Abstract
Abdominal aortic aneurysm (AAA) is an important vascular disease carrying significant mortality implications due to the risk of aneurysm rupture. Current management relies exclusively on surgical repair as there is no effective medical therapy. A key element of AAA pathogenesis is the chronic inflammation mediated by inflammatory cells releasing proteases, including the enzyme dipeptidyl peptidase IV (DPP-IV). This review sought to recapitulate available evidence on the involvement of DPP-IV in AAA development. Further, we assessed the experimental use of currently available DPP-IV inhibitors for AAA management in murine models. Embase, Medline, PubMed, and Web of Science databases were utilised to access the relevant studies. The review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA). A narrative synthesis approach was used. Sixty-four studies were identified from the searched databases; a final 11 were included in the analysis. DPP-IV was reported to be significantly increased in both AAA tissue and plasma of patients and correlated with AAA growth. DPP-IV inhibitors (sitagliptin, vildagliptin, alogliptin, and teneligliptin) were all shown to attenuate AAA formation in murine models by reducing monocyte differentiation, the release of reactive oxygen species (ROS), and metalloproteinases (MMP-2 and MMP-9). DPP-IV seems to play a role in AAA pathogenesis by propagating the inflammatory microenvironment. This is supported by observations of decreased AAA formation and reduction in macrophage infiltration, ROS, matrix MMPs, and interleukins following the use of DPP-IV inhibitors in murine models. There is an existing translational gap from preclinical observations to clinical trials in this important and novel mechanism of AAA pathogenesis. This prior literature highlights the need for further research on molecular targets involved in AAA formation.
Introduction
An abdominal aortic aneurysm (AAA) is a focal dilatation of the abdominal aorta with a diameter greater than 3 cm or 50% larger than its normal size. 1 Its prevalence is 4–8% in screening studies, affects mostly males above 65 years old, and carries a high risk of mortality due to rupture.2–6 Currently, the management of AAA relies exclusively on screening, surveillance, and prophylactic surgical repair at AAA diameter ⩾ 5.5 cm. 7 The unmet need for medical therapy to halt the progression of small aneurysms, thus alleviating the need for surgery, provides an impetus for exploratory research for possible molecular targets for AAA medical therapy. 8 In a prior international survey of vascular surgery professionals, novel medical treatments to attenuate AAA growth or novel medications to ‘regress’ AAA size are considered among the top priorities of research in AAA. 9
The pathological hallmark of AAA is chronic inflammation with proteolytic extracellular matrix (ECM) degradation, vascular smooth muscle (VSM) cell apoptosis and elastin destruction. 10 The inflammatory microenvironment in AAA is laden with cellular infiltrates such as B and T cells that release proteases, including dipeptidyl peptidase IV (DPP-IV) and a related protein, attractin, thought to have a role in AAA pathogenesis.11,12
DPP-IV (CD26) is a glycoprotein and a member of the proline-specific peptidases family of related proteins. 13 As a type II transmembrane serine protease and an exopeptidase, DPP-IV is involved in cleavage and inactivation of various substrates including incretins such as glucagon-like peptide (GLP-1 and 2), peptides such as neuropeptide Y, and cytokines including tumour necrosis factor (TNF) and interleukin 6 (IL-6).14,15 Attractin (mahogany peptide), is a DPP-IV-like enzyme and also a member of the proline-specific peptidases family. 16 Also, it is a glycoprotein known to have an intrinsic DPP-IV activity and shares a similar extensive physiologic feature profile to DPP-IV.17–21 Both DPP-IV and attractin exist in two forms: membrane-bound and soluble.15,18 Interestingly, although the soluble form of DPP-IV has been shown to result from proteolytic cleavage of the intracellular portion of the membrane-bound form, the soluble form of attractin, present only in humans, is a result of alternative RNA splicing.18,22,23 Both attractin and DPP-IV have a ubiquitous distribution in human tissues and are involved in multiple physiological processes, including immune regulation and inflammation, cell adhesion, metabolism, neuroendocrinology, and cardiovascular effects (Figure 1). Attractin is also involved in other additional processes such as myelination and pigment synthesis.21,24–28
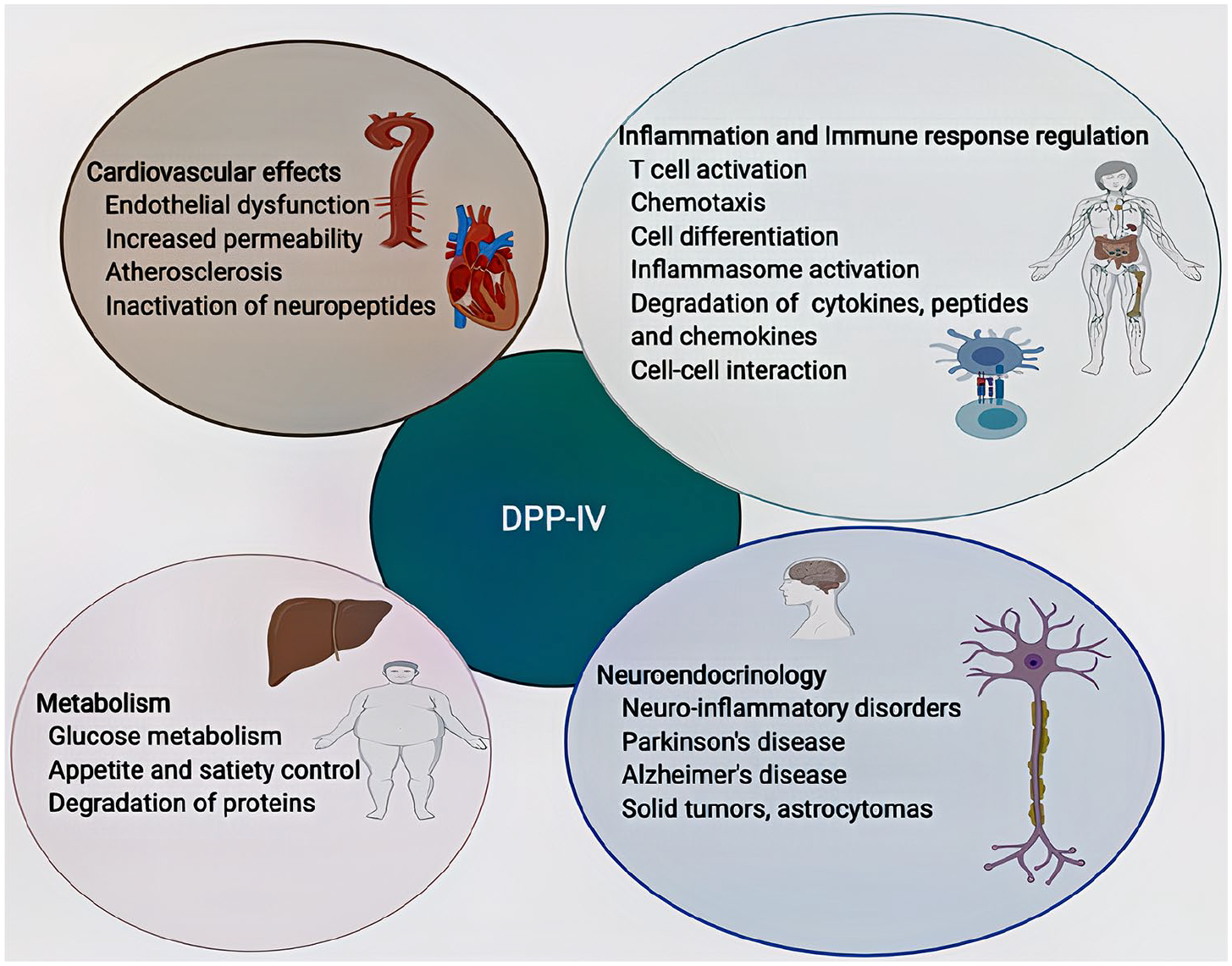
Diagram illustrates the involvement of Dipeptidyl peptidase IV (DPP-IV) in multiple physiological and pathological processes. DPP-IV regulates the inflammatory and the immune responses, controls glucose metabolism through its proteolytic effects on incretins and have a wide range of cardiovascular effects, including endothelial dysfunction and increased vascular permeability. Further, DPP-IV plays a role in the neuroendocrine system and has been implicated in neuroinflammatory and neurodegenerative disorders such as Alzheimer’s and Parkinson’s disease.
