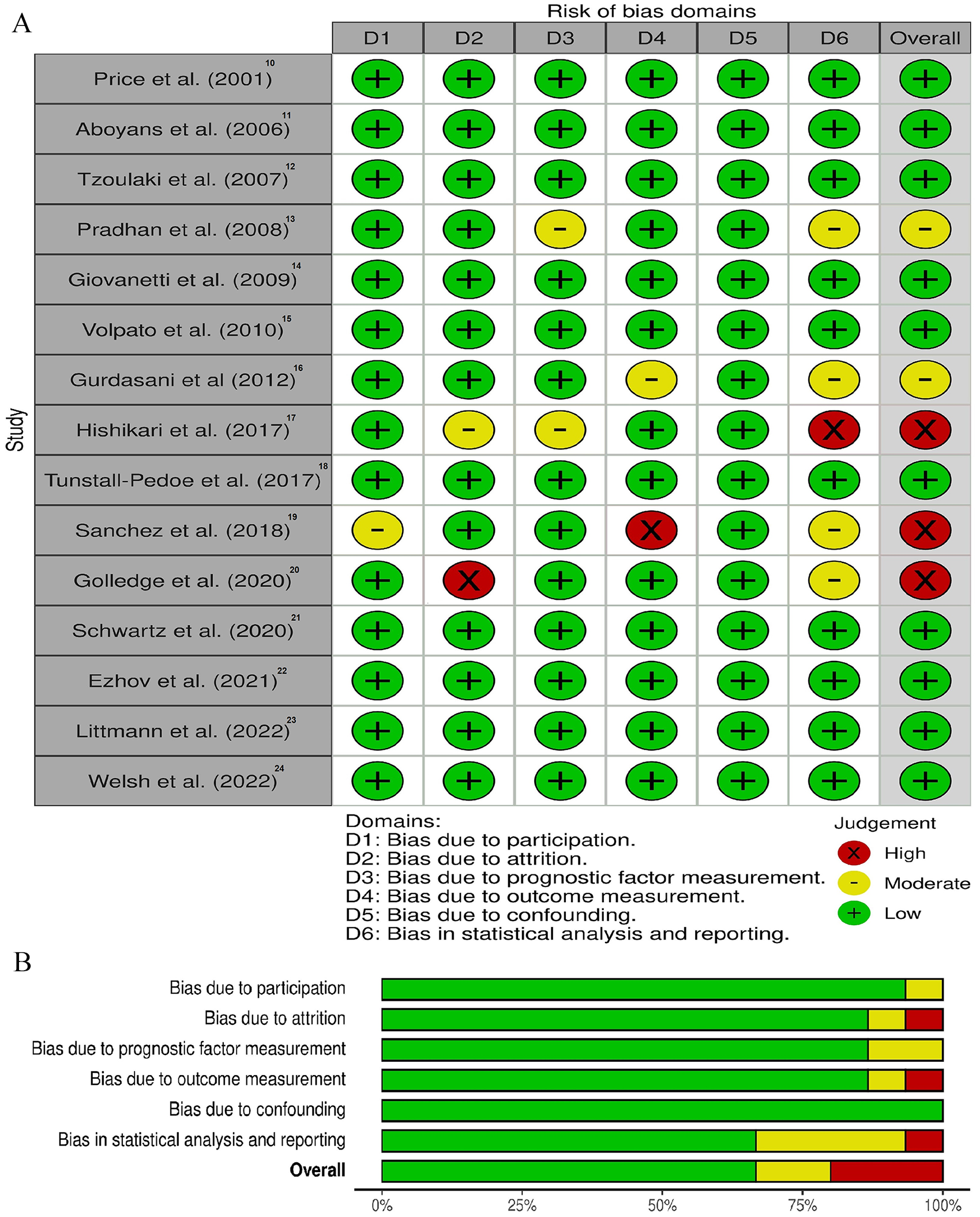
Individual
The characteristics of the studies included in this systematic review are shown in online Supplementary Table 1. The effect measures reported by the analyzed studies can be seen in Figure 3.
