Abstract

Keywords
Following the demonstration that thrombosis involving the superficial veins of the lower extremities (SVT) is a less benign disease than previously thought, 1 several controlled studies have consistently shown that fondaparinux in preventive doses, low-molecular-weight heparins (LMWHs) in intermediate doses, and rivaroxaban in preventive doses are effective and safe strategies for preventing extension or recurrence of the disease, as well as progression to the deep vein system and migration to the pulmonary circulation.2-6 In all these clinical trials, patients with the thrombus head within 3 cm from the saphenous–femoral junction were not eligible for recruitment, as they were perceived as being at a higher risk of venous thromboembolic complications in the absence of an active drug, nor were they in studies addressing the treatment of deep vein thrombosis (DVT) because of the lack of involvement of the deep vein system. Accordingly, which is the most proper therapeutic conduct in these patients is unknown. International guidelines are elusive in this regard.7,8 Although most physicians end up managing these patients with therapeutic doses of anticoagulants because of the feared risk of progression to the deep vein system, as far as we know there is no evidence of treatment failure coming from the long-term follow-up of patients managed with lower doses. Here we report the findings from an international registry.
The Computerized Registry of Patients with Venous Thromboembolism (RIETE) (
Out of 1320 patients with isolated SVT (i.e., without simultaneous involvement of the deep venous system, as assessed by systematic bilateral ultrasonography) who were enrolled in the RIETE registry between March 2015 and June 2021, 374 (28.3%) had a thrombosis involving the most proximal tract of the greater saphenous with the thrombus head being within 3 cm from the saphenous–femoral junction. Of these patients, 227 (60.7%) were managed with full-dose LMWH or therapeutic doses of fondaparinux overlapped with and/or followed by vitamin K antagonists or direct oral anticoagulants. The remaining 147 patients (39.3%) were managed with preventive doses of fondaparinux or intermediate-dose LMWH. Table 1 illustrates the main demographic and clinical characteristics of the recruited patients, as well as the main risk factors of thrombosis, which were fully comparable between the two study groups. As expected, the duration of treatment differed considerably between the two groups of patients, as a length of treatment shorter than 3 months was scheduled in less than 17% of those managed with therapeutic doses as compared to 50% of those managed with preventive ones.
Main demographic and clinical characteristics of the study patients.
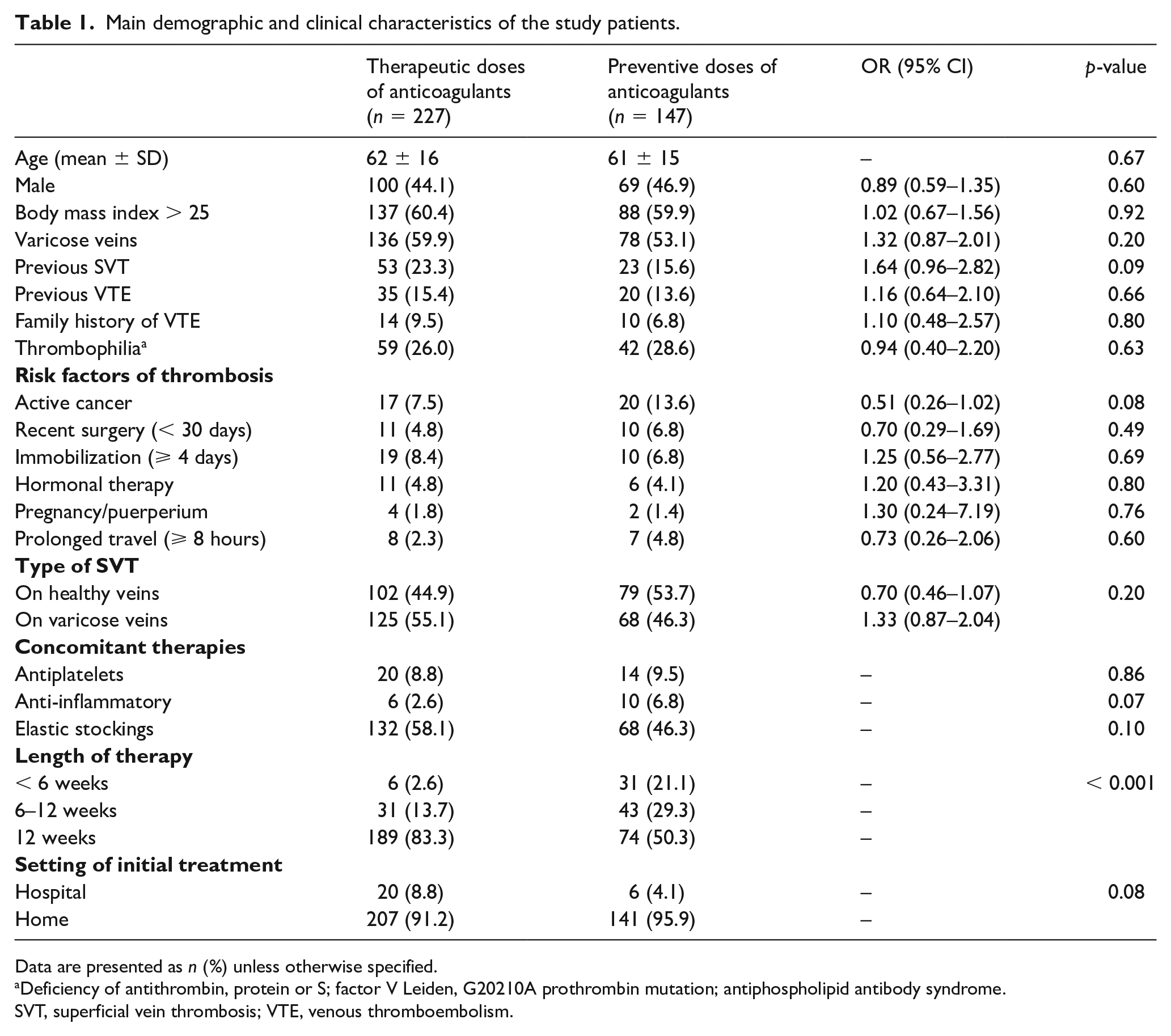
Data are presented as n (%) unless otherwise specified.
Deficiency of antithrombin, protein or S; factor V Leiden, G20210A prothrombin mutation; antiphospholipid antibody syndrome.