DPP-IV is known to act through the incretin pathway by proteolytically degrading endogenous GLP-1, an incretin hormone released by enteroendocrine L cells in the distal intestines. 29 Administration of DPP-IV inhibitors halts the GLP-1 biodegradation process, thus allowing GLP-1 to assert its effects, including insulin biosynthesis and secretion, endothelial and cardio-protection, appetite and satiety control, and immune and inflammatory response regulation.30–33 Various studies have reported low incidences of AAA among patients with diabetes, which has partly been attributed to the use of antidiabetic drugs such as metformin, DPP-IV inhibitors, and GLP-1 analogues.34–40 Although the use of DPP-IV inhibitors and GLP-1 analogues was previously reported to have reduced overall cardiovascular events, hence sparking interest in their vasculoprotective and anti-inflammatory effect, and a potential utility outside diabetes, subsequent clinical trials investigating cardiovascular outcomes with their use did not conclusively support these findings. 41
DPP-IV and attractin are thought to exert their inflammatory effect by acting upstream of the inflammasome cascade.42,43 The inflammasome complex consists of three components, the NOD-like receptor protein 3 (NLRP3), an adaptor protein known as apoptosis-associated speck-like protein (ASC), and an effector, caspase-1. 44 DPP-IV inhibition of GLP-1 leads to the activation of the inflammasome pathway with the release of proinflammatory cytokines IL-1β, chemokines, reactive oxygen species (ROS) and matrix metalloproteinases (MMPs).45–47 Inhibition of the inflammasome activity using specific inhibitors of NLRP3 and IL-1β was shown to reduce the release of cytokine MMP-9 and ROS release with resultant reduction in elastin degradation, and neutrophil and macrophage infiltration, which are key processes in AAA pathogenesis.48–54
Identifying molecular targets of AAA pathogenesis that can be used to develop effective drugs for AAA management remains a top priority in vascular surgery. Although numerous studies support the hypothesis that DPP-IV and attractin are involved in the immune and inflammatory response, sparse and isolated evidence is currently available on the involvement of these two proteins, specifically in AAA pathogenesis. Also, little is known about the potential use of DPP-IV inhibitors in AAA management. In light of these gaps, this current review sought to recapitulate available evidence of the involvement of DPP-IV and attractin in the AAA pathogenesis. Further, we assessed findings from the experimental use of DPP-IV inhibitors in the management of AAA in murine models.
Methods
Study design
This was a review of original studies investigating the role of DPP-IV and attractin in AAA pathogenesis and the experimental use of DPP-IV inhibitors in AAA in murine models. To guide the review process, the objectives, outcome measures, inclusion and exclusion criteria, and analysis methods were developed in a protocol before the formal review commenced.
Search strategy
Embase, Science Direct, PubMed, and MEDLINE databases were searched comprehensively for studies. Furthermore, the reference lists of the identified studies were screened for potentially relevant studies. All identified studies were collated, and duplicates eliminated. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) protocol was used to narrow down to key relevant studies. 55 The following search terms were used: (‘Dipeptidyl peptidase IV’ OR ‘DPP-IV’ OR ‘DPP-4’ OR ‘DPP-IV inhibitors’ OR ‘GLP-1’ OR ‘Attractin’ OR ‘ATRN’ OR ‘Mahogany’ OR ‘Sitagliptin’ OR ‘Vildagliptin’ OR ‘Saxagliptin’ OR ‘Linagliptin’ OR ‘Teneligliptin’ OR ‘Alogliptin) AND (‘Abdominal aortic aneurysm’ OR ‘aortic aneurysm’ OR ‘AAA’).
Eligibility criteria
Studies that measured the levels of DPP-IV and attractin in patients with AAA and the experimental use of DPP-IV inhibitors in AAA management in murine models were included. Only full articles were considered. Editorials, case reports, series, and letters were excluded. All studies published until Dec 31, 2020 were included.
Article selection and data extraction
Two independent reviewers (EN and PL) undertook a systematic search on the databases. Article selection was made on the basis of title and abstract. All excluded studies and the reasons for exclusion were discussed and agreed on by the two reviewers. In the event of a disagreement between the two reviewers, a third researcher was asked to adjudicate. A data extraction tool was developed, which included author name, year of publication, study design, sample size, study objectives, method of AAA induction, type of DPP-IV inhibitor used, and key observations.