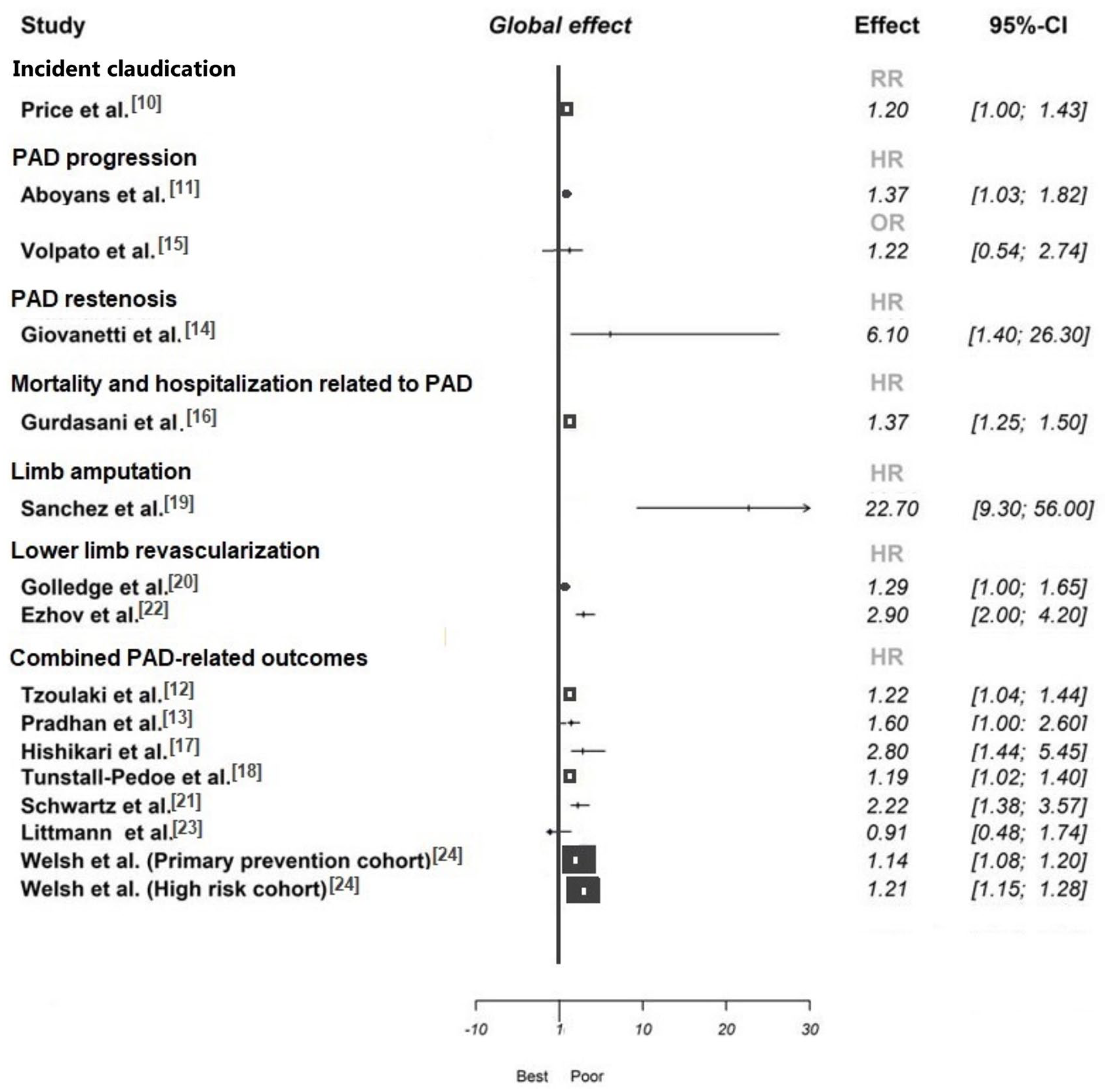
Effect of Lp(a) levels on peripheral artery disease outcomes.
Incident claudication
Price et al. 10 showed that raised Lp(a) levels at baseline were associated with an increased risk of incident claudication (RR: 1.20, 95% CI: 1.00–1.43) in caucasian men and women aged 55–74 years.
PAD progression
Aboyans et al. reported that higher Lp(a) level (per 1 mg/dL) was associated with a HR for PAD progression of 1.41 (95% CI: 1.02–1.88), as defined as a > 10% ABI decline. 11 Unlike that study, Volpato et al. 15 reported that the highest quartile of Lp(a) (32.9–175 mg/dL) compared to the lowest quartile (0–35 mg/dL) was not associated with an increased risk of manifesting an ABI value < 0.9 over time (OR: 1.22, 95% CI: 0.54–2.74).
PAD restenosis
One study evaluated the rate of primary patency after 6 months in symptomatic patients with PAD who underwent elective revascularization. 14 The authors showed that restenosis at 6 months was significantly related to Lp(a) > 30 mg/dL (HR: 6.10, 95% CI: 1.40–26.30).
