Abstract

Vascular endothelial dysfunction is an early manifestation of cardiovascular disease and is associated with incident cardiovascular events. 1 Brachial artery reactivity testing (BART) is used to noninvasively assess responsiveness to reactive hyperemia (flow-mediated dilation [FMD]) and evaluate nitric oxide [NO]-dependent large vessel endothelial function.2–4 Although BART is the gold standard for the noninvasive evaluation of endothelial function, the technique requires substantial time and technical expertise for implementation. Sidestream darkfield (SDF) imaging, in contrast, is a rapid, noninvasive modality to assess abnormalities of the microcirculation and evaluate the thickness and permeability of endothelial glycocalyx, a proteoglycan-rich protective barrier coating the luminal surface of the vascular endothelium. 5 SDF imaging has been proposed as a valid method to assess microvascular endothelial integrity by the European Society of Cardiology.5,6 However, the relationship between macrovascular and microvascular function is uncertain. We sought to determine associations between BART and abnormalities of the microcirculation by SDF imaging.
The study was approved by the NYU School of Medicine Institutional Review Board and all participants provided written informed consent. Adults aged ⩾ 18 years with and without systemic lupus erythematosus (SLE) were prospectively enrolled into a study investigating platelet activity and vascular function. Demographic data and clinical comorbidities were recorded. All participants underwent SDF microscopy of the sublingual microvasculature to measure the microvascular percent red blood cell (RBC) filling, the proportion of time in which microvascular segments are perfused, and the perfused boundary region (PBR), the extent to which RBC penetrate the glycocalyx in vessels 5–25 µm in diameter. 7 Increased PBR indicates the glycocalyx is thin, with impaired barrier function and greater susceptibility to vascular endothelial damage. Lower % RBC filling indicates poor microvascular perfusion. To evaluate the reproducibility and within-subject variability of sublingual SDF imaging over time, a separate cohort of four healthy individuals underwent serial measurements (on days 0, 14, and 20). BART was performed in all participants. Brachial artery diameter was measured with high-resolution vascular ultrasound before and after transient arterial occlusion for 5 minutes (endothelium-dependent FMD) and after sublingual nitroglycerin (endothelium-independent nitroglycerin-mediated dilation [NMD]), as previously described. 2 Reduced FMD reflects impaired endothelial-dependent vasodilation indicative of vascular dysfunction. NMD serves as an index of endothelium-independent vasodilation. 4 Categorical variables are reported as frequencies and percentages and continuous variables are shown using mean (SD) and median (IQR). Associations between measures were assessed by Spearman’s correlations. Statistical tests are two-sided and p < 0.05 was considered statistically significant.
A total of 53 participants underwent BART and SDF imaging (on the same day in 98% of cases). The mean participant age was 40.6 ± 14.7 years, 96.2% were women, and 45.3% were White; SLE was present in 77.4%, hypertension in 24.5%, dyslipidemia in 7.5%, diabetes mellitus in 3.8%, and established atherosclerotic cardiovascular disease in 3.8%. The median FMD was 3.1% (IQR 1.6–5.3%), median NMD was 29.9% (IQR 23.3–36.0%), median PBR was 1.90 µm (IQR 1.72–2.10 µm), and median % RBC filling was 73.6% (IQR 68.5–78.0%). There was an inverse correlation between measures of FMD and PBR (r = −0.29 [95% CI −0.02 to −0.53], p = 0.034; Figure 1A) and a positive correlation between FMD and % RBC filling (r = 0.31 [95% CI 0.03 to 0.54], p = 0.024; Figure 1B). No correlation between NMD and PBR or % RBC filling was observed (Figures 1C and 1D). Among four healthy individuals with multiple measures over time, SDF imaging demonstrated good reproducibility with an 8.1% and 4.1% intra-assay coefficient of variation for PBR and % RBC filling, respectively.