Quality assessment
Assessment of risk of bias of the experimental studies included in this review was assessed using the Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) Risk of Bias (RoB) tool. 56 This tool is adapted from the Cochranes RoB tool but is specifically tailored for methodological quality assessment of animal studies. The SYRCLE RoB tool has criteria that assesses selection, performance, detection, attrition, and reporting bias.
Data synthesis
Reported findings from the selected studies were collated in a table format (Table 1 and Table 2) and a narrative synthesis approach was taken to analyse the findings from the studies. No further statistical analysis or meta-analysis was done due to the heterogeneity of the studies.
Summary of studies investigating the role of DPP-IV and attractin in abdominal aortic aneurysm pathogenesis.
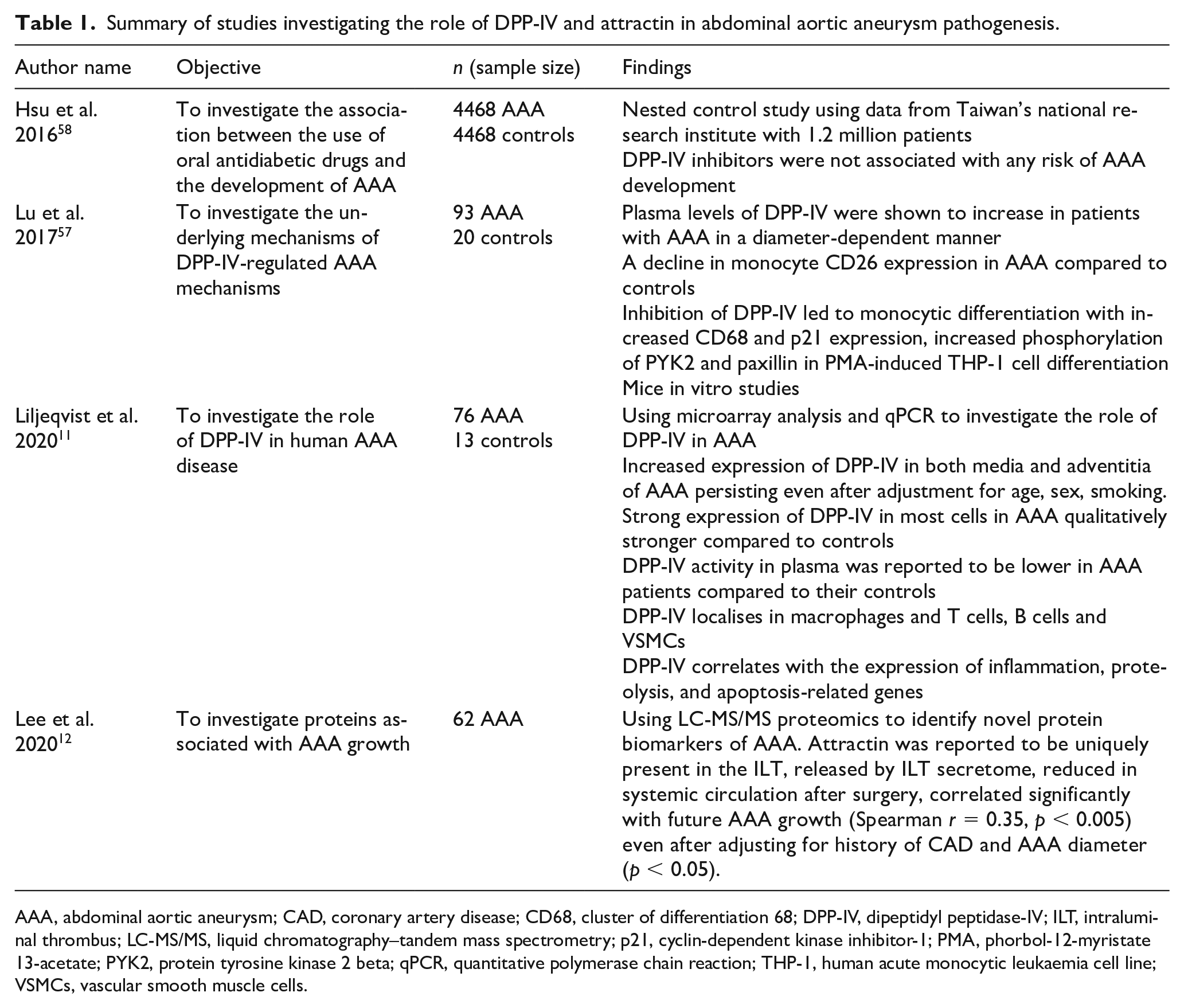
AAA, abdominal aortic aneurysm; CAD, coronary artery disease; CD68, cluster of differentiation 68; DPP-IV, dipeptidyl peptidase-IV; ILT, intraluminal thrombus; LC-MS/MS, liquid chromatography–tandem mass spectrometry; p21, cyclin-dependent kinase inhibitor-1; PMA, phorbol-12-myristate 13-acetate; PYK2, protein tyrosine kinase 2 beta; qPCR, quantitative polymerase chain reaction; THP-1, human acute monocytic leukaemia cell line; VSMCs, vascular smooth muscle cells.
Summary of studies investigating the experimental use of DPP-IV inhibitors for management of AAA in murine models.