Mortality and hospitalization related to PAD
Gurdasani et al. analyzed men and women aged 39–79 years in a large cohort. 16 Lp(a) was associated with PAD-related hospitalization and mortality; the fully adjusted HR for PAD for a 2.7-fold increase in Lp(a) (equating to a 1 SD increase in Ln[Lp(a)]) was 1.37 (95% CI: 1.25–1.50).
Limb amputation
Stable outpatients with symptomatic PAD were analyzed by Sanchez et al. 19 Over a mean follow-up of 36 months, 58 patients developed subsequent lower limb amputation. Multivariable analysis confirmed that patients with Lp(a) levels ⩾ 50 mg/dL were at an increased risk for limb amputation (adjusted HR: 22.75, 95% CI: 9.25–55.97).
Lower limb revascularization
Golledge et al. 20 showed that patients with PAD with Lp(a) ⩾ 50 mg/dL had a greater requirement for lower limb peripheral revascularization (HR: 1.29, 95% CI: 1.00–1.65). Similarly, another study 22 reported that a Lp(a) level above 30 mg/dL was significantly and independently associated with repeated revascularization during 3-year follow-up (HR: 2.90, 95% CI: 2.00–4.20).
Combined PAD-related outcomes
Tzoulaki et al. 12 reported that in caucasian men and women aged 55–74 years, Lp(a) was little affected by cardiovascular risk factors and displayed significant associations even in the multivariable analysis with the combined endpoint of incident claudication, CLI, or surgical intervention (HR: 1.22, 95% CI: 1.04–1.44). Another prospective study found borderline nonsignificant results for the association between Lp(a) levels and the combined endpoint of incident claudication and/or peripheral arterial surgery (HR: 1.60, 95% CI: 1.00–2.60) when evaluating female health professionals > 45 years. 13
A large cohort study evaluating men and women aged 25 to 75 years without cardiovascular disease showed that Lp(a) levels (every 5% increased) were associated with a 19% increased risk of developing the combined endpoint of incident claudication, CLI or surgical intervention (amputation or vascular surgery). 18 Another small study 17 found that the incidence of major adverse limb outcomes (amputation or major reintervention) was almost three times more frequent in patients with PAD and Lp(a) values > 40 mg/dL compared to individuals with Lp(a) < 40 mg/dL (HR: 2.80, 95% CI: 1.44–5.45). Schwartz et al. 21 showed that in statin-treated patients with recent acute coronary syndrome the risk of PAD outcomes (CLI, limb revascularization or amputation) was related to Lp(a) levels (highest quartile vs lowest quartile – HR: 2.22, 95% CI: 1.38–3.57). Unlike this report, in the study published by Littmann et al., 23 no association between Lp(a) and PAD outcomes (atherosclerosis of native arteries of the extremities or aorta, vascular intervention, or amputation) was observed. Finally, Welsh et al. 24 showed that in primary prevention patients aged 37–73 years, the Lp(a) level was associated with a HR for PAD outcomes (atherosclerosis, arterial embolism, thrombosis, and other disorders of arteries) of 1.14 (95% CI: 1.08–1.20). Similar findings were reported by the same authors when analyzing a high-risk population (patients aged 37–73 years with baseline cardiovascular disease and/or taking a statin). After adjustment for classical risk factors, 1 SD increment in log Lp(a) was associated with a HR for PAD outcomes of 1.21 (95% CI: 1.15–1.28).
Discussion
Updated evidence is shown in this systematic review that examines the relationship between Lp(a) levels and the incidence of PAD-related outcomes.