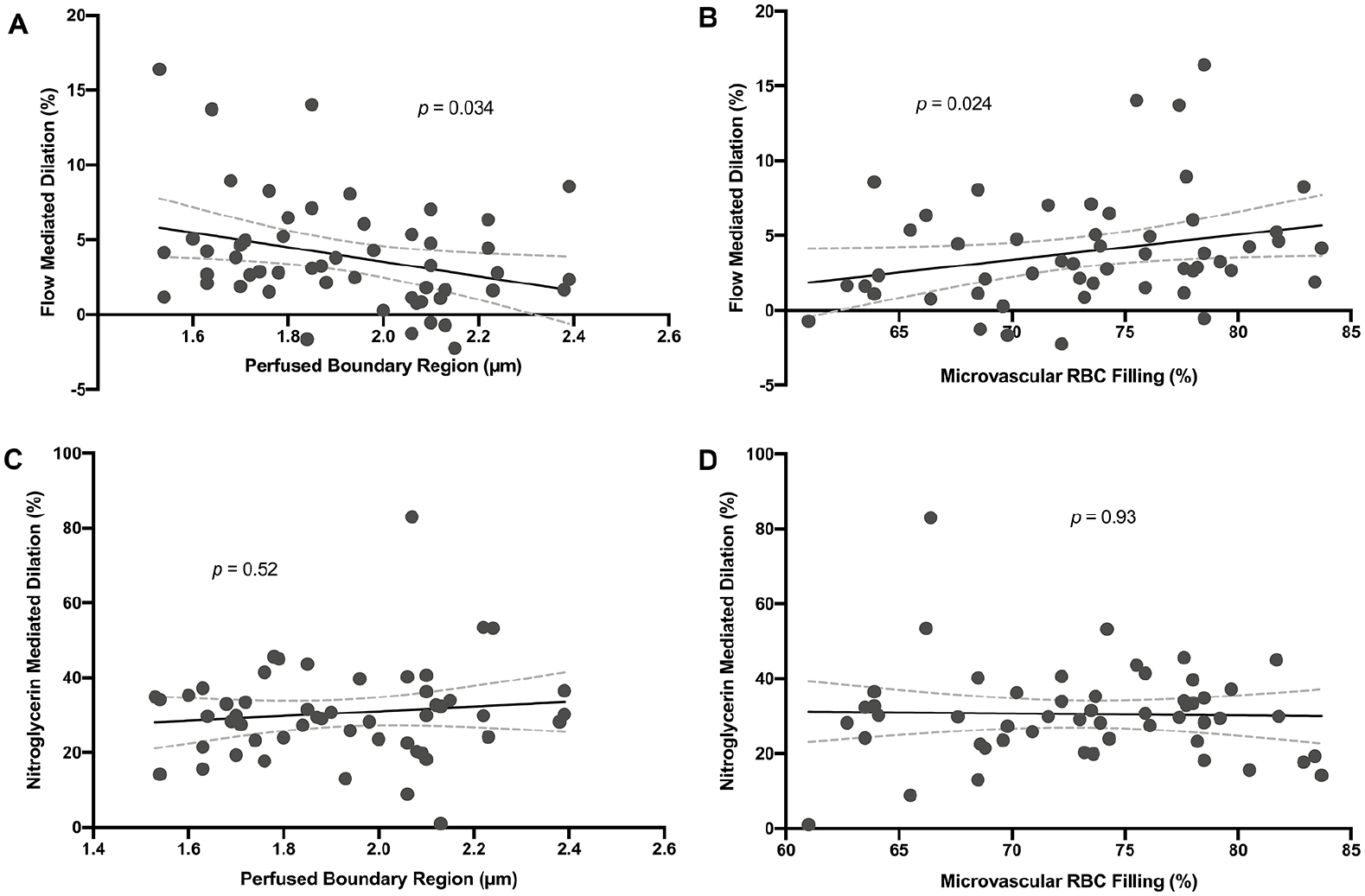
Correlations between measures of FMD and PBR
In this observational study of 53 adults who underwent vascular function testing with sublingual SDF imaging and BART, we observed microvascular RBC filling and microvascular endothelial glycocalyx thickness were significantly associated with large-vessel endothelial function. These data demonstrate that vascular endothelial dysfunction impacts both microvascular arterioles (~5 to 25 µm in diameter) and large caliber brachial arteries (⩾ 4 mm). Notably, there was no association between the endothelial-independent NMD and microvascular measures, reinforcing the observation that these microvascular properties are proxies for endothelial-dependent vascular function. Sublingual SDF microvascular imaging provides an easily implemented, noninvasive method to assess vascular endothelial function and may serve as a surrogate marker for BART in research and clinical settings. The generalizability of our findings is potentially limited by selection bias due to predominant enrollment of women with autoimmune disease. Risk factors for coronary artery disease were prevalent in our study sample, but the presence of subclinical atherosclerotic vascular disease was not systematically evaluated. Lipids, inflammatory cytokines, and other biomarkers associated with vascular endothelial function were not measured. Still, these considerations are unlikely to affect the observed relationship between BART and SDF imaging.
In conclusion, we observed correlations between microvascular glycocalyx thickness, microvascular RBC filling, and brachial artery FMD. Additional studies in diverse populations are warranted to determine whether sublingual microvascular SDF imaging may be a valid surrogate measure for noninvasive assessment of vascular endothelial function.
Footnotes
Declaration of conflicting interests
Dr Smilowitz serves on an advisory board for Abbott Vascular. The remainder of the authors report no conflicts of interest to disclose.
Funding
This study was funded by the National Heart, Lung, and Blood Institute of the National Institutes of Health (R01HL139909 to JPB and JSB). Dr Smilowitz is supported by the National Heart, Lung, and Blood Institute of the National Institutes of Health under Award Number K23HL150315. Dr Berger is funded, in part, by the National Heart, Lung, and Blood Institute of the National Institutes of Health (R35HL144993). The National Heart, Lung, and Blood Institute of the National Institutes of Health did not have any direct role in the conduct or reporting of the study findings.