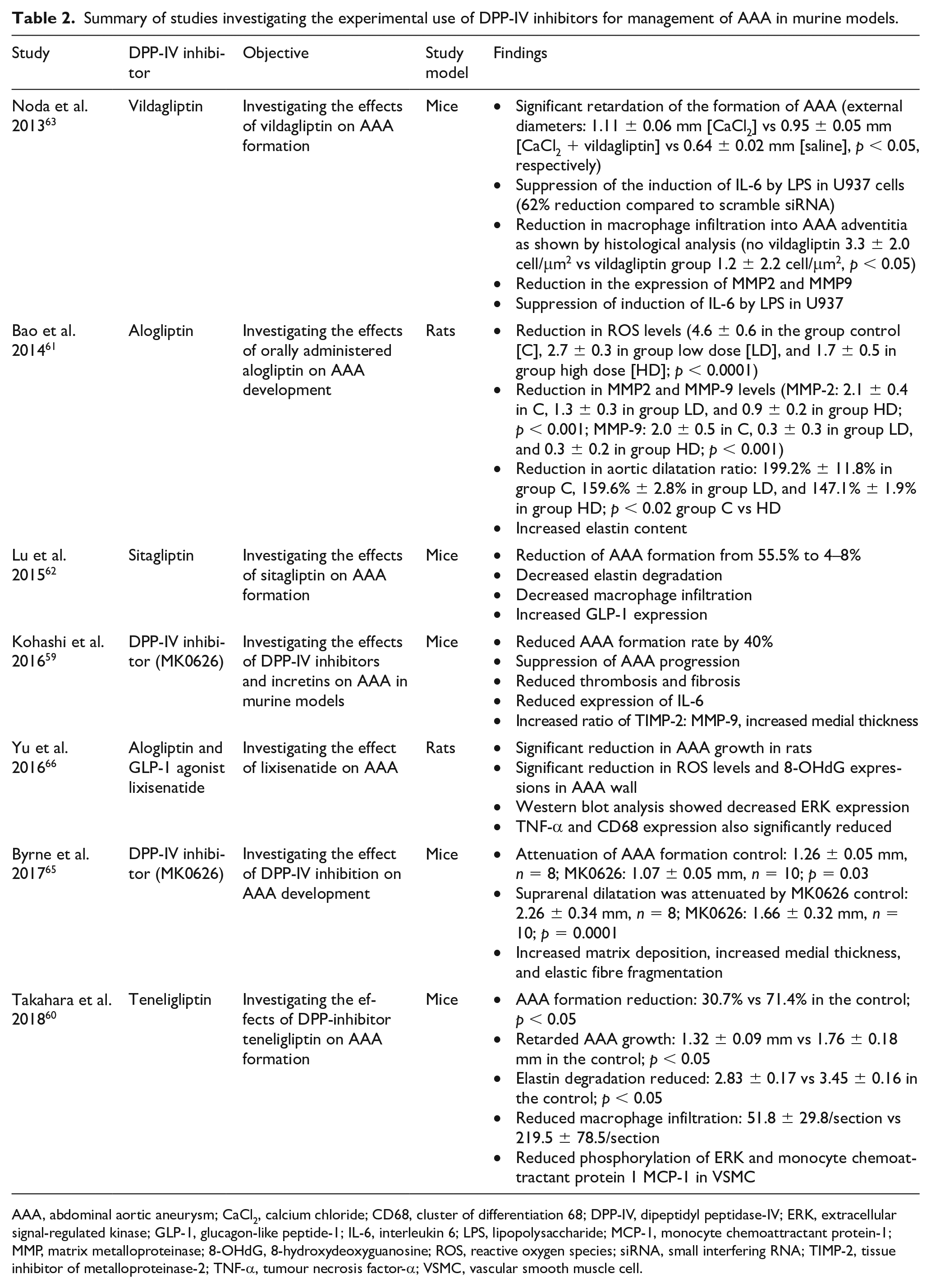
AAA, abdominal aortic aneurysm; CaCl2, calcium chloride; CD68, cluster of differentiation 68; DPP-IV, dipeptidyl peptidase-IV; ERK, extracellular signal-regulated kinase; GLP-1, glucagon-like peptide-1; IL-6, interleukin 6; LPS, lipopolysaccharide; MCP-1, monocyte chemoattractant protein-1; MMP, matrix metalloproteinase; 8-OHdG, 8-hydroxydeoxyguanosine; ROS, reactive oxygen species; siRNA, small interfering RNA; TIMP-2, tissue inhibitor of metalloproteinase-2; TNF-α, tumour necrosis factor-α; VSMC, vascular smooth muscle cell.
Results
Literature search results
A total of 64 articles were obtained from database searching. Upon removal of duplicates, 52 studies remained. We further excluded 41 studies that did not address the questions at hand. In the final analysis, 11 (17.2%) of the total identified studies from the search were included. Four studies were conducted in humans investigating the role of DPP-IV or attractin in AAA pathogenesis. The other seven studies were conducted in murine models and investigated the use of DPP-IV inhibitors for AAA management. The PRISMA flow diagram of all the studies is presented in Figure 2. A summary of all the studies is provided in Tables 1 and 2. The risk of bias assessment scores for the individual studies investigating experimental use of DPP-IV inhibitors in AAA is summarised in Table 3.
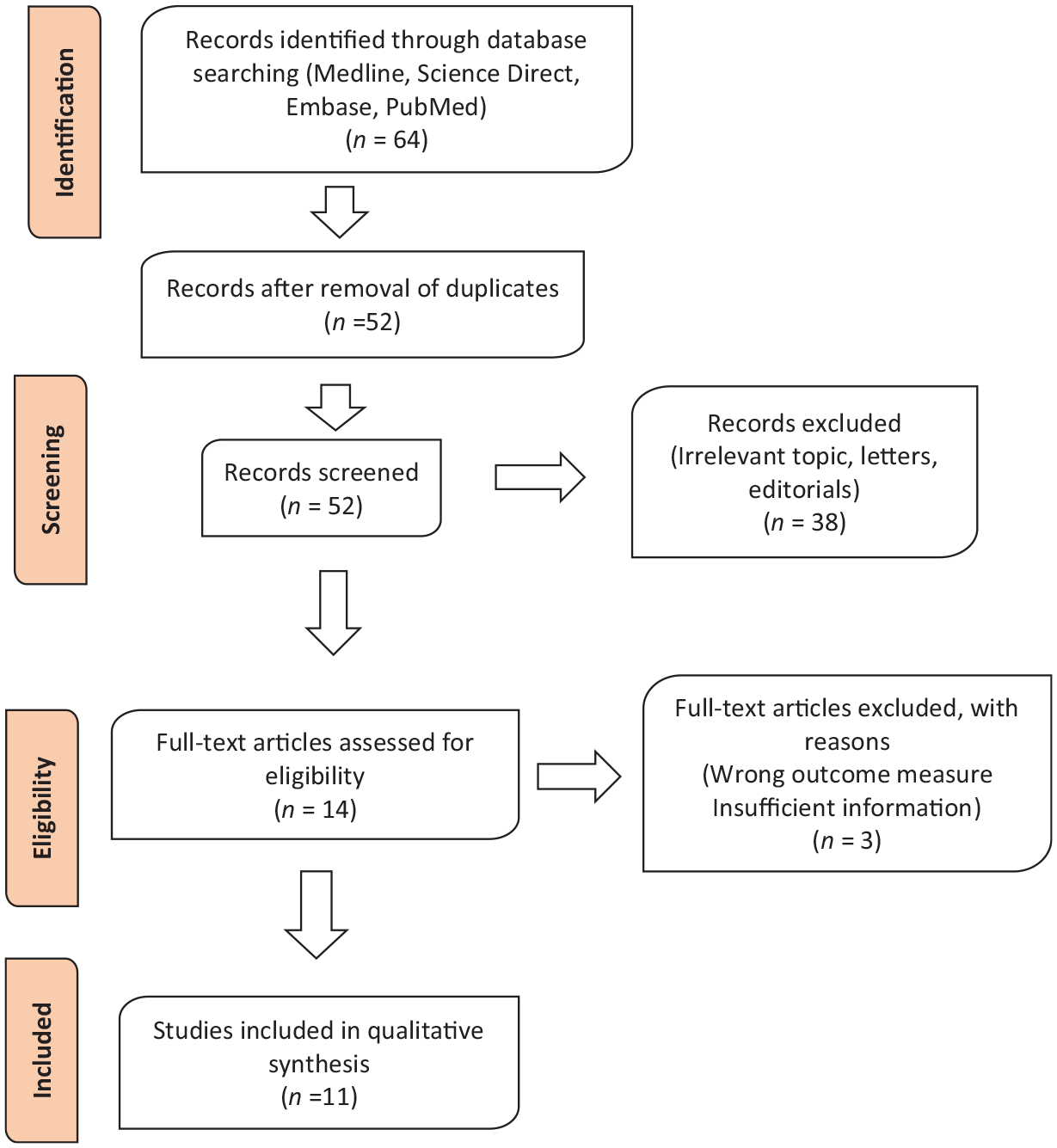
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram of studies identified during the systematic search.