Multiple pathophysiological mechanisms have been proposed to explain the increased vascular risk of Lp(a). This lipid marker is more atherogenic than LDL, at least in part because it carries more oxidized phospholipid than LDL. 27 The oxidized phospholipid induces endothelial activation by upregulating chemokines and adhesion molecules. Also, Lp(a) particles do migrate more easily into the subendothelial space compared to LDL particles. This could in part be explained by the longer plasma residence time of Lp(a), which results in a more oxidized state. 28 In addition, previous reports suggest that the apo(a) component of Lp(a) is highly homologous to plasminogen, being able to inhibit activation of plasminogen to plasmin by endogenous tissue plasminogen activators as well as competing for binding of plasminogen and plasmin to established fibrin clots, showing a clear antifibrinolytic effect. 29 This dual pathophysiological mechanism, promoter of atherosclerosis and simultaneous generator of a pro-thrombotic state, is very similar to what occurs in CLI. A recent histopathological study in patients with CLI showed the presence of thromboembolic phenomena not associated with atherosclerosis, especially in arteries below the knee. 30 This pro-thrombotic pathophysiological mechanism associated with Lp(a) could be of particular relevance since a recent clinical trial in which patients with PAD treated with aspirin and low doses of rivaroxaban had fewer vascular outcomes than those treated with aspirin alone. This benefit was even greater in patients with PAD than in those with coronary artery disease.31,32
The relationship between Lp(a) and coronary artery disease was addressed in numerous studies; despite this, only a limited number of studies have evaluated the association with PAD outcomes. Our review suggests that an independent positive association between Lp(a) levels and the risk of future PAD outcomes has available evidence to support this; both in the primary prevention population and in high-risk populations, such as patients with a history of PAD. This systematic review explored 15 studies, finding that most of them reported a significant association between elevated Lp(a) levels and the risk of PAD outcomes. In that sense, the elevated Lp(a) levels were associated with a higher risk of incident claudication (32%), PAD progression (41%), PAD restenosis (about sixfold), death and hospitalization related to PAD (37%), limb amputation (about 22-fold), and lower limb revascularization (up to about threefold). In addition, our findings showed that the presence of elevated Lp(a) values were associated with a higher risk of combined PAD outcomes, in a range between 14% and about 280%, despite adjusting for traditional risk factors. The great heterogeneity of the results could be explained by the different populations evaluated, as well as by the different cut-off points of Lp(a) analyzed. Only two studies reported that elevated Lp(a) levels were not predictive of PAD outcomes. The study by Volpato et al., 15 which evaluated lower limb outcomes as the primary endpoint, did not find a statistically significant association. In the aforementioned study, the findings could be explained by the small number of patients included, the elderly population, or simply by chance. Given that most cardiovascular risk factors have attenuated relative rates at older ages, the average age observed in this study (73.7 years) perhaps influenced the final result. 33 The study published by Littmann et al. did not find a favorable result either. 23 In this case, it is possible that the relatively low incidence of PAD outcomes in the study has rendered statistical power too low to detect a significant difference.
Unfortunately, the wide variety of methods used and the small number of studies analyzed in this systematic review make it difficult to explore the association between Lp(a) assays and the outcomes. Currently available assays have not been subjected to a global standardization regime. 34 In addition, there is no precise recommendation to choose a certain method, although some guidelines suggest prioritizing the measurement of the number of particles concentrations (nmol/L) instead of measuring the Lp(a) values as mass concentrations (units of mg/dL). 8
To date, we do not know if lowering Lp(a) levels with a pharmacological intervention reduces vascular outcomes. Niacin reduced Lp(a) by 20% to 25%. However, clinical trials with these agents did not show reduction in major cardiovascular outcomes. 35 In contrast to niacin, PCSK9 inhibitors have been shown to reduce Lp(a) levels and decrease cardiovascular outcomes.36,37 LDL-cholesterol was reduced by approximately 60% with the mentioned drugs; therefore, it is controversial to attribute the reduction in outcomes to the decrease in Lp(a). Promising therapies for Lp(a) include antisense oligonucleotides that selectively bind mRNA coding for apolipoprotein (a). 38 However, its potential role in the treatment of PAD is still unclear and clinical studies will be necessary.
Study limitations
This systematic review has some limitations. First, there was clinical heterogeneity due to the characteristics of the populations and different follow-ups. Second, the endpoints evaluated were different when comparing the studies. Third, our study included different technology for Lp(a) measurements. In this context, the use of conversion factors to compare measurements with different units is inappropriate. 39 Fourth, the studies included in our analysis were mostly observational. Consequently, the presence of biases and confounders was highly expected. In this sense, most of the multivariate models were not adjusted for the medication received by the patients. We understand that this variable was not considered and could influence the results. Finally, quantitative analysis (meta-analysis) was not possible due to heterogeneity of the populations included, the different measurements of Lp(a) used, and the type of PAD outcomes reported. However, we have analyzed the best evidence available to date.
Conclusion
In this systematic review, we demonstrated that the high Lp(a) levels were associated with a marked risk of PAD outcomes. Physicians involved in the follow-up and treatment of patients with PAD should act vigorously on modifiable risk factors, especially if they show hyperlipoprotein(a)emia. If the reduction of Lp(a) levels with pharmacological interventions leads to a clinical benefit, it should be demonstrated with randomized studies.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221091320 – Supplemental material for Elevated lipoprotein (a) levels and risk of peripheral artery disease outcomes: A systematic review
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221091320 for Elevated lipoprotein (a) levels and risk of peripheral artery disease outcomes: A systematic review by Walter Masson, Martín Lobo, Leandro Barbagelata, Graciela Molinero, Ignacio Bluro and Juan P Nogueira in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