SVT, superficial vein thrombosis; VTE, venous thromboembolism.
All patients were followed up for 3 months. During this study period, among the 227 patients who were managed with therapeutic doses of drugs, thromboembolic episodes developed in three (1.3%): ipsilateral DVT in one, nonfatal pulmonary embolism in one, and recurrent SVT in one; among the 147 who were managed with preventive doses of drugs, thromboembolic episodes developed in four (2.7%): ipsilateral DVT in two, nonfatal pulmonary embolism in one, and recurrent SVT in one (odds ratio (OR), 0.48; 95% CI, 0.11 to 2.17; p = 0.33). Major or clinically relevant nonmajor bleeding complications, defined according to the International Society on Thrombosis and Haemostasis (ISTH) classification, developed in 3/227 patients (1.3%, including one major bleeding) of the former group, and in 1/147 (0.7%) of the latter (OR, 2.0; 95% CI, 0.20 to 19.41; p = 0.56). Deaths for reasons other than thromboembolic or hemorrhagic complications occurred in 1.3% and 0.7%, respectively.
Based on these findings, the use of subtherapeutic doses of anticoagulants for the initial and long-term management of patients with SVT involving the saphenous–femoral junction is unlikely to be less effective than that of therapeutic doses. Indeed, although there was a trend favoring the therapeutic approach in terms of thromboembolic episodes during the 3-month follow-up, the difference was not statistically significant. Although the allocation to therapeutic or subtherapeutic doses was not randomized, the comparability of the baseline and clinical characteristics of the study patients makes the evaluation of the study outcomes reliable. Not surprisingly, the risk of either major or clinically relevant nonmajor bleeding complications was slightly higher among patients treated with therapeutic doses.
These findings should be intended as hypothesis-generating, as they come from a registry where the allocation to either anticoagulation regimen was done at the discretion of the attending physicians. However, they suggest that in patients with SVT involving the saphenous–femoral junction, the efficacy of preventive doses of antithrombotic drugs may not be lower than that of therapeutic doses. A randomized clinical trial addressing the comparison between therapeutic and prophylactic doses of antithrombotic drugs in these patients is strongly warranted.
Footnotes
Appendix
Acknowledgements
We express our gratitude to Sanofi Spain, LEO Pharma, and Rovi for supporting this Registry with an unrestricted educational grant. We also thank the RIETE Registry Coordinating Center, S&H Medical Science Service, for their quality control data, and logistic and administrative support, and Prof. Salvador Ortiz, Universidad Autónoma Madrid, and Silvia Galindo, both Statistical Advisors in S&H Medical Science Service for the statistical analysis of the data presented in this paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