Risk of bias assessment using the SYRCLE tool.
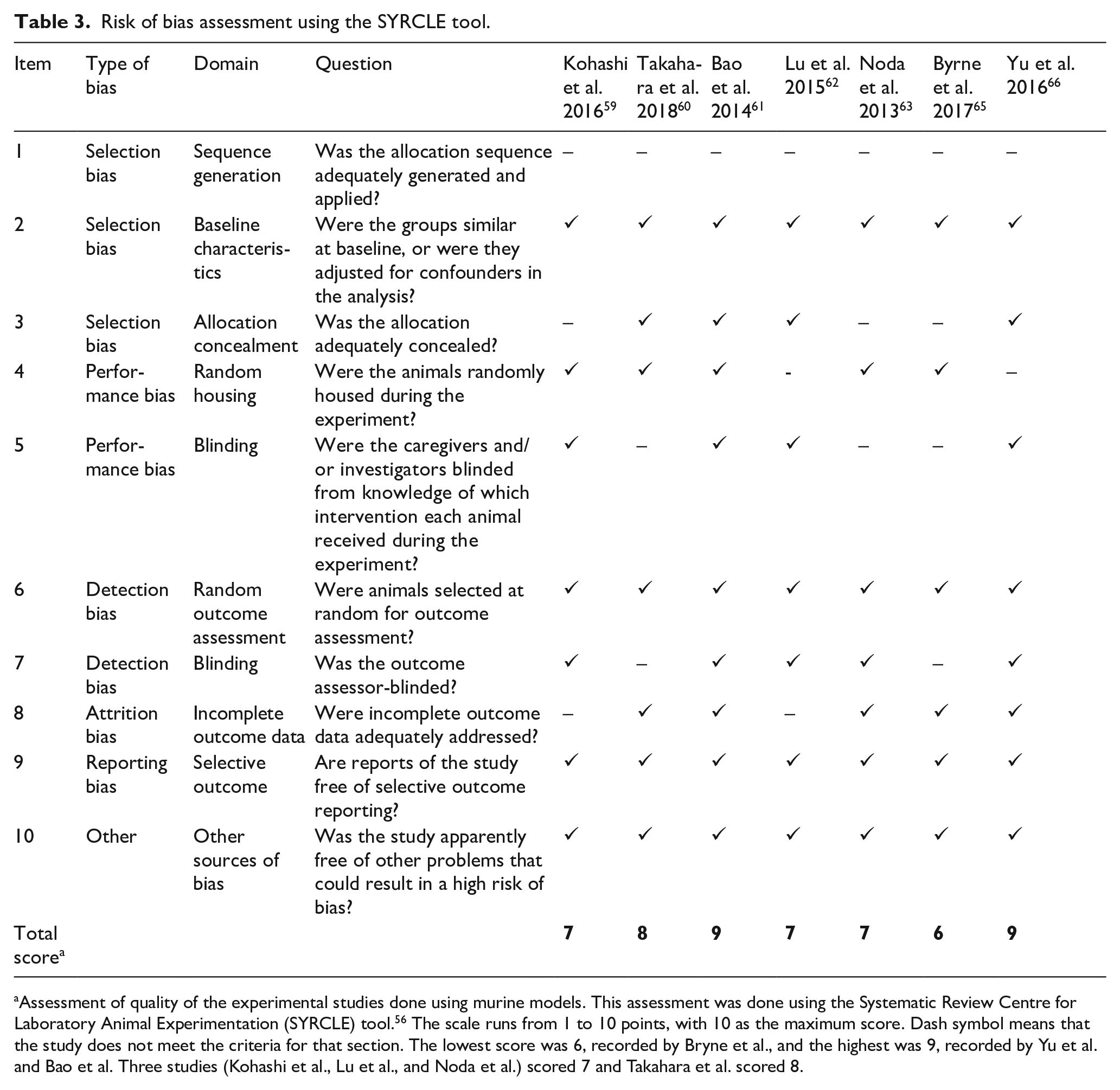
Assessment of quality of the experimental studies done using murine models. This assessment was done using the Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) tool. 56 The scale runs from 1 to 10 points, with 10 as the maximum score. Dash symbol means that the study does not meet the criteria for that section. The lowest score was 6, recorded by Bryne et al., and the highest was 9, recorded by Yu et al. and Bao et al. Three studies (Kohashi et al., Lu et al., and Noda et al.) scored 7 and Takahara et al. scored 8.
DPP-IV and attractin in AAA
Four studies investigating the potential involvement of DPP-IV or attractin in AAA were identified. Lu et al.’s work investigated the underlying mechanisms of DPP-IV in AAA among 113 participants (93 AAA and 20 controls). 57 Using computer tomography (CT) analysis, patients with AAA were further divided into small AAA (< 5 cm = 16) and large AAA (⩾ 5 cm, n = 77) groups. They reported increased plasma levels of DPP-IV in an AAA diameter-dependent manner among patients with AAA (control: 2.3 ± 1.5; small AAA: 10.0 ± 10.9; large AAA: 32.2 ± 15.0). On the other hand, plasma GLP-1 levels decreased in a diameter-dependent manner (control: 3.7 ± 2.4; small AAA: 2.1 ± 0.9; large AAA: 1.8 ± 1.1). There was also an associated decline in monocytic differentiation with the expression of CD68 in patients with AAA compared to controls. 57 Inhibition of DPP-IV induced several effects such as monocytic differentiation with increased CD68 and P21 expression, increased phosphorylation of protein-tyrosine kinase (Pyk2) and paxillin in phorbol-12-myristate-13-acetate (PMA)-induced THP-1 cell differentiation.
Liljeqvist et al. investigated the role of DPP-IV in human AAA disease. 11 Eighty-nine participants were recruited into the study (76 AAA and 13 controls from abdominal aortic tissue from organ transplant donors). Using microarray analysis, they reported significantly increased expression of DPP-IV in both media (p < 0.001) and adventitia (p < 0.001) of AAA tissue persisting after adjustment for age, sex, and smoking compared to the controls. These observations were further validated by quantitative polymerase chain reaction (qPCR) (media p < 0.01, adventitia p < 0.01). 11 There was also an increased expression of DPP-IV across cells in AAA compared to age-matched controls. However, unlike in the Lu et al. study, 57 this study reported a lower DPP-IV activity in plasma of patients with AAA compared to their age and sex-matched controls. Using immunohistochemistry (IHC), DPP-IV was shown to co-localise in macrophages, and B, T, and VSM cells. Gene-set enrichment analysis showed that DPP-IV expression correlated with genes that regulate inflammation, apoptosis, and proteolysis, ECM remodelling and angiogenesis. 11
Hsu et al. investigated the association between the use of oral antidiabetic drugs and the development of AAA using a nested case–control study using data from 1.2 million patients with diabetes from Taiwan’s national research institute. 58 They identified 4468 patients with AAA and an equal number of age-matched controls. Although some antidiabetic drugs, including metformin, sulfonylureas, and thiazolidinediones, were associated with a lower risk of AAA formation, DPP-IV inhibitors were not associated with any risk of AAA development. There was a 28% reduced risk of developing AAA in those taking metformin compared to their controls (CI: 0.64–0.80). 58
The work from Lee et al. observed the presence of proteins associated with AAA growth using liquid chromatography–tandem mass spectrometry (LC-MS/MS). 12 In a panel of proteins, attractin was identified to be uniquely present in the intraluminal thrombus (ILT) and released by ILT secretome. Also, a reduction of attractin in systemic circulation after surgery was reported. Attractin significantly correlated with future AAA growth both in slow and fast AAA growth groups of patients. The combination of attractin and AAA antero-posterior diameter (APD) as two-input variables in a growth prediction algorithm was superior to APD as the only input variable for predicting the future 12-month AAA growth: APD alone: Area Under the Receiver Operating Characteristics (AUROC) 0.76 (95% CI: 0.59 to 0.87) vs APD + attractin: AUROC 0.85 (95% CI: 0.69 to 0.93), p < 0.001, for slow/no growth; APD alone: AUROC 0.52 (95% CI: 0.35 to 0.66) vs APD + attractin: AUROC 0.76 (95% CI: 0.59 to 0.87), p = 0.001, for fast growth. 12
Experimental use of DPP-IV inhibitors and GLP-1 receptor agonists in murine models
Several experimental studies have investigated the utility of currently available DPP-IV inhibitors in the management of AAA in murine models.59–66 Kohashi et al. studied the effect of MK0626, a nonselective DPP-IV inhibitor on angiotensin II-induced AAA, in managing AAA in mice. 59 There was a 40% reduction in AAA formation with significant suppression of AAA progression (Ang II, 3.7 ± 1.0; Ang II + MK0626, 2.8 ± 0.8; p < 0.05) associated with DPP-IV inhibition. The use of MK0626 also reduced the expression of proinflammatory cytokine IL-1β and MMP-9. Interestingly, incretins did not have any effect on AAA, and the administration of incretin receptor blockers did not reverse the effects of DPP-IV inhibitors. Similarly, Byrne et al. reported that MK0626 attenuated AAA formation in mice treated for 2 weeks with oral beta-aminopropionitrile (BAPN) and 4 weeks of angiotensin II via mini-osmotic pump (control: 1.26 ± 0.05 mm, n = 8; MK0626: 1.07 ± 0.05 mm, n = 10; p = 0.03). 65
Takahara et al. investigated the effect of yet another DPP-IV inhibitor, teneligliptin, on AAA in mice. 60 Teneligliptin administration significantly reduced AAA formation (30.7% vs 71.4% in the control p < 0.05), suppressed AAA growth (p < 0.05), and decreased elastin degradation (p < 0.05) and macrophage infiltration into the aortic wall (p < 0.05). There was also an associated reduction in phosphorylation of extracellular signal-regulated kinase (ERK) and mRNA expression of monocyte chemoattractant protein-1 (MCP-1) in VSM cells. 60
Bao et al. explored the effects of orally administered DPP-IV inhibitor alogliptin on mice with AAA induced using elastase and calcium chloride. 61 AAA formation in the mice receiving alogliptin was significantly lower compared to controls (dilatation ratio: 199.2% ± 11.8% in controls, 159.6% ± 2.8% in the low-dose alogliptin group, and 147.1% ± 1.9% in the high-dose alogliptin group; p < 0.02 controls vs high dose). There was an accompanying reduction in ROS levels (p < 0.0001) and a significant reduction in the expressions of MMP-2 (p < 0.001) and MMP-9 (p < 0.001). Histological examination of aortic tissue from mice treated with alogliptin showed increased elastin formation. 61
Similar findings were observed by Morimoto et al. and Yu et al., where alogliptin was shown to increase the levels of GLP-1 6.3 ± 0.8 pmol/L in the control group, 7.6 ± 0.9 pmol/L in the alogliptin group, p < 0.05).64,66 The use of both the DPP-IV inhibitor alogliptin and the GLP-receptor agonist lixisenatide decreased the expressions of ROS and CD68 macrophage infiltration in aneurysm walls. 66 Western blot analysis showed decreased expressions of ERK, TNF-α, MMPs, and CD68. By the 28th day of administration, both alogliptin and lixisenatide had significantly reduced AAA growth in rats: 199.2 ± 10.0% in the control group, 155.1 ± 2.3% in the alogliptin group, p < 0.01; and 188.0 ± 5.6% in the control group, 162.0 ± 6.0% in the low-dose group, and 151.5 ± 4.9% in the high-dose group, p < 0.01, respectively. 66
Noda et al. investigated the effect of another DPP-IV inhibitor, vildagliptin, on AAA and its effect on monocyte inflammatory response and vascular remodelling in mice models. 63 AAA was induced in mice using calcium chloride. Vildagliptin significantly diminished the formation of AAA (external diameters: 1.11 ± 0.06 mm [CaCl2] vs 0.95 ± 0.05 mm [CaCl2 + vildagliptin] vs 0.64 ± 0.02 mm [saline], p < 0.05). Histological analysis revealed decreased macrophage infiltration into the adventitia (p < 0.05), and qPCR analysis showed that vildagliptin reduced expression of MMP-2, MMP-9, and IL-6. Further in vitro experiments demonstrated that DPP-IV inhibition using vildagliptin suppressed the induction of IL-6 by lipopolysaccharide (LPS) in U937 cells (model cell line used to study monocyte differentiation). 63
Lu et al. studied the effect of sitagliptin on angiotensin II-induced AAA in mice. 62 There was a drop in AAA formation from 55.5% to 4% in mice induced with angiotensin II. Histological analysis revealed decreased elastin degradation, macrophage infiltration, and increased GLP-1 expression associated with sitagliptin administration. 62
Discussion
The aim of this review was to collate and recapitulate available evidence on the involvement of DPP-IV and a related protein, attractin, in AAA pathogenesis. Further, we sought to assess the findings from the experimental use of DPP-IV inhibitors in AAA management in murine models. This review revealed evidence of the involvement of DPP-IV and attractin in the pathogenesis of AAA through propagating the inflammatory cascade that precedes ECM degradation, elastin destruction, and SMC apoptosis. This is further supported by observations from the seven studies on experimental use of DPP-IV inhibitors (gliptins).
Attractin and DPP-IV are not only increased in plasma and AAA tissues of aneurysm patients, they also seem to correlate with genes regulating inflammation and apoptosis which are key processes in AAA pathogenesis.11,12,62 In addition, their correlation with AAA growth highlights their potential utility as biomarkers for prediction of AAA growth.12,62 Although Liljeqvist et al. reported lower plasma activity of DPP-IV among patients with AAA compared to their age-matched controls, there was an increased level of DPP-IV in aortic tissues of the patients, which supports observations from previous studies implicating DPP-IV as a potential driver of the inflammatory microenvironment observed in AAA. 11
Murine model studies investigating the use of DPP-IV inhibitors (gliptins) reported significantly attenuated AAA formation with a reduction in elastin destruction, macrophage infiltration, MMPs, and ROS activity.59–66 DPP-IV seems to propagate the inflammatory microenvironment seen in AAA by activating the inflammasome pathway (NLRP3), promoting immune cell migration cell adhesion, macrophage aggregation, and the release of ROS, MMPs 2 and 9, and IL-6 (Figure 3).67–69 This is supported by other studies that have reported that DPP-IV inhibition attenuates inflammation with reduced expression of NLRP3, caspase-1, and IL-1β.42,43,45,49,70,71 Also, attractin seems to have a role in T cell maturation, differentiation, and enhancement of direct migration of T cells to sites of inflammation.17,68 This may explain the reported increase in DPP-IV and attractin levels during the natural progression of AAA as demonstrated by Lee et al. and Lu et al. and the effective AAA attenuation by use of DPP-IV inhibitors.11,12,59–66
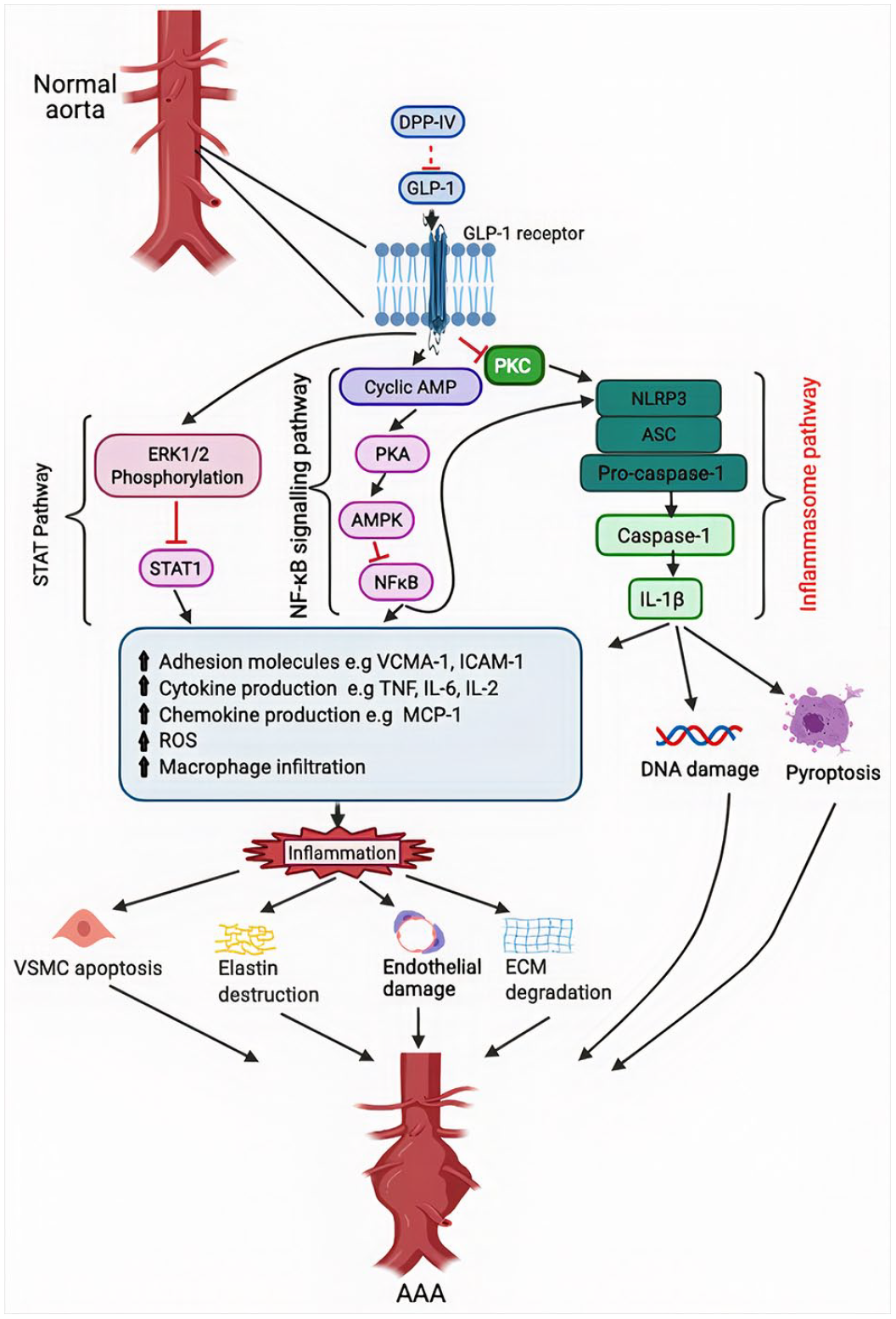
Schematic representation of the cascade initiated by dipeptidyl peptidase IV (DPP-IV) that propagates the inflammatory micro-environment leading to aneurysmal growth and eventual rupture. DPP-IV activates the inflammasome pathway through biodegradation of GLP-1. NLRP-3 activation further leads to the activation of pro-caspase-1 to caspase-1 which then activates interleukin 1-beta. IL-1-beta initiates downstream effects including ros release, macrophage infiltration, cytokine and chemokine release, DNA damage, and pyroptosis. DPP-IV biodegradation of GLP-1 also activates two other pathways, NFkB and STAT. These two pathways lead to similar downstream effects as the inflammasome pathway with resultant VSMC apoptosis, elastin destruction, endothelial damage, and extracellular matrix degradation. DPP-IV inhibitors work by blocking DPP-IV activity thus allowing GLP-1 to exert its anti-inflammatory effect by inhibiting PKC activation and subsequent expression of inflammasomes.
Besides DPP-IV and GLP-1 agonists, other oral antidiabetic medications, including metformin, have also been shown to reduce the incidence of AAA among diabetic patients, as reported by Hsu et al. and other previous studies.35,58,72,73 The Hsu et al. study reported 28% lower odds of developing AAA in those taking metformin compared to their controls (CI: 0.64–0.80). This study, however, did not report any change in the incidence of AAA with the use of DPP-IV inhibitors. Although only 3.9% of participants were on DPP-IV inhibitors, this study was larger than most of the other studies in this review that supported the reported DPP-IV’s involvement in AAA. This underscores the need for more studies investigating the role of DPP-IV inhibitors in AAA, as current evidence is inconclusive. Interestingly, metformin seems to work predominantly through activation of the adenosine monophosphate-activated protein kinase (AMPK) pathway, with similar downstream effects including the reduced release of MMP 2 and 9, ROS, and macrophage infiltration, as seen with DPP-IV inhibitors.74,75
Observations from the preclinical studies in this review suggest that DPP-IV inhibitors (gliptins) seem to confer other cardiovascular benefits, including improvement in endothelial function, reduction in inflammatory markers, and oxidative stress. DPP-IV inhibitors are also thought to reduce cardiovascular risk factors through glucose control, favourable body weight profile, reduction in blood pressure, and circulating free fatty acids, leading to an overall reduction in cardiovascular events such as strokes and ischemic heart disease.76–81 It is postulated that these beneficial effects are due to the release of vasculoprotective endothelial progenitor cells (EPCs), stromal cell-derived factor 1 alpha (SDF-1α), and monocyte chemoattractant protein-1 (MCP-1).82–86 Although these preclinical and mechanistic studies have provided compelling evidence for potentially beneficial cardiovascular effects of DPP-IV inhibition, recent clinical trials including CAROLINA (Cardiovascular Outcome Study of Linagliptin vs Glimepiride in Patients with Type 2 Diabetes), EXAMINE (Examination of Cardiovascular Outcomes with Aloglitin versus Standard Care), and TECOS (Trial Evaluating Cardiovascular Outcomes with Sitagliptin) did not fully support these observations.87–89 Although the TECOS and EXAMINE trials did not support the results from the CAROLINA study of a possible risk of increased heart failure with the use of linagliptin, none conclusively supported the initial compelling observations of cardiovascular benefit from the preclinical studies.
Evidently, there is a significant translational gap from animal model findings to clinical trials, particularly in AAA management.90–92 This may be attributable to the difference in AAA induced in animal models using angiotensin II or calcium chloride versus the slow chronic inflammatory process observed in human AAA. 93 This is exemplified by the failed clinical trial targeting IL-1β, which is a downstream mediator in the inflammatory cascade initiated by DPP-IV. This clinical trial used IL-1β inhibitor ACZ885 (canakinumab) and was stopped for reasons of futility despite promising results in preclinical studies involving IL-1β inhibition.53,70,71,94 Although various DPP-IV inhibitors including sitagliptin, vildagliptin, alogliptin, and linagliptin have been shown to be effective in AAA in animal models, there has been very limited application of these drugs in AAA clinical trials, possibly due to the risk of hypoglycaemic events in nondiabetic patients with AAA due to insulin surge.
Conclusion
DPP-IV and its related protein attractin seem to play a crucial role in the pathogenesis of AAA through activation of the inflammasome cascade and correlate with AAA growth. This highlights their potential use as biomarkers for the prediction of AAA growth. Further, the experimental use of DPP-IV inhibitors in murine models demonstrates their effectiveness in reducing ECM degradation, elastin destruction, and VSM cell apoptosis by reducing the release of MMPs, ROS, and ILs, leading to reduced AAA formation. These observations offer promise in the search for realistic molecular targets for the development of effective medical therapy. However, there is a translational gap from experimental animal model observations to clinical trials. This calls for more explorative research into the role of DPP-IV-like enzymes in AAA pathogenesis and the potential use of nonsurgical treatment of AAA.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Elisha Ngetich is supported by the Rhodes scholarship. Pierfrancesco Lapolla is supported by The Foundation Blanceflor Boncompagni Ludovisi, née Bildt scholarship. Anirudh Chandrashekar is supported by the Sloane-Robinson/Clarendon scholarship. The remaining authors have no disclosures.
